Abstract
Objective:
Neurocognitive dysfunction is a common feature of bipolar disorder even in euthymia, and psychopharmacological treatment could have an effect on cognition. Long-term prescription of benzodiazepines in bipolar disorder is a common practice, and their effect on neurocognition has not been well studied in this population. The aim of this study was to evaluate the impact of concomitant benzodiazepine long-term use on neurocognitive function in stable euthymic bipolar disorder patients.
Methods:
Seventy-three euthymic bipolar disorder outpatients and 40 healthy individuals were assessed using a neurocognitive battery. Patients were classified in two groups according to the presence of benzodiazepines in their treatment: the benzodiazepine group (
Results:
Both bipolar disorder groups (benzodiazepine and non-benzodiazepine) showed an impairment in memory domains (Immediate Visual Memory [
Conclusion:
Although memory and processing speed impairments were found in bipolar disorder, regardless of their benzodiazepine treatment, benzodiazepine users presented additional neurocognitive impairments in terms of executive functioning. These findings support restricted prescription of benzodiazepines in individuals with bipolar disorder.
Introduction
Neurocognitive dysfunction is common in bipolar disorder (BD), even in asymptomatic patients. Features include impairments across inhibitory control, set shifting, verbal memory and sustained attention domains (Bora et al., 2009; Bourne et al., 2013). Neurocognitive deficits have been shown to be a major predictor of functional outcomes (Baune and Malhi, 2015; Tabarés-Seisdedos et al., 2008). Therefore, a goal of research should be identifying the potential risk factors that contribute to neurocognitive impairment so as to enhance functional recovery between affective episodes.
Among risk factors influencing neurocognition in BD, genetic vulnerability, subsyndromal depressive symptoms, history of psychotic symptoms, illness chronicity and comorbidities are well documented (Balanzá-Martínez et al., 2015; Berk et al., 2017). Recent research has suggested that polypharmacy and side effects of some psychotropic drugs used to treat BD may further compound these deficits (Balanzá-Martínez et al., 2010; Sabater et al., 2016). Several meta-analyses have examined the effects of concomitant lithium, anticonvulsants, antipsychotics and antidepressants on neurocognition (Bora et al., 2009; Bourne et al., 2013). However, cognitive effects associated with adjunctive benzodiazepines (BZDs) remain relatively unexplored in BD (Balanzá-Martínez et al., 2010; Correa-Ghisays et al., 2017; Martino et al., 2008). In one study, current use of BZDs in BD was related to impairments in attention, psychomotor speed and executive functioning (Martino et al., 2008). In addition, BZD use exerted a detrimental effect on manual motor speed, whose dysfunction is considered a neurocognitive endophenotype of BD (Correa-Ghisays et al., 2017).
Detrimental neurocognitive side effects of BZDs in other populations are well established. In healthy young individuals, short-term use of BZDs is associated with anterograde amnesia, diminished attention and psychomotor slowing (Woods et al., 1992). Similarly, long-term treatment has been associated with impairments in executive functioning, working memory, delayed recall or visuospatial domains (Boeuf-Cazou et al., 2011; Crowe and Stranks, 2018; Stewart, 2005). In elderly individuals, long-term BZD use has been related to a higher risk of dementia (Pariente et al., 2016). It remains uncertain whether this reflects direct pharmacological effects, or the role, of BZDs as markers of, e.g., insomnia or anxiety.
Despite the evidence of cognitive side effects of BZDs in different clinical populations, concomitant chronic use of BZDs is common in BD and their long-term impact in cognition remains unknown (Yatham et al., 2013). The aim of the present study was therefore to examine the effect of concomitant BZD use on neurocognition in stable euthymic BD patients who have had 2 years of sustained BZD treatment, compared to BD patients not treated with BZDs and a healthy control group. We hypothesized that both groups of euthymic BD patients would show a poorer neurocognitive performance than the control group, sharing basal cognitive disturbances attributable to the disease (Bora et al., 2009). In addition, we hypothesized that the BD group treated with BZDs would show a different neurocognitive profile to the BD group not treated with BZDs, with greater impairment in attention, memory domains, executive functioning and visuospatial domains, concordant with the limited studies of patients with BD who are users of BZDs (Correa-Ghisays et al., 2017; Martino et al., 2008) and general population studies of long-term users of BZDs (Boeuf-Cazou et al., 2011; Crowe and Stranks, 2018; Stewart, 2005).
Methods
Participants
A final sample of 73 BD outpatients from the Bipolar Disorders Unit at ‘La Fe’ University and Polytechnic Hospital (Valencia, Spain) and 40 healthy individuals recruited through advertising in the community were enrolled in this cross-sectional study. The Ethics Committee of ‘La Fe’ Health Research Institute approved this study (SM I 22/2011).
Eligible BD patients older than 18 years were included if they (a) had been clinically stable for at least 2 years (i.e. without a manic or depressive episode) – stability was defined at each routine medical visit by a score = 1 on two self-administered scales (a modified version of the Life-Chart (Livianos et al., 2003) and the Spanish Version of the Chinese Polarity Inventory (Benavent et al., 2004)) and also on the Clinical Global Impressions Scale for BD (CGI-BP) (Vieta et al., 2002; completed by the treating psychiatrist); (b) had been treated with the same mood stabilizer and with documented presence or absence of BZD treatment for at least 2 years; (c) did not have a comorbid axis I or II psychiatric diagnosis; and (d) had not received electroconvulsive therapy in the previous 2 years. Eligible controls were included if they had no history of psychiatric disorder. Finally, all eligible participants had to (a) be older than 18 years; (b) have no history of neurological or major medical disorders; (c) be not receiving treatments that could affect neurocognition (e.g. corticosteroids, antihistamines, anticholinergics and urinary antispasmodics); and (d) be euthymic at the time of the assessment.
To ensure eligibility criteria, BD patients were excluded if they did not fulfil the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (
A total of 386 BD participants and 47 controls were screened to obtain the final study sample (
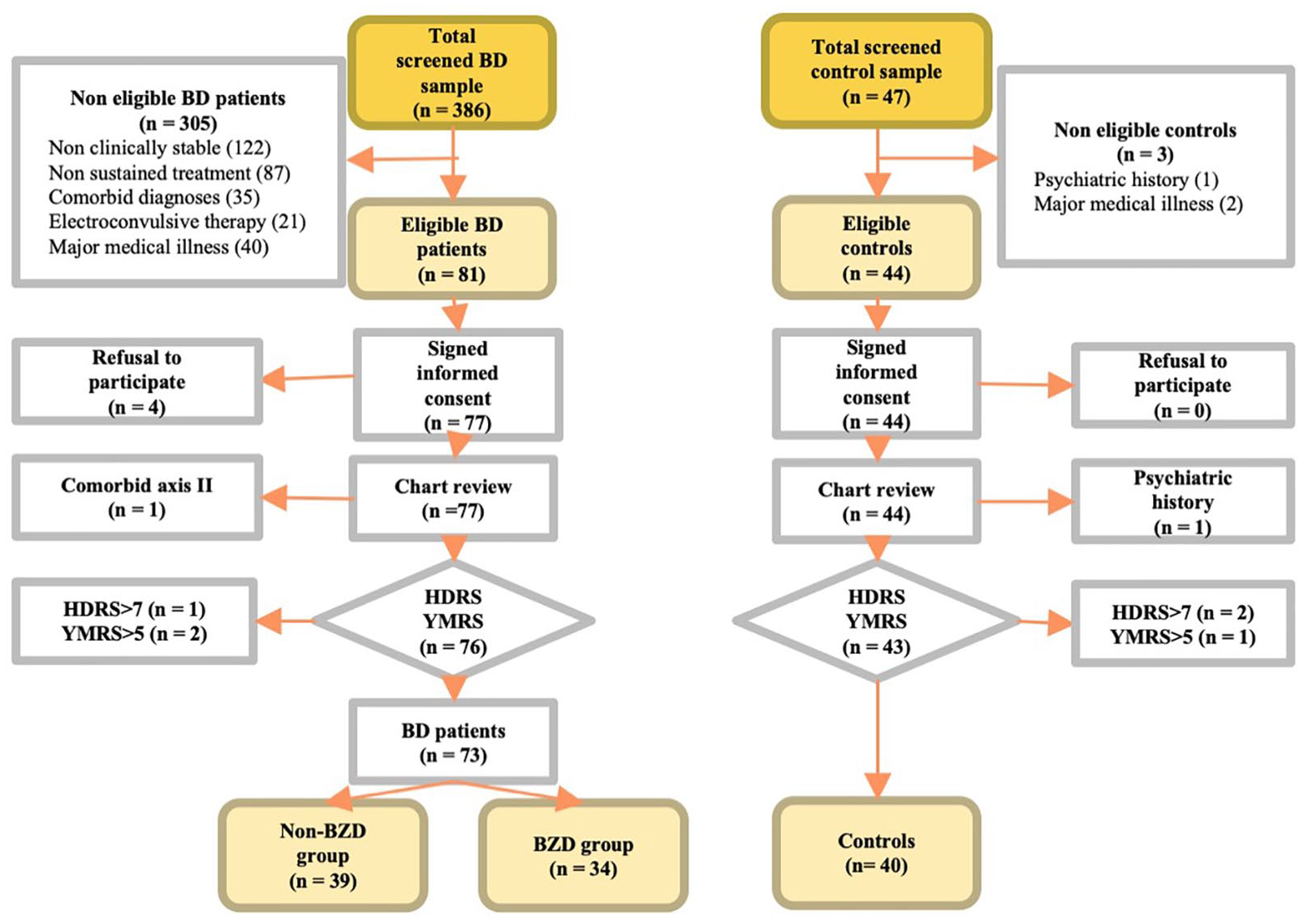
Flowchart of sample selection.
Materials
The following neurocognitive domains were assessed: global intelligence (Wechsler Adult Intelligence Scale-III [WAIS-III] (Wechsler, 2001)); attention (Digit Span, WAIS-III (Wechsler, 2001) and Trail Making Test Part-A [TMT-A] (Fernández et al., 2002)); processing speed (Colour of the Stroop Colour-Word Test (Golden, 2001)); memory (Wechsler Memory Scale-Revised [WMS-III-R] (Wechsler, 1997)); praxia (copy Rey Complex Figure Test [RCFT] (Pena-Casanova et al., 2009)); and executive function using the Interference of the Stroop Colour-Word Test (Golden, 2001), Wisconsin Card Sorting Test (WCST; Grant and Berg, 2001), Tower of Hanoi (TOH-4; Humes et al., 1997), Trail Making Test Part-B (TMT-B; Fernández et al., 2002) and Frontal Assessment Battery (FAB; Hurtado-Pomares et al., 2018). All the neurocognitive tests used had normative validation data for the Spanish population.
Procedure
After signing an informed consent form, all participants completed a demographic interview and the HDRS and YMRS, which were carried out by a postgraduate clinical psychology intern. In addition, current symptomatology was assessed with the Symptom Checklist-90-Revised (SCL-90-R) Spanish validated version (González de Rivera et al., 1989).
Moreover, BD participants individually completed a semi-structured interview to determine clinical illness variables, which were completed with their case note review. The following clinical data were collected: BD subtype, polarity of the index episode, history of psychotic symptoms, age at onset, number of episodes (mania and depression), duration of clinical stability (defined as time since the last affective episode) and the type of psychopharmacological medications in their treatment (i.e. lithium, anticonvulsants, antipsychotics antidepressants). In a second session on the following day, an experienced clinical psychologist conducted the neurocognitive testing.
Data analyses
We conducted an omnibus multivariate analysis of covariance (MANCOVA) with group (control, non-BZD and BZD) as an independent variable and neurocognitive domains as dependent variables. According to differences among groups (see Table 1), age, HDRS, YMRS, antipsychotic drugs and the number of depressive episodes were included as covariates to control for possible confounders. When the effects were significant, we conducted post hoc tests to control for type I errors. Bonferroni’s test was used to assess between-group comparisons. Finally, bivariate Pearson correlations were conducted to check the association between BZD doses (i.e. equivalent mean diazepam dose) and the performance of neurocognitive domains in the BZD group. Data were analysed using SPSS 26.0 for Windows.
Demographic and clinical data across groups.
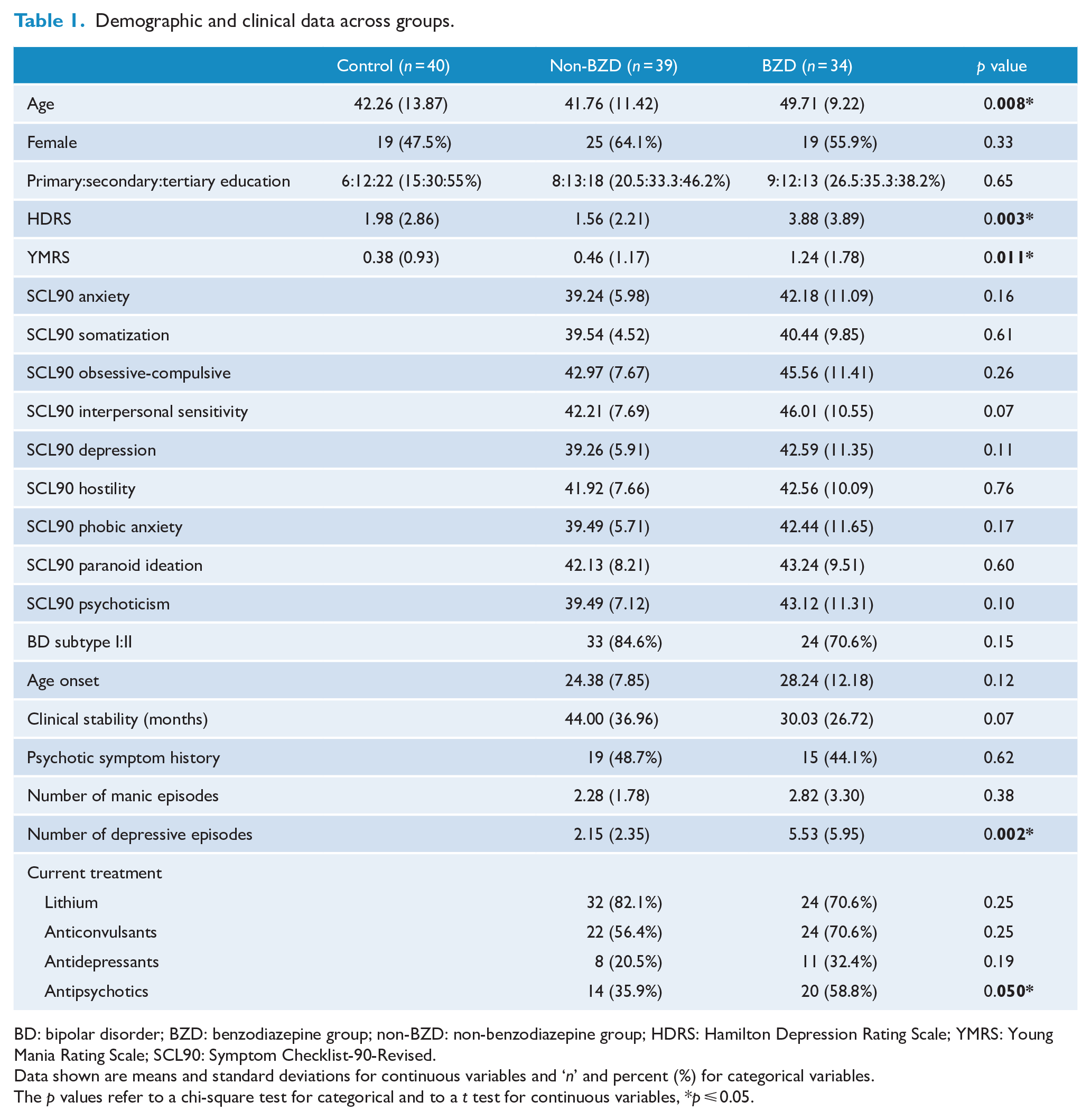
BD: bipolar disorder; BZD: benzodiazepine group; non-BZD: non-benzodiazepine group; HDRS: Hamilton Depression Rating Scale; YMRS: Young Mania Rating Scale; SCL90: Symptom Checklist-90-Revised.
Data shown are means and standard deviations for continuous variables and ‘
The
Results
Demographic and clinical details are presented in Table 1. Descriptive data regarding BZD prescription, types and doses are presented in Table 2. Table 3 shows neuropsychological assessment results across all groups (control, non-BZD and BZD) as well as MANCOVA analyses of group effect, using age, depressive and manic symptoms, antipsychotic drugs and number of depressive episodes as covariates. Results of ANCOVA of covariates are outlined, if significant, by superscript letters.
Types of BZDs used in the BZD group, reasons for prescription and equivalent mean diazepam dose.
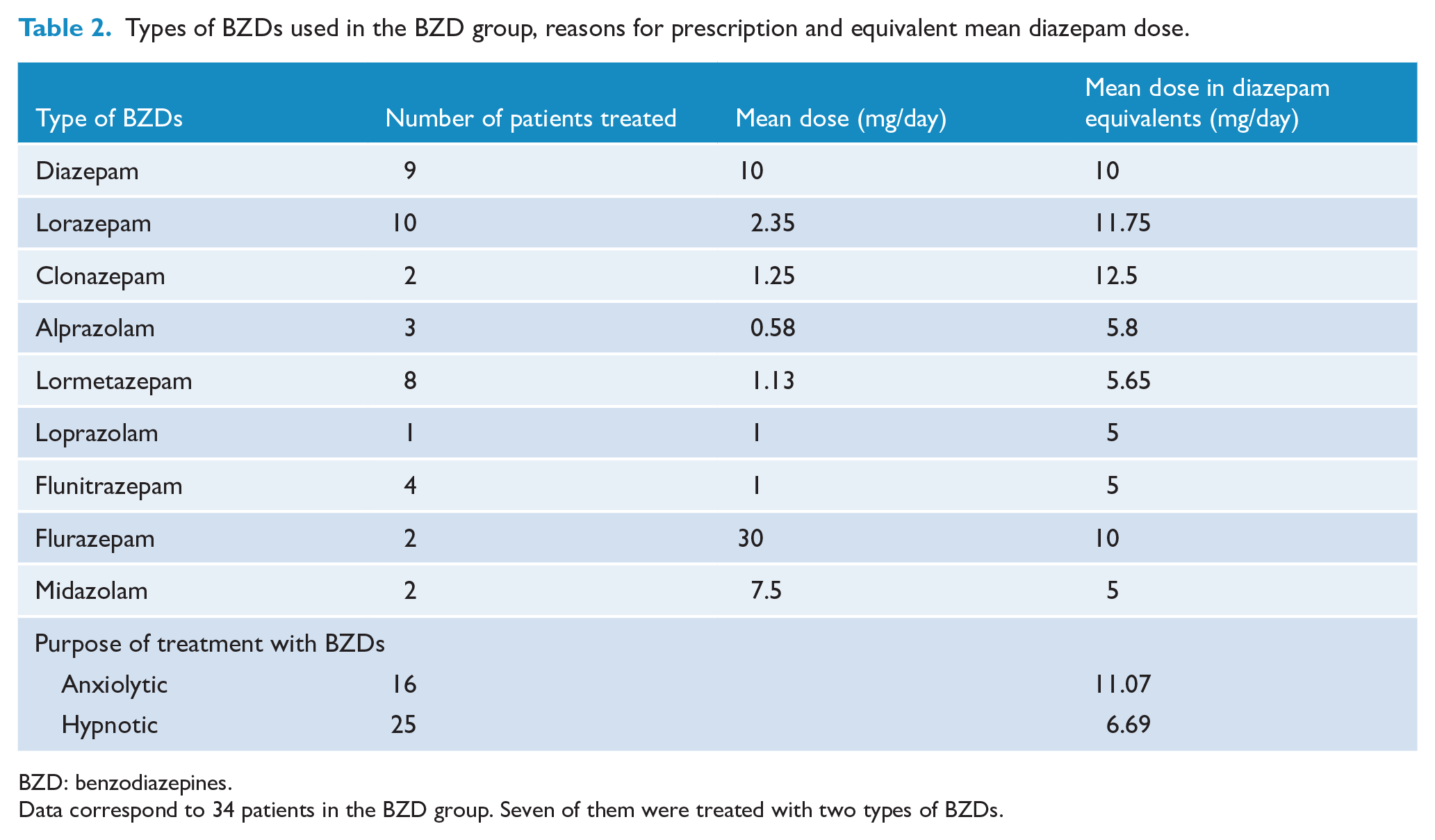
BZD: benzodiazepines.
Data correspond to 34 patients in the BZD group. Seven of them were treated with two types of BZDs.
Descriptive data on neurocognitive functioning across groups and MANCOVA of group effect, using age, depressive and manic symptoms, antipsychotic drugs and number of depressive episodes as covariates.
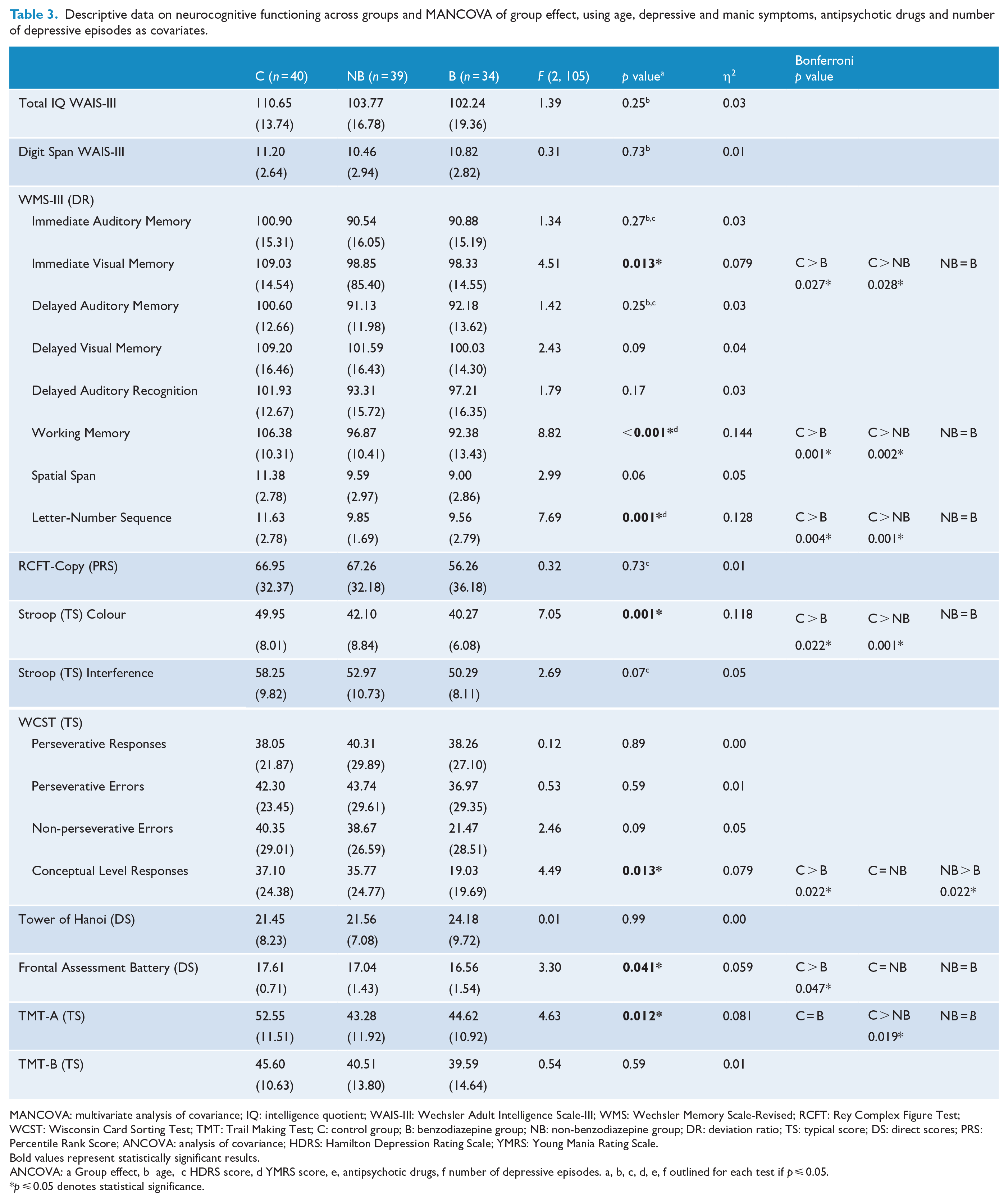
MANCOVA: multivariate analysis of covariance; IQ: intelligence quotient; WAIS-III: Wechsler Adult Intelligence Scale-III; WMS: Wechsler Memory Scale-Revised; RCFT: Rey Complex Figure Test; WCST: Wisconsin Card Sorting Test; TMT: Trail Making Test; C: control group; B: benzodiazepine group; NB: non-benzodiazepine group; DR: deviation ratio; TS: typical score; DS: direct scores; PRS: Percentile Rank Score; ANCOVA: analysis of covariance; HDRS: Hamilton Depression Rating Scale; YMRS: Young Mania Rating Scale.
Bold values represent statistically significant results.
ANCOVA: a Group effect, b age, c HDRS score, d YMRS score, e, antipsychotic drugs, f number of depressive episodes. a, b, c, d, e, f outlined for each test if
After controlling for potential confounders, analyses revealed statistically significant differences among groups on the combined neuropsychological dependent variables,
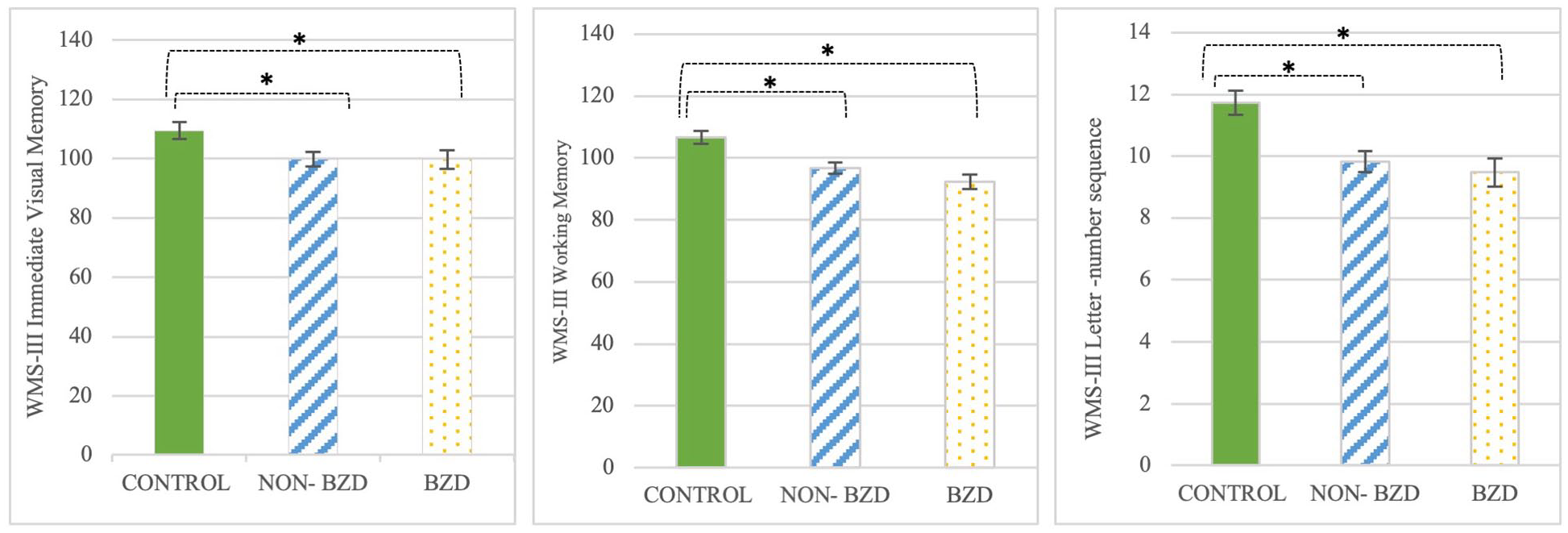
WMS-III subtest performance with differences between groups.
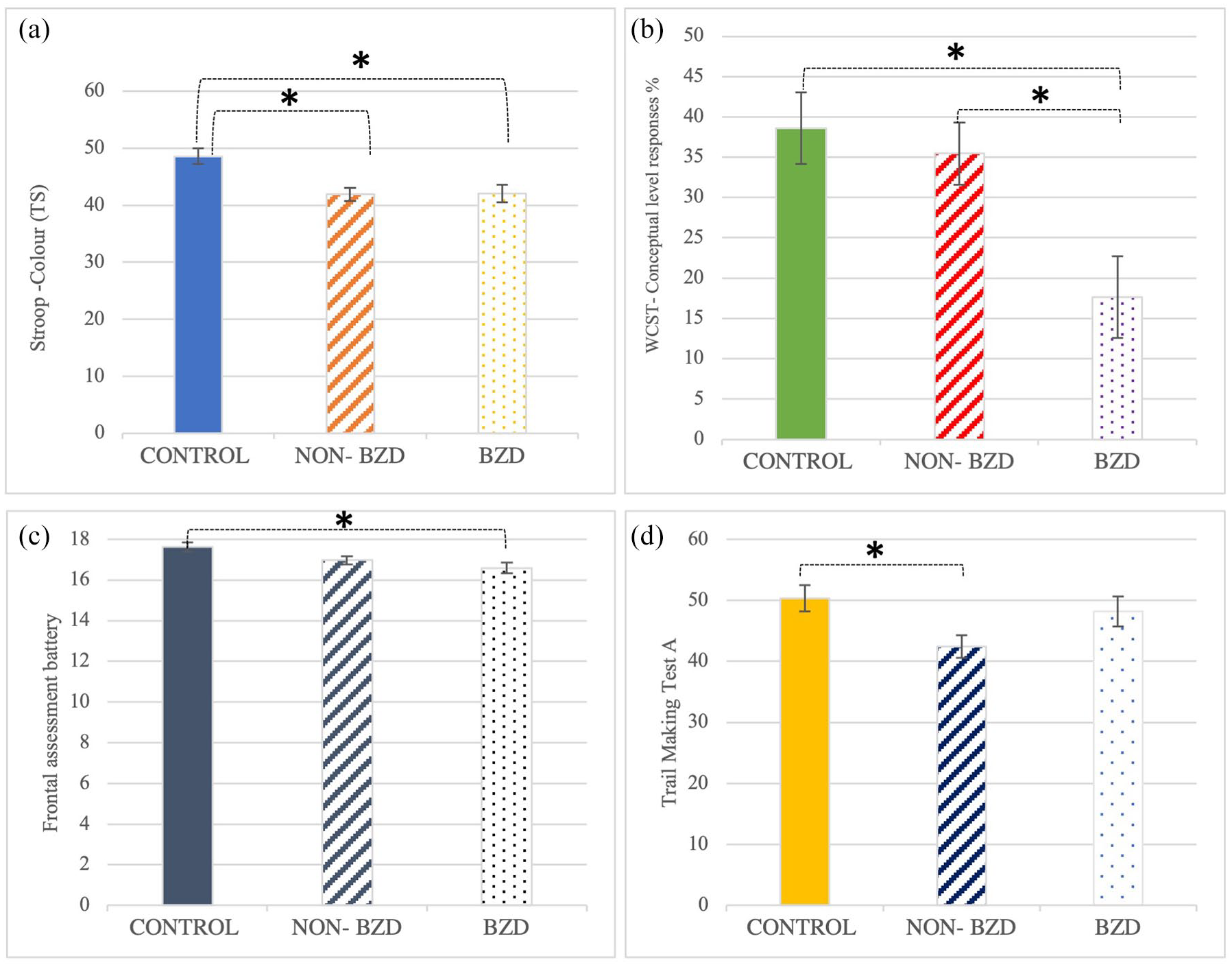
Neurocognitive performance with differences between groups in (a) Stroop Colour, (b) Conceptual Level Responses, (c) Frontal Assessment Battery and (d) Trail Making Test A.
When between-group comparisons were examined, four patterns of findings were observed: (a) a neurocognitive deficit in all BD participants (non-BZD and BZD) relative to the control group, (b) a neurocognitive deficit in BD participants who take BZDs relative to the control group, (c) a greater neurocognitive deficit in the BZD group relative to both the control and the non-BZD groups, and (d) a neurocognitive deficit in BD participants in the non-BZD group relative to the control group. First, Bonferroni’s test indicated that both clinical groups (non-BZD and BZD) showed worse performance than the control group in several WMS-III domains: Immediate Visual Memory (
As for the association between equivalent mean diazepam doses and performance of neurocognitive domains, higher BZD doses were linked to worse performance on WMS-III Immediate Auditory Memory (
Discussion
In our study, BD participants showed deficits in short-term visual memory and certain domains of executive functions and attention, specifically working memory and processing speed, compared to healthy individuals. Importantly, unlike non-BZD participants, those who took BZDs showed a greater deficit in some executive functions (i.e. frontal functioning) relative to the control group. Indeed, BZD patients showed worse executive functions in terms of abstract thinking than control individuals as well as non-BZD participants. Finally, praxia and certain executive domains (perseverative responses and the purpose of planning) were preserved in BD participants, regardless of BZD status. To the authors’ knowledge, this is the first study that addresses and highlights the particular cognitive long-term effects of concomitant BZD prescription in BD.
As for neurocognitive deficits in BD, regardless of BZD status, our findings indicated that short-term visual memory, some attention domains and executive functions of working memory were impaired in euthymic BD patients. Despite the variability in the affected cognitive domains among different studies (Bora, 2015; Bourne et al., 2013), neuropsychological performance in our BD participants was similar to the recent individual meta-analytic evidence on euthymic patients in which impaired cognitive domains indicate a fronto-temporal and fronto-lymbic involvement (Bourne et al., 2013).
BD participants who took BZDs, unlike those not taking BZDs, additionally showed greater impairment in certain executive functions such as frontal functioning or measures of abstract thinking. However, both clinical groups did not show differences in terms of delayed recall, visuospatial functioning or working memory as found in other studies with long-term BZD users (Boeuf-Cazou et al., 2011; Crowe and Stranks, 2018; Stewart, 2005). Exposure to BZDs in BD individuals has been correlated with worse performance in measures of psychomotor speed, attention and executive functions; however, mean doses were higher than those in our study, and duration of exposure was not specified (Martino et al., 2008). In this sense, studies with BZD abusers found diffuse cognitive dysfunction, with deficits in verbal memory, working memory, visuospatial memory and attention (Federico et al., 2017). In our sample, higher BZD doses were significantly associated with worse performances in auditory memory domains and interference; however, cognitive performance in these areas was not significantly different between groups. Of note, our study was carried out in BD participants with a close follow-up and adjuvant BZD treatment was prescribed by their treating psychiatrists, so higher mean doses than the ones used in our sample may in part explain the differences in the extent of cognitive dysfunction.
With respect to attention, the non-BZD BD group (but not the BZD group) performed worse than controls in TMT-A, but this difference was not seen on Digit Span (WAIS-III). Attention is one of the most affected cognitive domains in BD (Bourne et al., 2013) and can also be impaired because of the acute effect of BZDs, which is influenced by the elapsed time since the last dose was administered (Lucki et al., 1986; Stewart, 2005). However, interestingly, long-term users of BZDs are likely to develop tolerance to sedation or impaired attention, minimizing their influence on cognition (Buffett-Jerrott and Stewart, 2002). The tolerance phenomenon induced by long-term use of BZDs in our sample might explain the slight differences in this domain between non-BZD and BZD groups. Other studies in BD populations have suggested deleterious effect of BZDs on the manual motor speed domain of cognition, although this domain was not evaluated in our study (Correa-Ghisays et al., 2017).
Biological mechanism may explain the impact of long-term use of BZDs on neurocognition. While the γ-aminobutyric acid type A (GABA-A) receptor α1 subunit is related to anterograde amnesia and sedation, the α5 subunit has been found to modulate the temporal and spatial memory effects of BZDs (Tan et al., 2011). Global brain inhibition could hinder neuroplasticity compensatory mechanisms, exacerbating existent cognitive impairments (Pariente et al., 2016). In accordance, several cohort studies have examined the risk of dementia in BZD users in the general population, especially with high accumulated doses (Pariente et al., 2016; Wu et al., 2011). However, other studies of BZD long-term users indicated that the cognitive performance of individuals diagnosed with an anxiety-related disorder may not be different in long-term users of BZDs, former users and non-users (Lucki et al., 1986; Stewart, 2005). Discrepancies among studies may be influenced by methodological differences. Also, a protopathic bias cannot be excluded since the prescription of BZDs could indicate the existence of dementia prodromes (anxiety and insomnia; Pariente et al., 2016).
BZDs in euthymic BD are frequently prescribed for hypnotic or anxiolytic indications as in our cohort. Both sleep disruptions and comorbid anxiety are common in euthymic patients (Meyer et al., 2020; Pavlova et al., 2017). Moreover, these commorbidities are related to worse cognitive functioning (Bradley et al., 2020; Kauer-Sant’Anna et al., 2009). It is uncertain if the withdrawal of BZDs can improve cognitive performance (Baandrup et al., 2017), as it can also cause a transient worsening in symptomatology or even trigger affective relapses (Spoorthy et al., 2019). This issue comprises a therapeutic challenge because of the risk of iatrogenic mood effects or the fact that polypharmacy also contributes to cognitive impairment (Balanzá-Martínez et al., 2010; Spoorthy et al., 2019). This suggests the need to design studies that allow the weighing of risk and benefits so as to choose the best psychopharmacologic strategy.
The main strength of our study is the selection of a long-term euthymic sample with stable pharmacological treatment, as well as the systematic clinical evaluation and neuropsychological assessment. Differences among groups that could contribute to cognitive performance such as age, residual symptoms, number of depressive episodes and treatment with antipsychotic drugs were controlled for and adjusted within the statistical model. Other clinically relevant variables, such as general symptoms, history of psychotic symptoms or type of mood stabilizers, were equally distributed among groups. Thus, interpreted with caution and mindful of protopathic bias, our findings suggest that BZDs may contribute to a worse neurocognitive performance, regardless of polypharmacy.
Our study has several limitations to take into consideration. First, because of the cross-sectional design, the contribution of BZDs to neurocognitive performance can only be suggested. A prospective study would be necessary to explore cognitive changes over time and their relationship with treatment. Second, although the potential neuroprotective effect of lithium compared with other mood stabilizers and polypharmacy was adjusted for (Bourne et al., 2013; Sabater et al., 2016), the detrimental effect of BZDs on neurocognition may be associated with higher doses of medication (Balanzá-Martínez et al., 2010; Martinez-Aran et al., 2007). Third, anxiety is a possible effect modifier; nevertheless, SCL-90-R data show that there were no differences in symptomatology (e.g. anxiety, phobic anxiety, obsessive-compulsive; see Table 1). Fourth, as executive functioning is one of the most replicated impaired functions in BD (Bora et al., 2009), several tests were used to assess this domain. This aspect might increase the likelihood of finding differences among groups. Further research exploring whether these findings also apply to other cognitive domains is warranted. Prospective studies are needed to clarify the influence of low daily doses and sporadic intake of BZDs in this population. Finally, the lack of psychiatric prescriptions in the control group could also magnify the difference effect of pharmacologic treatment in BD groups, and the BZD intake in the BZD group. Thus, our results should be interpreted with caution, taking into account noted limitations.
Conclusion
Our findings support previous research regarding cognitive dysfunction in euthymic BD and suggest that BZDs may worsen these deficits. These data have clinical implications in the treatment of BD, as it seems prudent to restrict prescription of BZDs to PRN use, low doses and limited periods of time. In addition, it suggests that clinicians could implement alternate evidence-based strategies, such as psychotherapeutic approaches including cognitive behavioural therapy (CBT), mindfulness-based CBT, relaxation training and chronotherapeutic strategies (Gottlieb et al., 2019), or pharmacological strategies, such as adjusting the dose of mood stabilizer (Yatham et al., 2013). These data are also concordant with studies suggesting an adverse effect of BZDs on course and prognosis, and again suggest caution in BZD prescription (Bobo et al., 2015; Perlis et al., 2010).
Footnotes
Author Contributions
A.G.-B., L.L. and A.S. designed the study. P.N., Y.C., V.B.-M. and A.S. carried out preliminary review of the topic. A.S., P.S. and L.L. recruited the sample. A.S. and A.G.-B. carried out the neurocognitive assessment of the sample. Y.C. and A.G.-B. performed the data analysis. M.B., P.S. and S.D. interpreted the results and their potential limitations. V.B.-M., A.G.-B., P.N. and Y.C. wrote the draft. All authors supervised the final version of the article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Y.C. and P.N. have had support for conference from Janssen, Lundbeck, Angelini and Pfizer. P.S. has had engagements in the last 5 years with Janssen, Lundbeck, Sanofi, Servier, Rovi and Adamed. V.B.-M. has been a consultant, advisor or continuing medical education (CME) speaker in the last 5 years for Angelini, Ferrer, Juste, Lundbeck, Nutrición Médica and Otsuka. None are related to the current paper. M.B. has had engagements in the last 5 years with Janssen Cilag, Allergan, AstraZeneca, BioAdvantex, Bionomics, Collaborative Medicinal Development, Grunbiotics, LivaNova, Lundbeck, Merck, Mylan, Otsuka and Servier. S.D. has received grant support from the Stanley Medical Research Institute, National Health and Medical Research Council (NHMRC), Beyond Blue, Australian Rotary Health Research Fund (ARHRF), Simons Foundation, Geelong Medical Research Foundation, Harry Windsor Foundation, Fondation FondaMental, Eli Lilly, Glaxo SmithKline, Organon, Mayne Pharma and Servier; speaker’s fees from Eli Lilly; advisory board fees from Eli Lilly and Novartis; and conference travel support from Servier. A.S. and A.G.-B. have no potential conflicts of interest regarding this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: V.B.-M. is supported by the national grant (PI16/01770) from the Carlos III Health Institute. P.N. is supported by a Carlos III Health Institute (Spanish Ministry of Economy and Innovation) ‘Rio Hortega’ fellowship (CM19/00078). M.B. is supported by National Health and Medical Research Council (NHMRC) Senior Principal Research Fellowships (APP1059660 and APP1156072). A.G.-B. acknowledges a FIS Project (PI18/01352) and ‘Juan Rodés’ grant (JR17/00003) from the Carlos III Health Institute and cofinanced by the European Development Regional Fund ‘A way to achieve Europe’.
Previous Presentation
Partial results presented at the XXII Spanish National Conference of Psychiatry, Bilbao, Spain, 26–28 September 2019 (abstract, poster).
