Abstract
Background
Relapse rates in major depressive disorder (MDD) remain high even after treatment to remission. Identifying predictors of relapse is, therefore, crucial for improving maintenance strategies and preventing future episodes. Remote data collection and sensing technologies may allow for more comprehensive and longitudinal assessment of potential predictors.
Methods
The Canadian Biomarker Integration Network in Depression Wellness Monitoring for MDD (CBN-WELL) study was a prospective, multicentre observational study with an aim to identify biomarkers associated with relapse in patients on maintenance treatment for MDD. Participants had a DSM-5-TR diagnosis of MDD in remission and a Montgomery–Åsberg Depression Rating Scale (MADRS) score ≤14. Participants remained on their baseline medication regimens and were followed bimonthly for up to 2 years. Relapse criteria included MADRS > 22 for 2 consecutive weeks, suicidality or hospitalization, and initiation or change in medication for worsening symptoms. Data collection included clinical assessments, self-report questionnaires, and remote monitoring using wrist-worn actigraphs and smartphones.
Results
A total of 96 participants had follow-up data. Of these, 28.9% experienced a depressive relapse during the study period, with an average time to relapse of 211 days. Baseline depressive severity, as measured by MADRS, was higher in participants who relapsed compared to those who did not, but few other baseline clinical measures differentiated these groups.
Conclusions
Individuals with MDD in remission continued to have high relapse rates despite maintenance treatment. The paucity of clinical factors that predict relapse underscores the need for biomarkers. The CBN-WELL database can be used for future research to integrate multiple predictive factors and to identify objective measures to predict relapse in individuals.
Introduction
Major depressive disorder (MDD) is a common condition with an estimated global prevalence of about 1 in 20 people suffering from a current episode. 1 MDD is also a leading medical cause of increasing disability; the rate of disability-adjusted life years associated with depressive disorders has increased by more than 16% between 2010 and 2021. 2 Although many evidence-based treatments are available and the majority of people with MDD respond to initial treatment, these individuals are still at considerable risk of relapse and recurrence. 3 Studies indicate that between one-fifth to one-half of patients in remission from a depressive episode will suffer a recurrence within 2 years.4–10 Each relapse/recurrence is associated with an increased risk of future recurrence, 11 as well as reduced quality-of-life 12 and functioning. 13 Note that, generally, relapse is defined as an early return of symptoms of the original depressive episode, while recurrence refers to the later emergence of a new episode. However, because the definitions vary widely and there is no empirical evidence to differentiate between the original and a new depressive episode,14–16 we use the term relapse to indicate both relapse and recurrence.
Understanding the mechanisms underlying MDD relapse is critical, as this would allow for enhanced prediction of such events, creating opportunities for the development of more targeted treatments and improved preventative interventions.14,17 However, prior research has investigated a limited range of individual predictors, many of which are related to demographics and clinical history.7,9,18 For example, after treatment, a higher risk of relapse has been associated with the number and severity of previous episodes, the persistence of residual symptoms and a history of childhood maltreatment.9,15,19–23
In recent years, mobile and remote biosensors have become useful tools for assessing mental health conditions.24–26 Remote recording tools via the internet or smartphones allow patients to complete self-assessments at home27,28 and reduce participant burden and the need for frequent clinic visits. Remote biosensors can assess potential biomarkers such as motor activity and sleep patterns over long periods.29–31 These data can now be reliably recorded and may be important factors in depressive relapse in addition to, or combined with clinical information. However, the use of these technologies for predicting relapse requires longitudinal assessment over a protracted period, which can complicate the recording of potentially relevant data. 32 Hence, the feasibility of using these measures in this clinical context must be examined.
To investigate MDD relapse using these and other technologies, the Canadian Biomarker Integration Network in Depression (CAN-BIND) conducted a prospective, longitudinal, observational study, termed the CAN-BIND Wellness Monitoring for MDD (CBN-WELL) study. The primary aim of the study was to identify biomarkers of relapse in individuals with MDD who had responded to various treatments. The study collected a broad range of data by using remote monitoring and sensing in addition to collecting clinical data at regular study visits. We now report the methodology of CBN-WELL, as well as the primary clinical outcomes related to depression relapse, including the characteristics of this cohort, relapse rates, time to relapse, and initial comparisons of individual baseline measures between relapse and nonrelapse groups. For the latter, we also examined those with and without mild residual depressive symptoms during follow-up.
Methods
Participants and Setting
The CBN-WELL study (NCT02934334) is a prospective, multicentre, longitudinal observational study conducted from July 2016 to January 2019 to identify biomarkers of relapse in MDD. The protocol was designed to be similar to other observational studies of MDD relapse, such as the OBSERVEMDD study (NCT02489305), with common eligibility criteria, relapse criteria, outcome measures, and visit schedules.
Participants from a previous CAN-BIND study as well as other participants treated to remission were recruited at 5 clinical centres in Canada (Vancouver, Calgary, Toronto, Hamilton, and Kingston). Ethics approval was obtained from institutional review boards at each site and all participants provided written informed consent. Participants received financial compensation for their time in the study.
Entry inclusion criteria included: (1) outpatient age 18 to 65 years; (2) DSM-5 diagnosis of MDD in remission from their most recent major depressive episode (MDE) as determined by the Mini-International Neuropsychiatric Interview 33 ; (3) treated to remission from their most recent MDE; (4) Montgomery–Åsberg Depression Rating Scale (MADRS) 34 total score ≤14 at screening and baseline visits; (5) willing to complete self-report assessments via a study-specific smartphone and to wear a wrist-worn activity device for the duration of the study; and (6) sufficiently fluent in English.
Exclusion criteria included: (1) MDD with psychotic features (lifetime); (2) bipolar I or bipolar II diagnosis (lifetime); (3) schizophrenia, schizoaffective disorder, or any other primary psychiatric diagnosis other than MDD; (4) significant personality disorder (e.g., borderline and antisocial); (5) high suicidal risk, defined by clinician judgment; (6) alcohol or substance use disorder with at least moderate severity, within 6 months before screening; (7) significant neurological disorders, head trauma, or other unstable medical conditions; and (8) received an investigational drug (including investigational vaccines) or used an invasive investigational medical device within 90 days before screening, or currently enrolled in an investigational study.
Current or past treatments for MDD were not restricted, including antidepressant medication, psychotherapy, and neurostimulation. There were no study-related treatments or interventions. Concomitant medications, including vitamins, supplements, contraceptive agents, and over-the-counter pain medication for stable medical conditions, were allowed at the discretion of the study psychiatrist.
Procedures
The CBN-WELL study consisted of a screening phase of up to 2 weeks and an observational phase of variable duration of up to 2 years. During the observational phase, participants attended study visits at baseline and every 8 weeks thereafter and were assessed for indicators of relapse, including depressive symptom severity, medication status, and health service utilization, in addition to other measures (see Supplemental Table S1 for a listing of procedures and measures). Self-report questionnaires were administered using a tablet device or computer. Blood samples were also collected. If indicators of depressive relapse (see below) were present at a study visit, participants were scheduled for a relapse verification visit within 1 to 2 weeks, during which relapse criteria were confirmed.
Participants also received a study-specific smartphone (LogPad®) and a study-specific wrist-worn actigraphy device (GT9X Link®) that was worn throughout the study period. Weekly self-reports were completed remotely at home via LogPad®. LogPad® also collected speech and voice characteristics at 2-week intervals. Motor activity and sleep parameters were captured using the GT9X Link®, worn throughout the study period, with data downloaded at each study visit. Furthermore, a subset of participants with eligible smartphones were invited to download HealthRhythms®, a mobile app that continuously records passive data. 35 These participants used the mobile app continuously for the entire study period and uninstalled the app upon study completion or withdrawal.
Assessments
The main clinician-assessed measures included the MADRS and Clinical Global Impression Scale, Severity (CGI-S). 36 Self-rated measures included the Quick Inventory of Depressive Symptomatology, Self-Rated (QIDS-SR), 37 Generalized Anxiety Disorder Scale-7 (GAD-7), 38 Sheehan Disability Scale (SDS), 39 and Quality of Life, Enjoyment and Satisfaction Questionnaire (QLESQ). 40 Other assessment measures are listed in Supplemental Table S2.
Relapse Definition
Relapse during the observational period was defined in the initial protocol as any of the following: (1) MADRS total score ≥ 22 for at least 2 consecutive weeks (e.g., at both a study visit and relapse verification visit); (2) hospitalization for worsening of depression; (3) suicidal ideation with intent, or suicidal behaviour; or (4) other reasons (to be described by the investigator). Subsequently, before any analysis was conducted, these criteria were revised and expanded to include: (1a) MADRS total score ≥ 22 for at least 2 consecutive weeks and a CGI-S change from baseline ≥2; (5) medication change suggestive of worsening depression (e.g., increase in antidepressant dose or adding an adjunctive agent); or (6) initiation of a new treatment for depression.
All relapses were adjudicated using these criteria and verified, prior to any analysis, by consensus of a panel of investigators (CNS, KH, RWL, RVM, and RU) using all available data. This panel also classified nonrelapse participants as having mild residual depressive symptoms (defined as MADRS total score >14 and <22 at one or more bimonthly study visits) or not having residual symptoms (defined as MADRS total score ≤14 at every bimonthly study visit). Figure S1 in the Supplemental Materials provides examples of the data used to define the relapse, nonrelapse with residual depressive symptoms, and nonrelapse without residual symptoms groups.
Statistical Analysis
We examined differences in demographic and clinical variables between the relapse, nonrelapse and dropout groups using analysis of variance and chi-square tests. For participants who experienced a relapse event, only data collected up until relapse were included in the present analyses. To visualize the time to relapse, we conducted a Kaplan Meier survival analysis. Descriptive analyses were conducted in SPSS 29.0 and survival analyses were conducted in R version 4.3.1 using the ggsurvfit package. 41
Results
A total of 102 participants were screened for the study, with 100 participants completing the baseline assessment and 94 completing Visit 1 assessments (8 weeks after baseline). Four participants dropped out before any follow-up data were collected and were excluded, while the other 2 had subsequent follow-up data after Visit 1 and were included; hence, the total number of participants available for outcome analysis was 96. Table 1 summarizes demographic and clinical information for the total sample and stratified by participant group. A total of 28 individuals (29.2%) experienced a relapse during follow-up, with an average length of time before relapse of 211 days (SD = 121) (Figure 1). The criteria for relapse met by the participants were: criterion 1a (n = 16 out of 28, 57%), criterion 2 (n = 1, 4%), criterion 3 (n = 2, 7%), and criterion 5 (n = 9, 32%).
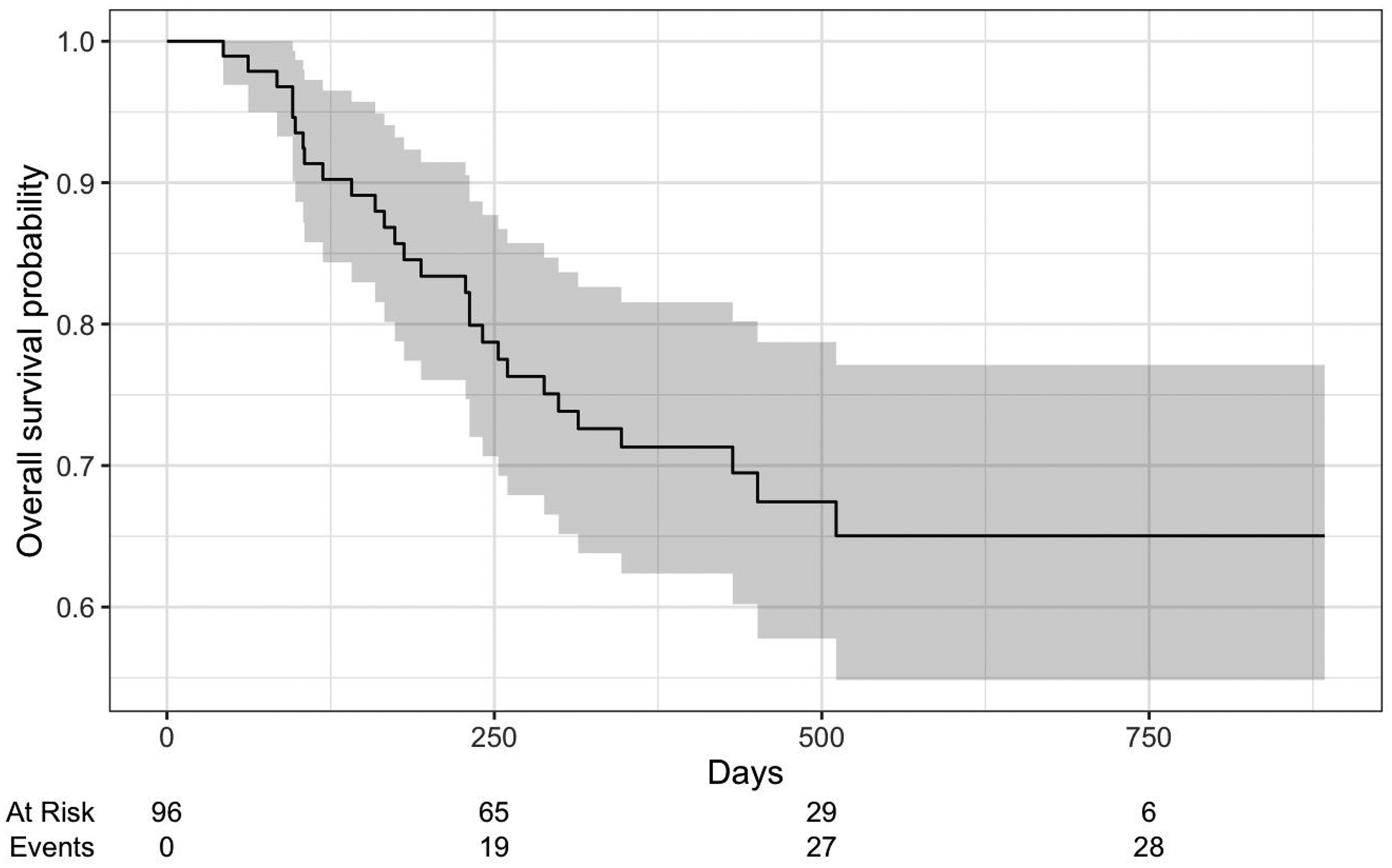
Kaplan–Meier survival curve for time to major depressive episode relapse.
Participant Characteristics for Relapse and Nonrelapse Groups.
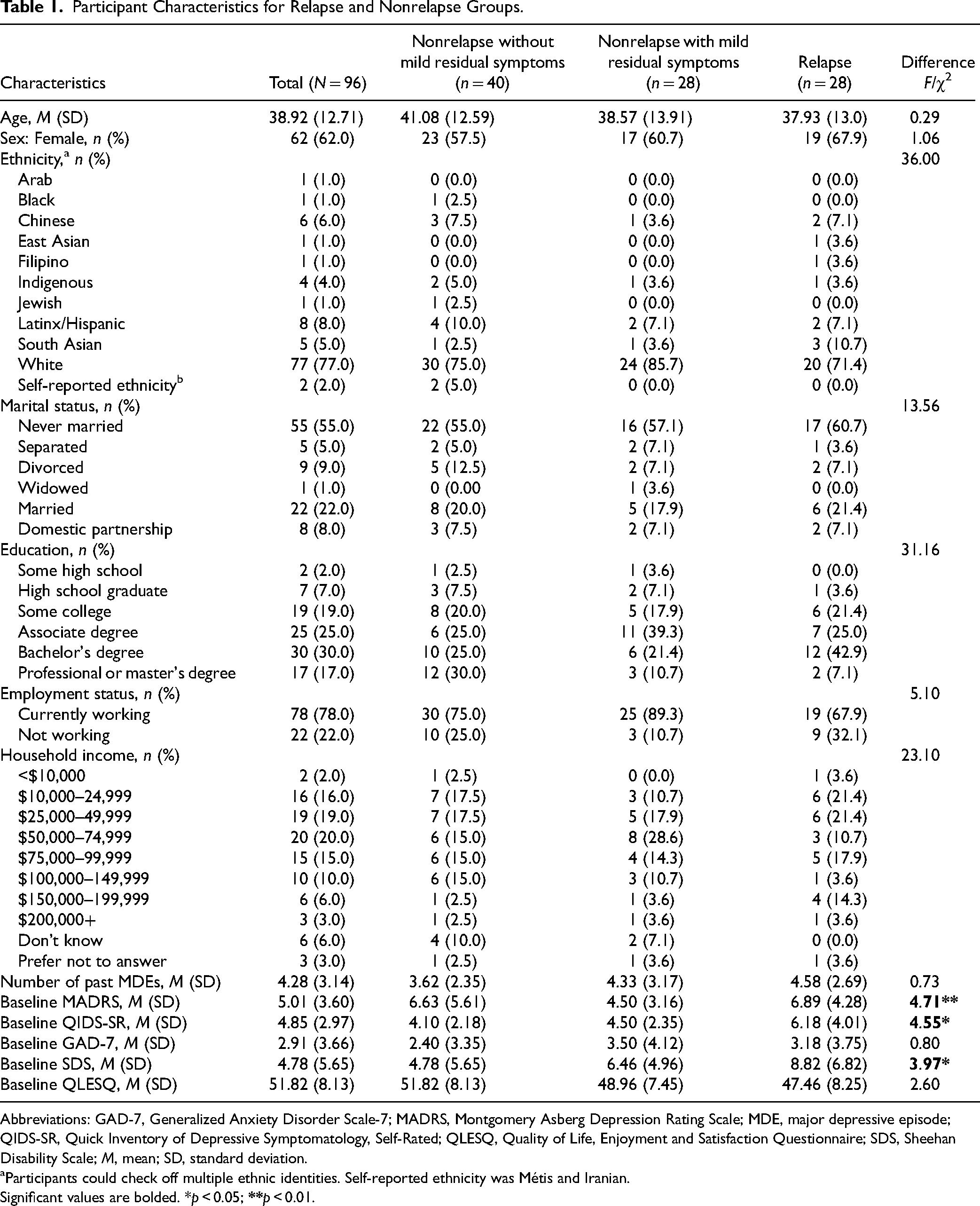
Abbreviations: GAD-7, Generalized Anxiety Disorder Scale-7; MADRS, Montgomery Asberg Depression Rating Scale; MDE, major depressive episode; QIDS-SR, Quick Inventory of Depressive Symptomatology, Self-Rated; QLESQ, Quality of Life, Enjoyment and Satisfaction Questionnaire; SDS, Sheehan Disability Scale; M, mean; SD, standard deviation.
Participants could check off multiple ethnic identities. Self-reported ethnicity was Métis and Iranian.
Significant values are bolded. *p < 0.05;
Of the 68 participants in the nonrelapse group, 28 (46.7%) were classified as having mild residual depressive symptoms during follow-up (i.e., those with MADRS >14 and ≤22 on at least 1 bimonthly study visit) while 40 (58.8%) were classified as not having residual symptoms (i.e., those with MADRS ≤14 at each bimonthly study visit). Those in the nonrelapse group were followed for an average of 451 days (SD = 203).
The 3 participant groups (relapse, nonrelapse with, and without mild residual symptoms) did not differ in terms of age, sex, ethnicity, marital status, education, employment status, household income, or number of previous MDEs, ps > 0.115. However, there was a significant group difference in baseline depressive symptom severity as assessed with the MADRS (F(3, 96) = 4.71, p = 0.004). Follow-up Tukey tests indicated that the relapse group had higher baseline MADRS scores (M = 6.89, SD = 4.28) compared to both the nonrelapse without residual symptoms group (M = 3.90, SD = 2.84, p < 0.049) and the nonrelapse with residual symptoms group (M = 4.50, SD = 3.16; p ≤ 0.049). Similarly, there were small overall differences among the groups in the QIDS-SR (F(3, 96) = 4.55, p = 0.015) and SDS (F(3, 96) = 3.97, p = 0.02). There were no other significant differences among groups (all ps ≥ 0.470).
Using the same definition for mild residual symptoms (MADRS total score >14 and <22 at 1 or more bimonthly study visits), 11 of the 28 participants (39.3%) in the relapse group showed a pattern of mild depressive symptoms in a study visit preceding the relapse event. There was no difference between the relapse and nonrelapse groups in the proportion of participants having mild residual depressive symptoms at a study visit (39.3% vs. 46.7%, respectively; χ2 = 0.03, p = 0.86).
Discussion
CBN-WELL is an observational study with the objective of identifying potential biomarkers of relapse in a well-characterized sample of participants in stable remission following treatment of MDD. Although most of the participants were on maintenance medications, we found that 28.9% experienced a depressive relapse during the 12-month follow-up period. This relapse rate is similar to those reported in other clinical settings7,22 and in meta-analyses of RCTs,6,8 which have shown that those who remain on antidepressants following remission had relapse rates of 21% to 49%.
The high relapse rates during maintenance treatment speak to the importance of identifying predictors of relapse. Except for a small difference in the baseline scores in the MADRS, QIDS-SR, and SDS (all of which were within the remission range), there were no baseline clinical characteristics that differentiated those who relapsed from those who did not. This is consistent with other studies where few clinical factors predicted relapse in patients after treatment for MDD.15,20–23
We also examined the pattern of remission during follow-up in the nonrelapse group. We found that almost half (46.7%) had periods with mild residual depressive symptoms, that is, they had at least 1 bimonthly study visit where their MADRS score ranged between 14 and 22. Residual depressive symptoms have been identified as a predictor of relapse in previous studies, but many participants with residual symptoms in this study did not have a subsequent relapse event. Similarly, 39.3% of those who relapsed also showed this pattern of elevated MADRS score on a study visit within 8 weeks prior to their relapse event; however, this was not a significantly higher proportion than those who did not relapse. This suggests that the presence of residual depressive symptoms, as indicated by total MADRS scores, is not an accurate predictor of who will go on to a relapse event.
While individual baseline measures may not be accurate predictors of relapse, combinations, longitudinal changes, or relationships between several measures may be more informative. 42 For example, we found that the correlation between depressive symptoms (as assessed by the QIDS-SR) and anxiety symptoms (as assessed by the GAD-7) examined longitudinally across time, termed the depression–anxiety coupling, was a robust predictor of relapse in this cohort. 43 This demonstrates how the rich and comprehensive CBN-WELL database can be leveraged to explore many potential clinical predictors and combinations of predictors, as novel indicators of future relapse.
Most clinical outcome measures are based on subjective patient reports, whether by self-rated questionnaires or by interviews for clinician-rated scales. These are subject to issues such as recall bias and negative cognitive bias. Thus, there is a need for more objective measures to predict relapse during maintenance treatment. Remote sensing measures, such as actigraphy, may be a promising objective measure of behaviour that can be standardized to an individual's baseline. 29 We have developed a method for longitudinal long-term tracking of actigraphy data using an open-source data processing pipeline that accounts for issues such as missing data, nonwear periods, and sleep-wake scoring, 44 making it suitable for long-term relapse prediction studies. Another objective measure used in CBN-WELL is a computerized assessment of implicit self-depressed cognitive associations, the Depression Implicit Association Test (DIAT). 45 We found that greater self-depressed associations at baseline and longitudinal increases across follow-up predicted a shorter time to relapse in this cohort. 46 The DIAT is a promising example of a simple, low-cost screening tool to identify patients at risk of relapse.
A strength of CBN-WELL is that our protocol (eligibility criteria, relapse criteria, outcome measures, and visit schedule) was aligned with other multicentre observational studies investigating digital biomarkers of relapse, such as the OBSERVEMDD study (NCT02489305). This allows for pooling of data to increase power, or for using one study as an independent external validation sample for predictors found in another study. For example, the predictive ability of actigraphy combined with symptom self-ratings was examined in a personalized N-of-1 relapse prediction model framework in OBSERVEMDD (n = 286). 47 The relapse prediction performance in the OBSERVEMDD cohort (balanced accuracy of 73.5%) was replicated in the independent CBN-WELL cohort (71%). 47 External validation in an independent sample is a powerful feature for biomarker validation but is still uncommonly applied in mental health studies. CBN-WELL provides a unique opportunity for future work to leverage this approach in predicting depressive relapse, which has the potential to yield stronger and more reliable conclusions in comparison to earlier work. 42
Finally, our study sample includes a diverse group of participants with MDD who were in variable periods of remission after treatment with a range of depression interventions, including medications, psychotherapy, and neurostimulation, and who were on different medication regimens during maintenance. This is both a strength and a limitation; although the heterogeneity of our sample may make it more difficult to identify a common predictor of relapse, it also represents a real-world population in which any potential biomarker will need to be validated. Other limitations include our broad criteria for a relapse event, which included medication changes for worsening symptoms; it is possible that these participants would not have met full relapse criteria with watchful waiting, in the same way, that many participants with residual symptoms did not go on to experience a full relapse.
In conclusion, CBN-WELL is a multicentre observational study of potential predictors of relapse for a well-characterized sample of participants with MDD in stable remission following treatment. The relapse rate in this cohort over a 1-to-2-year follow-up was high (28.9%), and very few baseline clinical factors were predictive of relapse, consistent with results from previous studies. Only small differences in baseline depressive and anxiety symptom scores differentiated those who relapsed from those who did not. However, the rich CBN-WELL database, the use of remote and sensing technology for outcome assessment, and the alignment of the protocol with other observational studies will enhance the discovery of predictors of relapse that have implications for optimizing early intervention and prevention efforts. Future studies should focus on examining the combinations between measures, longitudinal changes in measures, and the use of objective assessments for relapse prediction.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437251337603 - Supplemental material for Predicting Relapse of Depressive Episodes During Maintenance Treatment: The Canadian Biomarker Integration Network in Depression (CAN-BIND) Wellness Monitoring in Major Depressive Disorder Study: Prédire la rechute d’épisodes dépressifs pendant le traitement d’entretien : Une étude de suivi du bien-être dans les troubles dépressifs majeurs du Réseau canadien d’intégration des biomarqueurs pour la dépression (CAN-BIND)
Supplemental material, sj-docx-1-cpa-10.1177_07067437251337603 for Predicting Relapse of Depressive Episodes During Maintenance Treatment: The Canadian Biomarker Integration Network in Depression (CAN-BIND) Wellness Monitoring in Major Depressive Disorder Study: Prédire la rechute d’épisodes dépressifs pendant le traitement d’entretien : Une étude de suivi du bien-être dans les troubles dépressifs majeurs du Réseau canadien d’intégration des biomarqueurs pour la dépression (CAN-BIND) by Raymond W. Lam, Katerina Rnic, John-Jose Nunez, Keith Ho, Joelle LeMoult, Abraham Nunes, Trisha Chakrabarty, Jane A. Foster, Benicio N. Frey, Kate L. Harkness, Stefanie Hassel, Sidney H. Kennedy, Qingqin S. Li, Roumen V. Milev, Lena C. Quilty, Susan Rotzinger, Claudio N. Soares, Valerie H. Taylor, Gustavo Turecki and Rudolf Uher in The Canadian Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AN, BNF, JJN, JL, KH, KLH, KR, LCQ, RU and SH disclose no relevant conflict of interests. CNS has received consulting honoraria from Eisai, Lundbeck, Otsuka, Diamond Therapeutics and Bayer and research grants from Ontario Brain Institute, SEAMO Innovation Funds, and Clairvoyant. JAF has served on the Scientific Advisory Board for MRM Health NL and has received consulting/speaker fees from Klaire Labs, Takeda Canada, WebMD, and Rothman, Benson, Hedges Inc. QSL is an employee of Janssen Research and Development, LLC. RWL has received honoraria for ad hoc speaking or advising/consulting, or received research funds, from Abbvie, Asia-Pacific Economic Cooperation, Bausch, BC Leading Edge Foundation, Brain Canada, Canadian Institutes of Health Research, Canadian Network for Mood and Anxiety Treatments, Carnot, CB Solutions, Healthy Minds Canada, Janssen, Lundbeck, Michael Smith Foundation for Health Research, MITACS, Neurotorium, Ontario Brain Institute, Otsuka, Shanghai Mental Health Center, Vancouver Coastal Health Research Institute, Unity Health, and VGH-UBCH Foundation. RVM has received consulting and speaking honoraria from AbbVie, Eisai, Janssen, Lallemand, Lundbeck, Neonmind, and Otsuka, and research grants from CAN-BIND, CIHR, Janssen, Nubiyota, and OBI. SHK has received research funding or honoraria from Abbvie, Abbott, Brain Canada, Boehringer-Ingelheim, Canadian Institutes of Health Research, Janssen, Lundbeck, Lundbeck Institute, Merck, Neurocrine, Ontario Brain Institute, Otsuka, Pfizer, SPOR (Canada's Strategy for Patient-Oriented Research), Sunovion and Servier; and holds stock/stock options in Field Trip Health. SR has grant funding from the Ontario Brain Institute and holds a patent on “Teneurin C-Terminal Associated Peptides (TCAP) and methods and uses thereof.”
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The CBN-WELL clinical study (NCT02934334) was carried out as a collaboration between Janssen Research & Development, LLC, USA and the Canadian Biomarker Integration Network in Depression (CAN-BIND), with funding support from the Ontario Brain Institute, the Ontario Research Fund – Research Excellence Program, and Janssen Research & Development, LLC. CAN-BIND is an Integrated Discovery Program carried out in partnership with, and financial support from, the Ontario Brain Institute, an independent nonprofit corporation, funded partially by the Ontario government. The opinions, results, and conclusions are those of the authors and no endorsement by the Ontario Brain Institute is intended or should be inferred. Additional CAN-BIND funding was provided by the Canadian Institutes of Health Research, Janssen, Lundbeck, Bristol-Myers Squibb, Pfizer, and Servier. Funding and/or in-kind support is also provided by the investigators’ universities and academic institutions. KR was funded by a Killam Postdoctoral Fellowship, a Social Sciences and Humanities Research Council (SSHRC) Postdoctoral Fellowship, and a Michael Smith Health Research BC Trainee Award. JJN was partially supported by a Marshall Fellowship Award from the UBC Institute of Mental Health. JL is partially funded by a Michael Smith Health Research Scholar Award. The funding sponsors had no role in the study design, conduct, analysis, interpretation, and write up or publication.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
