Abstract
Background
Stress and traumatic experiences are well-established risk factors for psychiatric disorders. Stressful events can induce symptoms of anxiety and depression and may lead to overt psychosis, especially when there is an innate biological vulnerability. This study explores the role of the stress-regulating endocannabinoid system, specifically the activity of the enzyme fatty acid amid hydrolase (FAAH), a key regulatory enzyme for endocannabinoids, in association with stress by analysing data from healthy individuals and patients with psychosis.
Methods
We performed a post-hoc exploratory analysis on 65 positron emission tomography scans using the selective FAAH radioligand [11C]CURB, encompassing 30 patients with psychosis (6 female) and 35 healthy controls (19 female). The study aimed to examine the association between FAAH activity and stressful life events, assessed through the Recent Life Events, Survey of Life Experiences, and Hassles and Uplifts Scale.
Results
There was a significant difference regarding the number of recent stressors with higher levels in patients compared to healthy subjects (Survey of Life Experiences: t = 4.88, p < 0.001, hassles: t = 3.14, p = 0.003), however there was no significant relationship of brain FAAH activity and stressful life events in any of the applied scales across groups (Recent Life Events: F1,57 = 0.07, p = 0.80; Survey of Life Experiences: F1,57 = 1.75, p = 0.19; hassles: F1,56 = 1.06, p = 0.31). Linear mixed models performed separately for each group revealed that there was a positive association between FAAH activity and Recent Life Events in patients with psychosis only (F1,25 = 8.07, p = 0.009).
Conclusions
Our data reveal a significant disparity in recent stressors between the two groups, and a correlation between brain FAAH activity and stressful life events in patients with psychosis only. This suggests a complex interplay between stress and the endocannabinoid system.
Plain Language Summary Title
How Stress Affects the Brain’s Endocannabinoid System in Early Psychosis: A PET Study
Plain Language Summary
This study examines the relationship between stress and the endocannabinoid system (ECS) in individuals with early psychosis and healthy participants, using advanced PET imaging techniques. The endocannabinoid system is important for regulating mood, stress, and emotional responses, and previous research suggests it may be altered in people with psychotic disorders. By looking at how recent stressful experiences affect ECS activity, our work aims to understand whether the relationship of stress and the ECS is different in early psychosis compared to healthy individuals. Participants with early psychosis and healthy controls completed questionnaires about life stressors and received PET scans measuring the activity of the ECS. There was a significantly larger number of stressors in patients with early psychosis as expected. Furthermore, there was a positive relationship between recent life stressors and the activity of the ECS in patients with psychosis only. This could indicate a different regulatory response to stressors in the ECS in early psychosis. By investigating the role of the ECS in the context of recent stress, this study contributes to a deeper understanding of the complex mechanisms behind stress and psychotic disorders, with the aim of developing more effective treatment strategies.
Keywords
Introduction
Endocannabinoids exert similar effects in the human body as the well-known phytocannabinoids present in cannabis. They are involved in several human processes, such as relaxation, immune modulation, regulation of appetite, body temperature and neuronal function. Most importantly endocannabinoids regulate the response to stress and have a key role in restoring the body's energy stores after exhaustion or stress. 1 Endocannabinoids additionally contribute significantly to the regulation of cognitive and emotional processes, such as learning, memory formation and the extinguishing of fear responses, which are particularly relevant in the context of anxiety and trauma-related experiences.2–4 Anandamide and 2-arachidonoylglycerol are among the most extensively researched constituents of the endocannabinoid system, known for their affinity to bind to cannabinoid receptor types 1 (CB1) and 2 (CB2). These regulatory neurotransmitters are synthesized post-synaptically and engage with receptors located on the pre-synaptic membrane, effectively modulating the subsequent release of neurotransmitters. 5 Thereafter, they are degraded by specific enzymes such as fatty acid amid hydrolase (FAAH) or monoacylglycerol lipase (MAGL). 1 Upon experiencing stress, endocannabinoids are released to terminate the stress response and to facilitate recovery after the stressor has ended. 6 However, in the case of severe stressors the activity of the endocannabinoid system is downregulated in order to ensure an adequate fight or flight response. 7 This is supported by studies demonstrating that pharmacological disruption of endocannabinoid signalling in rodents leads to hyperarousal, enhanced stress response and anxiety.8,9 Vice versa higher activity of the endocannabinoid system is associated with better mood in individuals with traumatic experiences.10, 11 Enhanced endocannabinoid tone is furthermore responsible for fear-extinction due to its involvement in memory consolidation, retrieval and extinction. 12
It is well known that stress and traumatic events are risk factors for the development of psychosis. In psychosis, alterations of circulating endocannabinoids have been confirmed in multiple studies (see work by Garani et al. 13 for review). Additionally, while no variations were observed in the activity of the degrading enzyme FAAH in patients with psychosis as compared to healthy controls, a correlation was identified with symptom severity and the stage of illness. 14 Therefore, we wanted to investigate how stressful life events are associated with the activity of brain FAAH in patients with first episode psychosis (FEP) and healthy controls. We used the well-validated [11C]CURB positron emission tomography (PET) and we quantified stressful life events with three different stress-related psychometric scales to encompass as many possible facets of perceived stressful life circumstances as possible. We hypothesize that (1) patients with FEP will have more stressful life events compared to healthy controls and that (2) stressful life events will be positively associated with FAAH in patients with first-episode psychosis, premised on the hypothesis that a heightened number of stressors would lead to adaptive modifications within the endocannabinoid system, thereby influencing FAAH activity.
Methods and Materials
Participants
The study was conducted according to a repository protocol that enabled a re-assessment of pre-existing data, which was approved by the Centre for Addiction and Mental Health research ethics board and now approved under Clinical and Translational Sciences (CaTS) bioBank by the research ethics board of the Centre Intégré Universelle de Santé (CIUSSS), de l’Oule-de-Monceautaire de l’Ouest (Mental Health and Neuroscience). All participants were able to understand the study and provided consent (established using the MacArthur Competence Assessment Tool for Treatment (MacCAT-T 15 ) according to the Declaration of Helsinki. Data was collected from April 2014 to March 2020.
Participants aged from 18 to 40 years were screened negative for drug use and were excluded in case of the presence of a substance use disorder. Patients self-reported lifetime cannabis use was documented during a detailed interview, in which the amount of cannabis (g) used was estimated as accurately as possible. Healthy controls were excluded if they or a first-degree relative met criteria for a psychotic disorder. Patients with psychosis met criteria as specified by DSM-IV criteria (schizophrenia, schizoaffective disorder, schizophreniform disorder, delusional disorder or psychosis not otherwise specified). Patients with psychosis were assessed during a comprehensive clinical interview and clinical notes (when available) were taken. Their treatment history was recorded and their antipsychotic medication was converted to chlorpromazine equivalent. 16
Subjects were excluded if they were pregnant, breast feeding, had a medical or neurological disease, had a positive head trauma history, or in case of presence of contraindications for magnetic resonance imaging (MRI). Individuals (n = 65) from this cohort partially overlap with study samples from previously published work. 14
Assessment of Stressful Life Events
Recent Life Events
In this questionnaire, adapted from the List of Threatening Experiences from the work of Brugha et al., 17 participants were asked to report whether they experienced one out of nine life events within the last year to assess the psychological burdens stemming from illnesses, fatalities, severe financial distress, and similar major life events.
Survey of Recent Life Experiences
In addition to the above-mentioned scale, the 51-item Survey of Recent Life Experiences (SRLE) scale was applied.18, 19 This scale quantifies the levels of stress attributable to challenges encountered within the domains of employment, interpersonal relationships, social connections and familial dynamics over the preceding month.
Hassles and Uplifts
The Hassles and Uplifts scale 20 assesses smaller daily hassles and uplifts. Only the hassles sections of this questionnaire were used for the analysis, in which respondents are asked to assess the frequency and intensity of daily irritations and challenges across life domains including family relationships, financial stability, health, work, social interactions and personal habits, as of the preceding day of the PET scan visit. This assessment aims to quantify the cumulative effect of these minor stressors on an individual's psychological well-being.
Of the included subjects Recent Life Events data was available for 30 patients with patients with psychosis and 32 healthy controls. Survey of Recent Life Experiences was not available for one FEP patient, and one healthy control. For three healthy controls the Hassles and Uplifts scale was not available.
PET Using [11C]CURB
Subjects received a high-resolution PET on a CPS/Siemens HRRT (CPS/Siemens, Knoxville, TN) scanner, measuring radioactivity in a total of 207 slices with an interslice distance of 1.2 mm. An intravenous line was used to administer radioligand applications. An arterial line was used to take arterial blood samples from the contralateral arm. Thermoplastic masks were used and a head fixation was used to minimise head motion. A single photon source with 137Cs (t1/2 = 30.2 years, Eγ 662 keV) was used for transmission scans to attenuate correction of emission data. [11C]CURB was administered over a 60 s period (Holliston, MA, Harvard Apparatus). Data were recorded in list mode for 60 min. Full scanning procedures have been described elsewhere. 14
Arterial Sampling of [11C]CURB
Blood was taken for an arterial sample for the initial 22.5 min post tracer injection using an automated blood sampling system (Model PBS-101, Veenstra Instruments, Joure, The Netherlands). Blood was also taken at 3, 7, 12, 15, 20, 30 and 45, as well as 60 min post tracer injection using a manual blood sampling system. For the counting of radioactivity in total blood and plasma, Packard Cobra II or Wizard 2480 γ-counter was used (Packard Inc. Co.,Meridian, CT) and cross-calibrated with the PET system. A validated method was used for the determination of the parent radioligation and its metabolite fraction in each manual blood sample (except the sample at minute 15). For fitting the blood to plasma radioactivity ratios, a biexponential function was used. For determining the parent plasma fraction a Hill function was applied. Please refer to Rusjan et al. 21 to know how dispersive and metabolite corrected arterial plasma input function was generated.
Kinetic Analysis
To quantify [11C]CURB binding to FAAH, a 2-tissue-compartment model with irreversible binding compartment (2-TCMi) and arterial input functions was applied to 60 min data as previously validated. 21 The composite rate constant lambda k3 (λk3), provided by the 2-TCMi, is sensitive to changes in FAAH levels, 22 is highly reproducible 22 and is not dependent on regional cerebral blood flow, a common limitation of irreversible radioligands. 21 [11C]CURB PET image analysis and kinetic modelling pipeline (PELI v.2021.1 and FMOD v.1.7.3) was developed by Dr. Pablo Rusjan 23 and validated for [11C]CURB. 21 For quality control purposes, PET images and arterial blood modelling files are examined before and throughout the analysis pipeline.
The original PET and arterial blood files are centrally stored and initially annotated by PET technicians and radiochemists before being made available to PET scientists. Typical annotations include visual inspection of motion of the subject during the PET scan, missing manual blood samples, automatic blood sample system stoppages and/or alarms (status flags indicating whether peristaltic pump was still in function). Annotated issues were resolved, and files used for analysis were manually inspected before image and kinetic analysis. Mistakes occurring during pipeline processes (file reading, curve fitting, etc.) were marked and output to the PET analyst and, if the error was critical, the process was halted until the causative issue was resolved, when possible, and the pipeline re-initiated.
Magnetic Resonance Imaging
All study participants underwent MRI on a 3 T MR-750 scanner (General Electric Medical Systems, Milwaukee, WI) equipped with an 8-channel head coil as reported previously. 14
Region of Interest Analysis
On proton density-weighted images (fast spin echo imaging, echo time/repetition time/echo train length = MinFull/6 s/8, receiver BW ± 15.63 kHz, FOV = 22 cm, 256 × 256 sampling matrix, slice thickness = 2 mm, and a parallel imaging acceleration factor of 2) delineation of each region of interest (ROI) was executed and from these, extraction of time-activity curves was performed applying an in-house imaging pipeline. 21 Brain areas pre-published by our group and which are known to be high in FAAH 14 were selected for analyses: dorsolateral prefrontal cortex (DLPFC), mPFC (medial prefrontal cortex), temporal cortex (TC), anterior cingulate cortex (ACC), amygdala (AMY), hippocampus, associative striatum (AST), limbic striatum (LST), sensorimotor striatum (SMST) and cerebellum.
FAAH rs324420 Genotyping
FAAH genotype has a significant impact on [11C]CURB binding. 24 Therefore, the status of this polymorphism was determined for every subject as described in detailed previously. 24 Participants were excluded if genotype was not available (n = 1). From the three possible genetic constellations AA/AC/CC only three subjects fell into the AA group (two in the healthy cohort, and one patient with psychosis). Subjects carrying two A alleles are known to have significantly lower brain FAAH levels. 24 Thus, FAAH rs324420 genotype was entered as covariate in all statistical analyses.
Statistical Analyses
For statistical analysis and for plot generation R software (version 4.2.2 25 ) and the packages ggplot2 and nlme were used. To evaluate differences in demographics between groups, t-tests and chi-square tests were applied. To test for differences in stressful life events (Recent Life Events, Survey of Recent Life Experiences and Hassles) between groups, mixed linear models were used with score as dependent variable while test was entered as repeated measure fixed factor. Sex and age were entered as covariates. Post-hoc Tukey HSD tests for each stress scale were performed to evaluate group differences. Values of p < 0.05 were considered as significant.
Next, we aimed to investigate if there are interactions of group*SRLE, group*RLE or group*hassles on [11C]CURB λk3 values and thereafter to calculate the same model for the combined sample to see if there is an effect of stress (RLE, SRLE, hassles) on [11C]CURB λk3 values across groups. In the model we defined [11C]CURB λk3 as the dependent variable, group, ROI, FAAH rs324420 genotype—due its effects on the outcome measures—and score as fixed factors with subjects specified as random intercepts. A diagonal variance-covariance structure was specified. Common sources of confounds such as sex, age, antipsychotic medication, and previous cannabis exposure (lifetime in g) were also tested. For the exploratory hypotheses we calculated mixed linear models for each group and each stress scale separately to analyse the effect of stress on [11C]CURB λk3 values.
Results
Demographics and Clinical Characteristics
A total of 65 [11C]CURB PET subjects were analysed, of which 35 were healthy participants and 30 with a psychotic disorder. This dataset partially overlaps with a previously published dataset (n = 51 14 ). Demographics and clinical characteristics of the participants are presented in Table 1. Regarding the psychosis spectrum diagnosis, 22 patients had diagnosis of schizophrenia, five had diagnosis of schizophreniform disorder, the three remaining subjects were diagnosed with delusional disorder, psychosis not otherwise specified and schizoaffective disorder respectively. Furthermore, the psychosis cohort had significantly more individuals with previous cannabis experience and higher tobacco use. All included subjects tested negative for cannabis on the urine drug screen. As expected, patients with psychosis were exposed to significantly more life stressors compared to healthy controls testing across all three scales (group×test: F2,121.73 = 3.99, p = 0.02). The result did not change when age and sex were included. Post-hoc Tukey HSD tests revealed significant differences in Survey of Life Experiences (t = 4.882, p < 0.0001) and hassles (t = 3.14, p = 0.002) without a significant group difference for Recent Life Events (t = 1.54, p = 0.13).
Demographics and Characteristics of the Participants.
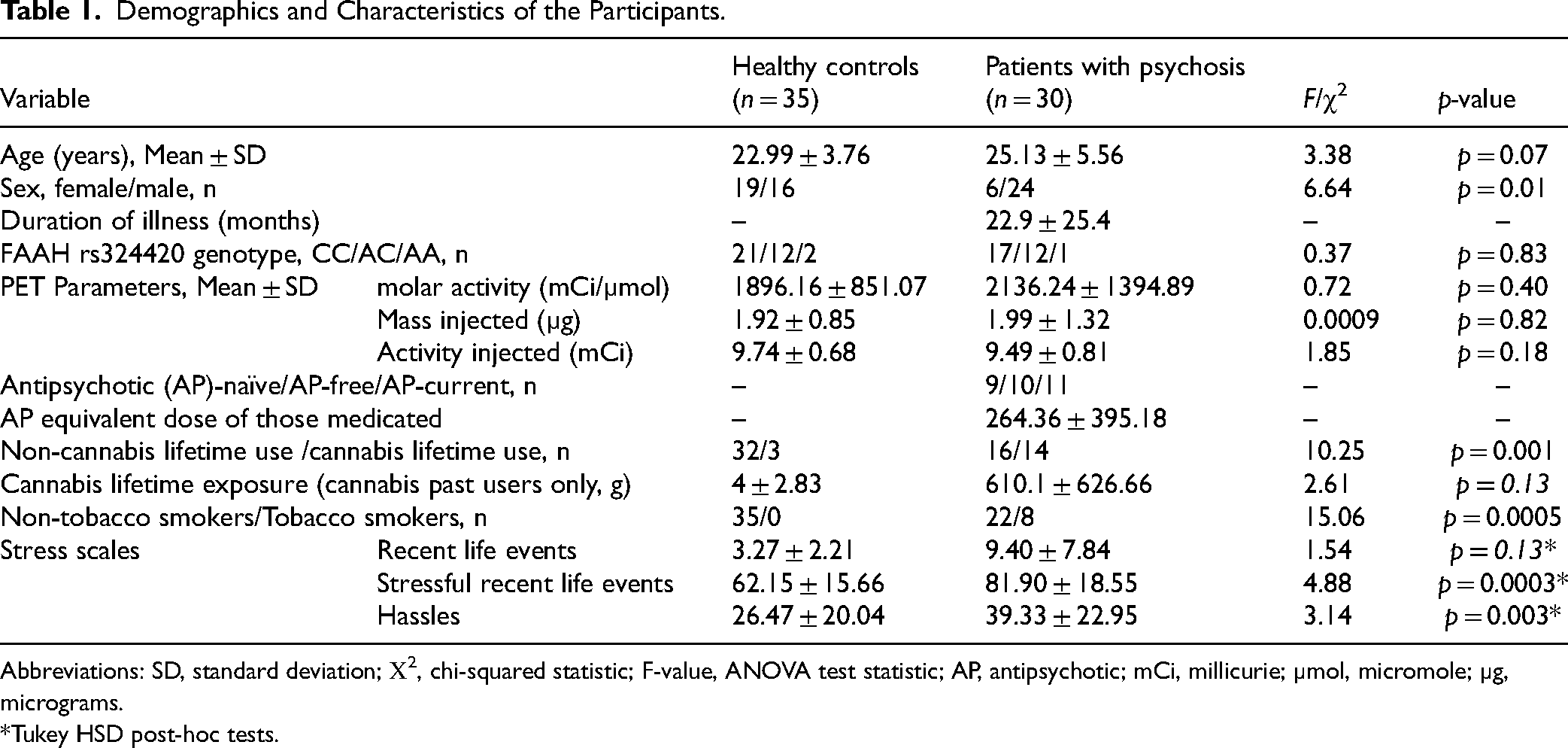
Abbreviations: SD, standard deviation; Χ2, chi-squared statistic; F-value, ANOVA test statistic; AP, antipsychotic; mCi, millicurie; µmol, micromole; µg, micrograms.
*Tukey HSD post-hoc tests.
FAAH Activity was Associated With Recent Life Events in Patients With Psychosis
In previous studies, our research group demonstrated that [11C]CURB λk3 is a sensitive index of FAAH activity. 22 Therefore, a higher [11C]CURB λk3 value indicates higher FAAH activity. This allows for a more comprehensive understanding of the enzyme's role in the endocannabinoid system, particularly in the context of psychosis. Here, we found no association between FAAH activity and Recent Life Events score (F1,59 = 2.8, p = 0.1) controlling for ROI (F9,558 = 159.63, p < 0.0001) and C385A genotype (F2,59 = 34.38, p < 0.0001) across the whole sample. The association between FAAH activity and Recent Life Events score was also not different by group (group × Recent Life Events F1,57 = 0.01, p = 0.92) controlling for group (F1,57 = 0.50, p = 0.48), ROI (F9,558 = 159.63, p < 0.0001) and C385A genotype (F2,57 = 31.61, p < 0.0001; see Figure 1). Of note, when the model was repeated for the patients with psychosis only, there was a significant association between FAAH activity and Recent Life Events score (F1,26 = 7.98, p = 0.009) controlling for ROI (F9,261 = 79.09, p < 0.0001) and C385A genotype (F2,26 = 6.67, p = 0.005). The results did not change when controlling for age, sex antipsychotic medication, cigarettes per day or life time cannabis consumption.
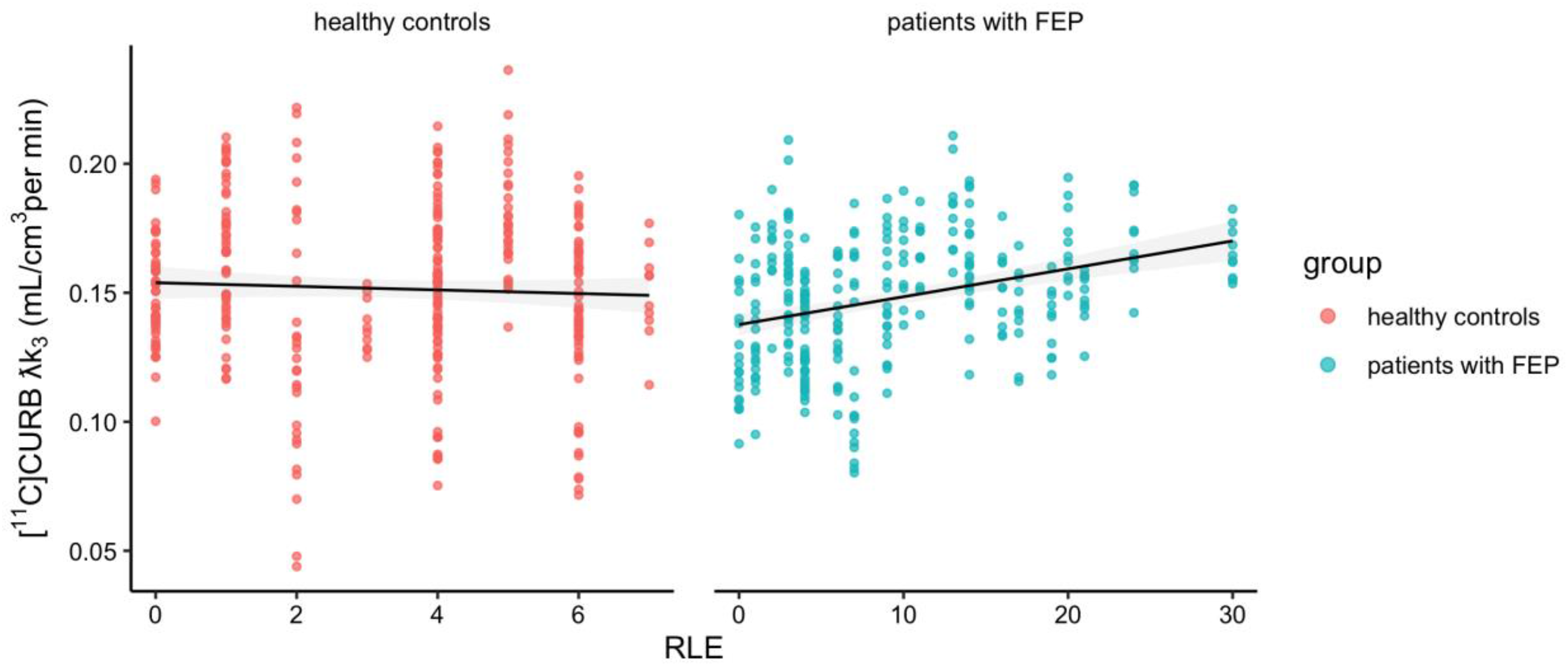
Association between brain [11C]CURB λk3 and recent life events.
The panels display scatterplots of individual data and the correlation between Recent Life Events scores brain FAAH activity as measured by [11C]CURB λk3 in both groups with all ROIs combined (i.e., as there was no group×score×ROI effect).
FAAH Activity was not Associated With Survey of Recent Life Experiences
There was no association between FAAH activity and Survey of Recent Life Experiences score across the whole sample (F1,59 = 0.002, p = 0.97), controlling for ROI (F9,558 = 153.52, p < 0.001) and C385A genotype (F2,59 = 42.11, p < 0.0001) and it was not different by group (group×Survey of Recent Life Experiences F1,57 = 0.09, p = 0.76) with no group effect (F1,57 = 0.06, p = 0.81), ROI (F9,558 = 153.51, p < 0.001) and C385A genotype (F2,57 = 40.54, p < 0.0001). The result did not change when controlling for age, sex, cigarettes per day, life time cannabis consumption or antipsychotic medication. When performing mixed linear models for each group separately, there was no significant association between FAAH activity and Survey of Recent Life Experiences score in neither of the groups.
FAAH Activity was not Associated With Hassles
There was no association between FAAH activity and hassle scores across the whole sample (F1,58 = 0.54, p = 0.47), controlling for ROI (F9,549 = 152.16, p < 0.001) and C385A genotype (F2,58 = 38.14, p < 0.0001) and it was not different by group (group × hassles F1,56 = 0.0005, p = 0.98) with no group effect (F1,56 = 0.027, p = 0.87), ROI (F9,549 = 152.16, p < 0.001) and C385A genotype (F2,56 = 36.01, p < 0.0001). The result did not change when controlling for age, sex, antipsychotic medication, cigarettes per day or life time cannabis consumption. Furthermore, separate analyses for each group did not yield significant associations between FAAH activity and hassles scores.
In sum, when mixed linear models were calculated for the patients with psychosis only, there was a significant association between FAAH activity and Recent Life Events score.
Discussion
To our knowledge this is the first account on the relationship between life stressors and brain FAAH activity as measured by [11C]CURB PET in patients with psychosis and healthy controls. While we measured significantly higher numbers of stressful life events and hassles in patients, there was no significant relationship with brain FAAH activity across groups. Calculating separate mixed linear models for exploratory purposes however revealed a significant positive relationship between brain FAAH activity and Recent Life Events scores exclusively in the group of patients with psychosis. We expected a higher number of life stressors to be associated with higher FAAH activity due to the fact that lowering the amount of circulating endocannabinoids enables an adequate stress response. 6 We were able to confirm our hypothesis at least with one of the used scales used in the patient group. It is worth speculating that the endocannabinoid-related stress homeostasis may still be intact in individuals experiencing their first episode of psychosis, which might explain the absence of FAAH differences in our previous work. 14
Regarding the lack of association between FAAH activity and other parameters, it is possible that FAAH activity is more sensitive to the occurrence of recent life events rather than the cumulative level of perceived stress. The complexity of the stress response may also mean that different aspects of stress (e.g., chronic vs. acute, perceived vs. actual) interact with FAAH activity in varying ways. The Recent Life Events questionnaire and the Survey of Recent Life Events both aim to quantify the impact of life stressors, but they do so in different ways. The Recent Life Events questionnaire primarily focuses on the occurrence and frequency of events, potentially capturing a broader spectrum of stressors that may trigger a biological response, such as changes in FAAH activity. In contrast, the Survey of Recent Life Events emphasizes the perceived severity and impact of these events on the individual. This distinction may explain why FAAH activity was more closely associated with recent life events, as the frequency of these events could be a more immediate trigger for changes in the endocannabinoid system, independent of the subjective stress levels reported by participants.
It is well established that stress can impact endocannabinoid tone and is considered a risk factor for psychosis. While alterations of peripheral endocannabinoids have robustly been found across studies, 13 alterations of brain FAAH activity may not necessarily be at the core of the neurobiology of psychosis. This is supported by previous work from our group showing no significant difference in brain FAAH activity between patients with psychosis and healthy controls in any of the regions of interest, even though a negative relationship with positive symptoms was observed, 14 which is interesting considering the positive relationship with recent stressors we observed in this analysis. Thus, it is essential to explore alternative explanations for the potential links between stress and psychosis, considering various mechanisms. A cautious conclusion drawn from our own findings and pre-existing literature could be that life stressors may increase risk for psychotic symptoms by altering dopaminergic signal transmission, as has been suggested in previous experiments,26–28 as well as by impacting endocannabinoid signaling, although with less impact at these earlier stages.
The question remains if interactions between CB1 receptors, assumed to be reduced in the brain of individuals with psychosis,29–31 and stressful life events might be relevant for psychosis as well. On this note it is assumed that chronic stress reduces CB1 receptor signaling.6, 32 However, to our knowledge there is no study investigating the interaction between CB1 receptors and stress in first-episode psychosis.
Inhibition of FAAH in order to increase endocannabinoid levels is currently being trailed as therapeutic strategy for stress-related psychiatric disorders. 33 The beneficial effects of FAAH inhibition on fear extinction, as well as stress- and affect-related behaviours, have been shown to be favourable in this regard. 34 Especially for post-traumatic stress disorder or other anxiety disorders, FAAH modulation is deemed to be a relevant treatment target.12, 35 It is important to note that next to FAAH, MAGL also significantly contributes to the metabolism of endocannabinoids. Notably, stress impacts the brain's endocannabinoid system by suppressing MAGL, which in turn boosts synaptic modulation within the hippocampus, suggesting a protective role against stress. 36 Furthermore, MAGL inhibitors show promise in reducing anxiety in stressed rodents, pointing to potential therapeutic uses. 37
We acknowledge that our healthy control group had a notably low occurrence of stressful life events, posing a challenge in investigating the correlation between FAAH activity and stress, as well as interpreting the results. Furthermore, a casual direction cannot be identified as the activity of the endocannabinoid system can lead to a different perception of stress especially in the context of a psychotic disorder. Furthermore, in future studies additional objective measures of stress such as cortisol values might be obtained and clinical high risk individuals should be included in future studies to investigate stress and FAAH regulation, as a risk factor for conversion to psychosis.
Conclusion
Stressful life events are a risk factor for psychosis, and are associated with psychopathology in patients. 38 Our data supports an association between life stressors and brain FAAH activity in patients with psychosis. Therefore, modulating FAAH activity may be relevant for treatments of stress-related disorders including in the treatment of stress alterations in psychosis.
Footnotes
Acknowledgments
We would like to thank Marcos Sanchez for statistical support and Rachel Tyndale for genotyping the initial cohorts (Watts 2020). We would also like to thank the participants and their families for their cooperation.
Data Access
Data can be made accessible upon reasonable demand.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RM was part of a SAB for BI in 2021. Without any implication to this work, AW has received financial compensation from Boehringer Ingelheim and Janssen for delivering lectures. All other authors report no biomedical financial interests or potential conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by a NARSAD Independent Investigator’s Grant (Grant No. 21977 [to RM]) and grants from the National Institute of Mental Health (Grant Nos. R21MH103717 and R01MH113564 [to RM]).
