Abstract
Background
During military deployment, stress regulation is vital to protect against the development of anxiety and trauma-related symptoms. Brain endocannabinoids play an important role in stress regulation and previous research has shown that genetic variations in the FAAH rs324420 polymorphism demonstrate protective effects during stress. In addition, this polymorphism shows interactions with the CRHR1 and CNR1 polymorphisms on anxiety. The present study examines whether genetic variations of the FAAH, CRHR1 and CNR1 polymorphisms interact with the development of anxiety and trauma related symptoms in military veterans.
Methods
Veterans (N = 949) who went on military deployment and experienced a stressful event were genotyped for FAAH rs324420, CRHR1 rs110402 and CNR1 rs2180619. Anxiety and trauma symptoms were measured pre-deployment and 6 months after deployment. Anxiety was measured with the anxiety subscale of the Symptom Checklist-90 (SCL-90) and trauma with the Self-Rating Inventory for PTSD (SRIP).
Results
Covariance Pattern Models demonstrated no significant relation of genetic variations in FAAH rs324420 on anxiety and PTSD symptoms from pre-deployment to 6 months after military deployment. Additionally, we investigated interactions between the FAAH s324420, CRHR1 rs110402 and CNR1 rs2180619 polymorphisms. This also demonstrated no significant effects on anxiety and PTSD symptoms pre- to post deployment. However, the covariate of childhood trauma that was included in the models was significant in all these models.
Conclusion
Genetic variations in FAAH rs324420 and its interactions with CRHR1 rs110402 and CNR1 rs2180619 are not related to the development of anxiety and trauma-related symptoms. The study however, indicates the importance of considering childhood trauma in the investigation of the effects of polymorphisms that are related to the endocannabinoid system on the development of anxiety and PTSD symptoms.
Introduction
Post-traumatic stress disorder (PTSD) is a debilitating disorder that can develop after being exposed to or by witnessing a traumatic event. 1 Certain professions have a higher risk for the development of PTSD, such as soldiers, firefighters, and first responders.2,3 Individuals with these occupations often deal with stressful events in which adaptive stress regulation and emotional processing are vital to protect against the development of trauma and anxiety-related symptoms. For example, prevalence rates demonstrated that approximately 9% of soldiers develop PTSD symptomatology six months after returning from military deployment.4,5 Converging evidence from animal and human studies suggest that brain endocannabinoids play an important role in regulating stress and emotional processing, besides their already well-established role in the extinction of aversive memories.6–8
The yet known elements of the endocannabinoid system (ECS) are the cannabinoid receptor 1 (CB1) and cannabinoid receptor 2 (CB2), the endogenous cannabinoids anandamide (AEA) and 2-arachidonoylglycerol (2-AG), and the catabolic enzymes for the degradation of these cannabinoids, fatty acid amide hydrolase (FAAH) for AEA and monoacylglycerol lipase (MAGL) for 2-AG. 9 The well-established role of the ECS in the extinction of fear is repeatedly demonstrated in animal and human studies. Studies showed that blocking or genetically deleting the CB1 receptor resulted in a failure to extinguish fear.6,10,11 On the other hand, augmenting endocannabinoid signaling by CB1 agonists or the pharmacological blockade of the enzyme FAAH enhanced fear extinction reconsolidation.6,8,12 However, the ECS also plays an important role in the regulation of stress. Inhibition of the FAAH enzymes prevents the reduction in AEA that is normally accompanied by stress and anxiety. 13 Therefore, FAAH inhibitors are a promising new agent in the treatment of anxiety and stress-related symptoms in PTSD.14,15 One study demonstrated that the use of a FAAH inhibitor (PF-04457848) was able to improve the recall of fear extinction memories in healthy volunteers. 15 Additionally, in women with PTSD, genetic variation in the FAAH rs324420 gene influences fear learning. 16 However, a recent phase II trial aimed at reducing the severity of PTSD symptoms with a FAAH inhibitor failed to demonstrate a reduction in symptoms. 17 Additionally, research suggests that FAAH inhibition is not anxiolytic per se but protects against the anxiogenic effect of stress during high environmental aversiveness.13,18–20
In humans, several studies have focused on a single nucleotide polymorphism in the FAAH gene because of its potential protective effects on stress and anxiety during high environmental aversiveness, as demonstrated in animal studies.13,18–20 The polymorphism FAAH rs324420, A-allele, has a frequency of approximately 25% in populations of Caucasian ancestry and is associated with reduced FAAH activity and elevated levels of AEA.21–23 Several studies demonstrated that A-allele carriers were associated with decreased anxiety, enhanced fear extinction learning and extinction recall, decreased threat-related amygdala reactivity, increased fronto-amygdala connectivity, and protected against stress-induced decreases in AEA and negative emotional consequences of stress.16,24–29 Interestingly, these studies did not assess or correct for childhood trauma in their analysis, despite evidence showing that it can cause alterations in the ECS.30–32 Taken together, these studies support the role of decreased FAAH activity and increased AEA levels in buffering stress responses besides its enhancement of fear extinction.
In dealing with stress and anxiety corticotropin-releasing hormone receptor 1 (CRHR1) also plays a significant role. Therefore, recent studies do not focus solely on the FAAH rs324420 polymorphism, but also on its interaction(s) with different CRHR1 polymorphisms.33,34 The ECS plays an important role in the activation and regulation of Hypothalamic–Pituitary–Adrenal (HPA) responses to stress. Namely, stress-related reduction in AEA is driven by activation of FAAH within the basolateral amygdala (BLA).7,35 This in turn activates the HPA axis. The association between FAAH rs324420 genotypes and amygdala habituation, which is thought to be associated with anxiety, was shown to depend on CRHR1 genotypes. 33 Blunted amygdala habituation was not directly affected by FAAH rs324420 AA/AC genotypes, but was observed in A-carriers that also had the CRHR1 rs110402 AA genotype. 33 Moreover, blunted left amygdala habituation mediated between these genotypes and increased risk for anxiety disorders. 33 Another study investigated the interactions on self-reported anxiety of the FAAH rs324420 genotypes with minor alleles of several other CRHR1 polymorphisms (ie rs110402 AA; rs242924 TT; rs7209436 TT). 34 This study concluded that FAAH rs324420 AA/AC genotypes and CRHR1 minor alleles were related to lower scores on the Beck Anxiety Inventory (BAI). Participants with the FAAH rs324420 CC genotype only reported lower anxiety when they also possessed a combination on three CRHR1 SNPs (rs110402 AA; rs242924 TT; rs7209436 TT).
Finally, variations in the CB1 cannabinoid receptor gene (CNR1) have been associated with anxiety and are of interest when investigating the effects of the FAAH rs324420 polymorphism, considering that FAAH impacts on the neurotransmitter AEA that binds to CB1 receptor. 36 A study on the role of the CNR1 rs2180619 polymorphism in fear learning demonstrated that G-carriers were associated with better fear extinction learning and less anxiety compared to A-homozygotes. 37 This seems to contradict the higher trait anxiety in G-carriers that also carried the s-allele of the serotonin transporter gene. 38 Although these findings are contradictory, further investigation into CNR1 genotypes and especially how they interaction with genetic variability in FAAH is needed to establish the role of the in anxiety more precisely. 39 It is especially important to investigate this interaction because the CB1 receptors play a significant role in anxiety behavior and are mediated by the FAAH enzymes. 40
So far, studies have mainly focused on populations that already have developed anxiety and trauma-related symptoms or in healthy individuals on stress and anxiety in an experimental setting.16,24–29 However, it remains unknown whether potential genetic variations of the FAAH, CRHR1 and CNR1 polymorphisms are protective in the development of anxiety and trauma-related symptoms in real life events. Therefore, the primary aim of the current study is to examine the relationship between genetic variations in the FAAH rs324420 polymorphism (ie AA and AC-allele carriers) and the development of anxiety and trauma-related symptoms after the experience of stressful events in military veterans who have been deployed in Afghanistan. Our secondary aim is to investigate whether the relation of the FAAH polymorphism on the development of anxiety and trauma-related symptoms interact with the CRHR1 rs110402 and CNR1 rs110402 polymorphisms after returning from military deployment.
Methods
Participants
For this study we analyzed data that was collected for the Prospective Research in Stress-Related Military Operations (PRISMO) study.41,42 PRISMO is a large prospective cohort study on the long-term effects of military deployment on mental health and the contribution of biological and psychological factors in the development of these mental health symptoms. Participants were Dutch military personnel who were deployed to Afghanistan between 2005–2008. We selected this subsample because we were interested in the relation of the differences in genotypes on anxiety and trauma symptoms during high environmental aversiveness. To select participants from the dataset we selected participants from the collected data who went on military deployment and had a minimum score of 1 (which means they experienced at least one stressful/traumatic combat related stressor) on the Deployment Experience Scale (DES). 5 The DES consist of 19 questions regarding events experienced during military deployment. Examples of combat related stressors were for example: witnessed people suffering, enemy fire, witnessed wounded, and a colleague injured or killed. The DES was assessed 1 month after military deployment. The study was approved by the Institutional Review Board of the University Medical Center Utrecht (15-705), The Netherlands. All participants provided verbal and written informed consent before participation in the study.
Questionnaires
The Symptom Checklist-90 (SCL-90) is a self-report questionnaire to measure a broad range of psychiatric symptoms.43,44 It measures different symptom dimensions but for this research question we were primarily interested in the anxiety subscale as an anxiety-outcome measure. The Dutch Self-Rating Inventory for PTSD (SRIP) was assessed to measure PTSD symptoms.45,46 The Early Trauma Inventory Short Form (ETI-SF) was used to assess traumatic experiences during childhood. The ETI-SF was used as a covariate in the analyses because of the known effects of childhood trauma on alternations in the ECS.30–32 Questionnaires were assessed 1 month pre-deployment and 6 months after military deployment. Except for the ETI-SF, which was only assessed pre-deployment.
Genotyping
Blood samples were obtained via venipuncture and standard protocol was used for DNA extraction. The concentration and quality of the DNA were examined using Nanodrop (Thermo Fisher Scientific, MA, USA). Genotyping was conducted using Illumina Human OmniExpress 24 v1.1. The genetic variations of the FAAH rs324420, CRHR1 rs110402, and CNR1 rs2180619 polymorphisms were extracted using PLINK software version 1.9. 47
Statistical Analysis
Before starting the analysis missing data from the SCL-90, SRIP, ERI-SF and DES were handled by Multivariate Imputation by Chained Equations (MICE). 48 The missing values in the data were assumed to be missing at random and all the variables and covariate used in the analyses were included in the imputation model. Details about the missing value analyses and multiple imputation procedure are shown in the Supplemental Data (1. Multiple Imputations).
Covariance Pattern Models were then used for the remaining analysis. Covariance Pattern Models are a form of linear mixed models that specifies a unique pattern of change over time in correlation among repeated measurements on the outcome measure. 49 We first analyzed the effect of FAAH rs324420 genotypes, CC, AC and AA, on development of anxiety and PTSD symptoms (separate analyses per outcome measure) from pre-deployment to 6 months after military deployment. In these analyses FAAH rs324420 (CC/AC/AA) was added as fixed effect and Time (anxiety or PTSD scores pre-deployment and 6 months after deployment) the dependent variable. The total score on the ETI-SF (childhood trauma) was used as a covariate in the analysis.
We then added the CRHR1 rs110402 (GG/GA/AA) and CNR1 rs2180619 (AA/GA/GG) genes to test interactions with FAAH rs324420 (CC/AC/AA). The Covariance Pattern Model now included the three polymorphisms as fixed effects and tested their (interaction) effects on the development of anxiety symptoms, also with childhood trauma as a covariate. Subsequently, this was repeated for the PTSD symptoms as an outcome measure. In case of any significant interaction effects or main effects we adjusted the model so that it would include only the significant interactions or main effect and then test this model again to see whether the effect remained. The Satterthwaite approximation was used in all models. This is recommended for small sample sizes or when the model has a complicated covariance type (ie unstructured as was used in the model). 50 This correction can result in atypical denominator degrees of freedom compared to traditional repeated measures models (eg, denominator degrees of freedom that may actually be higher than the number of subjects). 51 Analyses and the imputation of missing data were carried out with IBM SPSS Statistics (version 27). Pooled fixed effects from the Covariance Pattern Model analysis were calculated using the miceadds package in R (Version 4.3.1). 52 A p-value of p < 0.05 was considered statistically significant.
Results
Sample Characteristics
From the PRISMO sample 949 participants, reported experiencing at least one stressful event. In this subsample mean age was 28.61 years (SD = 8.99) and male/female ratio was 866/83 (91.3/8.7%). Other characteristics of the sample, with regard to military deployment are displayed in Table 1. All three polymorphisms (FAAH rs324420, CRHR1 rs110402 and CNR1 rs2180619) were in Hardy–Weinberg equilibrium, see Table 2.
Sample demographic characteristics.

Sample sizes might not add up to total participants due to missing data.
*Education (International Standard Classification of Education levels): Low = primary and lower secondary education; Moderate = upper secondary, postsecondary non-tertiary and short cycle tertiary education; High = bachelor, master and doctoral education.
Genotype frequencies and Hardy–Weinberg equilibrium.

Sample sizes might not add up to total participants due to missing data.
FAAH rs324420 Genotypes and Anxiety and PTSD Symptoms
The Covariance Pattern Model test of fixed effects on development of anxiety from pre-deployment to 6 months after military deployment for the different FAAH rs324420 (CC/AC/AA) genotypes was not significant, F(2, 63) = 2.635, p = .080. Also with regard to the PTSD symptoms the Covariance Pattern Model test of fixed effects demonstrated no significant effects, F(2, 405) = 1.048, p = .352, see also Table 3 for the pooled estimated marginal mean and standard errors. In both of the tested models the covariate of childhood trauma was significant, both models p < .001.
Pooled estimated marginal mean and standard error for the different FAAH rs324420 genotypes per outcome (anxiety and PTSD).

FAAH rs324420, CRHR1 rs110402 and CNR1 rs2180619 Interactions
The Covariance Pattern Model was then conducted first to examine the interaction effect of FAAH rs324420 (CC/AC/AA), CRHR1 rs110402 (GG/GA/AA) and CNR1 rs2180619 (AA/GA/GG) genotypes on PTSD symptoms and secondly to examine the effects on anxiety symptoms six months after military deployment. The tests of fixed effects demonstrated no statistically significant three-way interaction between FAAH rs324420 (CC/AC/AA), CRHR1 rs110402 (GG/GA/AA) and CNR1 rs2180619 (AA/GA/GG) genotypes on PTSD symptoms six months after military deployment, F(7, 17278) = 1.154, p = .326. Also, the other interaction term and main effects were not significant, see also Supplemental Data (2. Tests of Fixed effects, Table 3). For the Covariance Pattern Model with anxiety as an outcome measure the interaction between the three genes was also not significant, F(7, 34) = 1.455, p = .216. However, this model demonstrated two significant main effects of FAAH (F(2, 107) = 5.275, p = .007) and CRHR1 (F(2, 716) = 4.213, p = .016). This was followed by an adjustment of the model so that it only included the main effects. However, when testing the effects in isolation they did not remained significant (CRHR1 F(2, 371) = .002, p = 1; FAAH (F(2, 63) = 2.635, p = .080). Again in all of our models the covariate childhood trauma reached a significance of p < .001.
Explorative Analysis on Childhood Trauma
In all our tested models childhood trauma was significant as a covariate. For exploratory purpose we tested the same Covariance Pattern Model as in our primary analysis. Childhood trauma was divided into three groups: no childhood trauma (score of 0 on the ETI-SF), low childhood trauma (ETI-SF score 1-3) and high childhood trauma (ETI-SF score 4 or higher), based on the median split and previous exploratory analysis on childhood trauma and FAAH rs324420 genotypes that used the same approach. 31
Because the AA genotype group (n = 30) would become too small when divided into no, low, and high childhood trauma categories, we decided to combine the AA group with the AC group. Since AA carriers often form a small group, they are frequently grouped together in such analyses.16,24–29 In the explorative analysis FAAH rs324420 (CC and AC/AA) and childhood trauma (no childhood trauma, low and high childhood trauma) were added as fixed effect and Time (anxiety or PTSD scores pre-deployment and 6 months after deployment) the dependent variable.
Both models demonstrated a significant interaction effect of FAAH rs324420 and childhood trauma on anxiety (F(5, 17) = 3.555, p = 0.02) and PTSD symptoms (F(5, 30) = 8.899, p=<.001). Table 4 and 5 shows the estimated fixed effects and standard error for the different groups in comparison to the reference group (high childhood trauma and AA/AC genotype). Both models demonstrated significant differences between the reference group and both genotypes with low childhood trauma and the CC genotype with no childhood trauma. These groups reported less anxiety and PTSD symptoms 6 months after military deployment.
Pooled estimated fixed effects and standard error for the differences in anxiety scores in relation to the reference group per genotype and childhood trauma category.

*significant with a p = < .05.
Pooled estimated fixed effects and standard error for the differences in PTSD scores in relation to the reference group per genotype and childhood trauma category.
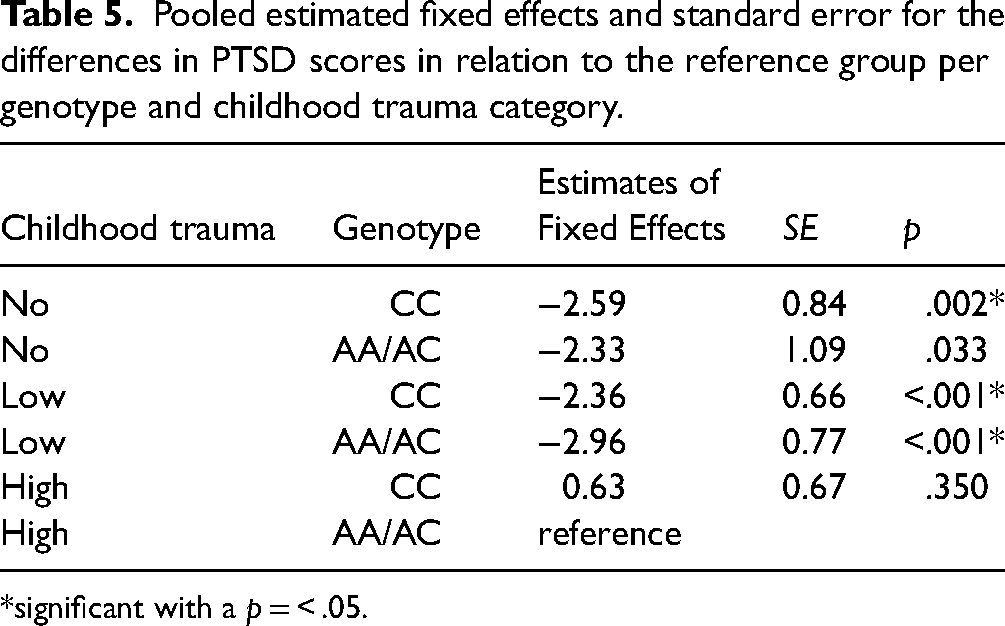
*significant with a p = < .05.
Discussion
Our study indicated that genetic variations in the FAAH rs324420 polymorphism are not related to the development of anxiety- and trauma related symptoms after the experiences of a stressful event in military veterans who were deployed in Afghanistan. Anxiety and PTSD symptoms did not differ pre- to 6 months post-deployment between the different FAAH rs324420 genotypes. Additionally, we investigated interactions between the FAAH rs324420, CRHR1 rs110402, and CNR1 rs2180619 polymorphisms. Again, interactions between FAAH rs324420, CRHR1 rs110402, and CNR1 rs2180619 on anxiety and PTSD symptoms six months after military deployment did not reach statistical significance. Interestingly, the covariate of childhood trauma was significant in all the models that we tested.
Contrary to our expectations based on prior studies, in our sample genetic variation in the FAAH rs324420 polymorphism did not relate to differences in anxiety and PTSD symptoms from pre- to post-deployment. Prior studies demonstrated differences in anxiety between A-allele carriers (individuals with AA or AC genotypes) in comparison with individuals with the CC genotype. These studies demonstrated that A-allele carriers of this polymorphism reported lower levels of anxiety, showed enhanced fear extinction and recall and decreased threat-related amygdala activity and this group seemed to be protected against negative consequences of stress.16,24–29,53 Contrary to previous studies that investigated genetic variations of the FAAH rs324420 polymorphism in experimental designs, our prospective study was the first to investigate stress and anxiety related to real life events. From each participant anxiety and trauma symptoms had been assessed before and after a stressful event (military deployment) and analyzed in relation to variations in the FAAH gene. It must be noticed, however, associations with the A-allele appeared to be more robust when assessed with experimental fear and stress tasks than on any of the subjective measures of anxiety and stress that have been used in the aforementioned studies. Furthermore, participants in our sample have been faced with high environmental stress due to being deployed, and witnessed people suffering and wounded, were target of enemy fire, or witnessed a colleague who was injured or killed. In experimental studies so far, not all have demonstrated effects of the FAAH polymorphism in the same direction. One study 53 used aversive pictures, which induces a strong emotional context relative to the emotional faces task used in the studies by Hariri and colleagues,25,33 and reported stronger fear responses in the A-carriers as indexed by startle potentiation. This was explained as potentially resulting from bidirectional effects of the ECS as a function of the intensity of the emotions experienced in a context. 53 Yet, the emotional intensity of watching negative pictures on a screen pales compared to the emotional context that the participants in this experiment experienced. On the other hand, the outcome measure was also not just intensity of emotion experienced at the time of the emotional event, but rather a farther removed consequence, ie, trauma and anxiety reported 6 months later. In short, the studies so far differ widely in study design and outcome parameters, and conclusions based on their comparison are at most preliminary.
As our secondary aim we investigated the interactions between the genetic variations in the FAAH rs324420, CRHR1 rs110402, and CNR1 rs2180619 polymorphisms on anxiety and trauma symptoms after military deployment. Again, interactions between FAAH rs324420, CRHR1 rs110402, and CNR1 rs2180619 on both anxiety and PTSD symptoms six months after military deployment did not reach statistical significance. However, in all these models childhood trauma as a covariate was significant. Interestingly, none of the aforementioned studies included childhood trauma as a covariate.16,24–29,53 Although it must be interpreted with caution because of its preliminary nature our exploratory analysis demonstrated interaction effects between FAAH rs324420 genotypes and childhood trauma on de development of anxiety and PTSD symptoms after military deployment. Traumatic experiences during childhood are related to the development of different psychiatric disorders like anxiety depression and schizophrenia later in life. 54 In addition, childhood trauma is also related to disturbances in development of the endocannabinoid and related systems. 32 For example, corticotropin-releasing hormone, a central regulator of the hypothalamic-pituitary-adrenal (HPA) axis, for which the CRHR1 rs110402 gene codes can be permanently disturbed by childhood trauma.55–57 Furthermore, clinical and preclinical studies demonstrated that trauma during childhood is related to an upregulation of endogenous cannabinoid and a down regulation of CB1 receptor availability. 32 One study that investigated the FAAH rs324420 polymorphism demonstrated that the chronically elevated AEA levels in A-carriers may be a risk factor in the case of chronic childhood adversity, having both is associated with higher levels of anxiety and depression. 31 These findings indicated that childhood trauma may interact in important ways with the ECS and needs to be taking into account when investigating the role of genetic variation on the development of anxiety and PTSD.
Our study has a couple of limitations that must be addressed. Firstly, our sample consisted of a predominantly male population (91.4%), although this is representative for the military. Additionally, we only examined veterans with a specific type of trauma exposure, namely related to combat. While this resulted in a homogeneous sample, future research with a larger group of women and different types of stressors is warranted, especially since it is known that females show differences in AEA levels and CB1 receptor density which is confirmed in both human and animal research.58–61 Secondly, our group of FAAH rs324420 AA carriers (n = 30) was small and no sample size calculation was conducted to confirm if the sample was large enough to find a significant effect. However, previous studies included a group of AA or AA/AC carriers ranging between 18–36 that resulted in significant results.16,24–29 Future studies would also benefit from using a prospective genotyping strategy to create balanced genotype groups, since the FAAH rs324420 A-allele was found to be associated with gene-dose-dependent increase in basal peripheral AEA levels. 26 In this way it is possible to investigate the AA and AC genotypes separately instead of putting them together in one group as is common practice in most studies. Thirdly, our study focused on a single polymorphism (FAAH rs324420) in combination with two other polymorphisms (CRHR1 rs110402 and CNR1 rs2180619). Future studies would also benefit from additionally investigating haplotypes, a set of DNA variants along a single chromosome that tend to be inherited together. 36 For example, the CB1 receptor site polymorphisms rs806379, rs1535255, and rs2023239 may be important as well. 62 Furthermore, more work is needed to investigate other signaling systems known to interact with the ECS, such as interactions between the 5-HTTLPR serotonin transporter gene and the CNR1 rs2180619 gene. 38 Fourthly, we did not determine AEA concentrations in our study, so we can only assume that AEA concentrations were higher in A-carriers. However prior preclinical and clinical research have consistently demonstrated higher AEA in A-carriers.15,21–23,26 Lastly, the analysis included anxiety and PTSD-related symptoms, and because these are self-report measures, there was no information about a possible diagnosis of an anxiety disorder or PTSD conducted by a psychologist. Additionally, no information was available about possible psychological or pharmacological interventions received between military deployment and the 6-month follow-up measure. Therefore, it is unclear what influence these interventions may have had on the anxiety and/or PTSD symptoms during this period. In addition it would be of interest to study other factors besides childhood trauma that could affect the ECS, such as gender and cannabis use history.63,64
Conclusion
FAAH rs32442, CRHR1 rs110402, and CNR1 rs2180619 were not associated with the development of anxiety- and trauma related symptoms after military deployment in Afghanistan. There was, however, a significant effect of childhood trauma as a covariate in all our models. Future research on the endocannabinoid system could benefit from assessing effects of genetic variations in the FAAH rs324420 polymorphism together with genotypes in the cannabinoid receptors, such as CNR1 rs2180619. This could lead to a better understanding of complexity and heterogeneity in endocannabinoid signaling. Finally, these results underline the importance of considering childhood trauma, and possible other factors that influence the endocannabinoid system, in the investigation of the effects of polymorphisms that are related to the endocannabinoid system on the development of anxiety and PTSD symptoms.
Supplemental Material
sj-docx-1-css-10.1177_24705470241285828 - Supplemental material for The Role of Genetic Variations in the FAAH rs324420 Polymorphism and its Interaction with CRHR1 rs110402 and CNR1 rs2180619 in Anxiety and- Trauma Related Symptoms After Military Deployment
Supplemental material, sj-docx-1-css-10.1177_24705470241285828 for The Role of Genetic Variations in the FAAH rs324420 Polymorphism and its Interaction with CRHR1 rs110402 and CNR1 rs2180619 in Anxiety and- Trauma Related Symptoms After Military Deployment by Nadia Leen, Antoin de Weijer, Marco Boks, Johanna Baas, Eric Vermetten and Elbert Geuze in Chronic Stress
Footnotes
Acknowledgements
We would like to thank Peter Zuithoff for his advice regarding the performance and interpretation of the statistical analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The Editor-in-Chief of Chronic Stress is an author of this paper, therefore, the peer review process was managed by Sage's in-house editorial team, and the submitting Editor was not involved in the decision-making process.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Dutch Ministry of Defence.
Author's Contribution
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
