Abstract
Objectives
The majority of patients with schizophrenia experience dramatic improvement in psychotic symptoms when treated with antipsychotic medication. Maintenance treatment can prevent relapses but problems with medication adherence limit effectiveness. Long-acting injectable antipsychotics (LAIs) provide an opportunity to establish adherence but challenges remain in ensuring that the dose selected is therapeutic. Therapeutic drug monitoring has not been established as valuable for LAIs in the maintenance treatment of schizophrenia. This exploratory study was undertaken to describe plasma paliperidone levels in outpatients treated with the LAI paliperidone palmitate and to determine whether paliperidone levels are associated with subjective experience on medication and side effects.
Methods
Twenty-one outpatients with schizophrenia receiving treatment with LAI paliperidone consented to participation in this study. Blood samples were obtained for measurement of paliperidone and prolactin levels at the first visit. A second paliperidone level was obtained at the time of the next injection for 18 of the participants. Clinical rating scales were administered at the first visit to assess illness severity, attitudes regarding medication, subjective well-being and side effects.
Results
Paliperidone levels were highly correlated at the two time points (ρ = .85;
Conclusions
We demonstrated that paliperidone levels can be measured reliably in patients receiving LAI paliperidone. Higher plasma levels were associated with higher prolactin levels and reduced sexual desire but not with measures of subjective experience on medications or other side effects. Measurement of paliperidone levels in patients treated with paliperidone palmitate may have the potential to minimize the dose of medication prescribed and, in turn, the severity of sexual side effects.
Plain Language Summary Title
Can the Dosing of Long-Acting Injectable Paliperidone for the Treatment of Schizophrenia Be Improved by Measuring Drug Levels?
Plain Language Summary
Why was the study done?
Antipsychotic medications are able to make the hallucinations and delusional thoughts go away for most people who have schizophrenia. Providing the medication in the form of an injection that can be given every few weeks or months can help people with schizophrenia stay well. It can be hard to know what dose of the injectable antipsychotic paliperidone will be best for each person. If the dose is too low, symptoms will return. If the dose is too high, side effects can be difficult to tolerate. This exploratory study was done to see whether measuring the amount of paliperidone in the blood can be used to make sure that the dose is likely to be effective and well tolerated.
What did the researchers do?
People with schizophrenia who were being treated with the long-acting injectable paliperidone had their blood drawn to measure the level of medication just before they received their next injection. Researchers also measured the symptoms and side effects that participants were experiencing to see whether people who had higher levels of paliperidone in their blood had more side effects.
What did the researcher find?
Even at the same dose of medication, some people had very small amounts of medication in their blood and others had much higher amounts. People with higher amounts in their blood experienced less sexual desire.
What do the findings mean?
The amount of paliperidone in the blood is a better measure of how much medication a person is getting than the dose they are given. Measuring paliperidone blood levels may lead to lower doses with fewer sexual side effects. It may make it easier for people with schizophrenia to continue on medication and to stay well.
Introduction
Most people with schizophrenia respond robustly to antipsychotic medication but are at very high risk of relapse if they stop taking them. 1 Long-term maintenance treatment with antipsychotic medication can dramatically reduce the risk of relapse. 2 When relapses do occur with maintenance treatment, poor treatment adherence is the most important predictor. 3 The use of long-acting injectable antipsychotic (LAI) medication provides an opportunity to establish adherence, which may account for superior efficacy in relapse prevention reported in some studies.4,5 Psychotic relapses may still occur in individuals with documented full adherence to both oral antipsychotics and LAIs. Analysis of the Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) study data revealed that higher rates of relapse were observed in those patients whose antipsychotic blood levels were below the calculated reference range compared to those at or above the reference range. 6 Antipsychotic plasma levels vary greatly across individuals treated with standard doses of both oral antipsychotics and LAIs, largely due to variability in hepatic metabolism and renal function. Therapeutic ranges for antipsychotic plasma levels have been described for many oral antipsychotics 7 but have not been established for LAIs. 8 Establishing the lowest effective plasma levels for relapse prevention with LAIs may also help in minimizing side effects that may contribute to problems with adherence. We carried out an exploratory study to describe the plasma paliperidone levels in outpatients receiving LAI paliperidone palmitate, and to investigate whether higher plasma paliperidone plasma levels are associated with lower subjective well-being, more negative attitudes towards medication and greater side effects.
Patients and Methods
All participants were receiving care from the Schizophrenia Outpatient Clinic at St. Joseph's Healthcare Hamilton in Hamilton, Ontario, Canada, a tertiary care medical centre fully affiliated with the Faculty of Health Sciences at McMaster University. This study was approved by the Hamilton Integrated Research Ethics Board and all participants provided written informed consent for their participation. Individuals were invited to participate if they were 18–75 years of age, were receiving maintenance treatment with long-acting paliperidone palmitate for a DSM-5 diagnosis of schizophrenia or schizoaffective disorder, were on the same dose for the previous two injections, were capable of consenting to study participation, and were able to understand and communicate in English. The study involved two visits when participants were scheduled to receive consecutive injections of paliperidone palmitate. For participants treated with the 1-month formulation of paliperidone palmitate, visits were scheduled either 3 or 4 weeks apart based on the individual participant's injection schedule. Plasma paliperidone levels and prolactin levels were drawn at Visit 1 and a second paliperidone level was drawn at Visit 2. Body mass index (BMI) was calculated as weight in kilograms divided by height in metres squared.
All blood samples were collected at the clinical laboratory at St. Joseph's Healthcare Hamilton, in Hamilton, Ontario, Canada. Plasma paliperidone (9-OH risperidone) levels were sent to be processed at the clinical laboratory at the Centre for Addiction and Mental Health in Toronto, Ontario, Canada. This laboratory-developed test was performed using liquid chromatography coupled with high-resolution mass spectrometry technology (Q-Exactive, Thermo Fisher Scientific). Specimens were spiked with deuterated (D4) 9-OH risperidone (Cerilliant), deproteinized and analyzed in SIM/dd-MS2 mode with 5 ppm mass accuracy against a 7-point calibration curve ranging from 5 to 410 nmol/L. Assay calibrators were prepared in blank human plasma from 1 mg/ml standard (Cerilliant). Quality Control material (MassCheck Neuroleptics) was from Chromsystems. Results were reported against a reference range of 19–211 nmol/L. Prolactin levels were measured ci16200 Architect (Abbott Diagnostics, Mississauga, Ontario). Testing was performed by certified laboratory staff at the Hamilton Regional Laboratory Medicine Program using internal routine standard-of-care processes.
At Visit 1, a series of rating scales were also completed. These included: Clinical Global Impression Scale–Severity (CGI-S), 9 Social and Occupational Functioning Scale (SOFAS), 10 Calgary Depression Scale for Schizophrenia (CDSS), 11 Subjective Well-being under Neuroleptics–Short Version (SWN-short version), 12 Drug Attitude Inventory (DAI-30), 13 Barnes Akathisia Rating Scale (BAS), 14 the Changes in Sexual Functioning Questionnaire (CSFQ-14), 15 Subjective Happiness Scale (SHS) 16 and Satisfaction with Life Scale (SWLS). 17
The SWN-short version is a 20-item self-report scale used to quantify the subjective experience of being on antipsychotic medication. Each item is rated on a 6-point Likert scale; a total score is calculated by summing scores for all items to yield scores that range from 20 to 120 points. 18
The DAI-30 is a 30-item self-report scale developed to quantify the attitudes and feelings that individuals with schizophrenia have on antipsychotic medication. Scores range from 30 to −30 with higher positive scores reflecting more favourable experience and negative scores reflecting more negative experience with antipsychotic medication.13,19
The BAS includes ratings for objective signs of restlessness, subjective restlessness, and a global clinical assessment of akathisia. As ratings of akathisia were very low in this sample, we considered akathisia as a binary variable that was present if the global clinical assessment of akathisia was greater than “questionable” in severity (i.e., mild to severe akathisia); 4/21 participants met this threshold.
The CSFQ-14 includes 14 questions in versions specific to male and female participants that are scored and added together to create a total score. Scores on the 14 questions yield five scales reflecting five dimensions of sexual functioning (Pleasure, Desire/Frequency, Desire/Interest, Arousal/Erection, Orgasm/Ejaculation) as well as three scales that correspond to phases of the sexual response cycle (Desire, Arousal, and Orgasm). Total scores on the CSFQ-14 can range from 14 to 70. Scores in the range of 47–70 and 41–70 are considered normal for men and women, respectively, with lower scores indicative of sexual dysfunction.
The SHS measures global subjective happiness using four self-report questions. These four questions are summed to provide a composite score for global happiness. Reliability and construct validity have been published. 16 The SWLS is completed by self-report and includes five statements regarding life satisfaction that are rated from 1 to 7 (“strongly disagree” to “strongly agree). We have used the SHS and SWLS in a previous study to assess happiness and life satisfaction in individuals with schizophrenia. 20
Associations between plasma paliperidone levels, prolactin levels, and clinical measures were assessed using Spearman rank correlations. Group differences were tested with the Mann–Whitney
Results
Twenty-one participants (11 female/10 male) provided informed consent for this study and had plasma paliperidone levels measured at Visit 1 of whom 18 had paliperidone levels repeated at Visit 2. Participants had a mean age of 41 years (SD = 13.9) and a mean duration of illness of 15 years (SD = 12.4). They had been treated with paliperidone palmitate for a mean of 18 months (SD = 11.4; range 2–47 months). Twenty participants were receiving the 1-month formulation of paliperidone palmitate every 4 weeks (
Demographics.
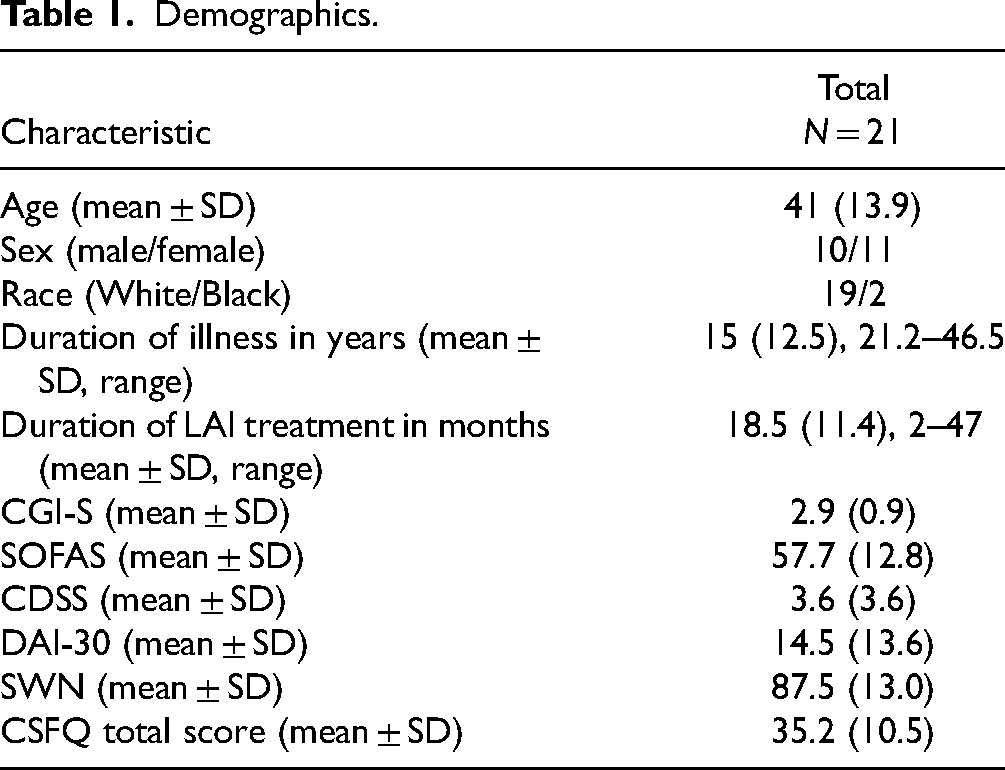
Mean paliperidone level at Visit 1 (
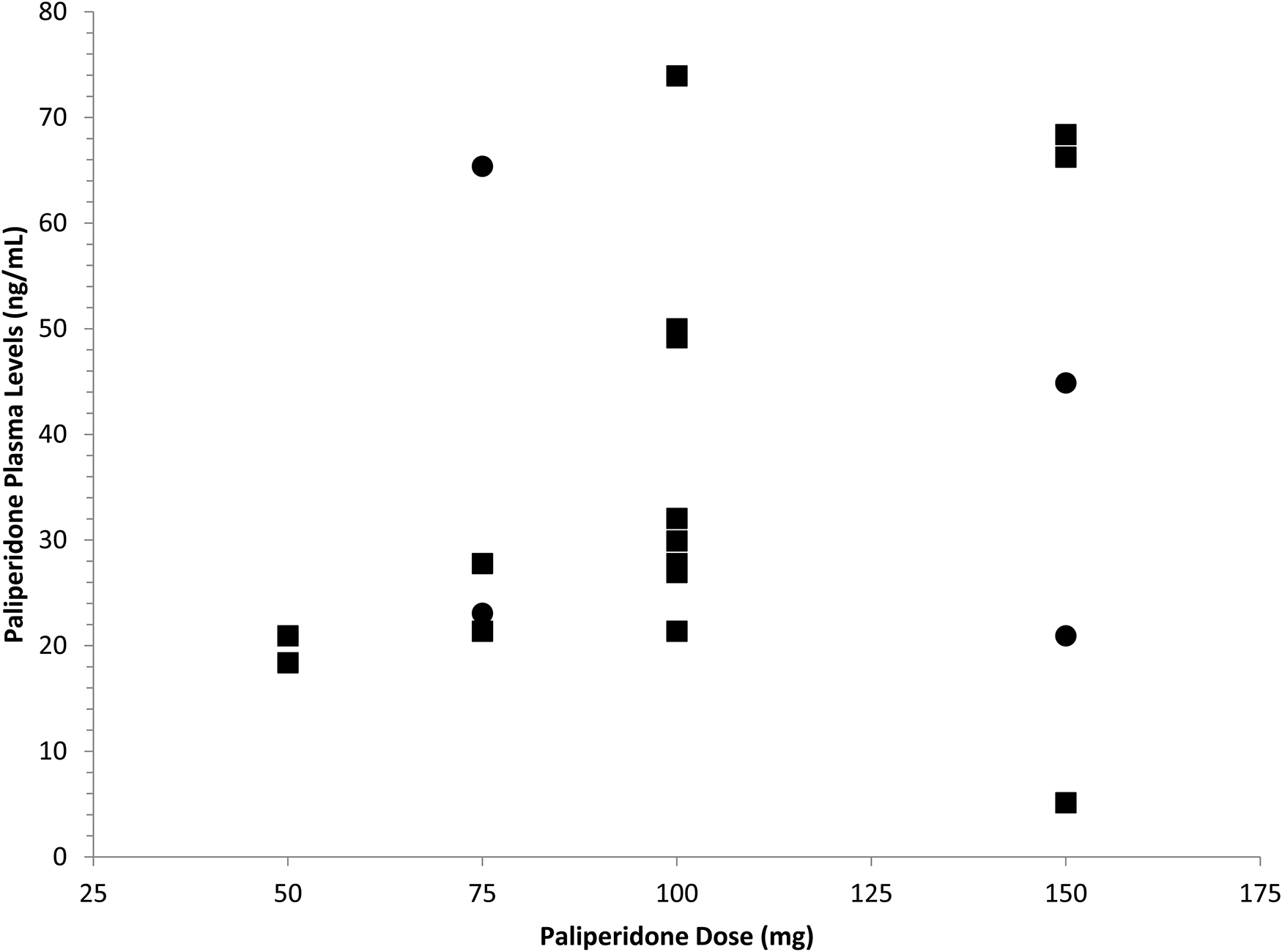
Trough paliperidone plasma levels for individuals receiving paliperidone palmitate every three weeks (●) and four weeks (▪).
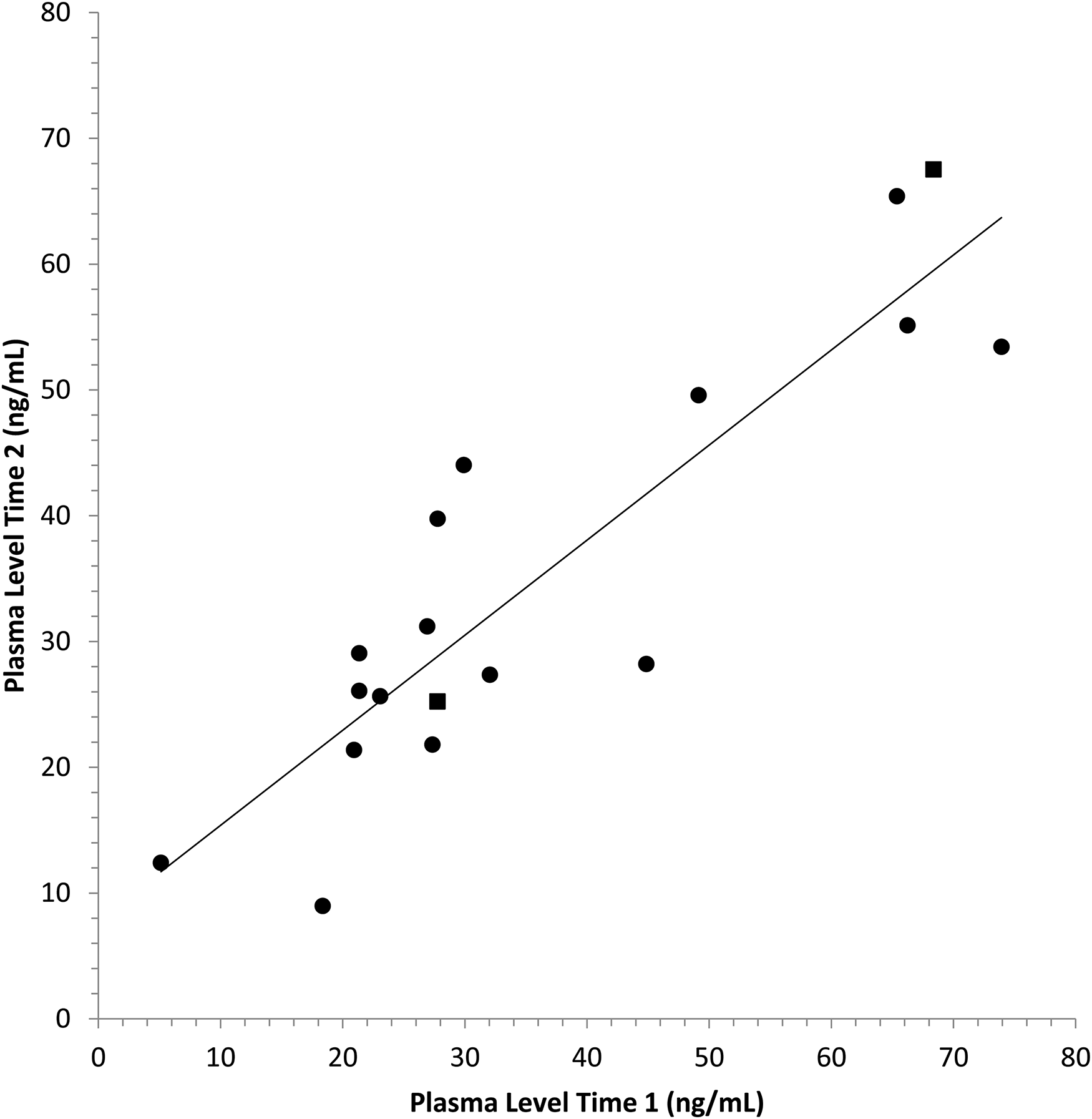
Paliperidone plasma levels measured at Visit 1 versus Visit 2 were significantly correlated (
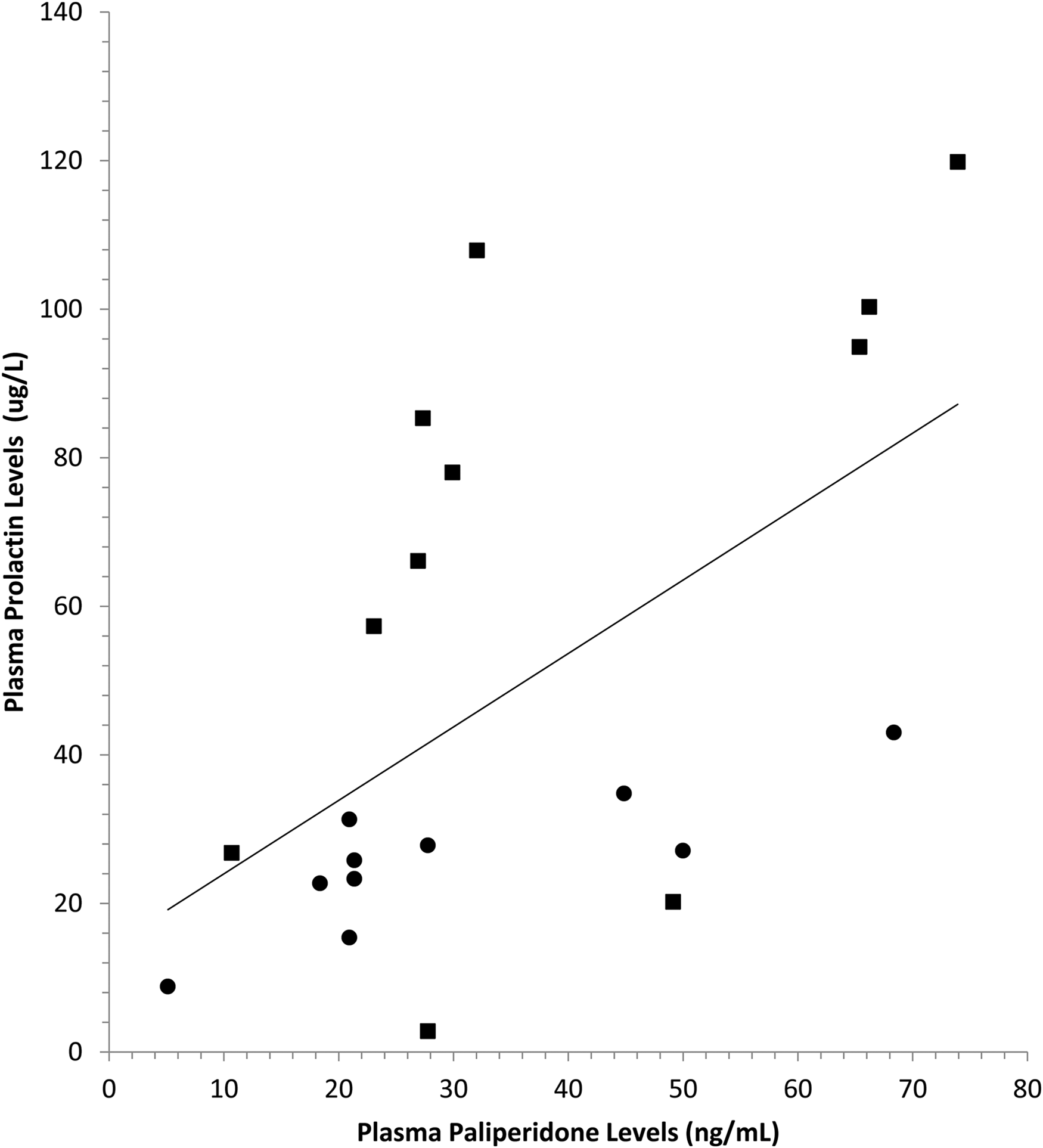
Plasma prolactin was significantly correlated with plasma paliperidone (
There was a trend for higher paliperidone levels to be associated with greater BMI but this did not reach statistical significance (ρ = .42,
Mean CSFQ-14 total score was 32.9 (SD = 12.7). The mean was 39.3 (SD = 8.8) for women and 27.0 (SD = 13.1) for men, both of which are below the threshold which is indicative of sexual dysfunction. CSFQ-14 total scores were not correlated with either paliperidone levels (ρ = −.40,
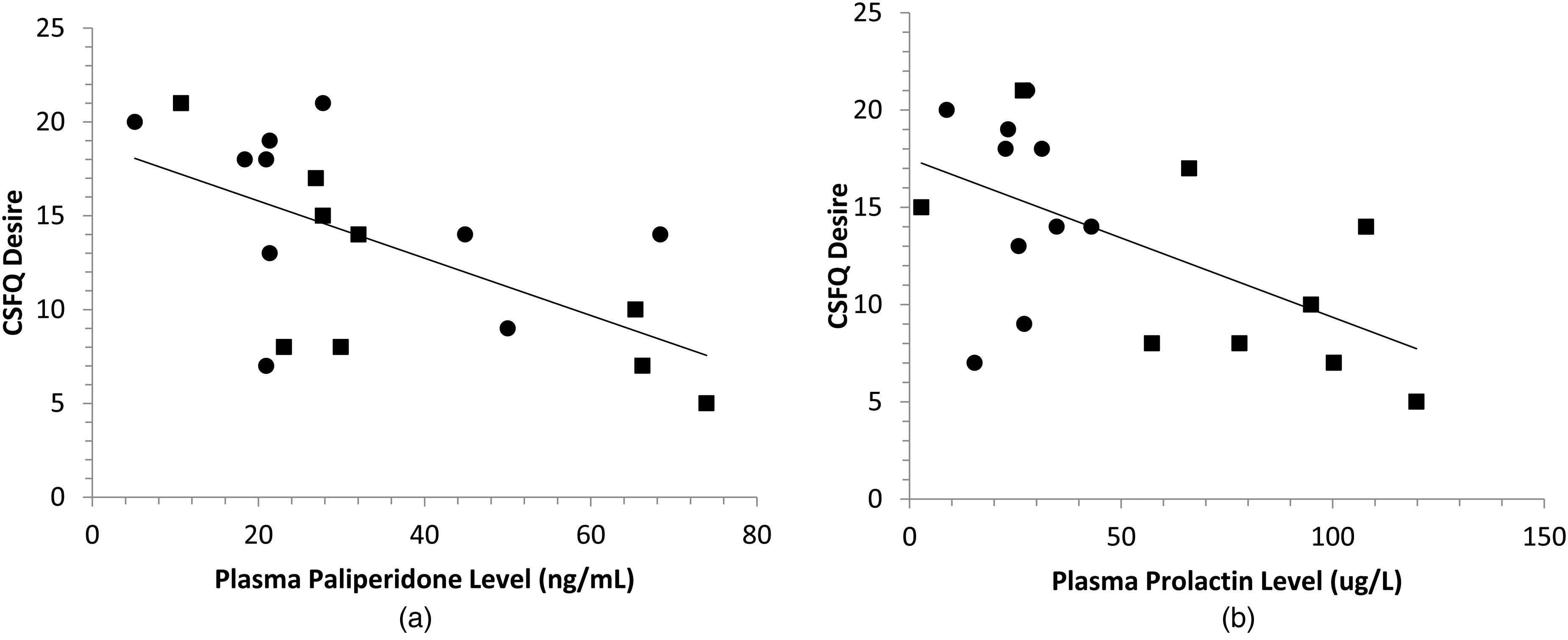
(a) Plasma paliperidone was significantly correlated with CSFQ Desire (
Discussion
In this study, we have demonstrated that paliperidone plasma levels can be measured reliably in patients being treated with the monthly formulation of LAI paliperidone and that higher paliperidone plasma levels are associated with higher levels of prolactin and reduced sexual desire.
In order for plasma antipsychotic levels to have a meaningful role in clinical practice, it is necessary to demonstrate that trough levels can be reliably measured over time. The consensus guidelines for therapeutic guidelines in neuropsychopharmacology consider a single plasma level to be insufficient for clinical decision-making. 7 It has remained unclear how consistent trough paliperidone levels can be expected to be in individuals treated with LAI paliperidone palmitate. This was clearly demonstrated in this dataset. It is of interest that standard clinical doses yielded paliperidone level that varied 15-fold between individuals. Trough plasma levels averaged 35 ng/ml which is comparable to the mean paliperidone level of 36 ng/ml reported by Nazirizadeh et al. 21 in a study of 217 patients treated with paliperidone palmitate. These plasma levels are also consistent with the therapeutic range of 20–60 ng/ml described for oral risperidone. 22 At Time 1, 3/21 participants had trough paliperidone levels that were below the therapeutic range described for oral paliperidone while 4/21 participants had levels above the therapeutic range. The mean plasma levels at trough were 75% higher than the lower limit of the therapeutic range, which approximates plasma levels associated with 65% dopamine D2 receptor occupancy in positron emission tomography (PET) studies. 23 This raises the possibility that lower doses of paliperidone palmitate may be therapeutic for some of these patients. Previous research 24 has demonstrated that many patients who have been effectively treated with long-acting risperidone have trough plasma level below 20 ng/ml, which suggests that plasma levels associated with 65% D2 occupancy may not be required throughout the injection cycle.
In clinical practice, measuring trough plasma levels in patients treated with paliperidone palmitate may be of value in individuals experiencing significant side effects. If the trough paliperidone level is found to be very high in patients who have responded well to paliperidone, then dosage reduction can likely be undertaken with little risk; if the trough paliperidone level is not elevated then dosage reduction may need to be carried out with greater caution as there will be a higher risk of loss of efficacy.
Antipsychotic medications are known to be associated with substantial weight gain with paliperidone considered to be of intermediate risk compared to haloperidol on the low end and olanzapine and clozapine on the high end. 25 In patients treated for a first episode of schizophrenia, haloperidol is associated with an average weight gain of 7.5 kg compared to 15.4 kg for those treated with olanzapine. 26 Higher antipsychotic doses are associated with greater weight gain 27 and this has been previously reported in individuals treated with paliperidone. 28 Participants in this study had a median BMI of 28.9 with a non-significant trend for those with higher paliperidone levels to have greater BMIs. In our sample, only four subjects had BMIs in the healthy range with seven in the overweight range, nine in the obese range and one in the severely obese range. Efforts to achieve the minimum effective dose of antipsychotic medication may well help to minimize the weight gain and metabolic problems associated with maintenance antipsychotic treatment.
Elevated prolactin levels have been reported with many antipsychotic medications and are known to be especially common in patients who are treated with risperidone and paliperidone. 25 Prolactin levels were significantly higher in women than in men in our sample (see Figure 4(b)), which is consistent with previous work reporting that elevated prolactin levels are more than twice as likely in women than in men treated with antipsychotic medication. 29 Elevated prolactin levels may result in amenorrhea and galactorrhea. It may also lead to sexual side effects in both men and women, which is consistent with our finding of reduced sexual desire with elevated prolactin levels. That prolactin levels were significantly correlated with plasma paliperidone levels suggests that monitoring plasma paliperidone levels in order to achieve plasma levels close to the threshold required for antipsychotic efficacy would also lead to reductions in prolactin levels.
We were not able to find an association between plasma levels and subjective well-being as measured by the SWN as has been previously described. 30 De Haan et al. 31 used single photon emission computed tomography to study 22 patients with schizophrenia treated with stable doses of olanzapine or risperidone for at least 6 weeks. They reported that higher striatal D2 occupancy was associated with worse subjective experience as measured by the SWN. This finding was replicated using PET by Mizrahi et al. 32 who studied 12 subjects with recent onset psychosis who were randomized to treatment with either high or low doses of olanzapine or risperidone for a period of 2 weeks. Striatal D2 receptor occupancy ranged from 50% to 91% in striatal areas and 4% to 95% in extrastriatal areas. The design of their study including a broad range of doses and brief period of treatment likely enhanced the potential to identify an association between D2 occupancy and SWN. In the present study, participants had been treated for a mean of 18 months, which presumably was a sufficient period of time to adjust doses for efficacy and tolerability, and may have limited the variance in plasma levels observed. This may also explain why we did not find plasma antipsychotic levels to be associated with severity of depressive symptoms, as De Haan et al. 31 had reported, or with other measures of illness severity, social and occupational functioning, happiness or satisfaction with life.
The lengthy mean duration of treatment with LAI paliperidone did not eliminate the findings that total scores of the CSFQ-14 were on average in a range indicating sexual dysfunction. While there may be many factors that contribute to sexual dysfunction in men and women with schizophrenia, the finding of an association between the CSFQ-14 Desire and higher levels of both paliperidone and prolactin suggests that treatment with paliperidone is a significant contributing factor. This is an important consideration when clinicians are trying to optimize the dosing of paliperidone for long-term maintenance treatment.
Interpretation of the study results is limited by a number of considerations. The sample size was modest, which limited the power to identify significant associations between plasma paliperidone levels and clinical measures. The limited racial diversity of this sample limits the generalizability of our findings. That many clinical centres lack the capacity to measure antipsychotic levels limits the application of our findings. As prolactin levels are known to fluctuate throughout the day, it would have been ideal to standardize the time that prolactin levels were drawn. This was not possible with this group of participants. It is notable that associations between prolactin levels and both paliperidone plasma levels and measures of sexual desire were found nonetheless. It will be important for future studies to standardize the timing for prolactin levels.
We elected to include individuals who were receiving oral antipsychotic medication as well as antidepressant and mood-stabilizing medication. Only 2/21 participants were receiving oral paliperidone and none were receiving risperidone so our finding of a high correlation between paliperidone plasma levels at Time 1 and Time 2 was not likely impacted by including these subjects. Concurrent medications could well have contributed to scores on the DAI, SWN and CSFQ. That 7/21 patients were on antidepressant medications, which often lead to sexual dysfunction, could also have contributed significantly to the low total scores on the CSFQ-14. When participants receiving antidepressants were removed from the analysis, the associations with CSFQ Desire remained statistically significant for both paliperidone levels (ρ = −.63,
Conclusions
Our findings support the view that paliperidone levels can be measured reliably at trough for patients receiving LAI paliperidone every 3 or 4 weeks. When patients are switched from oral to LAI paliperidone, there is a risk that some patients may be prescribed suboptimal doses that put them at risk of relapse; for others who are experiencing significant side effects with the transition, the finding of elevated plasma paliperidone levels may be helpful in guiding dosage adjustment. In our study, participants’ trough paliperidone plasma levels were, on average, in the middle of the therapeutic range described for oral paliperidone. Many patients, however, had plasma levels close to the 20 ng/ml level, which in PET studies leads to 65% dopamine D2 receptor occupancy, a level reported to be associated with antipsychotic response. Our findings raise the possibility that maintaining patients at levels just above the 20 ng/ml level may be sufficient for relapse prevention and may lead to less prolactin elevation and less impact on sexual desire. Whether these benefits would translate into improved adherence to maintenance antipsychotic treatment and fewer relapses remains to be established. Further research is required to define the minimum paliperidone plasma levels required for effective prevention of psychotic relapses in patients treated with paliperidone palmitate. 24
Footnotes
Acknowledgements
We would like to thank Helen Huyhn for her work in coordinating this study, Olivia Spandier for assistance with statistical analysis and manuscript preparation, Wei Wang PhD for statistical consultation, and the participants and staff from the Schizophrenia Outpatient Clinic at St. Joseph's Healthcare Hamilton whose involvement and support made this work possible. This work was presented at the Schizophrenia International Research Society (SIRS) Conference in Florence, Italy (4–8 April 2018). Data on paliperidone dose and plasma levels were included in the systematic review of therapeutic drug monitoring studies for long-acting paliperidone. 8
Data Availability
Anonymized data used in this study can be requested through the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by an educational grant from Janssen Canada.
