Abstract
Objectives:
To understand the impact of 3-monthly treatment with paliperidone palmitate on patient management, including non-adherence and relapse, from a psychiatrist and nurse perspective for 73 patients enrolled in a patient familiarisation programme (PFP) in New Zealand.
Methods:
An online questionnaire was sent to clinicians with at least 6 months of regular interaction with PFP patients. Questions addressed treatment effectiveness and patient management changes. Analyses are descriptive only and do not represent patient or carer perspectives.
Results:
Seven psychiatrists, representing 58 of 73 (79.5%) of patients, and 17 nurses responded to the survey. Psychiatrists were satisfied with efficacy and tolerability and relapse prevention. Treatment goals were either ‘met’ (2/7; 28.6%) or ‘exceeded’ (5/7; 71.4%). The focus on adherence issues decreased and the focus on life areas and recovery goals increased.
Conclusions:
From the clinician perspective, 3-monthly paliperidone palmitate offers patients the potential to remain adherent and improve social functioning.
A good relationship between psychiatric healthcare professionals and patients is critical. 1 Developing this therapeutic relationship can be challenging and complicated by patient factors such as reduced awareness of their illness or an unwillingness to communicate. 2 An improved therapeutic relationship can improve patient outcomes, including adherence to therapy. 3
Avoidance of relapse in schizophrenia is a health imperative. People who experience relapse have poorer function and diminished cognitive capacity. 4 Extending the half-life of long-acting injectables (LAIs) not only reduces the risk of relapse 5 but has the potential to reduce non-adherence by increasing the time between injections. Invega Trinza® is a 3-monthly injection of paliperidone palmitate, used in the maintenance treatment of schizophrenia in adult patients who have been adequately treated with the monthly paliperidone palmitate formulation (Invega Sustenna®).6,7
Invega Trinza is registered by Medsafe in New Zealand; a funding application has been submitted to the Pharmacology and Therapeutics Advisory Committee. An Invega Trinza patient familiarisation programme (PFP) was launched in New Zealand in 2017. Medication administration was in accordance with the registered indication in New Zealand. We surveyed psychiatrists and nurses at participating PFP centres to understand the real-world impact of 3-monthly paliperidone palmitate on patient outcomes. It should be noted that patients and carers were not contacted for their opinions as part of this survey.
Methods
Between 7 September 2017 and 7 March 2018, 73 patients from seven District Health Boards (DHBs) in New Zealand were enrolled in the PFP. Patients provided their verbal and written consent to participate in the PFP; this was noted in individual patient clinic records. In April 2019, an online questionnaire was sent to 12 psychiatrists and 39 nurses who had at least 6 months of regular interaction with PFP patients. The questionnaire was based on that used in the Australian 3-monthly paliperidone palmitate PFP. 8 A complete list of response options is found in Table 1. Ethical approval was not required as patients were not surveyed and no patient-level data were collected.
End of PFP psychiatrist and nurse survey questions

PFP: Patient familiarisation programme.
Eligible patients included well, stable patients on Invega Sustenna; patients requiring decreased frequency of injections due to work commitments; recalcitrant patients resistant to treatment; itinerant patients and patients subject to compulsory treatment orders consistent with Section 29 of the Mental Health Act. 9 Patients subject to compulsory orders had to voluntarily agree to enrol in the PFP, demonstrating a level of shared decision-making ability.
To maintain anonymity, the distribution, response collection and summary of results of the questionnaire was managed by Atlantis Healthcare. No formal statistical testing was performed; analyses are descriptive only.
Results
Seven of 12 psychiatrists and 17 of 39 nurses who were sent the survey link provided responses. Overall, psychiatrist responses represented 52 of 73 patients (79.5%) at six of seven DHBs involved in the PFP. An average of seven vials per PFP patient were dispensed between the dates 7 September 2017 to 29 November 2019 (range: 1–15 vials). It was not possible to identify the number of vials dispensed on average to the patients represented by prescriber responses.
Patient experience (clinician-reported)
The majority of psychiatrists (6/7; 85.7%) and nurses (9/17; 52.9%) reported a decreased focus on medication adherence and medication issues, with five (5/7; 71.4%) stating that this had significantly decreased. Seven nurses (7/17; 41.2%) said this focus had remained unchanged, and one (1/17; 5.9%) stated that the focus had somewhat increased.
All psychiatrists believed that they had been able to increase the focus on other, non-medication related life areas and recovery goals during patient interactions; of these, four (4/17; 57.1%) stated that this focus had significantly increased. Ten nurses (10/17; 58.8%) reported an increased focus on other life areas and recovery goals; however, seven (7/17; 41.2%) thought the focus had not changed.
Survey responses indicated that discussions with patients had focused on meaningful activity (employment, education, volunteering), wellness planning, psycho-education and relationships (Figure 1).
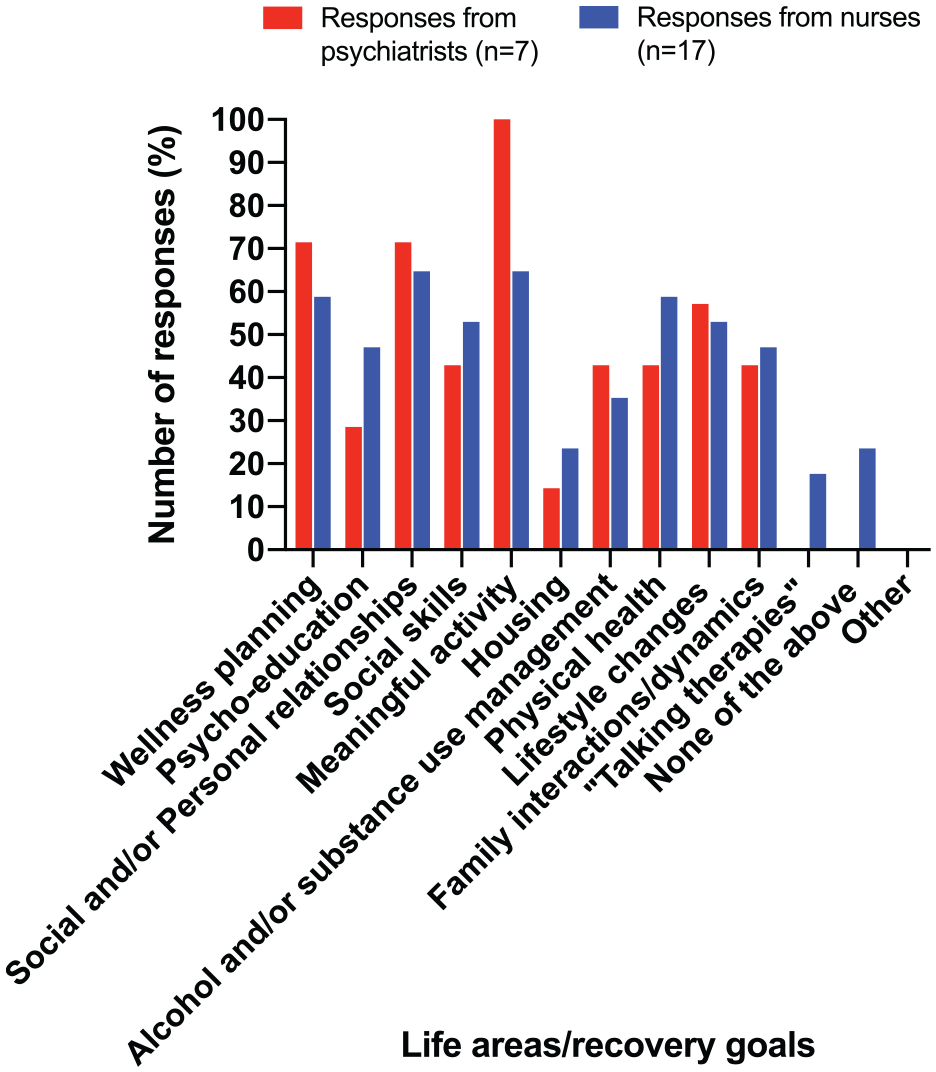
Responses from psychiatrists and nurses for survey questions relating to life areas/recovery goals for patients receiving INVEGA TRINZA®.
Clinician experience
Survey questions related to identifying a patient’s suitability for treatment, the effectiveness (efficacy and tolerability) of the 3-monthly regimen, satisfaction with relapse prevention (reduced risk of hospitalisation) and whether or not treatment goals were met were posed to psychiatrists alone. All psychiatrists were confident in identifying whether a patient was suitable for treatment; most were very or completely confident (3/7 each; 42.9%). All psychiatrists were satisfied with the effectiveness of 3-monthly paliperidone palmitate; see Figure 2.
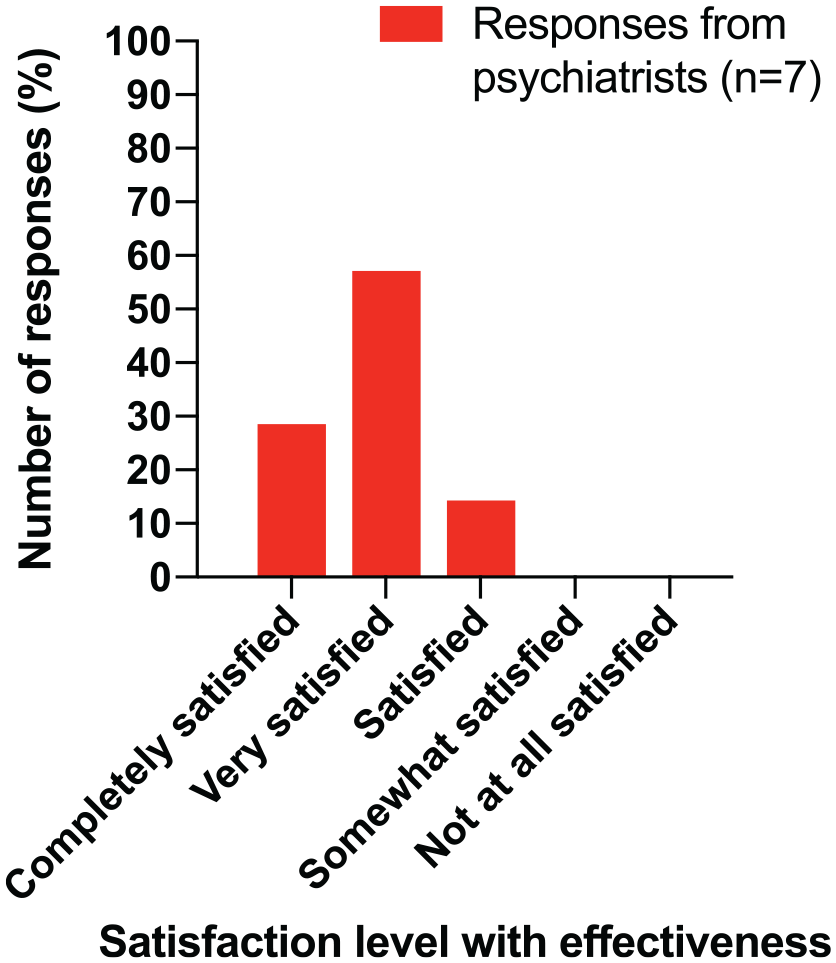
Psychiatrist satisfaction with effectiveness (efficacy and tolerability) for patients taking INVEGA TRINZA®.
Similar high levels of prescriber satisfaction were reported with the use of 3-monthly paliperidone palmitate in the prevention of relapse; see Figure 3.
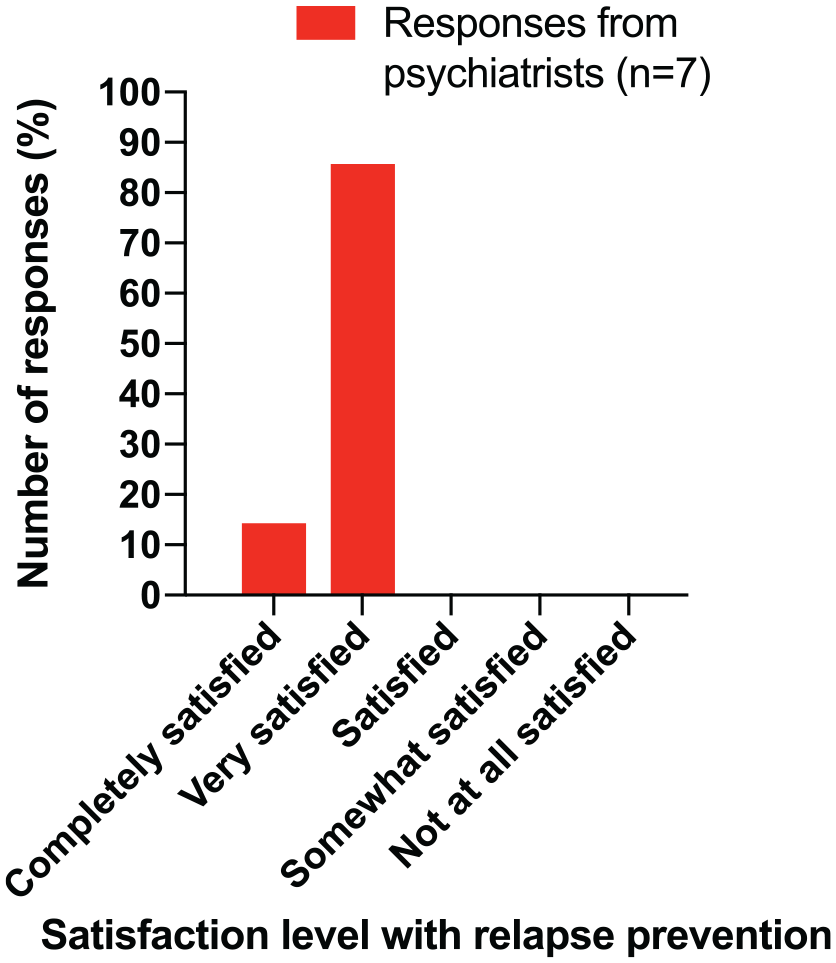
Psychiatrist satisfaction with relapse prevention (reduced risk of rehospitalisation) for patients taking INVEGA TRINZA®.
Clinicians were asked to report their confidence with the administration of 3-monthly paliperidone palmitate, comfort levels with patient management given the longer dosing interval, and whether the frequency of patient interactions had changed. All were confident with administration to varying degrees; the majority reported high levels of confidence and only one psychiatrist (1/7; 14.3%) and one nurse (1/17; 5.9%) reported that they were only somewhat confident. Although psychiatrists were themselves unlikely to physically administer the injection, it was their responsibility to ensure the prescribing and administration were correctly performed.
All psychiatrists were either very or completely comfortable with patient management given the longer dosing interval. Nurse responses were split between somewhat comfortable (1/17; 5.9%), comfortable (6/17; 35.3%) and very or completely comfortable (10/17; 58.8%).
Most psychiatrists reported that the frequency of patient interactions remained unchanged (6/7; 85.7%), and only one (1/7; 14.3%) said it had somewhat decreased. Approximately half of nurses also reported no change to the frequency of nurse–patient interactions (8/17; 47.1%), however, nine (9/17; 52.9%) reported a decreased frequency.
Discussion
The views garnered in this survey were those of healthcare providers only; no information was requested of patients or carers, so the results discussed may differ greatly from those from the patient perspective. From the provider perspective, clinical expectations were met in the 3-monthly paliperidone palmitate PFP. Prescribing psychiatrists reported that the treatment goals were met or exceeded, and all were satisfied with treatment efficacy and relapse prevention.
Integration into work, social and leisure activities is a key concern when treating people with schizophrenia. A recent meta-analysis showed that LAIs were superior to both placebo and oral antipsychotic medications in improvements in functional outcomes, 10 thus aiding in meeting the challenge of patient integration. Survey results indicated a positive shift in patient–clinician dynamics when patients received Invega Trinza. Patients themselves have also reported the importance of a combination of good therapeutic conversations, trusting relationships with healthcare professionals and antipsychotic treatment over time. 11
A reduced risk of relapse not only improves patient outcomes but brings with it potential cost savings to the New Zealand healthcare system. An Australian study found that hospitalisation due to relapse is associated with an increase in costs, which appear to persist over a 12-month period. 12 A recent study in the United States projected reduced hospitalisation-associated costs for patients switching from oral antipsychotics to 3-monthly paliperidone palmitate. 13 Additional research has shown that patients stabilised on a LAI are likely to want to continue treatment with the LAI. 14
A locally relevant publication reporting prescriber attitudes and knowledge of risperidone LAI reported limited use of the LAI; it was viewed as a last resort and there was a perception that LAIs somehow encroached upon the quality of the therapeutic relationship. 15 These views are likely to apply to other LAI formulations and may potentially restrict applying the benefits of LAIs such as 3-monthly paliperidone palmitate to the New Zealand population. Patients may also be resistant to receiving LAIs feeling there is a stigma attached to their use. 16 Utilising the principles of the Listen-Empathize-Agree-Partner® (LEAP) approach can be very beneficial in enhancing acceptance of medical treatments and services. 17
Limitations of the current study included a lack of data from patients and carers on their experience with 3-monthly paliperidone palmitate and a lack of patient demographic and prior and current dosing data. Not all psychiatrists involved in the PFP responded; however, there was good patient representation from those who did respond.
Conclusions
Using 3-monthly paliperidone palmitate has the potential to improve patient outcomes by increased adherence, reduced risk of relapse and an increased focus on life areas and recovery goals. Global data have described the significant cost of relapse and has projected reductions in associated costs with the use 3-monthly paliperidone palmitate; therefore, there is the potential for cost savings in New Zealand. Medical education should focus on increasing prescriber knowledge regarding the utility of LAIs; this may help to combat any remaining negative connotations surrounding their use for both prescribers and patients. Future research is needed to investigate the patient and carer experience and perspectives on the use of 3-monthly paliperidone palmitate.
Footnotes
Acknowledgements
The authors thank Rachelle Steele, BPharm(Hons) of WriteSource Medical Pty Ltd, Sydney, Australia, for providing medical writing support/editorial support by preparing the manuscript outline, developing the first draft and collating and incorporating author comments. Medical writing support was funded by Janssen Australia and New Zealand in accordance with Good Publication Practice (GPP3) guidelines (![]() ).
).
Disclosure
CC and WM were treating psychiatrists in the New Zealand PFP, who have received an honorarium for attending a Janssen Invega Trinza® advisory board meeting in 2018.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Janssen Australia and New Zealand.
Author contribution and statement
Both authors made a substantial contribution to the conception and design of the survey and interpretation of the data. Both authors critically revised the manuscript for important intellectual content and approved the final version to be published. Both authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriate investigated and resolved.
