Abstract
Objective
One in every 4 individuals born with a 22q11.2 microdeletion will develop schizophrenia. Thirty years of clinical genetic testing capability have enabled detection of this major molecular susceptibility for psychotic illness. However, there is limited literature on the treatment of schizophrenia in individuals with a 22q11.2 microdeletion, particularly regarding the issue of treatment resistance.
Methods
From a large, well-characterized adult cohort with a typical 22q11.2 microdeletion followed for up to 25 years at a specialty clinic, we studied all 107 adults (49 females, 45.8%) meeting the criteria for schizophrenia or schizoaffective disorder. We performed a comprehensive review of lifetime (1,801 patient-years) psychiatric records to determine treatments used and the prevalence of treatment-resistant schizophrenia (TRS). We used Clinical Global Impression–Improvement (CGI-I) scores to compare within-individual responses to clozapine and nonclozapine antipsychotics. For a subgroup with contemporary data (n = 88, 82.2%), we examined antipsychotics and dosage at the last follow-up.
Results
Lifetime treatments involved on average 4 different antipsychotic medications per individual. Sixty-three (58.9%) individuals met the study criteria for TRS, a significantly greater proportion than for a community-based comparison (42.9%; χ2 = 10.38, df = 1, p < 0.01). The non-TRS group was enriched for individuals with genetic diagnosis before schizophrenia diagnosis. Within-person treatment response in TRS was significantly better for clozapine than for nonclozapine antipsychotics (p < 0.0001). At the last follow-up, clozapine was the most common antipsychotic prescribed, followed by olanzapine, risperidone, and paliperidone. Total antipsychotic chlorpromazine equivalent dosages were in typical clinical ranges (median: 450 mg; interquartile range: 300, 750 mg).
Conclusion
The results for this large sample indicate that patients with 22q11.2 microdeletion have an increased propensity to treatment resistance. The findings provide evidence about how genetic diagnosis can inform clinical psychiatric management and could help reduce treatment delays. Further research is needed to shed light on the pathophysiology of antipsychotic response and on strategies to optimize outcomes.
Plain Language Summary Title
Real-world treatment of schizophrenia in adults with a 22q11.2 microdeletion
Plain Language Summary
There is little uptake of clinical genetic testing in psychiatry. A major question has been whether genetics can provide information that is relevant for the psychiatrist's clinical treatment decisions. The most common of the rare genetic changes that are associated with increased risk for schizophrenia (SZ) is a deletion on the long arm of chromosome 22 (22q11.2 microdeletion). This study examined real-world clinical data amassed for 107 individuals with SZ and microdeletion 22q11.2. Using conservative criteria and the lifetime treatment history, the research team compared two groups, with and without treatment resistance. Nearly 60% of individuals with SZ and 22q11.2 microdeletion had treatment resistance, a significantly greater proportion than the comparison sample, and greater than usual expectations of about 30%. The subgroup with treatment resistance had been tried on an average of 5 antipsychotics, significantly more than the no treatment resistance group. Dosages for the many antipsychotics used appeared to be in the usually reported range for SZ. This suggests that effectiveness and tolerability of the medications for individuals with 22q11.2 microdeletion may be comparable to those for others with SZ. Clozapine, the antipsychotic recommended for individuals with treatment-resistant SZ, showed greater effectiveness than other antipsychotics within individuals, although average dosage was in the low end of the range typically used. This study shows evidence of a connection between genetic variation and clinical intervention in SZ. The results add to those of recent studies suggesting that individuals with a 22q11.2 microdeletion and similar clinically relevant genetic changes are hidden within treatment-resistant SZ populations. The findings support the clinical utility of molecular diagnosis for SZ, and the potential value of this genetic model for studying SZ and its treatment.
Introduction
The 22q11.2 microdeletion is the strongest known molecular risk factor for schizophrenia, conveying >20-fold increased risk over population expectations, and affecting approximately 0.5% to 1% of individuals with schizophrenia.1,2 With an estimated live birth prevalence of one in 2,148, 3 the 22q11.2 microdeletion is the most common of clinically relevant copy number variations (CNVs) that collectively account for an estimated one in every 25 individuals with schizophrenia.4,5
Antipsychotic medications are the mainstay of treatment for schizophrenia in the general population.6–8 However, an estimated 30% of individuals have treatment resistance: inadequate improvement in psychotic symptoms after 2 or more antipsychotic trials.6,9 Recent reports have suggested a possible association between treatment resistance and CNVs,10–12 with previous studies of 22q11.2 microdeletion also speculating about possible enriched treatment resistance.13,14 However, none of these studies has involved more than 20 individuals with 22q11.2 microdeletion and schizophrenia, and a recent review deemed the evidence for treatment resistance to be inadequate. 15 While there is no question that molecular genetic diagnosis changes overall management, 2 many questions remain about antipsychotic treatment of schizophrenia and treatment resistance for individuals with a 22q11.2 microdeletion.
The current study used real-world data to delineate antipsychotic treatment and treatment resistance in a community-based cohort of 107 adults with schizophrenia and 22q11.2 microdeletion. We predicted a higher prevalence of treatment resistance in these individuals, compared to estimates available for a comparable general schizophrenia population sample.
Methods
This study used a retrospective observational design involving a well-characterized community-based adult (age ≥18 years) cohort (n = 402) with typical (most commonly 2.5 Mb LCR22A to LCR22D) 1 chromosome 22q11.2 microdeletion.16,17 We included all n = 107 (26.6%) meeting DSM-5 criteria for schizophrenia (n = 90) or schizoaffective disorder (n = 17), 18 collectively termed schizophrenia. The cohort was ascertained through a specialty clinic for adults with 22q11.2 microdeletion, at the transition from paediatrics and through referrals (genetic, psychiatric, or other).16,17 Patients were followed for up to 25 (median: 6.8; interquartile range (IQR): 3.0, 14.7) years. Informed consent was obtained in writing, with study approval by hospital research ethics boards affiliated with the University of Toronto. Treatment for n = 20 was previously reported. 13
Age was recorded as at study end (March 1, 2023) or death (n = 17). We recorded age at molecular diagnosis of 22q11.2 microdeletion, presence of major congenital heart disease 19 and moderate/severe intellectual disability. 17
We comprehensively reviewed available (1,801 patient-years) psychiatric records, clinical summaries, pharmacy records, and patient/caregiver reports. 20 Multiple clinicians were involved in treatment decisions, with choices based on their clinical judgment. We recorded the age at the onset of psychotic illness requiring treatment, and regularly scheduled antipsychotics prescribed, including dosage, side effects, and reasons for discontinuation.
Definition of Treatment Resistance and Other Markers of Clinical Severity
The most common definition of treatment-resistant schizophrenia (TRS) is the failure of ≥2 nonclozapine antipsychotics (adequate dose/duration).6,9,21 We chose a commonly used conservative proxy22,23: any clozapine trial or offer/recommendation,24–26 and/or a clinical diagnosis of TRS with ≥2 nonclozapine antipsychotics.
We also noted other markers of severe/difficult-to-treat illness: lifetime use of electroconvulsive therapy (ECT), 6 or ≥2 concurrent antipsychotics.27,28
Comparison With Prevalence of Treatment Resistance in Community-Based Schizophrenia Sample
The prevalence of TRS ranges greatly, depending on the definition used and the population studied (e.g., higher for inpatients than outpatients).6,29 We searched the literature for a large community-based (outpatient) sample to use as a comparison to the 22q11.2 microdeletion cohort, seeking one as comparable as possible on key parameters (age, duration of illness, and outpatient status), and definition of TRS used. We identified a study reporting on a random cluster sample of 1,739 outpatients with schizophrenia and analysed for TRS based on eligibility for treatment with clozapine. 30 This sample was of similar mean age (40.0, SD 11.6 vs. 40.5, SD 12.3 years; t = −0.43, p = 0.67) and illness duration to the 22q11.2 microdeletion cohort, though had proportionately more males (70% vs. 54.2%, p ≤ 0.0001) and fewer of European ancestry (52% vs. 82%, p ≤ 0.0001).
Assessment of Treatment Response to Clozapine
We used the Clinical Global Impression–Improvement (CGI-I) scale to assess within-individual treatment response for 30 participants with sufficient pre- and post-treatment data for clozapine and a nonclozapine antipsychotic,13,31,32 selecting the latter through random-generation (SAS 9.3, SAS Institute, Cary, NC). CGI-I ratings of 1, 2, 3 represent very much, much, minimally, improved, 4 no change, 5–7 minimally, much, and very much worse, respectively. 31 The CGI-I was completed independently by 2 psychiatrists (LV and ASB), using full data (LV), and 60 (2-per-participant) anonymized clinical vignettes prepared on pre- and post-treatment mental state and functioning from all records, blinded to antipsychotic and participant (ASB). CGI-I-rating interrater-reliability showed substantial agreement (weighted Cohen's kappa 33 0.69, 95% CI, 0.58 to 0.80).
Contemporary Subsample (n = 88) and Treatment at Last Follow-up
Given changes in prescribing over the prolonged time frame, we examined a contemporary subsample at the last follow-up. We derived a subset of 88 (82.2%) participants by restricting those with treatment data from January 1, 2012, to the study end, and recorded antipsychotics at the last follow-up (median-year: 2020; IQR: 2018, 2021). We converted dosages to chlorpromazine equivalent (CPZE) using chlorpromazineR (version 0.2.0).34–37 We designated primary treatment as clozapine or nonclozapine antipsychotic with the highest CPZE when >1 was concurrently prescribed. We summed the total CPZE per individual. We also noted individuals receiving ECT.
Analyses
For descriptive statistics, main analyses and subanalyses, we used χ2, Fisher's exact, and Wilcoxon 2-sample tests, as appropriate, for categorical and rank-ordered, or continuous but nonnormally distributed variables. We analysed within-individual CGI-I scores for clozapine and nonclozapine antipsychotics using a Wilcoxon signed-rank test. In the main analyses of lifetime treatment, we compared TRS and no-TRS subgroups. For the contemporary subset (n = 88), we compared antipsychotic dosage at the last follow-up between TRS and no-TRS groups. Statistical analyses were performed using SAS 9.3 (SAS Institute, Cary, NC). Two-tailed p-values <0.05 were considered statistically significant.
Results
Table 1 summarizes demographic, clinical, and genetic variables for the 107 adults with 22q11.2 microdeletion and schizophrenia in the study. Individuals were prescribed on average 4 antipsychotics over their lifetime of schizophrenia treatment (median age at onset 20.0 years, median duration of illness 17.7 years; Table 1). All 25 second- and first-generation antipsychotics recorded for the cohort are summarized in Supplemental Tables S1a and S1b, respectively, with dosages, side effects, and reasons for discontinuation provided for antipsychotics documented in ≥10 individuals.
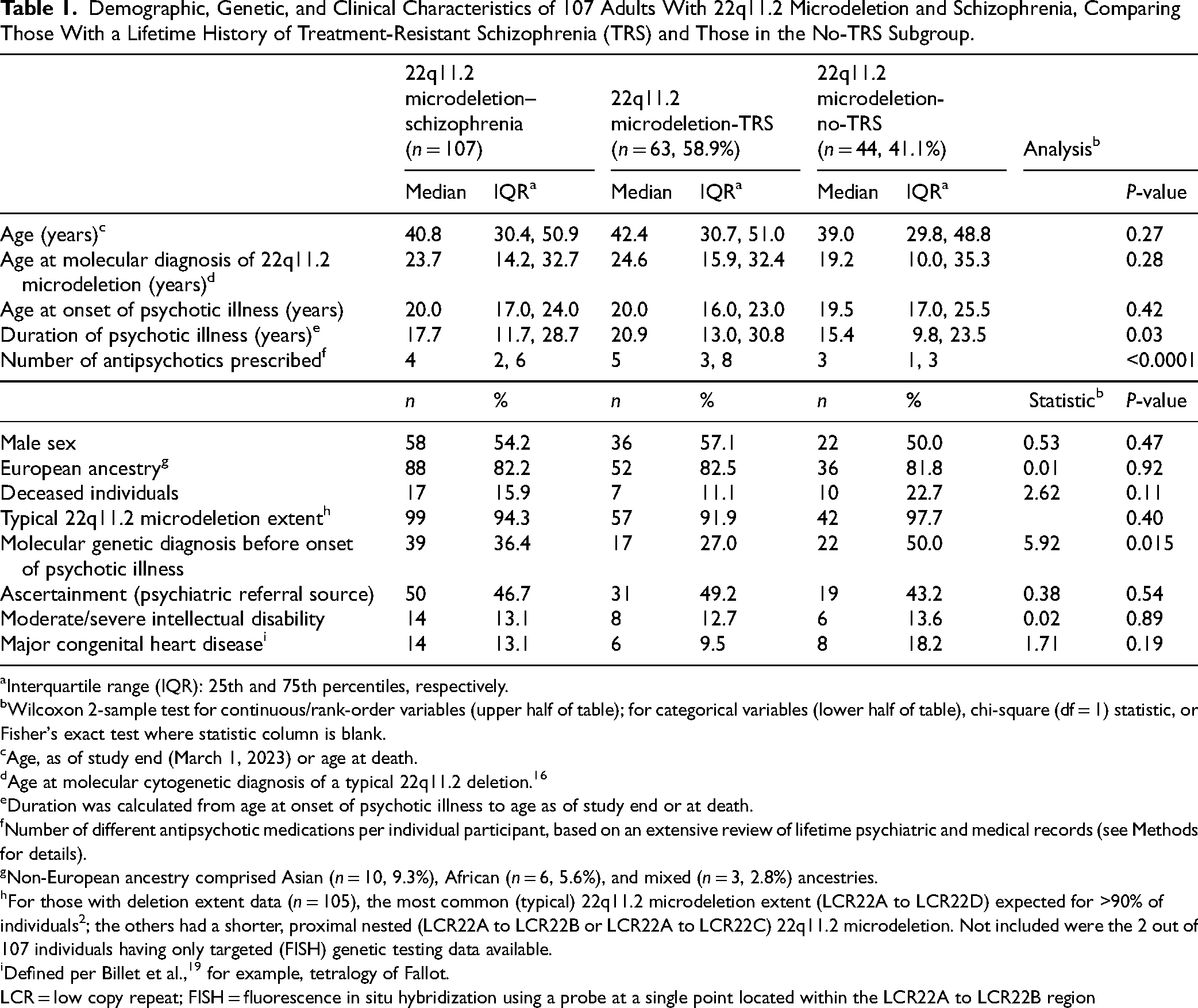
Demographic, Genetic, and Clinical Characteristics of 107 Adults With 22q11.2 Microdeletion and Schizophrenia, Comparing Those With a Lifetime History of Treatment-Resistant Schizophrenia (TRS) and Those in the No-TRS Subgroup.
Interquartile range (IQR): 25th and 75th percentiles, respectively.
Wilcoxon 2-sample test for continuous/rank-order variables (upper half of table); for categorical variables (lower half of table), chi-square (df = 1) statistic, or Fisher's exact test where statistic column is blank.
Age, as of study end (March 1, 2023) or age at death.
Age at molecular cytogenetic diagnosis of a typical 22q11.2 deletion. 16
Duration was calculated from age at onset of psychotic illness to age as of study end or at death.
Number of different antipsychotic medications per individual participant, based on an extensive review of lifetime psychiatric and medical records (see Methods for details).
Non-European ancestry comprised Asian (n = 10, 9.3%), African (n = 6, 5.6%), and mixed (n = 3, 2.8%) ancestries.
For those with deletion extent data (n = 105), the most common (typical) 22q11.2 microdeletion extent (LCR22A to LCR22D) expected for >90% of individuals 2 ; the others had a shorter, proximal nested (LCR22A to LCR22B or LCR22A to LCR22C) 22q11.2 microdeletion. Not included were the 2 out of 107 individuals having only targeted (FISH) genetic testing data available.
Defined per Billet et al., 19 for example, tetralogy of Fallot.
LCR = low copy repeat; FISH = fluorescence in situ hybridization using a probe at a single point located within the LCR22A to LCR22B region
Lifetime History of Treatment Resistance in Individuals With 22q11.2 Microdeletion and Schizophrenia, and Response to Clozapine
There were 63 (58.9%) individuals who met the criteria for TRS, 61 based on clozapine recommendation/trial, and 2 on a clinical diagnosis of TRS. The proportion with TRS in the 22q11.2 microdeletion cohort was significantly greater than reported for a large community schizophrenia sample (42.9%, χ2 = 10.38, df = 1, p < 0.01). 30
The 22q11.2 microdeletion-TRS group had a significantly longer median duration of psychotic illness (20.9 years vs. 15.4 years, p = 0.03) and a greater median number of lifetime antipsychotics prescribed (5 vs. 3, p < 0.0001) than the no-TRS group (Table 1). Interestingly, in the no-TRS group, a larger proportion had 22q11.2 microdeletion detected before onset of schizophrenia, than the TRS group (Table 1). There were no other significant between-group differences in demographic, genetic, or clinical variables, including proportion with moderate/severe intellectual disability (Table 1). Eleven (10.3%) individuals had received a course of ECT over their lifetime of treatment (Supplemental Table S1a).
Of the 61 individuals recommended to have clozapine, 49 (80.3%) subsequently had a clozapine trial. The response to clozapine was significantly better than to nonclozapine antipsychotics within individuals (median CGI-I: 2; IQR: 2, 2; median CGI-I: 3; IQR: 3, 4, respectively; S = 141.5, p < 0.0001), for the subset (30 out of 49) with before and after treatment data available.
Most Recent Documented Treatment for a Contemporary Subgroup (n = 88)
Treatments at the last follow-up in the 22q11.2 microdeletion contemporary subgroup (n = 88 out of 107, 82.2%) are shown in Figure 1. The median total antipsychotic dosage per individual was 450 (IQR: 300, 750) mg CPZE, in line with average dosage ranges commonly used in general population schizophrenia.34–37 This included individuals treated concurrently with one or more antipsychotics or ECT (Figure 1). For most (n = 78 out of 88, 88.6%), the antipsychotic was a second-generation compound. Of the 12 primary antipsychotics recorded at the last follow-up, clozapine was the most commonly used (n = 29 out of 88, 33.0%; median clozapine dosage: 275 mg; IQR: 200, 350 mg), followed by olanzapine, risperidone, and paliperidone (Figure 1). Twelve (13.6%) individuals received a long-acting injectable (LAI) agent (Figure 1).
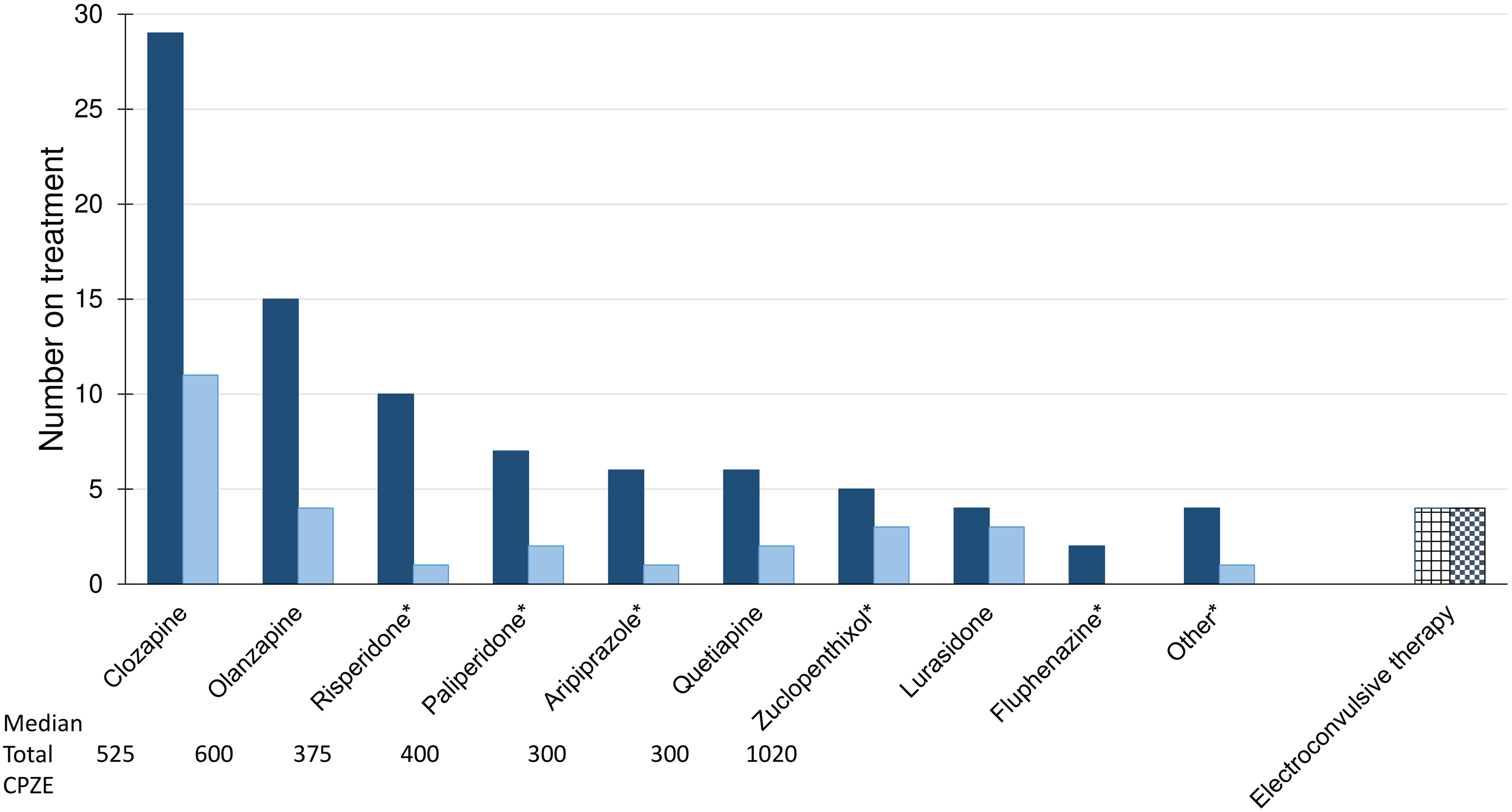
Treatments documented at the last follow-up for 88 adults with schizophrenia and 22q11.2 microdeletion. Shown is treatment information available at the last follow-up for the contemporary subsample of 88 individuals with schizophrenia and a 22q11.2 microdeletion (see Methods for details). Dark solid bars indicate results for the primary antipsychotic treatment (for all n = 88), and light solid bars the subset of individuals concurrently prescribed a secondary/additional antipsychotic. Primary treatments were deemed to be clozapine, or the nonclozapine antipsychotic with the highest CPZE dosage when more than one nonclozapine antipsychotic was prescribed (see Methods for details). An asterisk (*) indicates a long-acting injectable antipsychotic for one or more individuals (total, 12 out of 88 individuals). Median total CPZE dose (in mg) per individual (i.e., primary and secondary antipsychotics) is shown for the 7 most commonly used primary antipsychotics (i.e., involving ≥5 individuals). The category “Other” represents one individual each with primary antipsychotic ziprasidone, loxapine, or perphenazine, and one with severe Parkinson's disease who was receiving no antipsychotic treatment (but who, after the study end, was prescribed low dose clozapine). With respect to the 11 out of 29 individuals on clozapine who were concurrently prescribed another antipsychotic, at the last follow-up the CPZE dosage was higher for clozapine than for the other antipsychotic in 6 cases. Adjunctive/secondary antipsychotic medications included those named in the figure as primary antipsychotics, and also brexipiprazole, methotrimeprazine, chlorpromazine, and flupentixol. On the right (grid patterned bar) are individuals receiving ECT at the last follow-up, all concurrently prescribed primary antipsychotic treatment (dark checkered bar), 2 receiving quetiapine, one olanzapine, and one clozapine, each represented to the left within the n = 88 (dark solid bars).
Demographic, clinical and genetic variables, and lifetime results for this subset, both overall and for TRS versus no-TRS comparisons (Supplemental Table S2), appeared similar to those for the full sample (Table 1). Of the 88 individuals, 54 (61.4%) were in the TRS group (Supplemental Table S2). Total CPZE dosages at the last follow-up were significantly higher for the TRS group than for the no-TRS group (median 525 vs. 360 mg, respectively, Z = −2.20, p = 0.03). Antipsychotic polypharmacy at the last follow-up was documented for 28 (31.8%) individuals, including 11 (37.9%) of the 29 receiving clozapine (Figure 1). These 29 individuals represent a majority of those ever tried on clozapine within this subset (29 out of 42, 69.0%) (see footnote in Supplemental Table S1a).
Discussion
This naturalistic observational study of schizophrenia treatment focuses on 22q11.2 microdeletion, a clinically detectable genetic change that is one of the strongest known risk factors for schizophrenia.5,18,38 Capitalizing on a large well-characterized community-based cohort, we report several novel findings. Dosages for the many antipsychotics used in this 22q11.2 microdeletion cohort appear to be in the usual therapeutic range reported for schizophrenia, suggesting similar efficacy and tolerability profiles as in the general population. About 60% of individuals had TRS, indicating a high likelihood of developing treatment resistance in the 22q11.2 microdeletion population.6,30 Other indicators of treatment challenges included 3+ antipsychotic trials, antipsychotic polypharmacy, and ECT. Response to clozapine showed greater within-individual improvement compared to other antipsychotics. A majority of those ever receiving clozapine had ongoing use, with an average dosage in the lower end of the typical range.13,39 A provocative finding was that individuals diagnosed with 22q11.2 microdeletion before the onset of psychotic illness were underrepresented in the TRS group, suggesting possible advantages to early genetic diagnosis. These results shed further light on the clinical utility of molecular diagnosis for schizophrenia,4,40 and on the potential value of this genetic model for studying schizophrenia and its treatment.38,41–44
Treatment, Treatment Resistance, and Intolerance in 22q11.2 Microdeletion– Schizophrenia
Notably, dosages of nonclozapine antipsychotics used in this 22q11.2 microdeletion cohort were comparable to those commonly reported for schizophrenia in the general population.34–37 Of the wide variety of antipsychotics used, second-generation antipsychotics were most common (Supplemental Table S1a; Figure 1), in line with international prescribing trends. These results are consistent with a recent review that recorded treatments reported in the literature for psychosis, including a subset diagnosed with schizophrenia, in individuals with a 22q11.2 microdeletion or related syndrome. 15
A novel finding was that an estimated 60% of those with 22q11.2 microdeletion–schizophrenia met conservative criteria for treatment resistance (eligibility for clozapine), a significantly greater prevalence than for a large outpatient sample of schizophrenia. 30 This is also greater than figures for TRS reported in most studies of schizophrenia or as summarized in schizophrenia clinical practice guidelines.6–8 Even individuals with 22q11.2 microdeletion in the no-TRS group had been treated with an average of 3 different antipsychotics. Although there has been speculation about antipsychotic treatment intolerance in 22q11.2 microdeletion, 15 the dosage results of the current study go against this as a main driver of TRS and clozapine use. Collectively, the findings suggest that a 60% TRS estimate for 22q11.2 microdeletion may be considered a minimum, and/or that some level of treatment intolerance may be a factor in eventually moving to clozapine. Tolerability of antipsychotics and treatment choices, however, appear likely to vary in a manner similar to that for any patient with schizophrenia. 45
Clozapine in 22q11.2 Microdeletion–Schizophrenia
Results on within-individual response for this 22q11.2 microdeletion cohort suggest the superiority of clozapine over other antipsychotics, consistent with decades of literature on clozapine efficacy. 39 The results were also broadly consistent with a previous report involving 20 of the total 107 individuals studied here, using a different study design, that found comparable clinical improvement but at a lower median dose of clozapine compared to a Canadian community schizophrenia sample without a 22q11.2 microdeletion. 13 In the current study, clozapine dosage was in the lower range of that generally recommended for schizophrenia (200–500 mg). 35 A recent review of 25 articles involving 37 individuals with 22q11.2 microdeletion, most with psychotic illness, reported a mean clozapine dose of 202 mg, 46 further supporting dosing at the lower end of typical ranges.
Notably, the majority of individuals with 22q11.2 microdeletion tried on clozapine remained on clozapine. However, the lifetime median of 5 antipsychotics in those with TRS suggests there are likely issues related to delays and underutilization of this special antipsychotic comparable to those in general clinical practice. 6 This is despite the many known advantages of clozapine treatment, including lower mortality for clozapine-treated individuals. 47 The current study was underpowered to replicate this finding for individuals with a 22q11.2 microdeletion, as indicated by the nonsignificantly lower proportion of individuals who had died in the TRS group than in the no-TRS group (Table 1). Addressing clinician, patient, and caregiver fears and concerns about clozapine is likely to be as important in 22q11.2 microdeletion as for schizophrenia in the general population.47–49 We have written previously about clozapine safety and related risk reduction measures for 22q11.2 microdeletion, including seizure prevention, using vitamin D and prophylactic anticonvulsants (Table 2),2,13 in addition to the strict general monitoring system safeguarding against neutropenia risk. 47
Clinical Considerations for the Treatment of Schizophrenia in Adults With a 22q11.2 Microdeletion.

DSM = The Diagnostic and Statistical Manual of Mental Disorders; CNV = copy number variation; CBC = complete blood count; TSH = thyroid stimulating hormone; PTH = parathyroid hormone; BMI = body mass index; HbA1c = haemoglobin A1c.
Molecular Genetic Subtypes of Schizophrenia and Treatment Resistance
Our finding of elevated prevalence of TRS in 22q11.2 microdeletion is consistent with recent data available on CNVs that convey an increased risk for schizophrenia. 5 One study of patients with TRS reported an increased prevalence of clinically relevant CNVs, including 4 individuals with 22q11.2 microdeletion. 11 Another reported increased prevalence of TRS in individuals with CNVs (n = 15 with typical 22q11.2 microdeletion). 12 A review of case reports involving 8 individuals with a 3q29 microdeletion and schizophrenia suggested possible enrichment of TRS, efficacy of clozapine, and no relation to intellectual disability. 10 Here, we similarly found no relationship between moderate/severe intellectual disability and treatment resistance in 22q11.2 microdeletion. Collectively, these results provide support for the potential utility of clinical genetic diagnostics in schizophrenia, that is, genome-wide microarray for detecting pathogenic CNVs.4,40,50 Clinically detectable schizophrenia-relevant CNVs, including 22q11.2 microdeletion, may be considered to be genetic markers of potential TRS.
Clinical Considerations for Treatment of Schizophrenia in 22q11.2 Microdeletion
The results of the current study appear broadly consistent with clinical practice guidelines for schizophrenia, in general, and for 22q11.2 microdeletion.2,6–8 The findings support the likelihood that most issues that are important for the general population of genetically undiagnosed schizophrenia are also relevant for 22q11.2 microdeletion–schizophrenia. This would include treatment adherence, and underutilization despite known benefits of LAI antipsychotics to reduce the risk of relapse and hospitalization. 51 A recent Canadian study estimated the use of LAIs at 6.5%, 52 and our results showed 13.6% prevalence in 22q11.2 microdeletion (Figure 1).
For individuals with a 22q11.2 microdeletion, as for any patient with schizophrenia, there are no perfect choices in regard to antipsychotics, with all choices requiring consideration of the balance between benefits and risks. The benefits of antipsychotics for the treatment of schizophrenia are clear.6–8,39 The risks – and feasible management of these risks – are also clear for antipsychotics, almost all now in long-term use, including 35 years of experience with clozapine. 39
Table 2 outlines clinical considerations for managing schizophrenia in the context of a 22q11.2 microdeletion. Importantly, 22q11.2 microdeletion-associated conditions are responsive to standard treatment strategies for each, including schizophrenia, thus there are longstanding recommendations to follow routine clinical practice guidelines for individual conditions.1,2,6–8,53–55 The main caveat is that in all cases attention be paid to the multisystem nature of 22q11.2 microdeletion-related comorbidities.2,17,20,53 There is also a general emphasis on the importance of family or other caregivers in obtaining a history for diagnosis, monitoring treatment response, facilitating medication management, and ensuring appointment attendance. 2
The 22q11.2 microdeletion conveys increased risks for multiple conditions, whether or not antipsychotics are used, and perhaps regardless of the specific antipsychotic. 2 Notably, the onset of Parkinson's disease, obesity, hypertriglyceridemia, and type 2 diabetes is on average several decades earlier in 22q11.2 microdeletion than in the general population. For some individuals, and particularly for metabolic conditions, onset may precede, coincide with, or be detected soon after the onset of schizophrenia (Table 2).2,56–60 Thus, it will be important to consider the propensity to, and/or presence of, pre- and post-treatment neurologic and cardiometabolic risks, and to monitor for and mitigate these risks (Table 2).2,59,60 Looking ahead, new strategies such as concomitant metformin with antipsychotic treatment may be particularly relevant for individuals with a 22q11.2 microdeletion. 17
Diagnostic Considerations Relevant to Schizophrenia and the 22q11.2 Microdeletion
In the current study, clinical genetic testing for the 22q11.2 microdeletion occurred before the onset of schizophrenia in a significantly greater proportion of individuals in the no-TRS group than in those with TRS (Table 1, Supplemental Table S2). This suggests the possibility that earlier genetic diagnosis of 22q11.2 microdeletion can help facilitate anticipatory care and thus more timely diagnosis and effective management of schizophrenia. 2 Potential clinical relevance includes reducing the duration of untreated psychotic illness, and reducing the time to clozapine, given the evidence of increased TRS.
In the general population, poorer outcomes of schizophrenia are associated with a longer duration of untreated psychotic illness, 61 and longer time to clozapine prescription,48,49 however, there remain ongoing challenges with addressing these.47–49,61 Similarly, in 22q11.2 microdeletion, and despite the >20-fold increased risk for schizophrenia, misattribution of symptoms to intellectual/learning disabilities, challenges in obtaining a longitudinal history, and/or reluctance to make a diagnosis of schizophrenia or TRS, may cause diagnostic delays and/or confusion about optimal management. 2 These will be important issues to address in clinical care and to assess in a prospective study.
For 22q11.2 microdeletion, a genetic diagnosis can also facilitate the identification of treatable comorbidities, including those directly applicable to psychiatric care. 2 A relevant example related to antipsychotics is the opportunity to forestall the misattribution of Parkinson's disease symptoms to extrapyramidal side effects, and thus prevent the serious consequences of diagnostic and treatment delays (Table 2).59,60
Notwithstanding these advantages, there remain challenges with implementing genetic diagnostics in psychiatry. Many individuals who meet the criteria, especially adults, have not had standard clinical genetic testing to identify the 22q11.2 microdeletion. 50 Barriers to implementation vary from location to location but lack of knowledge, training and practice in ordering genetic tests, and associated fears and concerns (in contrast to other more commonly used tests such as neuroimaging) are common in many areas of medicine. 50
Strengths and Limitations
To our knowledge, this is the largest report of treatment of schizophrenia in individuals with 22q11.2 microdeletion. Strengths include a well-characterized cohort with molecularly confirmed typical 22q11.2 microdeletion and application of standard diagnostic criteria for schizophrenia. The duration of illness excluded early phase psychotic illness, serving to further reduce diagnostic uncertainty. Observational data allowed determination of real-world treatments, and assessment of treatment resistance used a conservative definition, as recommended for retrospective studies. 9 Limitations mainly involve the retrospective and naturalistic nature of the study, involving a period of 25+ years, multiple clinicians, and variable availability and quality of records documenting lifelong antipsychotic treatment. These limitations precluded obtaining consistent data on adherence, detailed response, relative timing/order and rationale for all trials of treatment or their discontinuation. Also, the community-based schizophrenia sample used for the TRS comparison had no genetic test data available. 30 Some individuals could have had 22q11.2 microdeletion or another CNV with increased TRS propensity, thereby tending to diminish between-sample differences, but it is unlikely this would have substantively changed the results. There may have been unknown effects of ascertainment of the 22q11.2 microdeletion cohort. Nearly half of the individuals in this schizophrenia cohort were referred from psychiatric compared to other sources, for example, paediatrics/genetics. However, this variable showed no TRS-related between-group differences (Table 1). Although the cohort was ascertained through a specialty clinic for adults with 22q11.2 microdeletion, nearly two-thirds (63.7%) of the individuals studied had onset of schizophrenia, and thus antipsychotic treatment, before molecular diagnosis. Management could thus only be affected by adult specialty clinic services after that time, and after referral, as for all participants. Further research would be needed to assess the effects of specialty services on results.
Conclusions and Future Directions
The main contribution of this study is long-awaited evidence of a connection between genetic variation, TRS, and clinical intervention in schizophrenia. The results establish an increased propensity for treatment resistance for 22q11.2 microdeletion, adding to initial studies suggesting a relationship between TRS and clinically relevant CNVs.11–13 The findings also confirm antipsychotic dosage in the usual range with the possible exception of clozapine where somewhat lower doses are used. Individuals with 22q11.2 microdeletion are likely to be hidden within schizophrenia populations, 4 particularly in the large TRS subgroup. A genetic diagnosis may facilitate psychiatric management.2,40 Future studies are warranted to prospectively assess antipsychotic treatment, dosage, response, side effects and adherence to treatment in those with schizophrenia and a 22q11.2 microdeletion – a population likely to be excluded from most randomized control trials of schizophrenia, and research in general, given their medical and developmental comorbidities.4,62,63 22q11.2 microdeletion, enhanced by the many animal and cellular models available,1,2 is increasingly regarded as a valuable genetic model for schizophrenia pathogenesis.38,41,43,44 Results of this study indicate that this model may also help shed light on the pathophysiology of antipsychotic response and thus have utility for novel drug development and treatment modalities in an area of great need for many people with schizophrenia.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437241293983 - Supplemental material for Real-World Treatment of Schizophrenia in Adults With a 22q11.2 Microdeletion: Traitement dans le monde réel de la schizophrénie chez des adultes atteints du syndrome de microdélétion 22q11.2
Supplemental material, sj-docx-1-cpa-10.1177_07067437241293983 for Real-World Treatment of Schizophrenia in Adults With a 22q11.2 Microdeletion: Traitement dans le monde réel de la schizophrénie chez des adultes atteints du syndrome de microdélétion 22q11.2 by Lily Van, Tracy Heung, Nikolai Gil D. Reyes, Erik Boot, Eva W. C. Chow, Maria Corral and Anne S. Bassett in The Canadian Journal of Psychiatry
Footnotes
Acknowledgements
The authors thank the patients and their families for their participation, colleagues for referring patients, and research assistants, administrative staff, and students who assisted in the collection of data for the study over many years.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Canadian Institutes of Health Research (CIHR) [MOP-313331 and MOP-111238 to A.S.B.], and Dalglish Chair in 22q11.2 Deletion Syndrome at the University of Toronto and University Health Network (A.S.B.). The Dalglish Family 22q Clinic was founded by a grant from the W. Garfield Weston Foundation. The funding sources had no role in the study design, in the collection, analysis, and interpretation of data, or in the writing and submission of the report.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
