Abstract
Objective:
It has been proposed that different stages of the bipolar disorder might have distinct neurobiological changes. However, the evidence for this has not been consistent, as the studies in early stages of the illness are limited by small sample sizes. The purpose of this study was to investigate the gray matter volume changes in bipolar patients who recently recovered from their first episode of mania (FEM).
Methods:
Using a whole-brain voxel-based analysis, we compared the regional gray matter volumes of 61 bipolar patients who have recovered from their FEM in the past 3 months with 43 age- and gender-matched healthy participants. We also performed a series of subgroup analyses to determine the effects of hospitalization during the FEM, history of depressive episodes, and exposure to lithium.
Results:
No statistically significant difference was found between gray matter volumes of FEM patients and healthy participants, even at a more liberal threshold (P < 0.001, uncorrected for multiple comparisons). Voxel-based subgroup analyses did not reveal significant gray matter differences except for a trend toward decreased gray matter volume in left lateral occipital cortex (P < 0.001, uncorrected) in patients with a previous history of depression.
Conclusion:
This study represents the largest structural neuroimaging investigation of FEM published to date. Early stage of bipolar disorder was not found to be associated with significant gray matter volume changes. Our findings suggest that there might be a window of opportunity for early intervention strategies to prevent or delay neuroprogression in bipolar disorder.
Introduction
Bipolar disorder is a chronic and debilitating psychiatric condition characterized by recurring episodes of depression and mania. Despite decades of research, the neurobiological basis of this illness remains poorly understood. Recent advances in neuroimaging techniques have promised a noninvasive route to examine neuroanatomical correlates of psychiatric conditions. Existing neuroimaging studies of bipolar disorder have identified regional gray matter volume reductions mainly in the prefrontal-limbic network, a brain circuitry that plays an essential role in emotion processing and regulation. 1 However, most of these studies are limited by the use of heterogeneous samples which included patients at different stages of illness with confounding effects associated with illness duration, numbers of episodes, and prolonged exposure to medications. As such, it is unclear whether the reported morphological changes exist at the onset of the illness or develop over time as the illness progresses to more advanced stages. A recently proposed clinical staging model of bipolar disorder conceptualized bipolar disorder as a progressive condition that advances through multiple stages from a prodromal-subclinical phase to more severe and refractory presentations. 2 According to this model, different stages of bipolar disorder may be underpinned by distinct neurobiological substrates. 2,3 This theoretical conceptualization is supported by empirical evidence suggesting that advanced stages of bipolar disorder are associated with more extensive neuroanatomical alterations compared to the earlier stages of the illness. 4,5 All in all, given the clinical and neurobiological heterogeneity of bipolar disorder and potential confounding effects of variables related to the burden of illness, investigation of early stages of bipolar disorder would be necessary for a better understanding of the neurobiological underpinnings of this disorder.
To date, only a handful of studies using small sample sizes have reported on gray matter volume changes in bipolar patients during or shortly after the first episode of mania (FEM), and findings from these studies are conflicting and inconclusive. Although some studies suggested no volumetric difference between FEM patients and healthy participants, 6,7 others indicated regional gray matter volume reductions 8 –10 or increased gray matter volumes. 11,12 In this study, we aimed to investigate regional gray matter volume changes in early stage of bipolar disorder by analyzing data from the largest sample to date of FEM. We used voxel-based morphometry (VBM) which, in contrast to traditional region of interest analysis, provides an unbiased and fully automated voxel-by-voxel method for analyzing the entire brain. 13
Methods
Participants
The participants were identified from the Systematic Treatment Optimisation Programme for Early Mania (STOP-EM), which was a naturalistic, prospective study that examined the clinical outcome, cognitive performance, social functioning, and brain morphology in FEM patients. 14 Patients aged 14 to 35 years (inclusive) who experienced their first manic or mixed episode (FEM) within the previous 3 months were recruited into the study from Vancouver Coastal Health affiliated Hospitals and through referrals from the community physicians and psychiatrists from other parts of British Columbia. Written informed consent was obtained from each participant prior to their inclusion in the study. All participants underwent a comprehensive clinical interview by a board-certified psychiatrist. The diagnosis of bipolar disorder with recent manic or mixed episode was established according to available clinical information and was confirmed using the Mini-International Neuropsychiatric Interview 15 using the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision criteria. Patients with a primary diagnosis of bipolar I disorder with or without other comorbidities were included in the study. Those with a previous history of undiagnosed or untreated manic symptoms and those who met the diagnostic criteria for schizophrenia or schizoaffective disorder were excluded. Further, patients were excluded where there was diagnostic uncertainty.
Healthy participants aged 14 to 35 years were recruited from lower mainland of British Columbia using advertisements posted online and on public notice boards at the University of British Columbia, recreation centers, coffee shops, and grocery stores. Eligibility was confirmed based on the absence of mood, psychotic, anxiety, or substance diagnoses according to the Mini-International Neuropsychiatric Interview version 5.0. A detailed family history was also taken to ensure the absence of family history of mood or psychotic disorders.
From July 2004 to March 2016, 114 FEM patients and 68 healthy participants were enrolled in STOP-EM. A total of 68 patients and 47 age- and gender-matched healthy participants underwent a brain magnetic resonance imaging (MRI) scan at baseline.
Assessments
Clinical and sociodemographic variables were assessed using the following clinical rating scales: The Young Mania Rating Scale, 16 Montgomery–Åsberg Depression Rating Scale, 17 Brief Psychiatric Rating Scale, 18 and Positive and Negative Syndrome Scale. 19 These rating scales were completed at enrollment when patients were in remission from their FEM. All patients received psychiatric treatment according to the Canadian Network for Mood and Anxiety Treatments clinical practice guidelines for bipolar disorder. 20 –23
Data Acquisition of MRI
A Philips Achieva 3.0 Tesla scanner (Amsterdam, the Netherlands) was used to acquire high resolution T1-weighted axial magnetic resonance images using the imaging parameters as specified here: field of view = 20 × 25.6 cm, acquisition matrix = 256 × 256, isotropic voxels (1 × 1 × 1 mm3), autoshim, repetition time/echo time = autoset shortest, T/R head coil, flip angle = 8°, SENSE = 0, and 180 slices (1-mm thick contiguous) of the whole brain.
Preprocessing
Data of MRI were preprocessed and analyzed using FSL v6.0 (Analysis Group, FMRIB Software Library, Oxford, United Kingdom). Raw par/rec image data were converted to NIFTI format by using dcm2nii tool. Bias correction was done using MNIC N3 too. 24 Skull-stripping was done by in-house scripts (SPM8 new segment followed by morphological operations). Brain mask was edited manually. In-skull volume was used as total intracranial volume (TICV). Bias-corrected T1 scan and its skull-stripped brain volume were sent to FSL-VBM v1.1 pipeline 25 from FSL tools. 26 Briefly, a study-specific symmetric gray matter template was created by using matching subjects from each group. All gray matter images from FSL FAST (FMRIB's Automated Segmentation Tool) tissue segmentation were registered to the study-specific template by using FMRIB’s nonlinear registration tool 27 and were modulated for the contraction and enlargement due to the nonlinear transformation. The modulated gray matter images were smoothed with a Gaussian kernel with a sigma of 3 mm. Smoothed gray matter images were then used in the VBM analyses.
Statistical Analysis
Sociodemographic and clinical characteristics of the sample were reported using means and standard deviations for continuous variables and percentages for categorical variables. Independent sample t tests and chi-square tests were used to compare FEM patients and healthy participants for continuous and categorical variables, respectively. The threshold for statistical significance was set at P < 0.05 for all demographic and clinical comparisons. The statistical analyses were performed using R software (https://www.R-project.org/).
For the VBM analyses, we applied voxel-wise general linear model 28 on smoothed gray matter images using permutation-based nonparametric testing with Threshold-Free Cluster Enhancement thresholding option 29 and family-wise error rate (FWE) multiple comparison correction. First, to identify regional gray matter volume changes in early stage of bipolar disorder, we compared all FEM patients with healthy participants. We then performed an additional comparison between patients and healthy participants by including only those patients who required hospitalization during their FEM. Finally, in order to explore the impact of previous depressive episodes and exposure to lithium treatment on gray matter volumes, we performed a series of subgroup analyses by comparing regional gray matter volumes of healthy participants and FEM patients with and without previous depressive episodes and with and without lithium treatment. All VBM analyses were controlled for age, gender, and TICV. The significance level for VBM analyses was set at P < 0.05 after FWE correction. To avoid type II error, we also applied a more liberal threshold of P < 0.001 (uncorrected; voxels >40).
Results
Of the 68 patients who underwent brain MRI, 7 were excluded from analysis for the following reasons: poor scan quality, a brain lesion, and inability to come for a clinical assessment before or after the scan. Of the 47 age- and gender-matched healthy participants, 4 were excluded due to poor image quality.
Sociodemographic and Clinical Features
Demographic and clinical information are presented in Table 1. The FEM group and healthy participants were comparable in age, gender, ethnicity, educational, and marital status. They differed significantly from each other in terms of employment status (P = 0.001). Of the 61 FEM participants, 54 (88.5%) had been hospitalized during their FEM, 26 (42.6%) had been on lithium, and 33 (54.1%) had a past episode of depression.
Sociodemographic and Clinical Information.
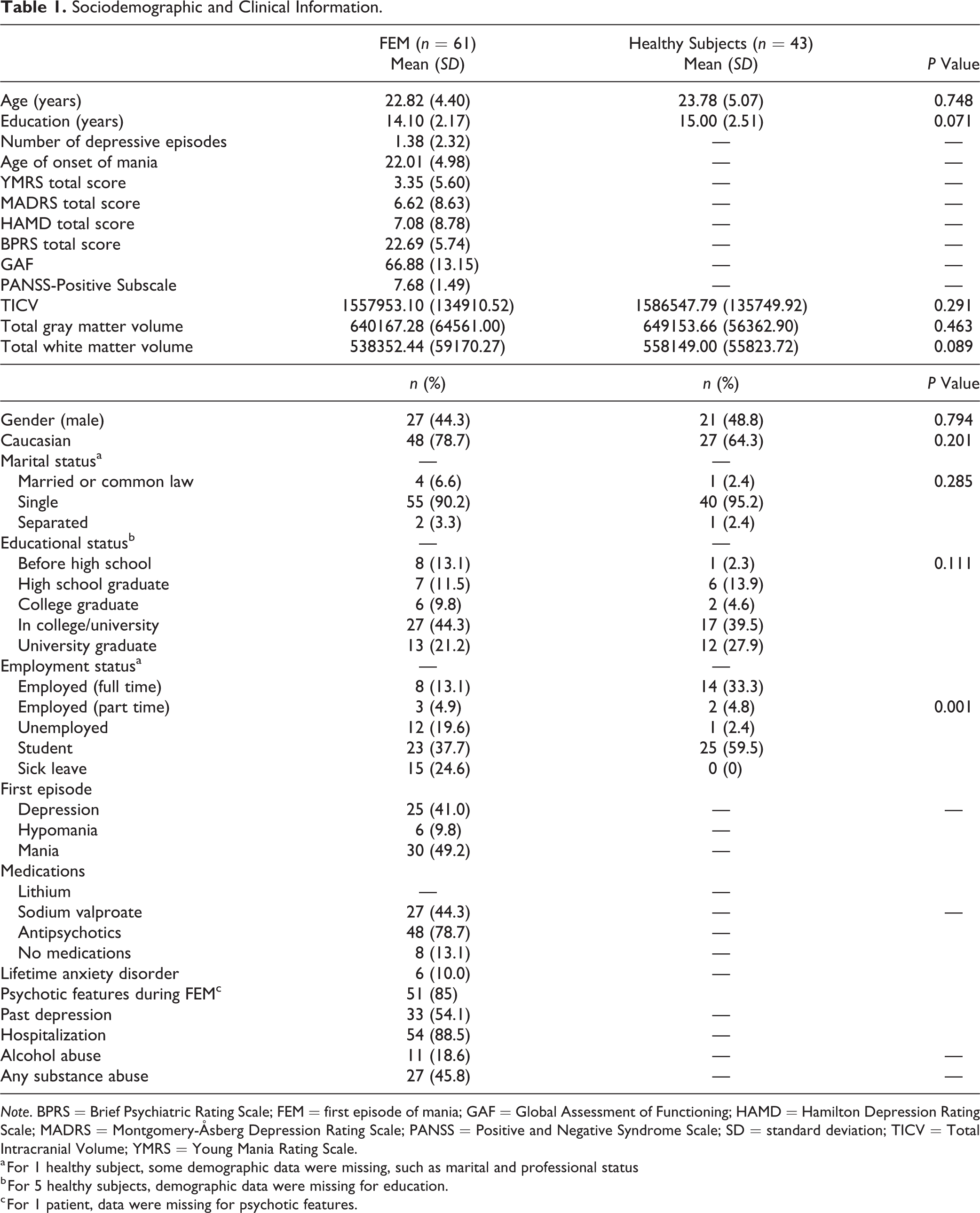
Note. BPRS = Brief Psychiatric Rating Scale; FEM = first episode of mania; GAF = Global Assessment of Functioning; HAMD = Hamilton Depression Rating Scale; MADRS = Montgomery-Åsberg Depression Rating Scale; PANSS = Positive and Negative Syndrome Scale; SD = standard deviation; TICV = Total Intracranial Volume; YMRS = Young Mania Rating Scale.
a For 1 healthy subject, some demographic data were missing, such as marital and professional status
b For 5 healthy subjects, demographic data were missing for education.
c For 1 patient, data were missing for psychotic features.
Voxel-Based Analysis
Pooled FEM versus healthy participants
Patients did not differ from healthy participants with regards to TICV (P = 0.291), total gray matter volume (P = 0.463), or total white matter volume (P = 0.089). The results of VBM analysis showed that FEM patients did not differ significantly from healthy participants in terms of regional gray matter volume, even at a more lenient statistical threshold of P < .001, uncorrected.
Subgroup analysis
No significant gray matter volume difference was found between hospitalized FEM patients (n = 54) and healthy participants. In comparison with healthy participants, FEM patients with previous depression (n = 33) had a trend toward decreased gray matter volume in a cluster (extent 93 voxels) comprising of left lateral occipital cortex (P < 0.001, uncorrected) as shown in Table 2. However, this difference ceased to be significant after correction for multiple comparisons. No significant gray matter volume differences were found between FEM patients with no previous depressive episodes (n = 28) and healthy participants. Similarly, no differences in gray matter volume was detected between patients who were treated with lithium (n = 26) and those without lithium treatment (n = 35) in comparison with healthy participants, even when we applied a more lenient statistical threshold of P < 0.001, uncorrected.
Clusters Showing Significant Between-Group Differences in the Voxel-Based Morphometry Analysis of Gray Matter Volumes.
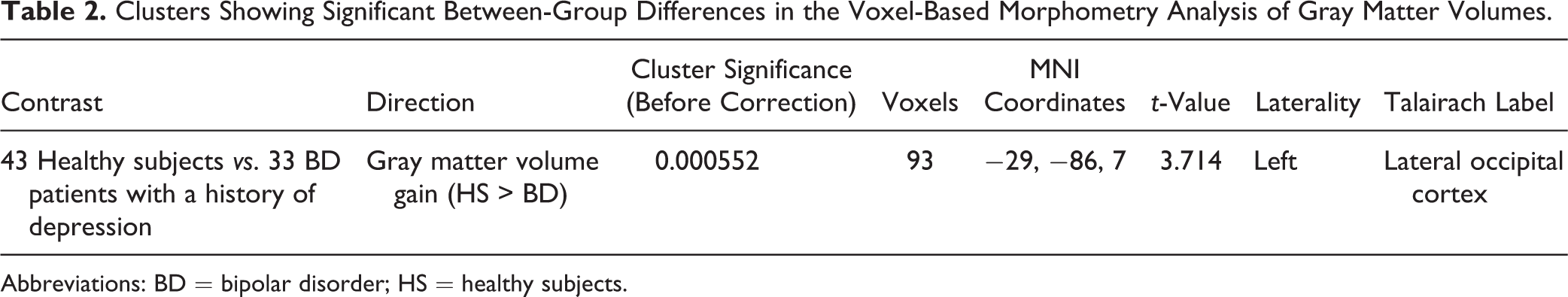
Abbreviations: BD = bipolar disorder; HS = healthy subjects.
Discussion
To our knowledge, this is the largest study of brain structure in FEM to date. We found no gray matter volume changes in patients with bipolar I disorder who had recently recovered from their FEM.
The few VBM studies on FEM reported contradicting results, which might be due to a combination of limiting factors such as small sample sizes, variations in patient populations, and image processing and data analysis. In addition, the relatively small sample sizes (ranging from 8 to 39 patients) in those studies precluded the ability to account for potential confounding factors such as previous depressive episodes and exposure to lithium which has known neuroprotective and neurotrophic effects. 30 Our findings are consistent with a recently published VBM study, 6 which found no significant gray matter volume difference between 31 hospitalized patients with FEM and 31 healthy controls. On the other hand, our results contradict a number of earlier studies that reported gray matter volume changes in patients with FEM. Methodological and population differences are likely to account for the discrepancies in results. For instance, a VBM study of 33 patients with FEM indicated increased gray matter volume in several brain regions including cerebellum, cingulate cortex, and temporal cortex. 12 However, there is a possibility of a type I error since the authors used uncorrected statistical significance threshold. Also, since majority of patients in that study were in a manic or mixed state at the time of scanning, interpretation of the findings can be limited by the mood state. Another first-episode bipolar study by Janssen et al. 9 reported reduced gray matter volume in left medial frontal gyrus; however, patients in this study had childhood-onset mania (7 to 18 years) which may have different neuroanatomical correlates than FEM occurring when the brain is more developed. There is also a higher chance of diagnostic uncertainty and diagnostic shift in that age group. In addition, all patients in that study had psychotic symptoms which would limit the generalizability of the findings. Finally, Berk et al. 10 reported reduced gray matter volume in the orbitofrontal cortex, anterior cingulate cortex, inferior frontal gyrus, and the cerebellum in 26 patients with FEM compared to healthy participants. However, they used a diagnostically heterogeneous sample including bipolar I disorder, substance-induced mood disorder, and schizoaffective disorder.
While our findings suggest that gray matter volume is preserved at the early stage of bipolar disorder, VBM studies of chronic bipolar disorder have consistently reported regional gray matter volume reductions in prefrontal cortex, insula, and anterior cingulate. 31,32 In addition, a recent multisite structural MRI analysis of over 6000 bipolar patients and healthy participants indicated that longer duration of illness was associated with reduced cortical thickness in the occipital, left parietal, and right frontal cortices. 33 It can be argued that gray matter volume loss will occur during the later stages of the illness as a result of disease progression from an acute mood episode toward a chronic multiepisode illness. This view is consistent with the neuroprogression hypothesis of bipolar disorder. 34 According to this hypothesis, bipolar disorder is associated with progressive but potentially modifiable neuropathological alterations in the neural circuitry involved in mood regulation. This putative neuroprogressive process is thought to be accelerated by recurrence of mood episodes. 35 Although not conclusive, several lines of evidence support this hypothesis. For instance, a recent meta-analysis by Hallahan et al. 36 indicated a correlation between cerebral volume reduction and duration of bipolar disorder (see Schneider et al. 37 for a review). A more direct evidence of progressive brain changes was provided by Kozicky et al. 38 who reported gray matter volume reductions in left frontal and bilateral temporal regions in bipolar I patients who had a recurrence of a mood episode in the year following recovery from their FEM but not at baseline relative to healthy participants.
Despite using the largest sample size to date in a VBM analysis of FEM, it is possible that our investigation was not sufficiently powered to detect subtle volumetric differences between patients with first episode mania and healthy participants. To address this issue, we applied a more liberal statistical threshold to ensure we did not inadvertently miss existing gray matter volume differences between patients and healthy participants. Moreover, to account for the potential impact of clinical heterogeneity on the brain volume results, we performed a subgroup analysis by only including patients who were recruited from inpatient settings. The results remained unchanged following those adjustments.
Consistent with previous studies, more than half of our FEM sample had a previous history of depressive episode. Although the diagnosis of bipolar disorder can only be made when an episode of mood elevation is present, a history of depressive episodes prior to the FEM would create uncertainty regarding the actual onset of illness. To account for this uncertainty, we conducted subgroup VBM analyses on patients with and without previous depressive episodes. No significant volume differences were found between these subcategories of patients and healthy participants, suggesting that our findings are robust to the effect of previous depressive episodes.
Exposure to psychotropic medications is one of the major confounding factors in the neuroimaging literature. In bipolar disorder, the use of psychotropic medications in general and lithium in particular has shown to have a normalizing effect on brain structure which can increase the risk of type II errors, 30,39,40 although some authors suggest that the apparent normalizing effect of lithium might be caused by changes in the MRI signal rather than physical gray matter volume increase. 41 Regardless of the mechanism involved, treatment with lithium can complicate the interpretation of MRI data. Naturally, this confounding effect would be far more problematic in neuroimaging studies of chronic patient populations than those in the earlier stages of the illness with a more limited exposure to medications. However, even a short-term exposure to lithium (11 days) has been shown to have meaningful effect on the MRI signal, 41 therefore it is conceivable that the absence of gray matter abnormalities in our FEM sample is partly due to lithium. About 43% of patients in our study were being treated with lithium; excluding those patients from the VBM analysis yielded identical results suggesting that the lack of gray matter volume changes in FEM was not merely the artefact of treatment with lithium.
Consistent with other studies of bipolar disorder, most patients in our study experienced psychotic symptoms during their first manic episode. Neuroimaging studies of first-episode psychosis have consistently reported gray matter deficits in multiple brain regions compared to healthy participants and high-risk individuals. 42 However, it is not entirely clear the extent to which these structural changes represent the potential neurotoxic effects of psychosis or the underlying condition such as schizophrenia or bipolar disorder. In general, compared to schizophrenia, bipolar disorder has been consistently associated with less extensive areas of gray matter volume loss 43 which would indicate some degree of diagnosis-specific structural brain changes across the spectrum of affective and nonaffective psychoses.
Although it was not the focus of this article, we found decreased total white matter volume in FEM at a trend level (P = 0.089). This finding is consistent with other studies which have reported white matter abnormalities in FEM. 44,45 Future studies using imaging techniques optimized for white matter examination such as diffusion tensor imaging can determine the role of white matter pathology in the early stages of bipolar disorder.
The results from our study should be interpreted in light of the following limitations. First, almost all our patients were medicated at the time of scanning which may have influenced our results. Investigating medication-naive patients would eliminate the potential impact of medications on brain volumes, although practical and ethical considerations around patients’ safety would limit the feasibility of such investigations. Second, although acute mood symptoms at the time of scanning was minimal, not all patients were strictly euthymic at the time of scanning and therefore the potential impact of mood state on gray matter volumes cannot be entirely excluded. Third, our sample includes patients with a history of substance use disorders and anxiety disorders which could have affected our results. Finally, previous depressive episodes were determined retrospectively and thus subject to recall bias. Longitudinal studies of patients at risk for bipolar disorder with a first depressive episode would provide more accurate information about the onset of bipolar disorder.
Notwithstanding these limitations, our study provides new empirical evidence in support of preservation of gray matter volume in early stage of bipolar disorder. Although we did not directly explore the impact of early intervention on brain volumes in this study, our findings indicate a potential window of opportunity for early intervention to halt or delay gray matter volume loss which has been frequently reported in the later stages of the disorder. This perspective is consistent with findings from our preliminary analysis of longitudinal neuroimaging data indicating that bipolar patients who remained well during the first year after their FEM did not show any significant gray matter volume changes, whereas those who experienced a mood episode recurrences had gray matter volume reductions in frontal and temporal regions. 38
Conclusions
This study represents the largest structural neuroimaging investigation of FEM published to date. Contrary to studies of chronic bipolar disorder, findings from our investigation indicated that early stage of bipolar disorder may not be associated with significant gray matter volume changes. These results are consistent with a recently proposed concept of neuroprogression in bipolar disorder. Moreover, histories of previous depressive episodes and treatment with lithium were not associated with gray matter volume changes.
Although the clinical significance of regional gray volume changes in bipolar disorder is not entirely understood, a few studies suggested a correlation between gray matter volume and response to treatment. 40,46 In addition, the extent of gray matter volume reduction in bipolar disorder is more limited compared to schizophrenia which tends to have a less favorable clinical outcome. 43 Preservation of gray matter volume in the early stage of bipolar disorder therefore highlights the importance of early intervention strategies to prevent or delay neuroprogression in this population.
Footnotes
Acknowledgments
GS would like to acknowledge the Institute of Mental Health’s Marshall fellowship.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LNY been on speaker/advisory boards for or has received research grants from Alkermes, AstraZeneca, Bristol Myers Squibb, CANMAT, CIHR, DSP, Eli Lilly, GlaxoSmithKline, Janssen, the Michael Smith Foundation for Health Research, Pfizer, Servier, Sunovion, and the Stanley Foundation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The data for this study were obtained from the Systematic Treatment Optimisation Programme for Early Mania (STOP-EM) which was supported by unrestricted grant funding from Astrazeneca Canada (DC-990-205). Astrazeneca was not involved in conception, design, or analysis of data or decision to submit findings for publication.
