Abstract
Objective:
To update and extend our previous systematic review on first- (FGAs) and second-generation antipsychotics (SGAs) for treatment of psychiatric and behavioral conditions in children, adolescents, and young adults (aged ≤24 years). This article focuses on the evidence for harms.
Method:
We searched (to April 2016) 8 databases, gray literature, trial registries, Food and Drug Administration reports, and reference lists. Two reviewers conducted study screening and selection independently, with consensus for selection. One reviewer extracted and another verified all data; 2 reviewers independently assessed risk of bias. We conducted meta-analyses when appropriate and network meta-analysis across conditions for changes in body composition. Two reviewers reached consensus for ratings on the strength of evidence for prespecified outcomes.
Results:
A total of 135 studies (95 trials and 40 observational) were included, and 126 reported on harms. FGAs caused slightly less weight gain and more extrapyramidal symptoms than SGAs. SGAs as a class caused adverse effects, including weight gain, high triglyceride levels, extrapyramidal symptoms, sedation, and somnolence. They appeared to increase the risk for high cholesterol levels and type 2 diabetes. Many outcomes for individual drug comparisons were of low or insufficient strength of evidence. Olanzapine caused more short-term gains in weight and body mass index than several other SGAs. The dose of SGAs may not make a difference over the short term for some outcomes.
Conclusions:
Clinicians need to weigh carefully the benefit-to-harm ratio when using antipsychotics, especially when treatment alternatives exist. More evidence is needed on the comparative harms between antipsychotics over the longer term.
In Canada, prescribing of antipsychotics for children, adolescents, and young adults increased dramatically (upwards of 4-fold) between 1996 and 2011, 1 –4 with 12% of prescriptions provided to Canadian children younger than 9 years. 5 In 2013, second-generation antipsychotics (SGAs) constituted 97% of all antipsychotic prescriptions. 6 SGAs were frequently prescribed to augment stimulants. 1,7 Most antipsychotic prescriptions for children are off-label in Canada and in some cases are being used as first-line treatment despite recommendations for their use in second- or beyond-line therapies. 8
A shift away from first-generation antipsychotics (FGAs) toward SGAs has been driven by perceptions of their more favorable harm profile (e.g., less sedation and fewer extrapyramidal symptoms [EPS] and other adverse effects [AEs] caused by the persistent dopamine receptor blockade by FGAs). However, SGAs have been associated with AEs such as weight gain, elevated prolactin levels, and metabolic syndrome. 9,10 This risk profile is concerning because of known associations between obesity with type 2 diabetes, dyslipidemia, and hypertension. 11 While Canadian guidance exists for monitoring the safety of antipsychotics in children and youth, 12 ongoing systematic review on the efficacy and harms of SGAs and FGAs is needed to best inform treatment decisions. Compilation of evidence on harms across multiple conditions may also help inform situations in which harms data are insufficient for reviews underpinning condition-specific guidance. 13
In this systematic review, we examined the benefits and harms from the use of FGAs and SGAs for various psychiatric and behavioral conditions in children, adolescents, and young adults ≤24 years of age. Here, we report on the results for harms across all conditions; we will report on benefits within conditions in subsequent articles. This is an update and extension of a review commissioned by the Agency for Healthcare Research and Quality (AHRQ) and published in 2012. 14
Methods
Methods for this review are based on the AHRQ Methods Guide 15 and an a priori protocol (available at http://www.crd.york.ac.uk; CRD No.42016032943 and https://effectivehealthcare.ahrq.gov/topics/antipsychotics-children-update/research-2017). Panels of stakeholders, including a patient representative and clinical experts, as well as public commentary informed our methods, and full details on the methods and results are in the full report on AHRQ’s Effective Healtcare website (www.effectivehealthcare.ahrq.gov/reports/final.cfm). 16
Identification and Selection of Studies
A research librarian ran searches to April 2016 for studies published after 1987 in MEDLINE (Suppl. Material Appendix A), EMBASE, PsycINFO, CINAHL, ProQuest® Dissertations and Theses Global, and TOXLINE. We did not just update the previous search conducted in 2011 14 because of our broader scope including additional conditions (e.g., anxiety, substance use disorders) and medications (e.g., molindone). We scanned reference lists of included studies and relevant systematic reviews, searched clinical trial registries, and hand searched relevant journals and Food and Drug Administration (FDA) reports. Studies were included based on predefined eligibility criteria; Table 1 includes all criteria for both benefits and harms. The antipsychotic had to be approved by the FDA but not necessarily for the condition(s) studied in the articles. Additional outcomes were considered (see full report 16 ), although we focus here on the ones considered by our stakeholders to be the most important for decision making. For example, we focused on reports of abnormal serum lipids rather than mean changes in serum levels, which may not reflect a clinically relevant degree of harm. Based on clinical input, we assessed harms across all conditions rather than within each condition because of a lack of strong evidence of a difference in AEs based on indication.
Eligibility Criteria for the Review.

Abbreviations: ADHD, attention-deficit/hyperactivity disorder; ASD, autism spectrum disorders; CCT, controlled clinical trial; DICD, disruptive, impulse-control, or conduct disorders; FDA, Food and Drug Administration; FGA, first-generation antipsychotic; OCD, obsessive-compulsive disorder; PTSD, posttraumatic stress disorder; RCT, randomized controlled trial; SGA, second-generation antipsychotic
a Studies in adult populations were included if they provided data specific to younger ages (mean age + 1 standard deviation ≤24).
b Outcomes considered most important by our stakeholders. Additional outcomes were included, and results are in the full report (www.effectivehealthcare.ahrq.com).
For the database searches, 2 reviewers independently screened the titles and abstracts of all citations, and any deemed relevant by either reviewer based on broad criteria were reviewed in full text in duplicate using a standardized form. One reviewer conducted all other searches outlined in the above section. Disagreements on final inclusion of all studies were resolved through consensus or a third reviewer.
Data Extraction and Analysis
A standardized data extraction form was created and piloted with all reviewers. Thereafter, one reviewer extracted data and another verified data. We reference the primary publication of each study; all associated literature is tabulated in the full report. We recorded intention-to-treat results, and for continuous outcomes, we extracted change scores where possible. If standard deviations were not reported, we computed these. For dichotomous outcomes, we reported counts or proportions and sample size by study arm. We used the longest follow-up duration when data were available for more than 1 time point within each of our follow-up strata (e.g., results for both 1- and 3-month follow-up were within our 0- to <6-month stratum). Only numerical data for harms were extracted; that is, we made no assumptions about lack or presence of an AE if it was not reported. We extracted data (taking care to avoid duplication with other study reports) on harms from trial registries and regulatory agency reports of pediatric trials.
We synthesized data in the following order based on type of comparison: all comparisons combined (for select outcomes, see description of network meta-analysis), aggregate (across drug class) data for FGAs versus SGAs, individual FGAs versus SGAs, within-class comparisons between individual FGAs and individual SGAs (other drug or dose), and then aggregate and individual data for FGAs versus placebo/no treatment and SGAs versus placebo/no treatment. For harms, we combined data from all study designs; empirical evidence has found no difference in estimates of harms between meta-analyses of randomized controlled trial (RCT) and cohort study designs, 17 and cohort studies are commonly recognized as contributing relatively high-quality evidence applicable to real-world settings.
Various approaches to synthesizing the evidence are available, including direct pairwise meta-analysis and methods that combine direct and indirect evidence (i.e., network meta-analysis or mixed-treatment comparisons). 18,19 The summary effect from direct comparisons (e.g., an SGA vs. placebo, one SGA or FGA vs. another SGA or FGA) for one outcome at a similar time point is meaningful. However, such an approach does not allow for comparisons between drugs that may not have much direct evidence (e.g., drug A was compared with drug B and C, but drugs B and C were not directly compared). Where feasible, we conducted network meta-analyses. Detailed methods for our Bayesian pairwise and network meta-analyses are reported in the online Supplemental Material. We considered pooling studies when 3 or more studies reported on the same outcome.
We report mean differences (MD) for continuous outcomes including weight gain; for dichotomous outcomes, we report the relative risk (RR). Variance in meta-analysis results are reported with a 95% credible interval (CrI; used for Bayesian analysis), which can be interpreted similarly to a 95% confidence interval (CI). When the upper or lower limit of the interval crosses 0.0 (for MD) or 1.0 (for RR), the results are not statistically significant.
For prespecified population and intervention subgroups (Table 1), we extracted information on analyses conducted by study authors and also performed our own subgroup analyses based on study-level data. We employed univariate Bayesian meta-regression analyses for our outcomes with the most data (weight, weight gain of 7% or greater, somnolence, incidence of EPS in SGA vs. placebo/no-treatment comparisons) in terms of patient age, sex, antipsychotic treatment history (i.e., % naïve), and treatment duration.
Quality Assessment and Grading the Body of Evidence
Two reviewers independently assessed the risk of bias of all studies based on study design. 15,20,21 We also assessed the strength of the body of evidence, using 5 domains 22 : risk of bias, consistency, directness, precision, and reporting biases. For rare outcomes (i.e., <5%), findings were considered precise when the sample size (across studies) was at least 2000 patients, to estimate that adequate prognostic balance was achieved to detect at least a small difference. 23 Our overall assessments of high, moderate, low, or insufficient strength of evidence (SOE) indicate the degree of certainty we had in the effect estimate. Discrepancies between reviewers were resolved based on consensus. Because of limited guidance from the literature, we did not grade the SOE for our network meta-analysis findings.
Results
We identified 135 studies (57 new studies after the 2012 report 14 ) meeting our review criteria; of these, 126 (93%) reported on harms (Figure 1). 24 –149 Eighty-four (67%) were RCTs, 6 (5%) were controlled clinical trials (experimental studies without randomization), and 36 (28%) were observational studies. Most (about 70%) studies reported only on follow-up durations of less than 6 months. The risk of bias was high for 55% of trials. The main contributor to risk of bias was incomplete outcome data (i.e., <90% of patients included in analysis). Overall, the observational studies were of good quality; of 36 studies, 4 (11%) were rated as having high risk of bias, 12 (33%) as having medium risk of bias, and 20 (56%) as low risk of bias. The Supplemental Material provides a table of study characteristics including findings for risk of bias of each study (Appendix B).
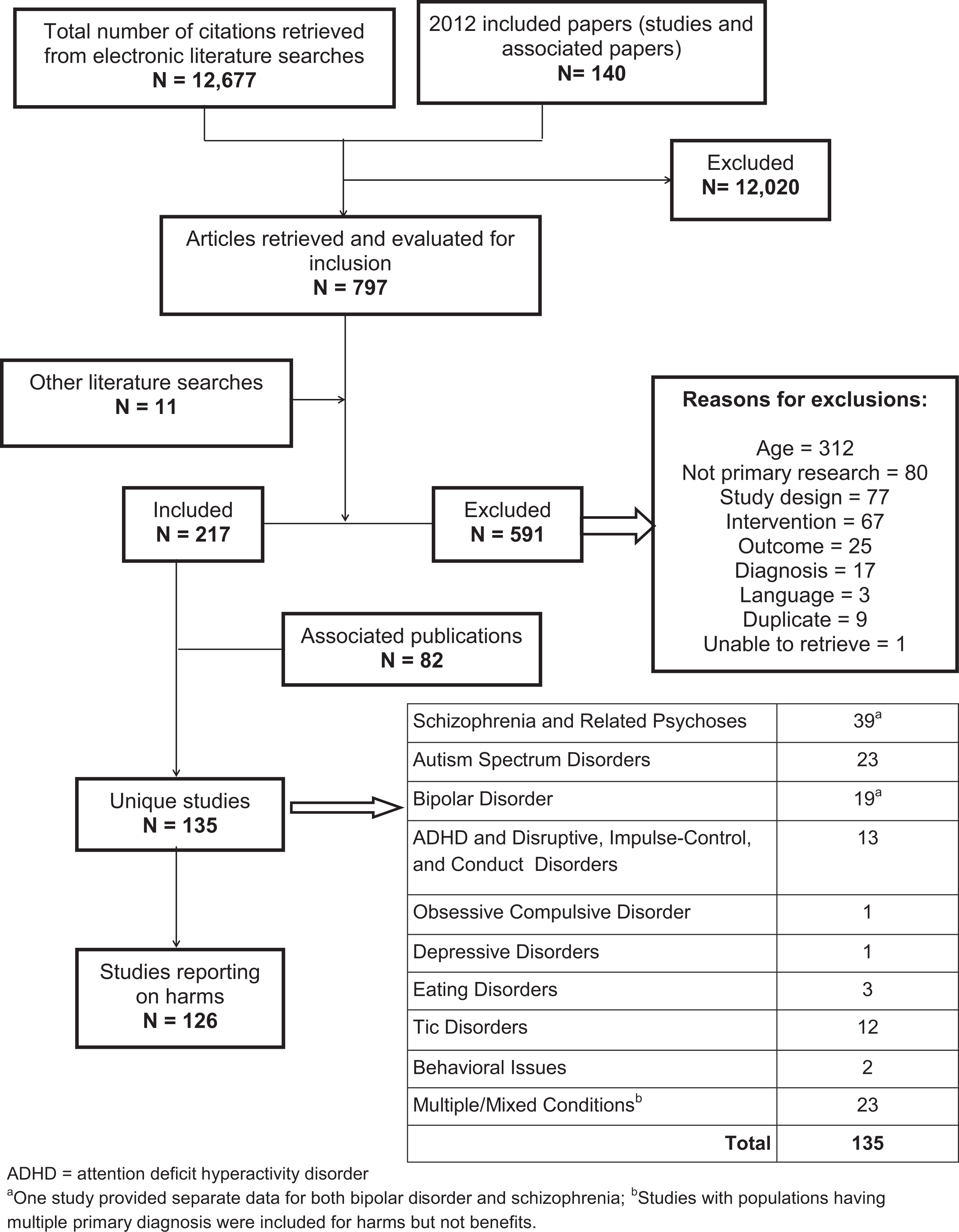
Flow of literature.
We present findings first from the network meta-analyses in which data across any study could be pooled to examine the impact of FGAs and SGAs on increases in weight and body mass index (BMI). Then, head-to-head comparisons between FGAs and SGAs were followed by head-to-head and placebo/no-treatment comparisons for FGAs and then SGAs. Tables 2 and 3 contain details for general AEs having moderate and low SOE, respectively. Results for major AEs are presented within the text only.
Findings with Moderate Strength of Evidencea: General Adverse Effects.
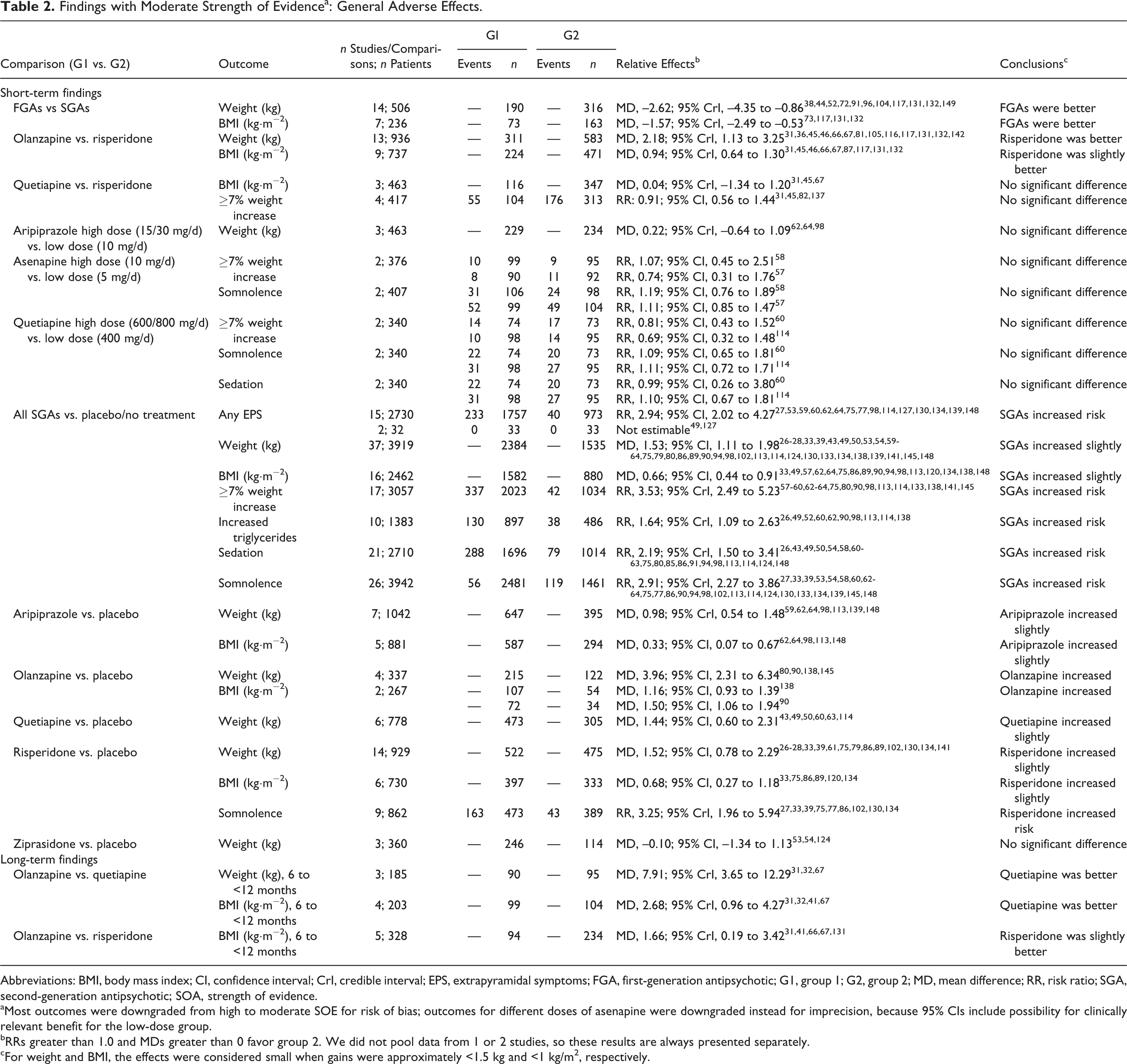
Abbreviations: BMI, body mass index; CI, confidence interval; CrI, credible interval; EPS, extrapyramidal symptoms; FGA, first-generation antipsychotic; G1, group 1; G2, group 2; MD, mean difference; RR, risk ratio; SGA, second-generation antipsychotic; SOA, strength of evidence.
aMost outcomes were downgraded from high to moderate SOE for risk of bias; outcomes for different doses of asenapine were downgraded instead for imprecision, because 95% CIs include possibility for clinically relevant benefit for the low-dose group.
bRRs greater than 1.0 and MDs greater than 0 favor group 2. We did not pool data from 1 or 2 studies, so these results are always presented separately.
cFor weight and BMI, the effects were considered small when gains were approximately <1.5 kg and <1 kg/m2, respectively.
Findings with Low Strength of Evidencea: General Adverse Effects.
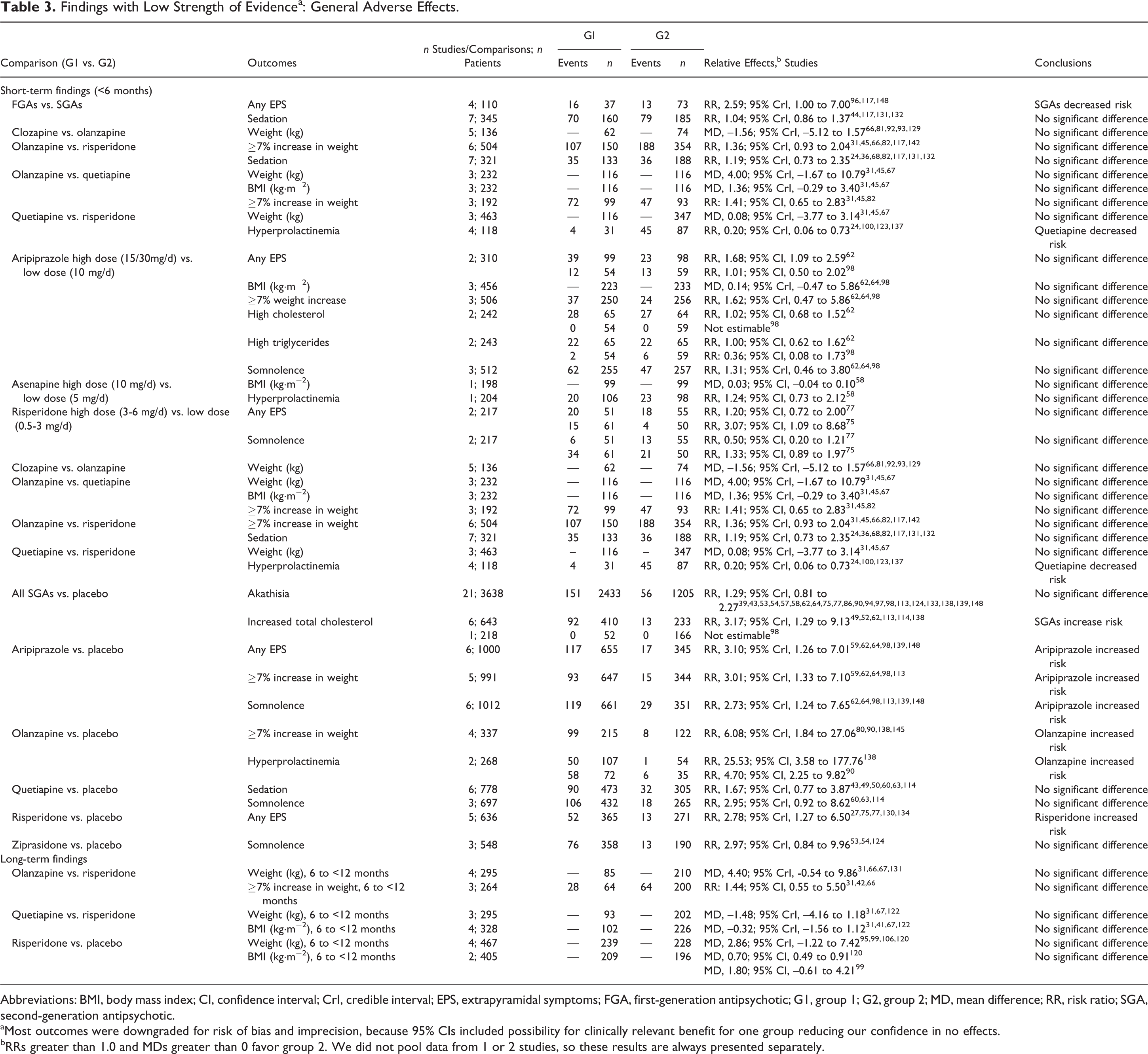
Abbreviations: BMI, body mass index; CI, confidence interval; CrI, credible interval; EPS, extrapyramidal symptoms; FGA, first-generation antipsychotic; G1, group 1; G2, group 2; MD, mean difference; RR, risk ratio; SGA, second-generation antipsychotic.
aMost outcomes were downgraded for risk of bias and imprecision, because 95% CIs included possibility for clinically relevant benefit for one group reducing our confidence in no effects.
bRRs greater than 1.0 and MDs greater than 0 favor group 2. We did not pool data from 1 or 2 studies, so these results are always presented separately.
Network Meta-analyses for Body Composition Outcomes across All Antipsychotics
Network meta-analysis was conducted for the outcomes of weight gain (Figure 2; n = 71 studies) and changes to BMI (Suppl. Figure S1; n = 35 studies). Results are presented in terms of a placebo referent, to rank the drugs based on a common comparator, but data from head-to-head comparisons were incorporated in the analysis. Results showed that patients taking most antipsychotics gain more weight than do patients taking placebo or not receiving antipsychotics. The relative harm from olanzapine (22 studies, n = 611; 4.12 kg, 95% CrI, 3.43 to 4.88) was most robust compared with aripiprazole (11 studies, n = 869; 0.88 kg, 95% CrI, 0.26 to 1.50), quetiapine (12 studies, n = 655; 1.25 kg, 95% CrI, 0.51 to 1.95), and risperidone (37 studies, n = 1535; 1.85 kg, 95% CrI, 1.40 to 2.35), because of the higher precision in these estimates compared with other drugs (e.g., pimozide, molindone, and lurasidone had fewer patients contributing to the findings). For BMI, olanzapine (16 studies, n = 470; 1.51 kg/m−2, 95% CrI, 1.28 to 1.84) remained worse than all other antipsychotics except for, possibly, clozapine (Suppl. Figure S1). We used data for longest treatment duration from each study for these analyses, but 81% (weight) and 71% (BMI) of studies were short term (often 6-12 weeks).
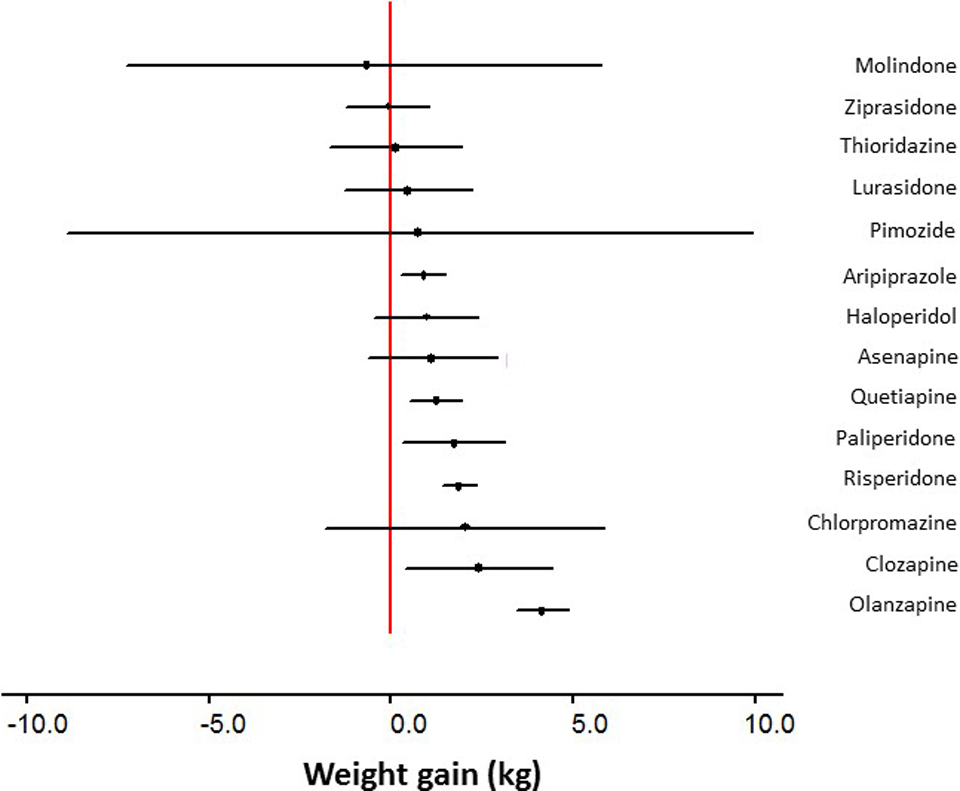
Plot of weight gain by first-generation antipsychotics (FGAs) and second-generation antipsychotics (SGAs) compared with placebo/no treatment This plot shows the findings from network meta-analyses combining placebo-controlled and head-to-head comparisons of FGAs and SGAs within one analysis. The effects shown represent the mean difference and credible intervals of each drug relative to placebo, which was used as the reference standard.
FGAs versus SGAs
Nine studies reported on major (4 long-term duration)* and 16 reported on general AEs (2 long-term) when comparing FGAs with SGAs. All findings on major AEs (i.e., neuroleptic malignant syndrome, seizures, cardiac arrhythmias, agranulocytosis, diabetes mellitus) had insufficient SOE. FGAs caused lower gains in weight and BMI than did SGAs (Table 2). Compared with SGAs, FGAs increased the short-term risk for experiencing any EPS symptom (Table 3). Low SOE found no significant difference between classes for sedation (Table 3). Because of risk of bias and imprecision due to very small samples sizes, evidence was insufficient for other across-class outcomes (e.g., akathisia, dystonia, hyperprolactinemia) and for all outcomes in individual drug comparisons.
FGAs: Comparisons between FGAs and between FGAs and Placebo/No Treatment
There was insufficient SOE for all major and general AEs in comparisons between different FGAs or between different doses of the same FGA and between FGAs (aggregate or individual) compared with placebo or no treatment. Single, very small studies 29,115,118,125,128,136 reported data for a small number of outcomes within the short- and long-term duration categories.
SGAs: Comparisons between SGAs
Sixteen (5 long-term) studies reported on major AEs and 37 (13 long-term) reported on general AEs.
For conditions for which SGAs were not considered primary treatment (i.e., exclusion of patients with schizophrenia, autistic spectrum disorders, tic disorders, and all patients talking clozapine), aripiprazole (hazard ratio [HR] vs. no antipsychotic, 7.72; 95% CI, 3.70 to 16.12) may increase the risk for developing diabetes more than risperidone (HR, 2.20; 95% CI, 1.14 to 4.26) in patients newly initiated and remaining on these drugs for at least a year (low SOE). 37 Data for other major AEs were insufficient, mostly from single studies having inadequate sample sizes for these rare events and moderate or higher risk of bias.
For weight and BMI, findings from pairwise comparisons between individual SGAs were similar to those seen from the network meta-analysis (Figure 1; Tables 2 and 3). There was less precision in some of the findings (due to smaller samples; Table 3), although for comparison with olanzapine in which no statistical significance was found, the limits of the 95% CrIs usually suggested more harm for olanzapine. Findings for clinically meaningful gains in weight (≥7% body weight) were of lower SOE; findings were not statistically significant but imprecise, with the 95% CIs including findings for meaningful harm from olanzapine.
Other findings with low SOE for individual SGA drug comparisons included no significant difference between olanzapine and risperidone for sedation and between quetiapine and risperidone for hyperprolactinemia (Table 3).
Different doses of aripiprazole resulted in no significant difference in short-term weight gain (Table 2). There was low SOE showing no difference between doses of aripiprazole over the short term for any EPS symptoms, BMI, the proportion gaining ≥7% weight, and somnolence (Table 3); the 95% CIs included values favoring the low dose. There was no difference for hypertriglyceridemia or increased total cholesterol (low SOE). There was no significant difference in the short term between low and high doses of asenapine for the proportion of patients gaining ≥7% weight, for risk of somnolence, or for hyperprolactinemia. Low and high doses of quetiapine were not significantly different for risk of gaining ≥7% weight, somnolence, or sedation over the short term (moderate SOE). There was no significant difference between low and high doses of risperidone for any EPS or somnolence (low SOE).
SGAs: Comparisons with Placebo/No Treatment
There was no significant difference in the short term across all SGAs compared with placebo/no antipsychotic for mortality (13 studies, n = 2447; 0 events in either group) † or for having a pathologically prolonged QT interval (14 studies, n = 2425; SGAs 14 of 1490 and placebo/no antipsychotic 9 of 935) ‡ (both moderate SOE). Compared with no antipsychotic treatment, patients newly initiated on SGAs may have an increased risk for developing diabetes over treatment of at least 1 year (low SOE; HR, 2.89; 95% CI, 1.64 to 5.10; 25.3 vs. 7.8 cases per 10,000 person-years follow-up). 37 Moreover, across all of the antipsychotics evaluated in this study (risperidone, olanzapine, aripiprazole, quetiapine, and ziprasidone), the risk remained elevated for up to 1 year following discontinuation of the drug. Other major AEs were assessed as having insufficient SOE due to rare events occurring in samples too small (<2000) to offer adequate power.
There was moderate SOE that SGAs were worse than placebo/no treatment for 7 general AE outcomes: any EPS symptoms, changes to body composition (weight, BMI, and ≥7% weight gain), increased triglycerides, sedation, and somnolence (Figure 3; Table 2). Low SOE found no significant difference for akathisia and that SGAs may increase the risk for hypercholesterolemia (Figure 3; Table 3).
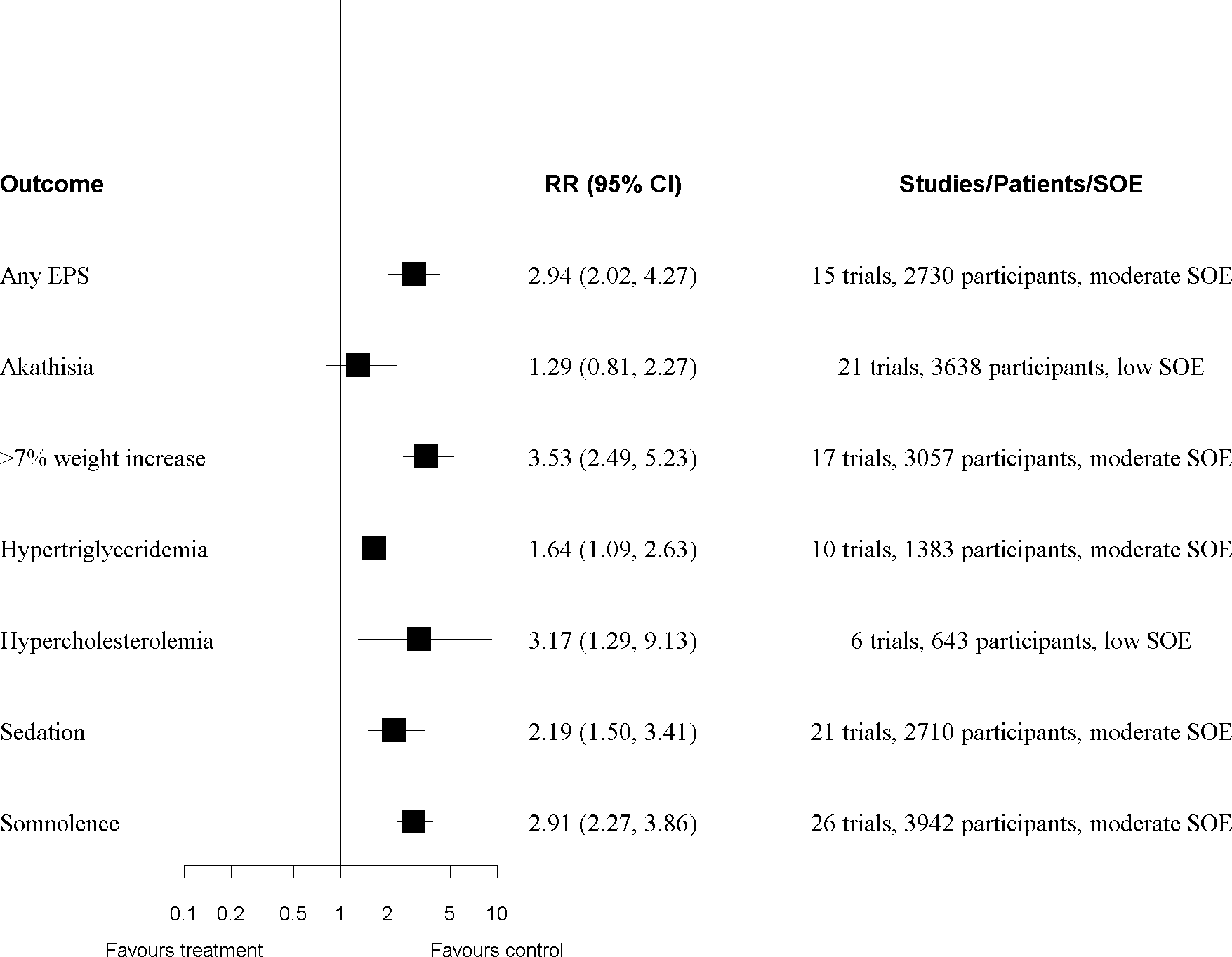
Results of meta-analyses for second-generation antipsychotics (SGAs) versus placebo/no treatment. EPS, extrapyramidal symptoms; RR, relative risk; SOE, strength of evidence (degree of certainty review authors had in the effect size [RR] based on study quality, consistency in effects, precision of findings, other limitations; no outcome had high SOE). Each row is from a different meta-analysis of studies comparing all SGAs versus placebo/no treatment. The number of studies and participants contributing to each meta-analysis is indicated.
For individual SGAs compared with placebo/no treatment, findings for weight and BMI were similar to those found by our network meta-analyses (Table 3). Risperidone and aripiprazole increased the risk for any EPS and for somnolence; findings for somnolence showing no significant difference for quetiapine and ziprasidone were low SOE because the possibility of meaningful harm from the SGA could not be ruled out. Although findings for hyperprolactinemia across all SGAs were insufficient, there was large heterogeneity between the SGAs, and findings specific to olanzapine were of low SOE, indicating this SGA increases the risk.
Subgroup Effects
Twenty-six studies reported on within-study subgroup analyses for harm outcomes (see full report). Findings were often inconsistent. Body composition, fasting glucose, and prolactin elevations do not appear to differ in patients taking SGAs and concomitant psychostimulants. 25,45 Dose of SGAs—particularly when considering cumulative doses—was found in 2 large, high-quality observational studies to increase the risk for metabolic effects (e.g., >10 mg/d olanzapine or >1.5 mg/d risperidone) 45 and development of diabetes (across all SGA doses vs. no antipsychotic: HR, 2.89 [95% CI, 1.674 to 5.10); ≥100-g dose equivalents of all SGAs: HR, 5.61 [95% CI, 2.38 to 13.24]; and ≥100 g risperidone: HR, 14.95 [95% CI, 4.60 to 48.64]). 37 Quetiapine 114 and risperidone 44,75 –77,120 appear to increase serum prolactin more in females than in males. Findings for effect moderation on risk for somnolence and neuromotor effects were mainly from single studies.
Only one subgroup effect was evident based on our meta-regressions using study-level data. Slightly greater weight changes can be expected over longer treatment durations (0.043 kg [95% CrI, 0.015 to 0.072] per week of additional treatment). Results for the study-level variables of mean age, sex (% male), and percentage with previous antipsychotic exposure were not statistically significant for weight or the other outcomes we examined in the regressions (greater than 7% increase in weight, somnolence, and EPS symptoms). Based on observations of the plots of data used for our meta-regressions, we could not see any trends indicating the effects varied by condition (Suppl. Figures S2-S5). The results for conditions for which these drugs are typically used in lower doses (e.g., attention-deficit hyperactivity disorder) than for other conditions (e.g., schizophrenia) appeared to be very similar when looking across studies. These study-level observations should be interpreted with caution.
Discussion
We systematically reviewed the harms of FGAs and SGAs across a wide array of psychiatric conditions in young people. As commonly recognized, FGAs as a class cause less weight gain and more EPS than SGAs. There may be no difference between the classes for sedation, and findings for many other harms were either not reported or of insufficient quality. SGAs have a poorer harms profile than placebo or no antipsychotic treatment for weight gain, somnolence, risk of high triglycerides, EPS, and sedation. Some differences exist between SGAs, with olanzapine being worse than several SGAs for body composition outcomes, although several comparisons had low or insufficient SOE. There is some indication that SGAs may increase the risk for hypercholesterolemia and type 2 diabetes compared with placebo or no antipsychotic treatment. The dose of SGAs may not make a difference over the short term for some outcomes, but there is some indication of worse effects over the longer term.
Overall, our conclusions for some harms are fairly similar to those reported by other review authors. 150 –154 However, our focus on clinically meaningful outcomes (e.g., hypercholesterolemia instead of mean cholesterol) and applying our criteria for SOE (in particular with respect to judgments of precision) limited our certainty about many effects, including between-drug differences about which other authors have made more definitive conclusions (see the full report 16 for details).
Findings are mainly applicable to acute, rather than maintenance, treatment. Patients with a history of various adverse events, including tardive dyskinesia, suicide-related behaviors, neuroleptic malignant syndrome, or abnormal laboratory values, were often excluded from studies. The mean age across the studies was greater than 8 years, and most studies did not include patients older than 18 years; therefore, the evidence is not directly applicable to young children or young adults. The applicability of study findings outside of research settings is also limited, as monitoring practices to ensure treatment adherence and timely dose adjustments based on response and tolerability assessments are less likely to occur. Although comprehensive and individualized monitoring for AEs has been recommended for several years, 155,156 evidence suggests that these practices remain inadequate 157 or may be difficult to maintain. 4
Limitations to this review are discussed in the Supplemental Materials. Results of the network meta-analyses should be considered exploratory in nature because of the use (i.e., modeling) of direct and indirect comparisons. Nevertheless, findings were quite consistent with those from the pairwise/direct comparisons described. This report was limited to comparisons between various antipsychotics and between antipsychotics and placebo or no antipsychotic treatment. As such, evidence on the use of other drug classes (e.g., anticonvulsants, mood stabilizers) that are frequently used in the treatment of these patient populations was not considered. We did not assess any outcome as having high SOE, although it should be recognized that attaining high SOE from trials of antipsychotics in children with psychiatric conditions is likely very difficult, and the overall evidence reviewed should not be interpreted as lacking in credibility.
Conclusion
Clinicians need to weigh carefully the benefit-to-harm ratio of antipsychotics when making prescribing decisions, especially in cases in which treatment alternatives exist and are supported by research evidence. More direct evidence on the harms of SGAs compared with FGAs, as well as from longer-term treatment studies, is required to advance clinical care. More evidence on sources of clinical heterogeneity, from gender, age, and other biological and environmental factors that may moderate the effects on harms, would be valuable.
Supplementary Material
779950_Supplementary_material - Harms of Antipsychotics in Children and Young Adults: A Systematic Review Update
779950_Supplementary_material for Harms of Antipsychotics in Children and Young Adults: A Systematic Review Update by Jennifer Pillay, Khrista Boylan, Amanda Newton, Lisa Hartling, Ben Vandermeer, Megan Nuspl, Tara MacGregor, Robin Featherstone, and Normand Carrey in The Canadian Journal of Psychiatry
Footnotes
Acknowledgments
We acknowledge with appreciation the staff at AHRQ, Laura Pincock and Christine Chang, for their efforts in helping us engage with key informants and the technical expert panel for this review; they also provided constructive comments on our draft full technical report (![]() ). In addition, we greatly value the time spent and excellent comments on our full technical report by EPC Associate Editor Marian McDonagh. We also thank the key informants and technical expert panel members (listed in the full report) who provided input into the review. Several technical expert panel members also provided peer review of the draft report submitted to AHRQ and available online. We acknowledge Syed Hamza Ahmed Jafri, BDS, MPH (University of Alberta), for his assistance with study selection, data extraction, and data analysis.
). In addition, we greatly value the time spent and excellent comments on our full technical report by EPC Associate Editor Marian McDonagh. We also thank the key informants and technical expert panel members (listed in the full report) who provided input into the review. Several technical expert panel members also provided peer review of the draft report submitted to AHRQ and available online. We acknowledge Syed Hamza Ahmed Jafri, BDS, MPH (University of Alberta), for his assistance with study selection, data extraction, and data analysis.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors have completed the Unified Competing Interest form at ![]() (available on request from the corresponding author) and declared that they have no potential conflicts of interest with respect to research, authorship, and/or publication of this article.
(available on request from the corresponding author) and declared that they have no potential conflicts of interest with respect to research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded under contract No. HHSA29020150001I from the Agency for Healthcare Research and Quality (AHRQ), U.S. Department of Health and Human Services (HHS). The authors of this article are responsible for its content. Statements in the article do not necessarily represent the official views of or imply endorsement by AHRQ or HHS. Dr. Newton is supported by a New Investigator Award from the Canadian Institutes of Health Research.
Role of the Funding Source
This topic was nominated by the American Academy of Child and Adolescent Psychiatry and selected by AHRQ for systematic review by an EPC. A representative from AHRQ served as a contracting officer’s technical representative and provided technical assistance during the conduct of the full evidence report and comments on draft versions of the full evidence report. AHRQ did not directly participate in the literature search, determination of study eligibility criteria, data analysis or interpretation, or preparation, review, or approval of the manuscript for publication.
Systematic Review Registration
PROSPERO No. 42016032943
Supplemental Material
Supplemental material for this article is available online.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
