Abstract
Objective:
To describe the extent of increase in use and the rate of continuation versus discontinuation of psychotropic agents before, during, and after pregnancy.
Methods:
Rates of psychotropic use (antidepressants, anxiolytic/sedative-hypnotics, antiepileptics, antipsychotics, lithium, stimulants) among women with a hospital-recorded pregnancy outcome were assessed using databases at the Manitoba Centre for Health Policy. Rate of use was defined as ≥1 prescription over the total number of pregnancies in the 3-12 months before pregnancy, 0-3 months before pregnancy, during pregnancy, or 3 months after pregnancy. Continued use was defined as ≥2 prescriptions with gap ≤14 days. Poisson regression was used to analyze trends.
Results:
Over the study period, a psychotropic drug was used before, during, or after pregnancy in 41,923 of 224,762 pregnancies. From 2001 to 2013, psychotropic use increased 1.5-fold from 11.1% to 16.2% (p < 0.0001) in the 3-12 months before pregnancy, 1.6-fold from 6.4% to 10.5% (p < 0.0001) in the 3 months before pregnancy, 1.8-fold from 3.3% to 6.0% (p < 0.0001) during pregnancy, and 1.5-fold from 6.2% to 9.5% (p < 0.0001) in the 3 months postpartum. Among the 13,579 women who received at least 1 psychotropic agent in the 3 months prior to pregnancy, 38.5% stopped the agent prior to pregnancy and only 10.3% continued use throughout pregnancy. Continued use throughout pregnancy was higher (56.9%) among the 6693 women who received at least 2 prescriptions for a psychotropic agent and were at least 80% adherent in the 3 months prior to pregnancy.
Conclusion:
The use of psychotropic agents increased over 12 years. The safety of continuing versus discontinuing these agents during pregnancy remains uncertain, but we observed a decrease in psychotropic drug use during the pregnancy period.
As many as 10% of women of childbearing age experience mental illness and may be exposed to a psychotropic medication prior to becoming pregnant. 1 –4 However, the relative risk of continuing versus discontinuing drug therapy during pregnancy on maternal and child health outcomes remains uncertain. 5 Although not treating mental illness in pregnant women may pose a higher risk of maternal relapse, poor obstetric care, and pregnancy complications, 6 the perceived risk of psychotropic drug use during pregnancy may contribute to nonadherence and discontinuation of these agents. 5,7
Clinicians are provided with limited guidance on how to best manage patients who are stable on medication during the perinatal period, and this decision is controversial and challenging for many clinicians. It is therefore important to study the pattern of psychotropic exposure in order to address the clinical implications associated with the use of these drugs. To date, only a few observational studies have examined the impact of continuing versus discontinuing psychotropic agents during pregnancy, and findings on the risk of continuing drug therapy during pregnancy are conflicting. 8 –11 Surveys have reported that up to 60% of women had poor adherence to chronic medication therapy during pregnancy. 5,7 Previous studies have examined the extent of psychotropic medication use and discontinuation during pregnancy 11 –24 ; however, few studies have used large administrative databases that distinguished between regular and intermittent users of these agents, and even fewer studies examined these trends in a Canadian population. We also have limited knowledge about the intentional use of certain psychotropic classes, such as stimulants, anxiolytics, and mood stabilizers, from a population perspective. Manitoba has one of the most comprehensive databases with which to study psychotropic drug use from a population perspective. We aimed to describe the extent of increase in use of select psychotropic agents before, during, and after pregnancy and to examine the rate of continued and discontinued use of these agents during pregnancy between 2001 and 2013.
Methods
Data Sources
This was a retrospective, population-based, cohort study to examine the frequency of psychotropic prescriptions dispensed before, during, and after pregnancy between April 1, 2001, and March 31, 2013. Data were obtained from the administrative database of the Population Health Research Data Repository located at the Manitoba Centre for Health Policy (MCHP), which includes administrative data for 1.2 million individuals living in Manitoba.
The Drug Program Information Network (DPIN) database was used to provide the outpatient prescription drug use of individuals living in Manitoba. DPIN captures the outpatient prescription dispensing record (this includes the name and strength of the medication, date of dispensing, days of supply, and quantity dispensed) from community pharmacies of all Manitobans regardless of drug coverage. Diagnoses were identified by ICD-9-CM or ICD-10-CA equivalent codes from hospitalization (hospital abstracts) files. Prescription data are linked to individuals with specific diagnoses from their contacts with the healthcare system (e.g., hospitalizations, prescriptions dispensed) through scrambled personal health numbers. Emergency data are available from April 1, 1999, to March 31, 2013, and were used to capture emergency visit encounters in Winnipeg for pregnancies that occurred between April 1, 2000, and December 31, 2012, to allow observation of emergency visits in the full year prior to pregnancy and 3 months after pregnancy. The Manitoba Health Insurance Registry provided demographic information at the beginning of each interval. Statistics Canada census data provided the average household income and income quintiles at the beginning of each index interval.
Cohort
Pregnant women were identified based on date of pregnancy outcome (index date) using ICD-9-CM (or corresponding ICD-10-CA) codes for livebirth, stillbirth (ICD-9-CM 656.4 or ICD-10-CA O36.4), and in-province specific hospitalization or healthcare database (ICD-9-CM V27 or ICD-10-CA Z37); terminations, molar, or ectopic abortions; and molar and ectopic pregnancies. Women with more than 1 pregnancy during the study period were included as multiple pregnancy observations. Date of conception was determined by subtracting gestational age at delivery from the date of birth. Out-of-hospital births, midwife care, and nurse practitioner care were not included in the analysis. Only those women registered with Manitoba Health for 365 days preceding the date of conception, through to at least 3 months following the index date (delivery or termination), were included (approximately 6.2% of all pregnancies during the study period were excluded for not meeting this criterion).
Psychotropic Medication Exposure
Psychotropic agents in this study included all available formulations of antidepressants (ATC N06A and N06CA), anxiolytic/sedative-hypnotics (N05B, N05C, and N03AE01), antiepileptic agents (N03A except N03AE01), antipsychotic agents (N05A except N05AN), lithium (N05AN), and stimulant agents (N06B). The annual rate of psychotropic use (among women with a hospital-recorded pregnancy outcome [2001-2013]) was assessed. Rate of use was defined as ≥1 prescription received by the total number of pregnancies in the 3 to 12 months before pregnancy, 3 months before pregnancy, during pregnancy (overall and separated by each trimester of pregnancy), and 3 months after pregnancy.
Among those with a livebirth, stillbirth, or intrauterine death pregnancy outcome, we identified those with at least 2 consecutive prescriptions for a psychotropic agent with a gap of 14 days or less between prescription fills in the 3 months prior to pregnancy and identified patients based on their pattern of continued or discontinued use based on 4 categories: (1) patients who did not receive a subsequent psychotropic medication in the first trimester; (2) patients who did not receive a subsequent psychotropic medication in the second trimester; (3) patients who received a subsequent psychotropic medication throughout their entire pregnancy (no gap of >14 days received in every trimester); and (4) patients who filled a subsequent psychotropic medication intermittently throughout pregnancy (may have gap >14 days). The gap of 14 days between prescription fills that was used to formulate the categories was established by estimating the end-date of each prescription using the supply (number of days) that was dispensed. As a secondary analysis, the average medication possession ratio (MPR) and the proportion of women who received a psychotropic agent with an MPR of 80% or more in the 3 months prior to pregnancy, during pregnancy, and for each category of use (discontinued before first trimester, discontinued before second trimester, continued use, intermittent use) were also determined to assess a change in adherence to medication. MPR was calculated using the days-supply received divided by the number of days in the observation period.
Statistical Analysis
SAS software for Windows, version 9.3, was used for all analyses. Summary statistics (mean and standard deviation for normally distributed data; median and interquartile range for skewed data) were used to describe prescription data. Poisson regression using a generalized estimating equation to account for repeated observations was used to analyze trends in rates of psychotropic drug use. Poisson regression models were used to compare the percentage of weighted pregnancies from 2001 to 2013 with at least 1 psychotropic medication for the 3-month prior period, for the during-pregnancy period, and for the 3-month postpartum period. Weighted number of pregnancies was used since not all women complete all trimesters. The weighted value was calculated by summing the actual number of days each woman contributed during a time period for all pregnancies, and the total number of days all pregnancies contributed to a time period was then divided by the possible number of days in that time period as the denominator (e.g., 91 days for each trimester). Patient characteristics of psychotropic medication users overall and by category of continued and discontinued use were described with descriptive statistics. Characteristics included maternal age (mean and proportion per age group [≤20, 21-29, 30-39, and ≥40 years]), income quintile, rural or urban residence, primiparous versus multiparous, healthcare use in the year prior to start of pregnancy (psychiatric, inpatient hospitalization, emergency visit), co-medications in the 3 months prior to pregnancy (anxiolytic/sedatives, antipsychotics, antidepressants, lithium, antiepileptics, stimulants, opioids), psychiatric diagnosis in 3 years prior to pregnancy (mood/anxiety, schizophrenia, substance use disorder, self-harm), and nonpsychiatric comorbidities in 3 years prior to pregnancy (cancer, diabetes, headache, epilepsy).
Ethics
This study was conducted in compliance with the Privacy of Health Information Act of Manitoba and was approved by the Research Ethics Board of the University of Manitoba and the Manitoba Health Information Privacy Committee.
Results
During the study period, there were 41,923 of 224,762 pregnancies in which at least 1 psychotropic drug was used in the 12 months before pregnancy, during the pregnancy, or 3 months after pregnancy. The rate of psychotropic use in the 12 months before pregnancy was higher (7.5%) than use in the other periods (Table 1). Anxiolytic/sedative-hypnotics and antidepressants were the most common psychotropic drugs used among these women. From 2001 to 2013, the weighted percentage of pregnant women who received at least 1 psychotropic medication prescription increased 1.5-fold from 11.1% to 16.2% (p < 0.0001) in the 3 to 12 months before pregnancy, 1.6-fold from 6.4% to 10.5% (p < 0.0001) in the 3 months before pregnancy, 1.8-fold from 3.3% to 6.0% (p < 0.0001) during pregnancy, and 1.5-fold from 6.2% to 9.5% (p < 0.0001) in the 3 months postpartum (Figure 1). While an increase in the use of psychotropic agents was observed from 2001 to 2013, the rate of use was always highest in the 3- to 12-month period before pregnancy and lowest in the second and third trimester of pregnancy (Figure 1). For further information on the trend in use peripregnancy by drug class over time, see the online supplement.
Overall psychotropic use (receipt of at least 1 prescription) before, during, and after pregnancy (N = 224,762).
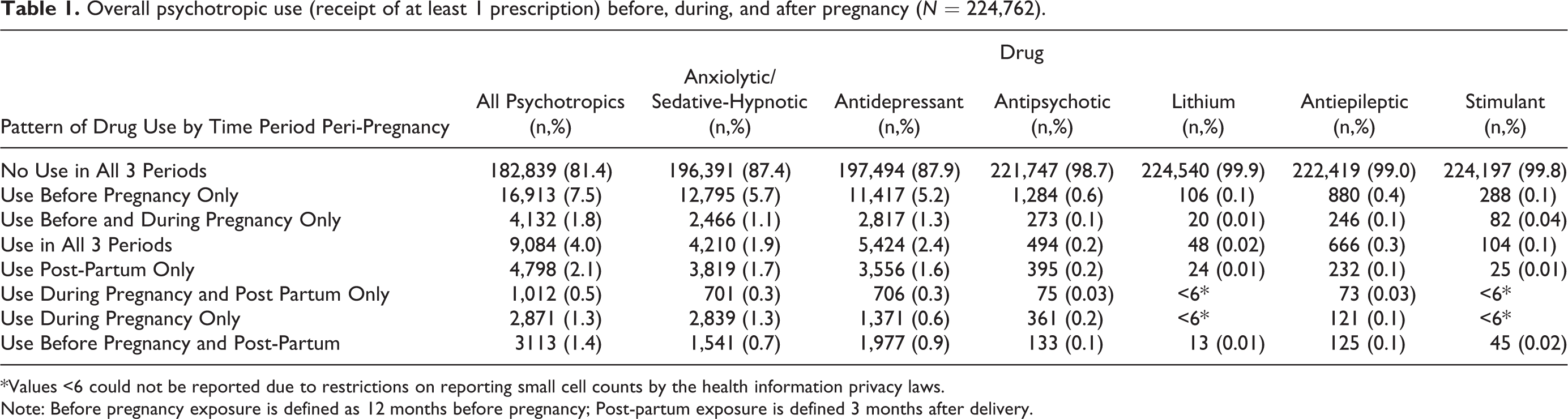
*Values <6 could not be reported due to restrictions on reporting small cell counts by the health information privacy laws.
Note: Before pregnancy exposure is defined as 12 months before pregnancy; Post-partum exposure is defined 3 months after delivery.
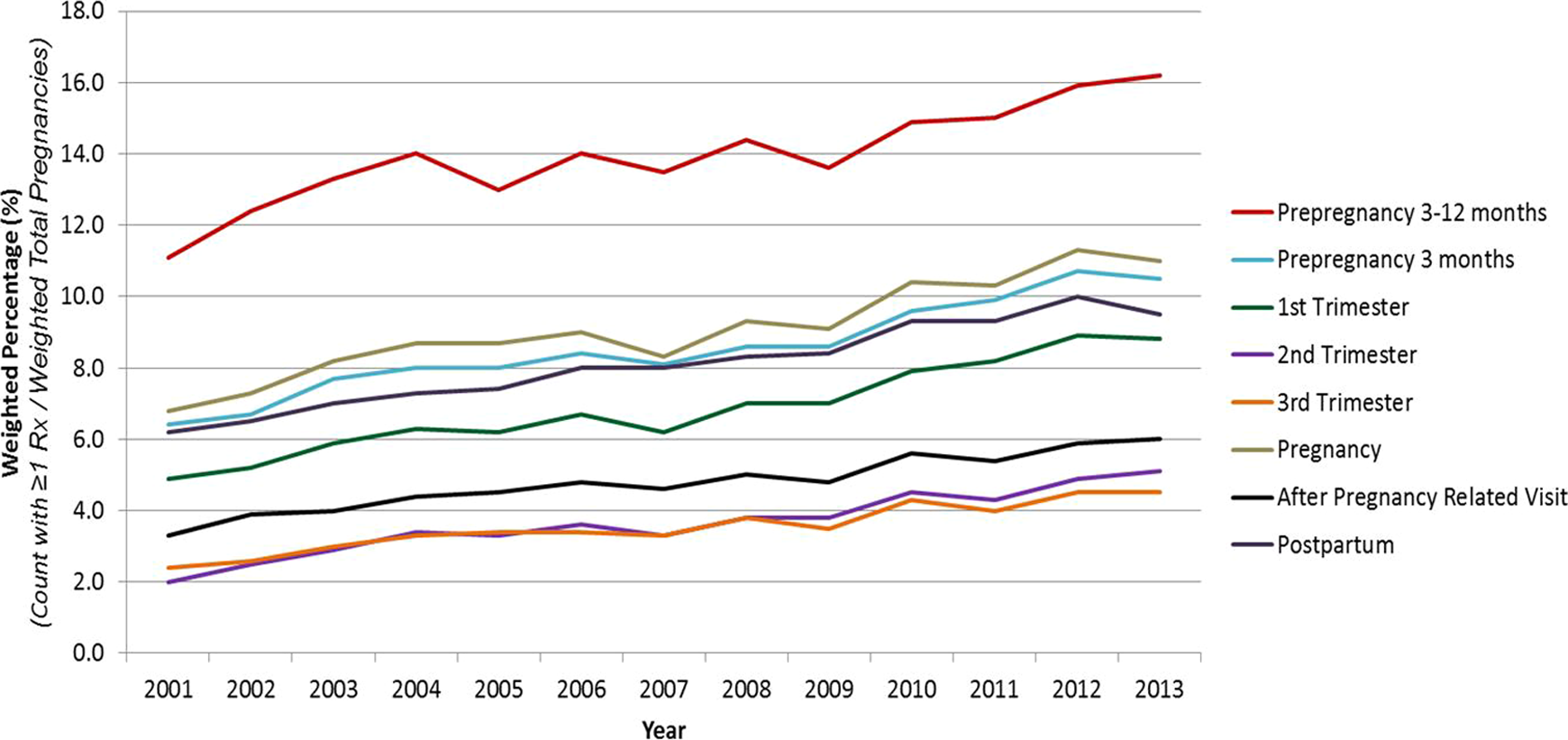
Weighted percentage of pregnant women who filled at least 1 prescription for a psychotropic medication (2001-2013).
The study identified 12,153 pregnant women with a birth outcome of livebirth, stillbirth, or intrauterine death who had at least 1 prescription for a psychotropic agent dispensed in the 3 months prior to the start of pregnancy. Mean age was 28 years (SD, 5.8 years). Baseline characteristics for patients overall, and for those who were categorized as discontinued, intermittent, and continued users of psychotropic agents, are described in Table 2.
Baseline demographic and clinical characteristics of pregnant women (birth outcome of livebirth, stillbirth, or intrauterine death only) who had ≥1 psychotropic drug prescription dispensed in the 3 months prior to start of pregnancy.
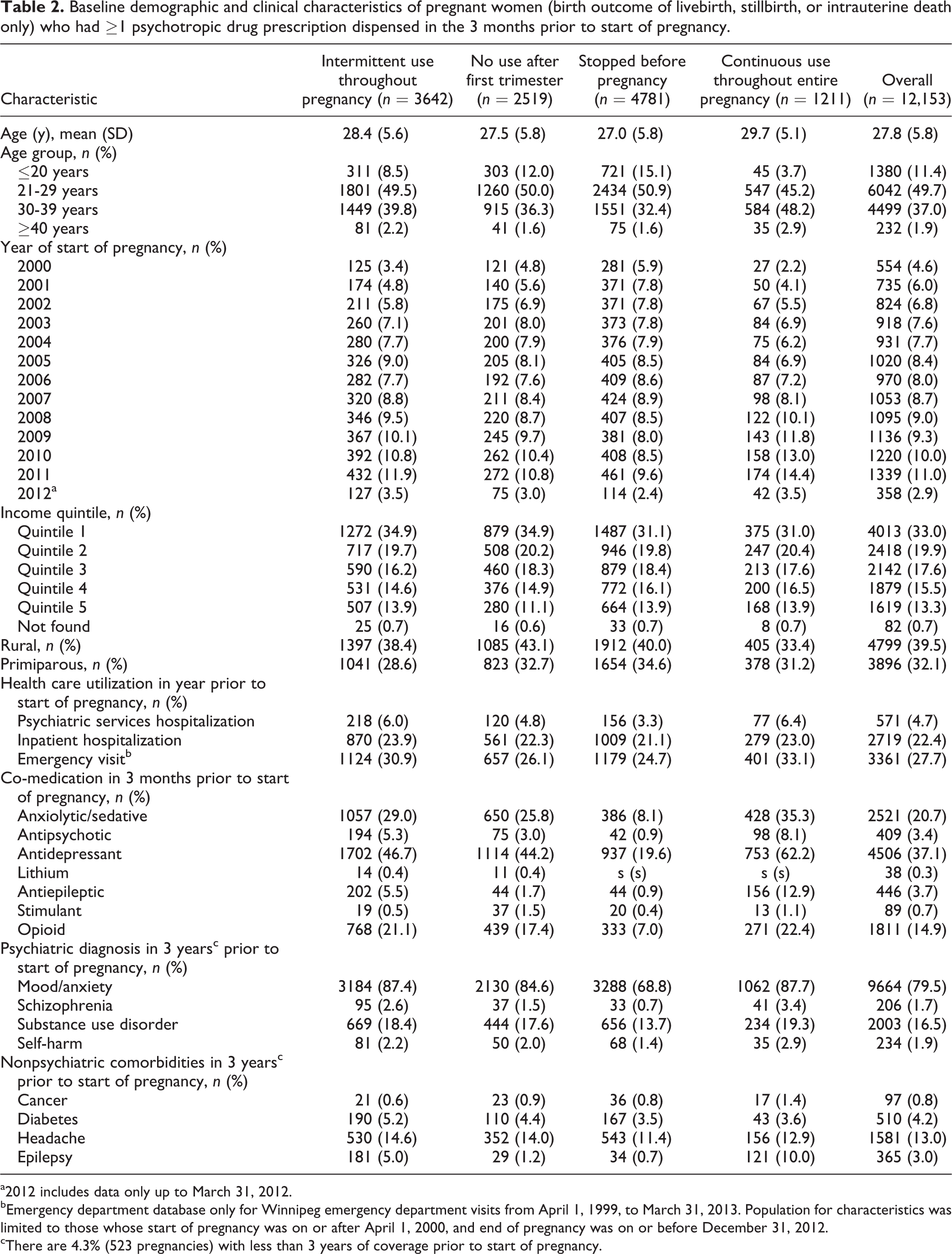
a2012 includes data only up to March 31, 2012.
bEmergency department database only for Winnipeg emergency department visits from April 1, 1999, to March 31, 2013. Population for characteristics was limited to those whose start of pregnancy was on or after April 1, 2000, and end of pregnancy was on or before December 31, 2012.
cThere are 4.3% (523 pregnancies) with less than 3 years of coverage prior to start of pregnancy.
Among those who filled at least 1 prescription for a psychotropic drug in the 3 months prior to pregnancy, 10.3% (n = 1401) of pregnant patients continued their psychotropic agent throughout pregnancy, and 38.5% (n = 5226) and 20.7% (n = 2812) stopped their psychotropic medication prior to their first and second trimesters, respectively. Approximately 30.5% (n = 4140) of patients were considered intermittent users of these agents throughout pregnancy. The proportions of patients who continued, discontinued, and intermittently used their agents by psychotropic drug class are shown in Figure 2. When the population was restricted to only those who filled at least 2 consecutive prescriptions (gap ≤14 days apart) in the 3 months prior to pregnancy who were at least 80% adherent (80% MPR) (n = 6693), a higher proportion of women continued use through the entire pregnancy (56.9%, n = 3808), and only 16.5% (n = 1105) and 15.2% (n = 1020) stopped their psychotropic medication prior to the first and second trimesters, respectively. Only 11.4% (n = 760) were considered intermittent users of these agents throughout pregnancy (Table 3). Antiepileptic drugs were among the psychotropic agents that were most likely to be continued throughout pregnancy. Among the 460 women who received at least 2 prescriptions for an antiepileptic drug and were 80% adherent in the 3 months prior to pregnancy, 63.7% continued their treatment throughout pregnancy (Table 3).
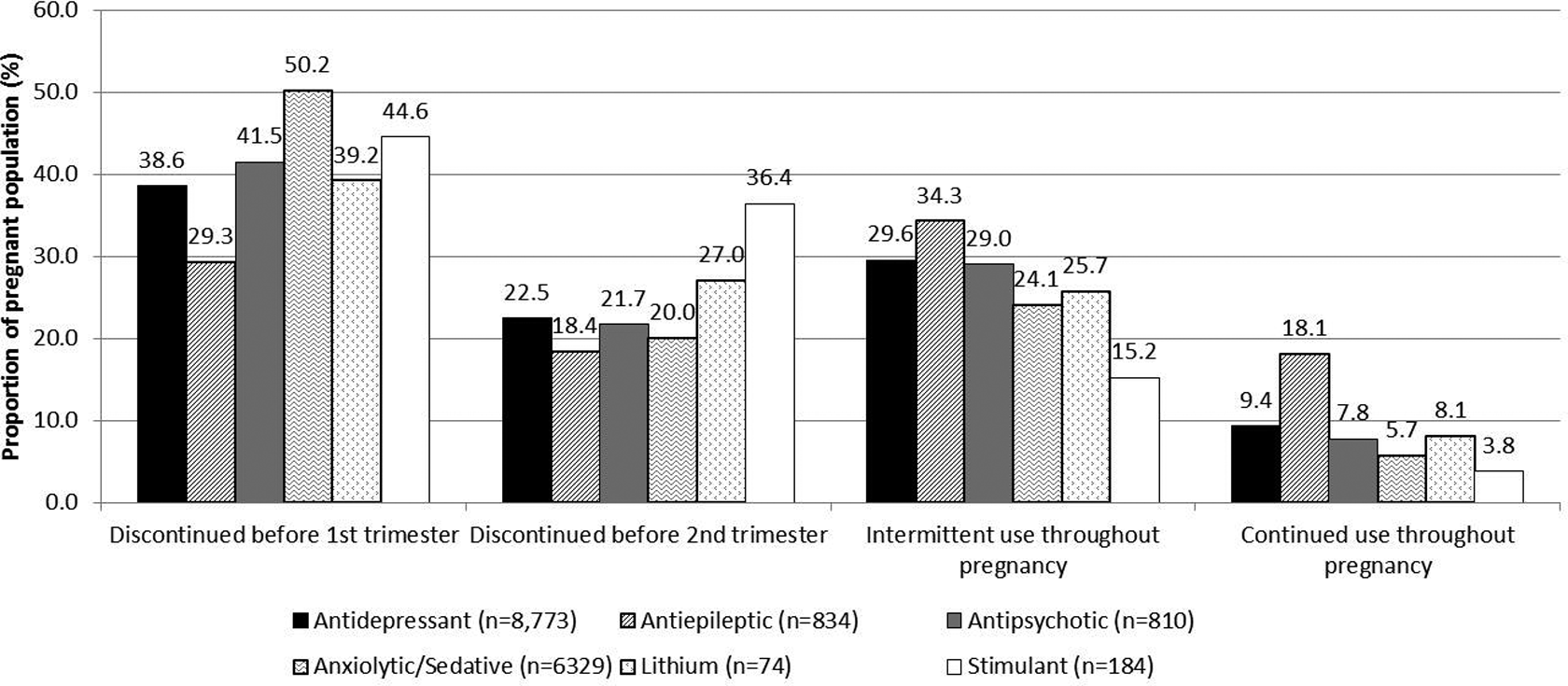
Comparison of discontinued (before first trimester and before second trimester), intermittent, and continued users among pregnant patients (birth outcome of livebirth, stillbirth, intrauterine death only) who had ≥1 psychotropic drug prescription dispensed in the 3 months prior to start of pregnancy. Denominator is population who received at least 1 prescription for each class of drug in 3 months prior to conception (shown in parentheses). Continuous use was defined as no gap greater than 14 days.
Pattern of continued and discontinued psychotropic use among pregnant women resulting in a livebirth, stillbirth, or intrauterine death (n = 6693)a.
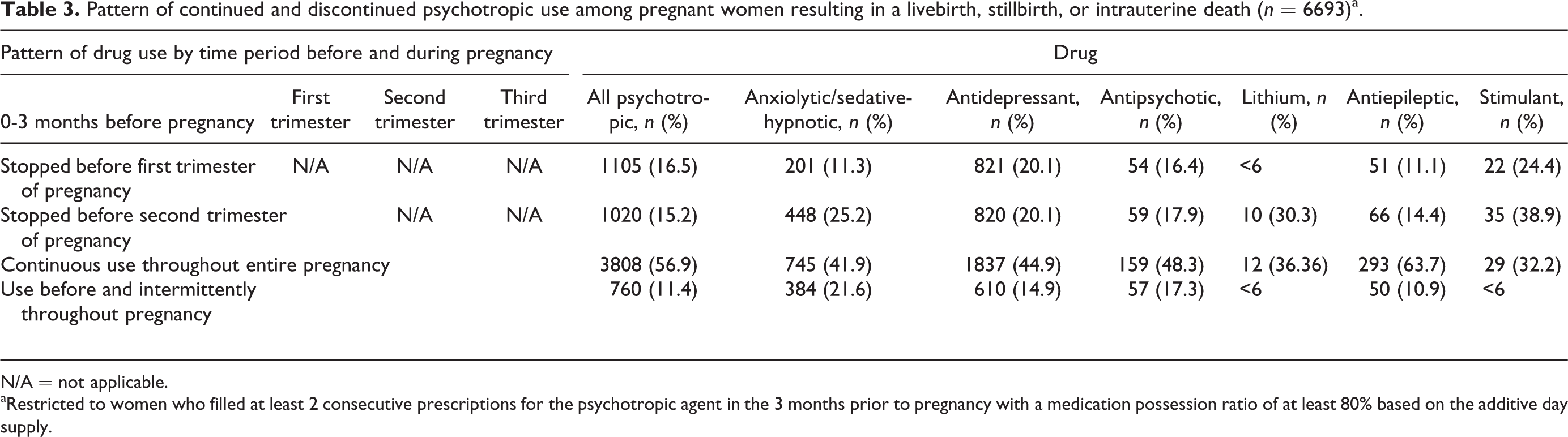
N/A = not applicable.
aRestricted to women who filled at least 2 consecutive prescriptions for the psychotropic agent in the 3 months prior to pregnancy with a medication possession ratio of at least 80% based on the additive day supply.
Discussion
This study presents population-based, real-world patterns of psychotropic medication use before, during, and after pregnancy. While an increasing trend in the use of psychotropic drugs was observed in all periods from 2001 to 2013, the use of these agents decreased considerably during the pregnancy period. However, the majority of women who were more likely to be adherent to their psychotropic medication prior to becoming pregnant were observed to continue their treatment with good adherence throughout pregnancy. This reflects current practice guidelines that recommend women who are stable on needed treatment to continue their therapy if the benefit of continuing outweighs the risk of discontinuing that agent. 25 –27 This pattern of use could also be an indication that women with more severe disorders or better controlled disorders are more likely to continue their medications.
Our findings were consistent with previous studies that examined the extent of psychotropic drug use and the rates of continuation and discontinuation of these agents during pregnancy. 11 –24 While antidepressants, 20,23,24 antipsychotics, 15,20,21 lithium, 18,23 and anticonvulsants 16,17,20,23 are commonly received before and during early pregnancy in the United Kingdom, Quebec (Canada), and the United States, a similar decrease in use was observed in late pregnancy in these studies. However, it is not known whether the women in these studies, whose use was based on the receipt of at least 1 antidepressant prescription, were adherent users of these agents prior to pregnancy. Moreover, our findings will build on the limited detailed data on the pattern of sedative-hypnotic and stimulant medication during pregnancy.
Our study had a number of strengths, including a large sample size and comprehensive database not restricted to income or age, which adds to the existing literature on psychotropic use during pregnancy. We also made a distinction between those who filled only 1 prescription prior to pregnancy and those who had good adherence to medication prior to pregnancy. There were a few limitations worth noting. We were unable to determine whether the decision to stop medication was based on the decision of the physician, patient, or both. We also did not identify the indication for the psychotropic agent. The number of patients who received lithium and stimulant medication in our sample was small. We also did not look at specific agents within each class of psychotropic agents, which warrants further investigation due to some inherent differences within each class. Data from these administrative databases do not capture those who do not use medical services during pregnancy or who deliver outside a hospital setting. We are only able to calculate the rate of use based on the dispensing of medications captured within these databases, which may not reflect actual drug consumption. However, the use of administrative databases is not prone to recall bias and encompasses prescription use for the entire population. Moreover, we did attempt to determine the rates of use based on patients who received at least 2 prescriptions with an MPR of at least 80% to capture those more likely to have consumed these agents. Further investigation is warranted to examine the impact of continuing versus discontinuing psychotropic agents during pregnancy on maternal mental health.
In conclusion, while the use of psychotropic agents increased over 12 years, we observed a decrease in use during the pregnancy period among women in a Canadian population; this is similar to the patterns of use observed in other countries. Continued use of psychotropic drug treatment was more likely in those who had good adherence to medication before pregnancy. These findings will provide the basis for future research examining the clinical implications of continuing versus discontinuing these agents during pregnancy.
Footnotes
Acknowledgements
The authors acknowledge the Manitoba Centre for Health Policy, University of Manitoba, for the use of data contained in the Population Health Research Data Repository (Health Information Privacy Committee [HIPC] No. 2013/2014-35). The results and conclusions are those of the authors, and no official endorsement by the Manitoba Centre for Health Policy, Manitoba Health, Seniors and Active Living, or other data providers is intended or should be inferred. Data used in this study are from the Population Health Research Data Repository housed at the Manitoba Centre for Health Policy, University of Manitoba, and were derived from data provided by Manitoba Health, Seniors and Active Living.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding for this study was provided by the Canadian Network for Observational Drug Effects Studies (CNODES), a collaborating centre of the Drug Safety and Effectiveness Network funded by the Canadian Institutes of Health Research.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
