Abstract
Seasonal variations in Ostracoda populations and the elemental composition of their carapaces were examined across diverse ecosystems of the Mandapam group of islands, on the southeast coast of India. Sediment samples from five islands-Shingle, Krusadai, Pullivasal, Poomarichan, and Hare-spanning coral reefs, seagrass meadows, sandy substrates, and mangrove environments, revealed 18 species across 16 genera with distinct shell ornamentations (smooth, reticulate, spinose, punctate, and ridged). Ostracod abundance peaked during the post-monsoon season, followed by the pre-monsoon and monsoon seasons. Elemental analysis of the carapaces showed the presence of Ca, C, O, Na, Mg, Al, Si, Fe, K, Nb, and Rb, with seasonal shifts—particularly Nb and Rb—highlighting the influence of monsoonal dynamics. Statistical assessments of adult/juvenile and valve/carapace ratios helped infer the sedimentation rates and the diagenetic processes. Smooth and porous forms were closely associated with coral habitats, while reticulate, punctate, and spinose forms dominated mangrove and seagrass environments. Overall, Ostracod distribution and shell chemistry reflect both ecological preferences and seasonal environmental changes.
Keywords
INTRODUCTION
Marine Ostracoda serve as proxies for marking environmental and ecological deterioration (Aljahdali et al., 2024; Ruiz et al., 2005; Salvi et al., 2015; Schmitz et al., 2024). These tiny bivalved crustaceans, belonging to the phylum Arthropoda, are ubiquitous across diverse aquatic environments (Huang et al., 2022; Pokrajac et al., 2024; Wang et al., 2025). Their remarkable resilience, having survived multiple extinction events throughout geological history, positions them as key subjects for understanding long-term ecological trends and environmental changes (Williams et al., 2015). Ostracoda are widely used in deciphering niche-specific habitats, and their occurrence in coral reefs, seagrass meadows, and mangrove forest areas has been thoroughly documented (El-Kahawy et al., 2021; Hussain et al., 2022; Radhakrishnan et al., 2022; Raynusha et al., 2020). Sediment characteristics exert a primary control on Ostracod assemblages, with variations in grain size, mineralogy, and organic content driving changes in community structure and ecological function (Radhakrishnan et al., 2022). Similarly, several studies have documented the various factors affecting the distribution of the fauna (Hussain et al., 2010; Radhakrishnan et al., 2023; Sridhar et al., 2019). Ostracod shell chemistry in both lacustrine and marine environments has primarily focused on magnesium (Mg) and strontium (Sr) as proxies for water chemistry and temperature (Holmes & Chivas, 2002). Seasonal environmental variations can be reflected in the trace element concentrations of Ostracod shells, corresponding to changes in ecological conditions (Xia et al., 1997). As environmental parameters shift, the elemental makeup of the shells adjusts accordingly, offering insights into past and present environmental dynamics.
By integrating the geochemical studies of carapace and seasonal distribution, this work focuses on the relationship of GoM Ostracoda with respect to the shifts in environmental factors, such as habitat-specific conditions, which affect the distribution and environment of Ostracoda. Ultimately, a better understanding of these patterns can enhance our ability to predict broader environmental and ecological shifts in response to both natural and anthropogenic pressures. However, despite these advances, significant gaps remain in our understanding of the spatiotemporal dynamics of benthic Ostracoda communities across these interconnected ecosystems. The present study aims to address this gap by deciphering the spatiotemporal variations in benthic Ostracod assemblages across multiple habitats within the Mandapam group of islands in the Gulf of Mannar Marine Biosphere Reserve (GoMMBR).
STUDY AREA (Gulf of Mannar Marine Biosphere Reserve)
The Gulf of Mannar Marine Biosphere Reserve (GoMMBR) is renowned for its intricate coral reefs, seagrass meadows, and mangrove forests, spanning approximately 140 km of coastline from Tuticorin to Pamban and comprising 21 islands arranged in four parallel groups along the coast (Kumaraguru et al., 2006). In recognition of its globally significant wetland habitats, the reserve was designated a Ramsar site in 2022. In the northern GoMMBR, the Mandapam group comprises various islands where corals, mangroves, and seagrass beds are prevalent. Both natural processes and human activities influence Ostracoda assemblages across these habitats. Ostracoda from different depths are extensively studied, and the faunal distribution and abundance are regulated by salinity, water depth, and calcium carbonate (Baskar et al., 2013; Hussain et al., 2007, 2022; Radhakrishnan et al., 2022, 2023; Sridhar et al., 2019).
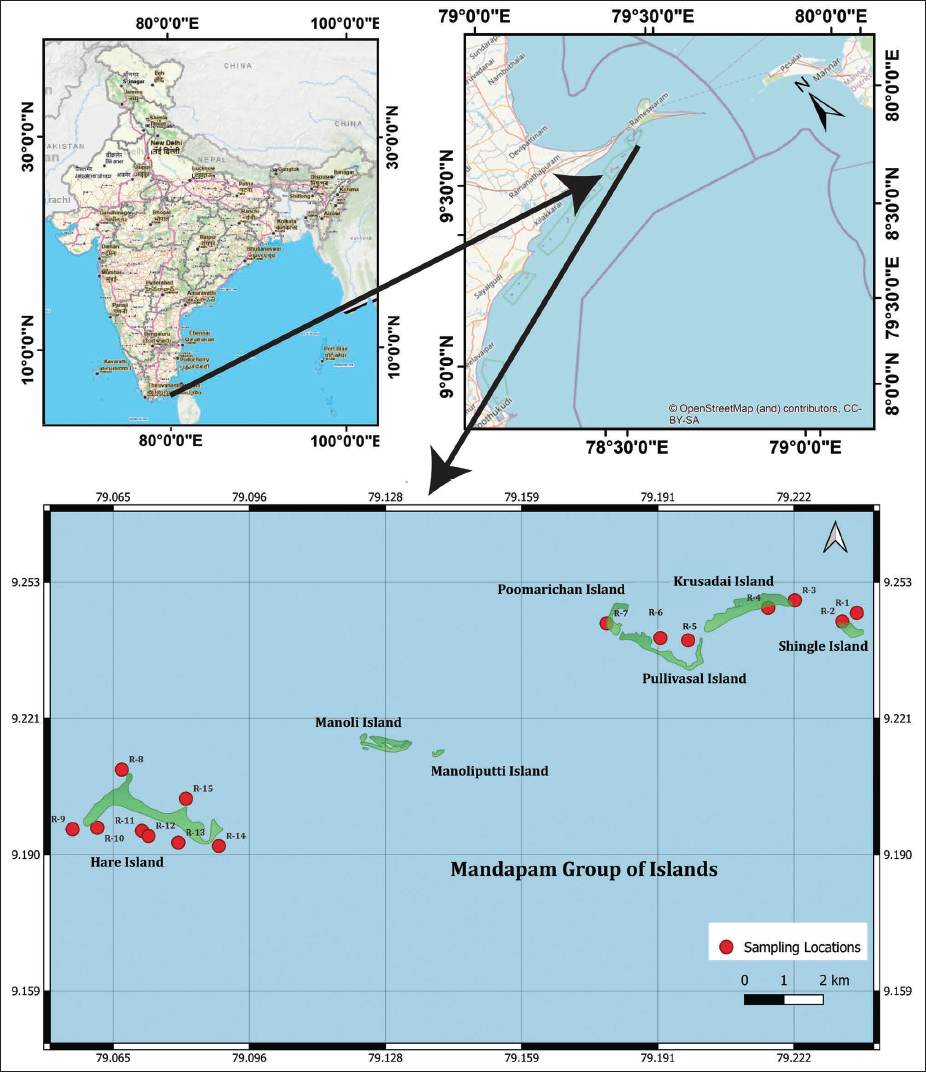
For the present study, 15 sediment samples were collected each season (a total of 45 samples from pre-monsoon, monsoon and post monsoon seasons): two from Shingle Island (R-1 to R-2), two from Krusadai Island (R-3 to R-4), two from Pullivasal Island (R-5 to R-6), one from Poomarichan Island (R-7) and eight from Hare Island (R-8 to R15).
METHODOLOGY
Field work
A total of 45 (15 locations per season) sediment samples were retrieved from coral, mangrove, and seagrass ecosystems on five coral islands; Hare (8 locations), Shingle (2 locations), Krusadai (2 locations), Pullivasal (2 locations) and Poomarichan (1 location) in the Mandapam group of islands from the GoMMBR (Figure 1). Sampling targeted three seasons: pre-monsoon (September–October), monsoon (December–January), and post-monsoon (February–March). Seabed sediments were collected by scuba diving at varying depths (2–5 m), ensuring minimal disturbance to the benthic environment.
Index map of the Mandapam Group of islands, GoMMBR, showing sampling locations and the study area.
Analytical protocols
Ostracoda were retrieved from sediment matrices using standardised protocols. Wet sediment samples were soaked overnight in a 30% hydrogen peroxide (H2O2) solution to oxidise organic matter, followed by rinsing through a 63-µm mesh sieve. The residues were subsequently oven-dried at 50°C. From each dried sample, 5 g of sediment was subsampled, and Ostracod valves were manually extracted using a 000-paint brush under a stereo zoom microscope (Optica, SLX-2). Additionally, scanning electron microscopy coupled with energy-dispersive X-ray spectroscopy (SEM-EDAX) was conducted on shells of each Ostracoda species retrieved from the five islands across three seasonal intervals, adhering to the protocols of Goldstein et al. (2003).
RESULTS AND DISCUSSION
Ostracod distribution
Ostracods were identified and classified following the taxonomy proposed by Hartmann and Puri (1974). A total of 18 species, belonging to 16 genera (Table 1), were recorded from the study area. These assemblages represent typical tropical shallow marine environments associated with coral reefs, mangroves, sandy substrates, and seagrass ecosystems. During the pre-monsoon season, coral-dominated habitats were primarily inhabited by Bairdoppilata (Bairdoppilata) alcyonicola (Maddocks, 1969), Neonesidea crasenticlavula (Maddocks, 1969), and Macrocyprina decora (Brady, 1866). The hard and sparse distribution of corals impacts Ostracod diversity and abundance.
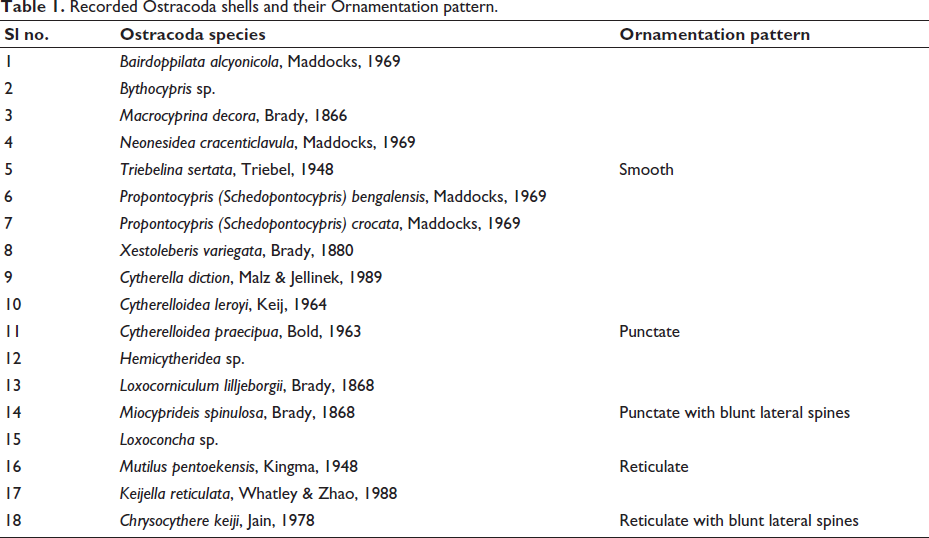
Recorded Ostracoda shells and their Ornamentation pattern.
The seagrass environment yielded the maximum abundance of Ostracoda, characterised predominantly by milky-white carapaces and a significant number of juveniles. In contrast, mangrove habitats were dominated by reticulate forms such as Keijella reticulata (Whatley & Zhao, 1988), Mutilus pentoekensis (Whatley & Zhao, 1988), and the punctate Loxocorniculum lilljeborgii (Brady, 1868). Sandy substrates showed the most significant overall Ostracod abundance, with specimens exhibiting a distinct white colouration during the pre-monsoon period. Notably, in seagrass beds, carapaces transitioned from an opaque, milky-white appearance during the pre-monsoon season to a more translucent, glass-like appearance during the monsoon season. On Hare Island, some juvenile forms from sandy substrates exhibited alar ornamentation; however, their species could not be identified due to their immature growth stage. B. (B.) alcyonicola (Maddocks, 1969) was the only species recovered from Shingle Island (R-2) during the monsoon season.
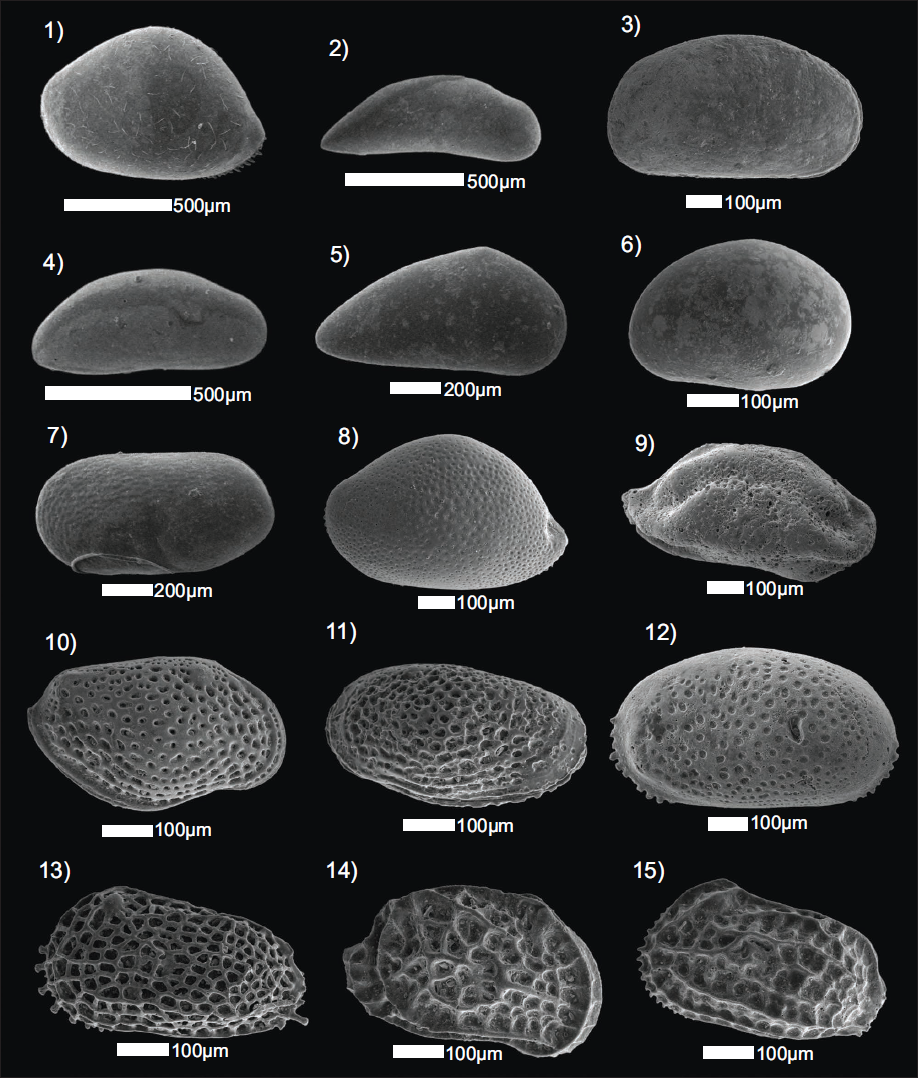
Loxoconcha sp. emerged as the dominant genus from the mangrove environment. Post-monsoon marks the abundance of smoothly ornamented Ostracoda forms in coral habitats, and most notably with a thin and glassy carapace. Punctate and reticulate ornamentation, which are typical in sandy substrates, are overwhelmingly populated. Mangrove forests support only sparse Ostracod populations, in contrast to seagrass meadows, which harbour a diverse assemblage dominated by juveniles. B. (B.) alcyonicola and Actinocythereis scutigera (Brady, 1868) are shallow-water, photic zone species that prefer low-turbulence environments. Despite their differing morphological traits, they are known to co-exist in similar habitats. However, the absence of A. scutigera (Brady, 1868) in all the sampled locations is notable and somewhat anomalous, as no adult specimens of this species were recovered during the present study. Scanning electron microscope (SEM) images of the ostracods collected from the study area are illustrated in Plate 1.
SEM images of Ostracoda carapace: (1) LV of Bairdoppilata (Bairdoppilata) alcyonicola (Maddocks, 1969); (2) RV of Macrocyprina decora (Brady, 1866); (3) RV of Bythocypris sp.; (4) RV of Propontocypris (Schedopontocypris) bengalensis (Maddocks, 1969); (5) P. crocata (Maddocks, 1969); (6) RV of Xestoleberis variegata (Brady, 1880), (7) LV of Cytherella dictyon (Malz & Jellinek, 1989), (8) LV of Neonesidea crasenticlavula (Maddocks, 1969), (9) RV of Triebelina sertata (Triebel, 1948), (10) RV of Loxocorniculum lilljeborgii (Brady, 1868), (11) RV of Loxoconcha sp., (12) LV of Miocyprideis spinulosa (Brady, 1868), (13) LV of Keijella reticulata (Whatley & Zhao, 1988), (14) RV of Mutilus pentoekensis (Kingma, 1948), (15) LV of Chrysocythere keiji (Jain, 1978) [LV-Left Valve, RV-Right Valve].
Ornamentation in the Ostracoda carapace
Valve chemistry, ornamentation, and Ostracoda shell preservation imply the physicochemical conditions of the environment in which the organism grows (De Deckker, 2002). Ornamentations in Ostracoda, particularly the pore patterns, aid in understanding the ontogeny, taxonomy, and phylogeny (Karanovic et al., 2017). Out of 18 species, 8 species are smooth ornate forms, 5 are punctate forms, 2 are reticulate forms, and one exhibits punctate ornamentation with blunt lateral spines (Table 1). Ostracod shell ornamentation is correlated with the nature of the substrate (Sridhar et al., 2016), and the pores in the shell serve as chemo-sensory organs (De Deckker, 2002). In the present study, smooth-shelled adult forms were found to dominate the coral environments. Reticulate and punctate forms were prevalent in the mangrove habitats, indicating a preference for this morphology in such settings. Sandy environments exhibited a lower abundance of adult ostracods, with glassy-shelled forms being the most prominent. In contrast, the seagrass habitats predominantly hosted milky-white forms, with a notable presence of juveniles.
Adult/juvenile and valve/carapace ratio
The adult/juvenile and valve/carapace ratios (Figure 2) are important statistical data in deciphering the relationship between Ostracoda and the sediments. A total of 2,131 Ostracoda shells were retrieved, with 1,541 adults and 590 juveniles. Out of 2,131 shells, 1,996 were valves, and there were 135 carapaces from all seasons. Post-monsoon assemblage has the maximum number of adult specimens. Four types of preservation patterns in the shells are noted from the area: Carapace with micro-hairs (sensilla) preserved, opaque carapace without appendages, and opaque and transparent valves. The specimens of B. (B.) alcyonicola, characterised by glassy, transparent shells, are identified from both coral and sandy environments, and have sensilla preserved within the shell. N. crasenticlavula. (Maddocks, 1969) Moreover, M. decora (Brady, 1866) is another Ostracoda form that has coral affinity. M. spinulosa (Brady, 1868) exhibits punctate ornamentation with blunt lateral spines and is dominant in a sandy environment. Loxoconcha sp., which have a reticulate type of ornamentation, are dominant in the seagrass environment, with a lower Ostracoda abundance. The shells recovered from the seagrass environment were milky-white adult forms. K. reticulata (Whatley & Zhao, 1988) and C. keiji (Jain, 1978) have reticulate ornamentation with blunt lateral spines, and the four species M. pentoekensis (Kingma, 1948), K. reticulata (Whatley & Zhao, 1988), L. lilljeborgii (Brady, 1868), C. keiji (Jain, 1978), and Loxoconcha sp. are dominant in the mangrove environment.
Adult/juvenile and valve/carapace ratio.
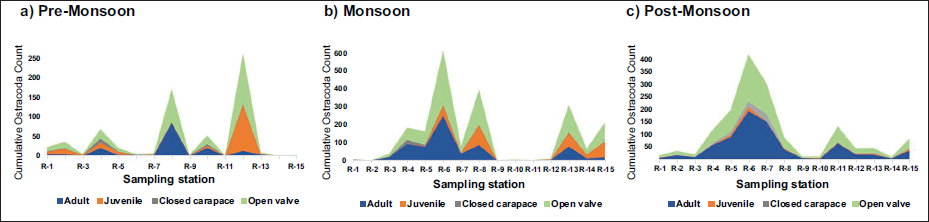
The sample R-6 from the coral environment at Pullivasal yields the maximum number of adults, 250 during the monsoon and 193 during the post-monsoon period, respectively. The exact location, R-6, has the maximum number of valves in the monsoon and post-monsoon periods. However, the pre-monsoon sample yields a different result. R-12 in Hare Island has the maximum adult forms and has the maximum number of valves. In an agitated environment, the shells open rapidly while the immediate burial in the sediment keeps the shells completely preserved. In the present study, valves outnumbered carapaces, which indicates a slower sedimentation rate.
Elemental analysis, along with heavy mineral data and ecotoxicological readings in Ostracod shells, is used to understand ontogenetic disturbances (Olga et al., 2025). The geochemical composition of Ostracod carapaces reflects both biomineralisation processes and environmental conditions (Frenzel & Boomer, 2005). Ca, C, O, and Mg predominantly form the calcium carbonate (CaCO₃) matrix of ostracods (Holmes & De Deckker, 2012). Na and Mg are incorporated during the biomineralisation process (Lowenstam & Weiner, 1989). Al, Si, and K are typically linked to clay minerals and silicate weathering (Taylor & McLennan, 1985). Fe also points to a terrigenous origin, or one that can be traced back to redox processes from the mangroves (Holguin et al., 2001). Nb indicates weathering of specific rock types in the surrounding regions (Nazeer et al., 2021). One factor contributing to the diversity and abundance of Ostracoda is the heterogeneity in elemental distribution. Since B. (B.) alcyonicola (Maddocks, 1969) is found on every island, elemental analysis is conducted on its shells. The presence of calcium (Ca), carbon (C), oxygen (O), sodium (Na), magnesium (Mg), aluminium (Al), silicon (Si), iron (Fe), potassium (K), niobium (Nb), and rubidium (Rb) is analysed (Figure 3). Calcium constitutes the primary component of the biomineralised valves, irrespective of the seasonal variations. Ca aligns with the mineralogy of Ostracoda valves, which are predominantly composed of chitin, protein, and low magnesium calcite (Gussone & Greifelt, 2019). Elevated Ca concentrations suggest minimal terrigenous or detrital contamination, consistent with deposition in environments conducive to robust carbonate precipitation, such as clear, open marine settings or well-developed coral habitats (Flügel, 2010). Nb was detected in the shells collected from Poomarichan and Shingle islands, and Rb was present in specimens from Krusadai Island during the pre-monsoon. However, during the monsoon, both Nb and Rb were absent in the carapaces across all five island sites. In the post-monsoon season, Rb reappeared in the carapaces from Shingle Island, while Nb remained undetected in shells from all five islands. Seasonal monsoonal dynamics significantly influence the elemental composition of Ostracoda shells, evidently affecting the incorporation of trace elements such as Nb and Rb. Al exhibited the lowest abundances during pre-monsoon and monsoon, whereas K was least abundant in post-monsoon samples. Elemental distribution patterns displayed spatial and seasonal variability. Core elements (C, Al, Si, Na, Mg, Ca) persisted across all seasons; however, monsoon samples displayed the most restricted elemental diversity, limited to these constituents. In contrast, pre-monsoon carapaces exhibited the broadest compositional range, with Fe, K, Nb, and Rb detected in addition to the core elements. Post-monsoon samples also showed elevated abundances of Fe, K, and Rb, though Nb was absent, suggesting seasonally mediated differences in environmental uptake or detrital influence.
Elemental composition in B. (B.) alcyonicola shells; (a) Pre-Monsoon, (b) Monsoon, (c) Post-monsoon.
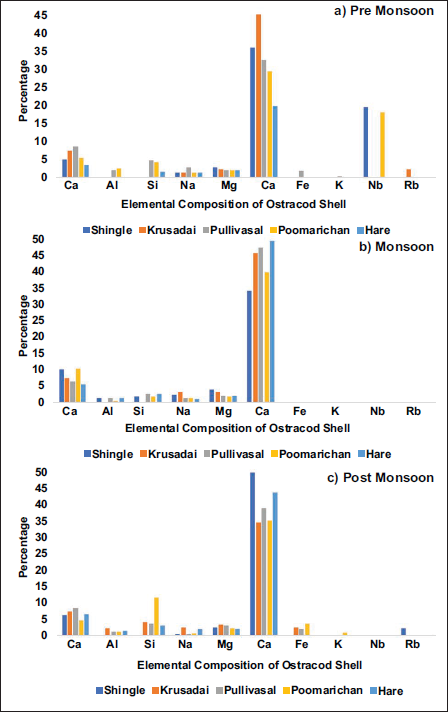
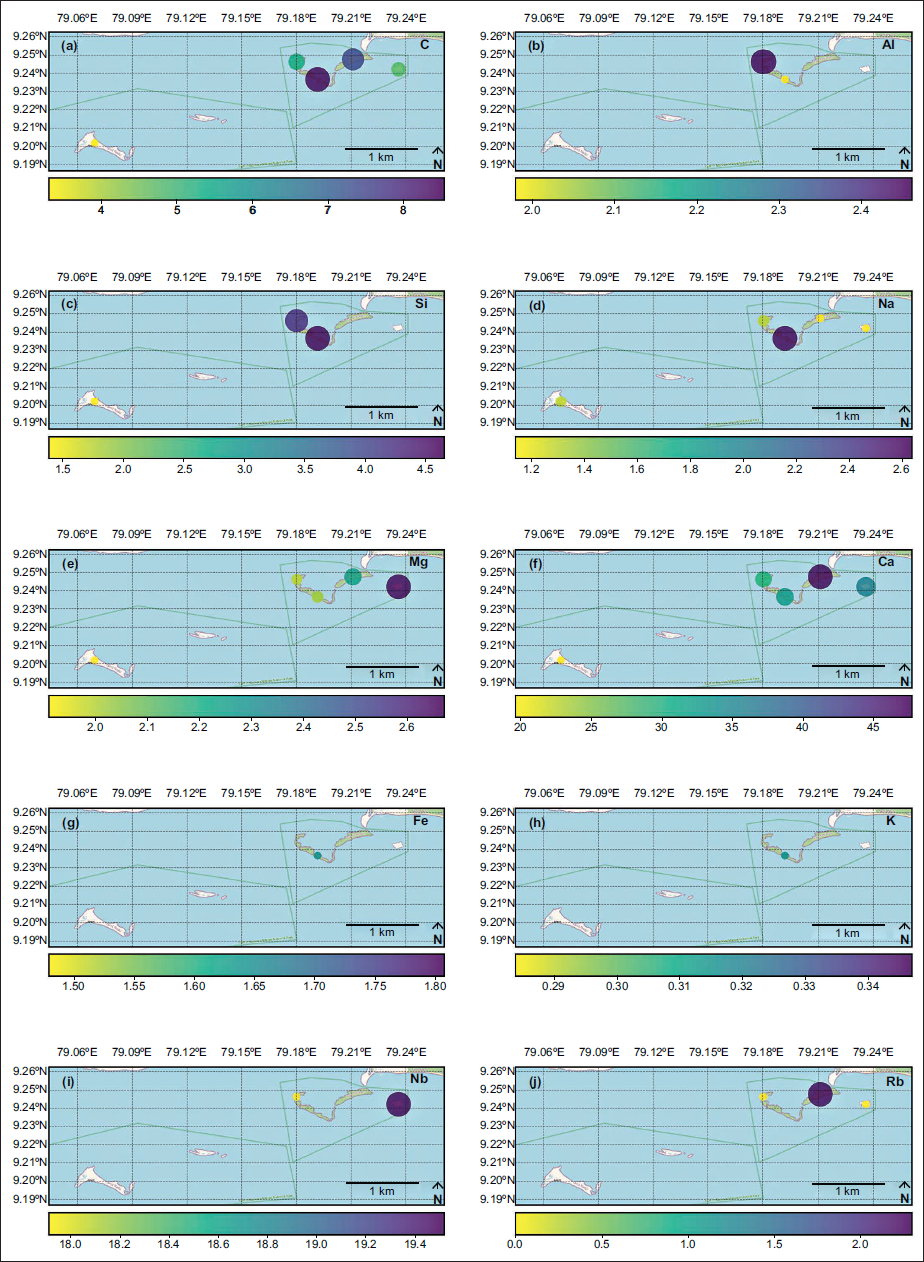
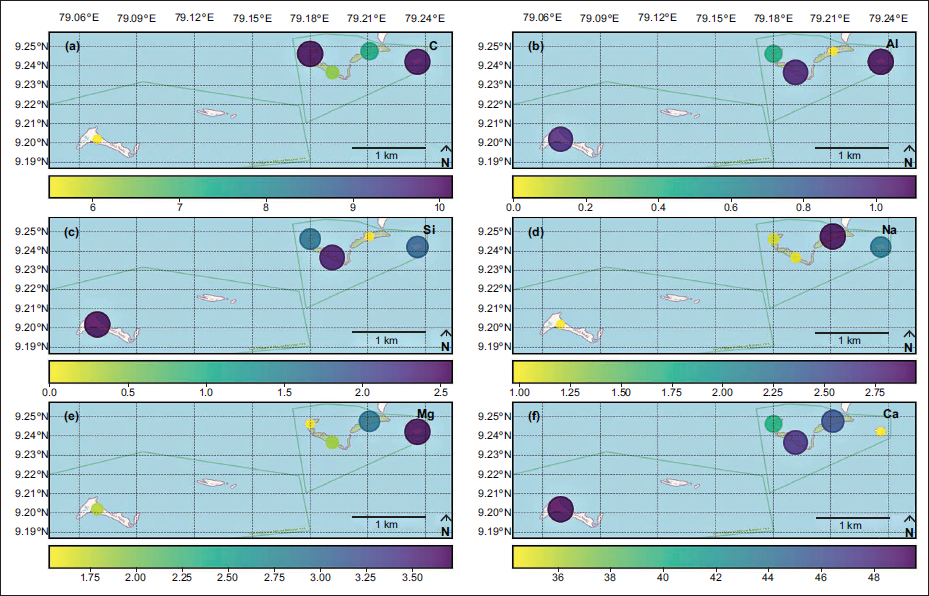
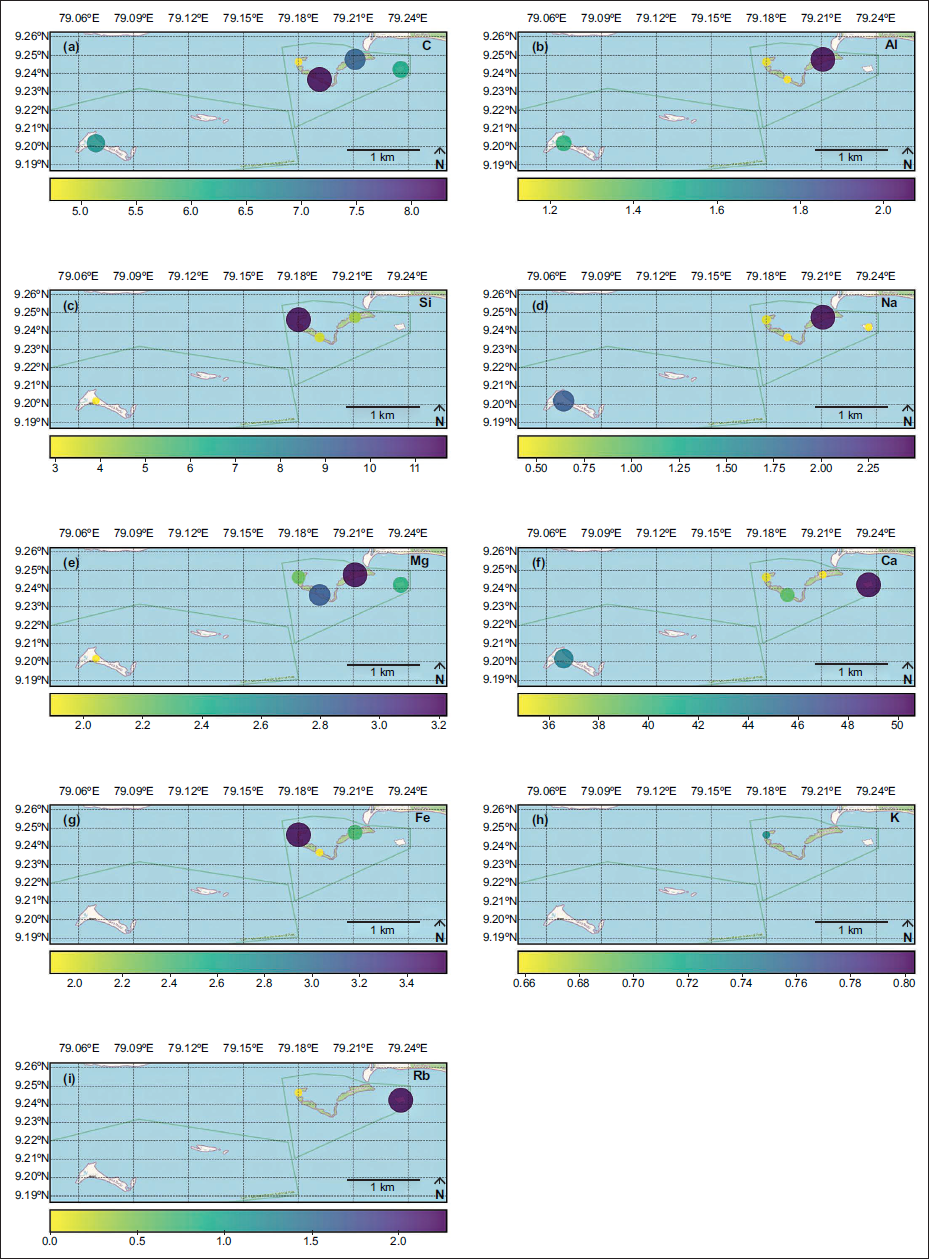
The spatial distribution of elements C, Al, Si, Na, Mg, Fe, K, Nb, and Rb plotted from B. (B.) alcyonicola was analysed, and the elemental composition of the shells was mapped for pre-monsoon (Figure 4), monsoon (Figure 5), and post-monsoon (Figure 6). Due to the prevalence throughout all islands, B. (B.) alcyonicola was selected as the species for elemental analysis. The analysis of the shells clearly documents that seasonal changes have an impact on the chemical composition of the fauna.
Spatial distribution of elemental composition of B. (B.) alcyonicola during pre-monsoon (values are in %).
Spatial distribution of elemental composition of B. (B.) alcyonicola during monsoon (values are in %).
Spatial distribution of elemental composition of B. (B.) alcyonicola during post-monsoon (values are in %).
CONCLUSIONS
The distribution, abundance, and diversity of Ostracoda are closely tied to the habitat type. Mixed sand–coral substrates proved the most favourable environment, supporting the highest faunal richness and population densities. Across seasons, B. (B.) alcyonicola consistently dominated coral habitats, while reticulate and punctate forms thrived in mangroves. Seagrass and sandy environments were predominantly populated by juveniles, with sandy areas exhibiting high abundance prior to the monsoon. Smooth-shelled adults dominated coral habitats, while reticulate and punctate forms characterised mangrove sites. Sandy substrates were primarily occupied by glassy-shelled specimens, while seagrass beds supported predominantly milky-white, juvenile-rich assemblages. Valves outnumbered intact carapaces, indicating slow sedimentation and bacterial‑mediated valve separation rather than rapid burial. This rarity of immediate burial reflects a low depositional rate in the study area. In mangrove settings, species diversity is reduced, and a return of glassy-shelled forms is observed, especially in coral and sandy areas during the post-monsoon period. The recovered assemblages consist of tropical shallow‑marine species characteristic of coral reefs, mangrove forests, sandy bottoms, and seagrass meadows. Elemental analysis confirms that Ostracoda valves are composed of biogenic low‑Mg calcite (Ca, C, O, Mg) with secondary incorporation of Na during shell formation. Terrigenous elements (Al, Si, K, Fe) reflect sediment provenance and redox conditions. At the same time, Nb and Rb display pronounced seasonal cycling, appearing pre‑monsoon, disappearing during the monsoon, and partially re‑emerging post‑monsoon. Monsoon currents thus exert a firm control on trace‐element uptake, narrowing the elemental suite to core constituents. Together, these findings demonstrate that both biomineralisation processes, hydrodynamic regimes, and seasonal fluctuations shape the geochemical signatures preserved in the Ostracoda shell.
Footnotes
Acknowledgement
The authors are grateful to the Chief Wildlife Warden of Tamil Nadu for the research permission in the restricted Marine Biosphere Reserve. The authors thank the Department of Marine Geology and Geophysics, Cochin University of Science and Technology (CUSAT), and the Geoscience Division, NCESS, Thiruvananthapuram, for the infrastructural support. The authors are grateful to IISc, Bengaluru, for providing with the SEM and EDS instrumentation support. Mr Rakhil Dev acknowledges the Department of Science and Technology (DST INSPIRE Division), Delhi, for the financial assistance.
Authors’ Contribution
Rakhil Dev: Fieldwork & Conceptualisation. R. Swathy: Formal analysis. Mohammed Noohu Nazeer: Draft preparation & Interpretation. Babu Nallusamy: Field work, methodology & formal analysis. S.G. Dhanil Dev: Statistical work & Review. S.M. Hussain: Supervision, Ostracoda identification & Editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Rakhil Dev was financially supported by the Department of Science and Technology (DST INSPIRE Division), Delhi.
