Abstract
Epithelioid angiosarcoma (EA) is a malignant tumor of endothelium origin that most commonly arises in the deep soft tissues of extremities but may occasionally be primary in skin, adrenal gland, and bone. A 72-year-old male presented with a painless enlargement of his thyroid for more than 10 days before hospitalization. A walnut-sized mass in the right thyroid was found simultaneously by palpation and Color Doppler Ultrasound. After a total thyroidectomy was performed, a mass with a size of 4.5 cm × 3.5 cm was found at the lower pole of the right thyroid gland. Histologically, the tumor was diffusely distributed in a sheet-like pattern, with tumor cells being epithelioid. There was extensive coagulative necrosis while no components of papillary carcinoma, follicular carcinoma and insular poorly differentiated carcinoma were noticed. CD31 and vimentin were positive for immunostaining. The diagnosis of primary epithelioid angiosarcoma of right thyroid was then given to the patient. Eleven months after the operation, the patient died from brain metastasis. It is suggested that primary epithelioid angiosarcoma of thyroid, as an extremely rare tumor, have no characteristic clinical manifestations, laboratory and imaging examinations, and the diagnosis mainly depend on its unique clinical pathological features. Although extensive surgical resection is the preferred treatment, the prognosis is still very poor.
Introduction
Angiosarcoma is a rare malignant neoplasm of endothelial origin, which often occurs in skin, viscera, and soft tissue, and is characterized by spindle cells of different degrees of atypia lining the vascular branches with a reticular structure. As a rare subtype of angiosarcoma, epithelioid angiosarcoma (EA) has obvious epithelioid appearance of tumor cells, which is common in the deep soft tissue of extremities and occasionally occurs in skin, adrenal gland, and bone,1–3 but it is extremely rare in thyroid. A case of primary epithelioid angiosarcoma of thyroid (PEAT) was diagnosed, but died soon after the operation. The case is presented, and the relevant literatures are reviewed in order to gain a deep understanding of the clinical characteristics, histological diagnosis, differential diagnosis, treatment and prognosis of this tumor.
Case presentation
A 72-year-old male patient presented with painless enlargement of the right thyroid for more than 10 days before hospitalization, accompanied by discomfort in the throat and low-pitched voice. As far as anamnesis was concerned, herniorrhaphy of left inguinal hernia was performed 5 years ago. And since sinus bradycardia with left axis deviation was diagnosed 12 years ago, no treatment was taken. The family history of hereditary disease was denied. At the time of hospitalization, the heart rate was 58 times per minute and blood pressure was 143/69 mmHg. Physical examination, a walnut-sized mass could be palpated in the right thyroid area of the neck, with unclear boundaries, soft texture, obvious tenderness, and moved up and down while swallowing. The skin on the surface was not ulcerated, and no abnormality was palpable in the left thyroid area of the neck. Blood routine tests showed that RBC was 4.28 × 1012/L (reference value 3.5–5.5) with WBC 6.48 × 109/L (4–10), PLT 129 × 109/L (85–303), and HGB 128 g/L (120–160). Biochemical examinations showed that cystatin C was 1.33 mg/L (0–1.03) with α-hydroxybutyrate dehydrogenase 228 U/L (72–182), sodium 140 mmol/L (132–145), chloride 102 mmol/L (90–108), magnesium 0.72 mmol/L (0.65–1.15), phosphorus 0.99 mmol/L (0.81–1.45), calcium 2.39 mmol/L (2.0–2.7), and potassium 3.9 mmol/L (3.5–5.3). Tumor marker tests showed that CA199 was 13.28 IU/mL (0–37) with CA125 5.59 IU/mL (0–35), AFP 2.36 IU/mL (0–6.05), and CEA 1.67 ng/mL (0–5.09). Thyroid function tests showed that T3 was 1.90 nmol/L (reference value 1.30–3.10 nmol/L) with T4 112.30 nmol/L (66.00–181.00 nmol/L), FT3 4.46 pmol/L (3.10–6.80 pmol/L), FT4 12.59 pmol/L (12.00–22.00 pmol/L), and TSH 2.91 mIU/L (0.27–4.20 mIU/L). Color Doppler Ultrasound demonstrated that a cyst-solid mass was located in the lower pole of the right thyroid with intact walls, poor or low echo, patchy calcification at the edge, no abnormal enlarged lymph nodes around, and that the trachea was pushed to the left with the narrow lumen (Figure 1). According to the manifestations, auxiliary examinations, and laboratory inspections, the malignancy of the right thyroid was given by a multiple-disciplinary team (MDT). The consent to treatment from the patient was signed. Then, a total thyroidectomy was performed.

By Color Doppler Ultrasound, a cyst-solid mass was found in the lower pole of the right thyroid with intact walls and poor or low echo.
The total thyroidectomy specimen was sent for pathological examination. Grossly, a nodular mass of 4.5 × 4.5 × 3.5 cm in size was seen at the lower pole of the right thyroid with clear margin. The cut surface of the mass was grayish-white to grayish-brown, soft and uniform in texture, with multifocal necrosis and hemorrhage of varying sizes, localized hardening and calcification. Histologically, hematoxylin-eosin (HE) staining showed that the original normal thyroid tissue was extensively destroyed while the tumor tissue was diffusely distributed in a sheet-like pattern. The ill-defined tumor cells were polygonal, round, short spindle-shaped and epithelioid, with eosinophilic cytoplasm. Some cells contained vacuoles or erythrocytes in their cytoplasm, and others were clustered together to form sinus-like lacuna. The nuclei were vesicular with significant atypia, and large red nucleoli. Pathological nuclear mitotic figures were easily seen. There was extensive coagulative necrosis, deposition of calcium salts, and hyaline degeneration. A few thyroid follicles were retained locally with some inflammatory cells infiltrating among them. No components of papillary carcinoma, follicular carcinoma, and insular poorly-differentiated carcinoma were seen. Reticular fiber staining showed the contours of immature blood vessels in the solid areas (Figure 2(a)–(d)).

Primary epithelioid angiosarcoma of thyroid. (a) Tumor cells were arranged in solid sheets with patchy necrosis (yellow arrow), HE ×100. (b) Tumor cells formed complex branched tubular structures or irregular sinus-like lacuna (yellow arrow), HE ×100. (c) Tumor cells were large and polygonal with abundant eosinophilic cytoplasm. Vacuoles containing red blood cells were present in the cytoplasm (yellow arrow) while the nuclei were vesicular with clearly visible nucleoli. Residual thyroid follicles were seen in the mid-right region, HE ×200. (d) In the solid area, complex branched tubular structures could be seen (yellow arrow), Reticular fiber staining ×200.
Upon immunohistochemical (IHC) detection, tumor cells were positive for CD31 and vimentin while were negative for cluster of differentiation 34 (CD34), factor VIII-related antigen (FVIIIRA), lymphocyte common antigen (LCA), high molecular weight keratin (CKH), low molecular weight keratin (CKL), B-cell lymphoma 2 (Bcl-2), epithelial membrane antigen (EMA), thyroglobulin, thyroid transcription factor 1 (TTF-1), human melanoma black 45 (HMB-45), synaptophysin (Syn), and calcitonin (CT; Figure 3(a) and (b)). Ki67 was about 75%. Then the same pathological diagnosis, primary epithelioid angiosarcoma of thyroid, was given to this mass by three experienced pathologists, respectively.
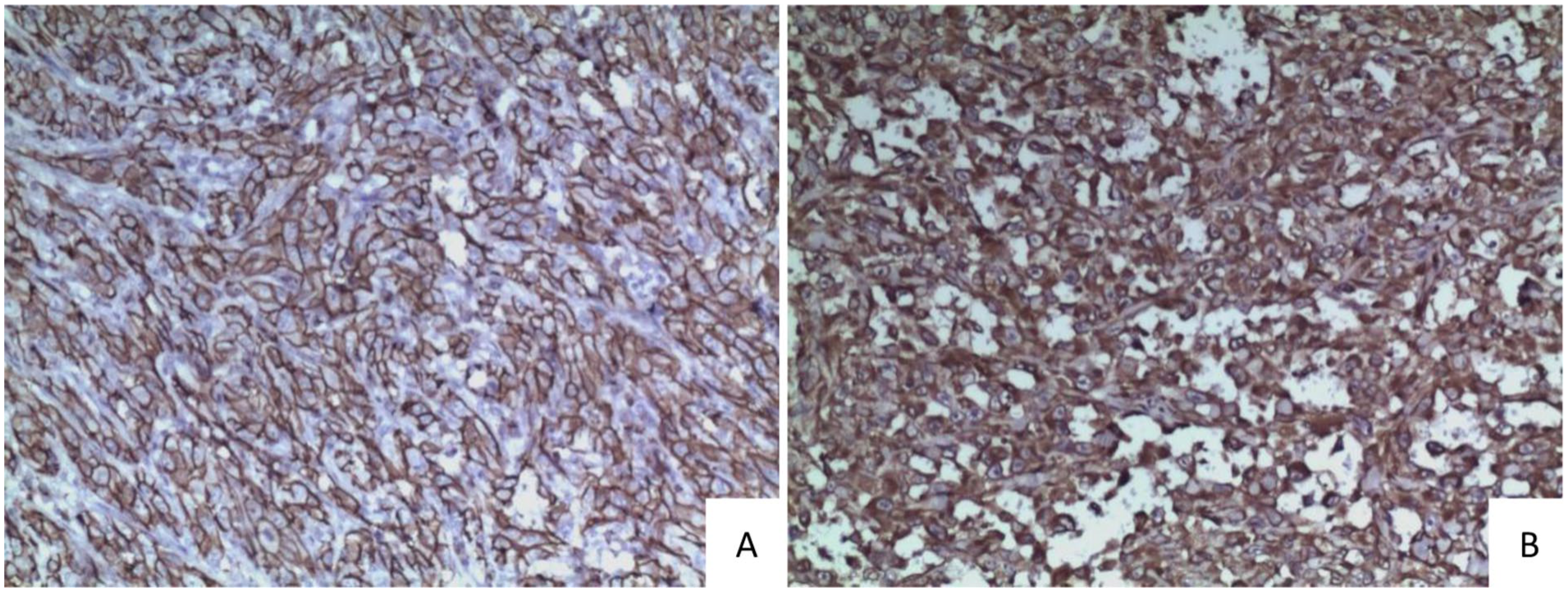
Primary epithelioid angiosarcoma of thyroid. Tumor cells were diffusely positive for CD31 (a) and Vimentin (b) upon immunohistochemical (IHC) staining, streptavidin-peroxidase method (SP) ×100.
At the follow-up 1 year after surgery, it was learned that the patient had died 11 months after surgery from “brain metastasis” according to CT examination at the local hospital in another city (imaging data unavailable). No brain biopsy or autopsy was performed. The reporting of this study conforms to CARE guidelines. 4 The written consent to participate was obtained from the patient’s legally authorized representative. All patient details have been deidentified.
Discussion
Angiosarcoma accounts for only a small fraction of all vascular tumors and less than 2% of all soft sarcomas. 5 The morphological features of angiosarcoma are highly variable, ranging from low-grade tumors resembling benign hemangiomas to high-grade tumors with poor vascular differentiation, which may present as papillary, branching lumen-like, or diffuse sheet-like patterns. Tumor cells can be spindle-shaped, oval, round, polygonal, or flattened epithelioid in appearance. Epithelioid angiosarcoma (EA) is one of these morphological subtypes.
Early reports of primary epithelioid angiosarcoma of thyroid (PEAT) were mostly from the Alpine regions of Europe, particularly in Switzerland, Austria, and Bavaria in Germany. Many patients had a long history of nodular goiter, so it was believed that the rich vascular stroma in nodular goiter was the pathological basis for the development of this disease.6,7 However, it was later found that it could also occur in patients from plain regions with no history of goiter. 8 In the past 30 years, 16 cases of PEAT have been reported in China, from both the southeastern coastal areas and the mountainous regions in the northwest. Similarly, some cases were accompanied by endemic goiter while others were not. So, it is considered that the pathogenic factors may be not solely related to iodine deficiency and that some unknown environmental factors may also be involved. Since 1996, universal salt iodization has implemented in China, and the iodine intake of residents has increased significantly. The patient reported here lived in a non-iodine-deficient region, without the history of nodular goiter and history of exposure to radioactive substances or chemicals, so the pathogenesis of PEAT may involve other unknown factors.
Epidemiological studies have found that the disease is most common in elderly women, with 79.2% of patients being over 60 years old and an average age of 66.1 years. The most common symptom is a rapidly growing neck mass, which may be a longstanding goiter or a newly arisen mass. Other common symptoms include dyspnea, dysphagia, cervicodynia, and weight loss. 9 Imaging examinations are nonspecific and are often used to determine the extent of the lesion. 10
Grossly, masses of PEAT often appear sponge-like with multifocal hemorrhages and unclear boundaries. Histologically, it mainly exhibits characteristics of differentiation toward endothelium as well as epithelioid features. In terms of organizational structure, the tumor appears in solid sheets or branching lumen-like forms. The solid areas resemble undifferentiated carcinoma with densely packed tumor cells and minimal stroma. Reticulin fiber staining clearly shows the contours of immaturely developed blood vessels. In the branching lumen-like areas, irregularly shaped or immature sinus-like lacuna or complex vascular branches can be observed, containing red blood cells and lined with a single layer or multiple layers of endothelium of varying degrees of atypia, with some areas showing papillary structures. The tumor is prone to irregular coagulative necrosis, accompanied by mixed inflammatory cell infiltration and fibrous tissue hyperplasia. The thyroid follicles are infiltrated and destroyed. Cytologically, tumor cells are large, round, oval, or polygonal, with abundant basophilic or eosinophilic cytoplasm, and red blood cells can be seen in cytoplasmic vacuoles. The nuclei are vesicular, located centrally or slightly off-center, with prominent nucleoli, and mitotic figures are easily observed. Immunophenotypes play important roles in diagnosis. CD31, CD34, Ulex europaeus agglutinin-1, FVIIIRA, and Fli-1 are commonly used endothelial markers. CD31 and Fli-1 are more sensitive than CD34 and FVIIIRA, and CD31 is the best endothelial marker in routinely fixed tissues. 11 Compared with the cases reported previously, the case reported here showed more complex vascular branching. Most notably, the tumor cells were positive for CD31, while negative for CD34 and FVIIIRA. The particular morphology and phenotype may be associated with poor differentiation of tumor cells. Under transmission electron microscopy, Weibel-Palade bodies may be observed in some tumor cells while desmosomes and tonofilaments cannot be seen. 12 At present, electron microscopy is not commonly used for the diagnosis of this disease.
PEAT often shows some histological features similar to those tumors such as thyroid anaplastic carcinoma, medullary carcinoma, malignant melanoma, epithelioid sarcoma, and epithelioid hemangioendothelioma, which need to be identified carefully. Anaplastic thyroid carcinoma may also appear in nest-like or sheet-like patterns. However, this neoplasm does not show features of vascular or endothelial differentiation such as complex vascular branching, immature sinus-like spaces, or erythrocytes within the cytoplasm. Meanwhile, thyroglobulin instead of endothelial marker is positive upon immunohistochemical staining. Although medullary thyroid carcinoma has been reported to exhibit sometimes pseudo-angiomatous features, it often shows low atypia, stromal amyloid degeneration and the expression of calcitonin (CT) instead of endothelial markers. Cytoplasm of malignant melanoma may contain melanin granules. The balloon-like tumor cells do not contain erythrocytes. Positive expression of S100, HMB-45, and Melan-A aids in diagnosis of malignant melanoma. Epithelioid sarcoma which commonly occurs in adolescents is rare in the neck, and may present pseudo-angiomatous structures, but does not express endothelial cell marker.13–15 Epithelioid hemangioendothelioma may show a myxoid or sclerotic stroma in which tumor cells are vacuolated and centered by red blood cells. 16 Based on conventional morphology, the diagnostic team had several different diagnoses, including undifferentiated thyroid carcinoma, medullary carcinoma, or malignant melanoma. But after the results of immunophenotype and reticular fiber staining came out, everyone reached a consensus that the diagnosis was PEAT.
The treatment of angiosarcoma may involves surgery, radiotherapy, and chemotherapy, either alone or in combination. Generally, extensive surgical resection combined with other adjuvant therapies is recommended. However, the effectiveness of adjuvant radiotherapy and chemotherapy requires to be further evaluated. 17 The prognosis of epithelioid angiosarcoma depends on the tumor’s location, size, stage, pleomorphism, and the number of mitotic figures. Other indicators of poor prognosis include bleeding, pain, and tumor diameter (greater than 5 cm). 18 Because the tumor grows rapidly and may spread quickly to lungs, bones, soft tissues, lymph nodes, or brain, EA generally has a very poor prognosis.19,20 Most PEAT cases primarily die from extensive metastasis with a postoperative survival period of 5–9 months. 15
Conclusion
It is suggested that primary thyroid epithelioid angiosarcoma, as an extremely rare tumor, has no characteristic clinical manifestations, laboratory and imaging examinations, and that the diagnosis mainly depends on its unique clinical pathological features. Although extensive surgical resection is the preferred treatment, the prognosis is still very poor.
Footnotes
Acknowledgements
The authors are grateful to Zhenbin Gong and Yinhong Gao (Department of General Sugery, San Ai Tang Hospital, Lanzhou) for their kind help, and to Xiaoqiang Wang, Jingling Wu (Department of Imaging, San Ai Tang Hospital, Lanzhou) for her kindly giving some good advice.
Author contributions
XH: the author was actively involved in writing the article, reviewed the literature, and wrote the final draft. LY: the author was actively involved in correcting language and grammar errors, and editing the Figures. GY: the author reviewed the slides, and interpreted imaging studies, and edited the final manuscript. WZ: the author was actively involved in writing the article, reviewed the literature, and wrote the initial draft of the manuscript. LM and FS: the authors provided clinical history, provided patient follow-up, gathered information. HW: the author reviewed the manuscript, and interpreted imaging. DM, LJ, and YL: the authors contributed to the gross and microscopic examination and reviewed the manuscript. All authors contributed substantially by gathering relevant data and clinical details and drafting and revising this manuscript. All authors have reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
The study was approved by the ethics committee of San Ai Tang Hospital and conducted in accordance with the ethical guidelines of the Declaration of Helsinki.
Consent to participate
A verbal consent to participate was obtained from the patients “carer.” A written consent was obtained from the patients “legally authorized representative.”
Consent for publication
All authors have reviewed and approved the final version of the manuscript to publish.
Trial registration
Not applicable, because this article does not contain any clinical trials.
