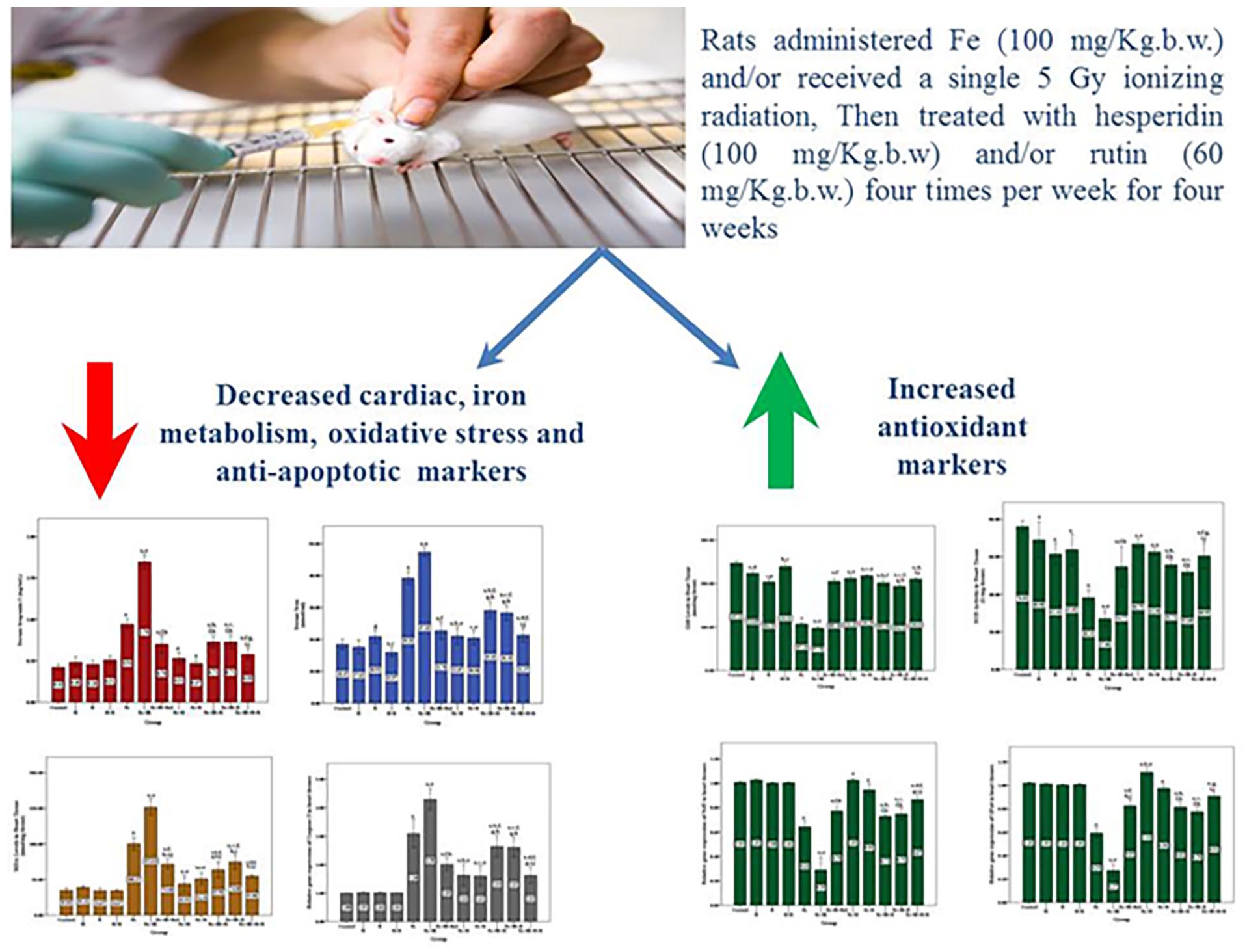
Abstract
Excess iron has been associated with cardiovascular diseases. Flavonoids are antioxidants and cardioprotectants. Therefore, the goal of the current study is to evaluate the anti-apoptotic, antioxidant, and iron-chelating qualities of two flavonoids, rutin (R) and hesperidin (H), as well as their potential to prevent induced ferroptosis in rats. It is an in vivo cross-sectional study, in which rats were divided into 12 groups; control, H, R, H + R, Fe, Fe + IR, Fe + IR + Ref, Fe + H, Fe + IR + H, Fe + R, Fe + IR + R and Fe + IR + H + R. Cardiac and serum iron levels, serum troponin I, creatine kinase-MB (CK-MB), total iron binding capacity (TIBC), transferrin, ferritin, and hepicidin were determined. Moreover, the levels of malondialdehyde (MDA), nitric oxide (NO) and glutathione (GSH) and the activities of superoxide dismutase (SOD) and glutathione peroxidase (GPx), were also determined. The expression levels of DMT1, ACSL4, GPX4, Nrf2, and caspase-3 genes were evaluated by RT-qPCR. Lastly, a histological analysis of the heart tissues from several groups of rats was conducted. After hesperidin and/or rutin treatment, our results revealed that cardiac markers (serum troponin I and CK-MB), iron metabolism markers (serum and cardiac iron, TIBC, ferritin, transferrin, hepicidin and DMT1 expression levels) and oxidative stress markers (MDA, NO and ACSL4 expression levels) were significantly (P ⩽ 0.05) reduced, while the antioxidant markers (GSH level, GPx and SOD activities and GPX4 and Nrf2 expression levels) were significantly (P ⩽ 0.05) increased. Also, hesperidin and rutin exerted its protective anti-apoptotic role by significantly (P ⩽ 0.05) decreasing caspase-3 expression levels. Hesperidin and/or rutin treatment can be proposed as a therapeutic candidate to attenuate ferroptosis.
Introduction
Since iron is vital for many biological processes such as mitochondrial respiration and oxygen transport during evolution, it is a mineral that almost all living cells and creatures require. 1 Because iron deficiency causes anemia and development stop, while iron overload is detrimental due to its redox reactivity, the human body’s iron levels are normally controlled by intricate mechanisms that balance iron absorption, transportation, usage, and excretion. 2 Ferroptosis is a distinct type of iron-dependent controlled cell death that is characterized by aberrant lipid peroxidation, impaired iron homeostasis, and diminished resistance to oxidative stress. According to recent researches, ferroptosis is a major pathogenic factor in a variety of illnesses, including viral, neurodegenerative, inflammatory, and cancer. 3 Additionally, it has been found that ionizing radiation (IR) has a number of detrimental biological consequences, including oxidative damage to biological components such proteins, lipids, DNA, and metabolites, which can lead to cell death through a number of mechanisms, including ferroptosis. 4
Although some studies found a correlation between body iron status and cardiovascular diseases (CVD) and support the role of iron in atherosclerosis, others found no association at all, making the relationship between iron overload and CVD—including coronary artery disease (CAD) and myocardial infarction (MI)—more complicated and unclear. 5 In 1981, Sullivan 6 first proposed the idea that iron overload may contribute to heart disease, as he reported that chronic iron deficiency or depletion has a key preventive effect against ischemic heart disease, while iron overload causes cardiovascular disease. Recently, Aierken et al. 7 reported that the inhibition of Slc39a14/Slc39a8 proteins reduce vascular calcification via alleviating iron overload induced ferroptosis in vascular smooth muscle cells. Moreover, several studies found a positive correlation between the risk of CVD and serum iron, ferritin, or transferritin saturation.8–12 Ferroptosis is now better recognized as a major mechanism mediating the etiology and development of a number of cardiovascular diseases than it was decade ago. Iron overload cardiomyopathy is also the predominant cause of death and a substantial comorbidity in patients with early hemochromatosis.13,14 As a result, there is currently a lot of focus on food bioactive component research in an attempt to develop new functional foods and nutraceuticals that target and/or reduce CVD risk factors.
Flavonoids are a ubiquitous class of polyphenolic substances typically found in seeds, fruit peels, bark, and flowers, and they are present in most plants. The beneficial pharmacological properties of flavonoids, such as cardiovascular protection and anticancer, antibacterial, and antimutagenic action, have been the focus of several studies conducted in recent years. 15 Hesperidin is a flavonoid belonging to the flavanone group that is found in a variety of plants, particularly citrus fruits. Its significance in health management has been acknowledged due to its antioxidant, anti-inflammatory, and anti-cancer properties, as well as its ability to stop several types of pathogenesis. 16 While rutin is one of the most intriguing naturally occurring flavonoids, which is found in a variety of plants, including apples, buckwheat, tea, and passionflower, has cardioprotective properties. 17 Therefore, the present study aims to evaluate the ameliorative effect of two flavonoids, hesperidin and rutin, and also compare their activities as iron chelating, antioxidant and anti-apoptotic agents against induced-ferroptosis due to iron overload and radiation in rats.
Materials and methods
Chemicals
For this study, the following supplies were acquired: hesperidin and rutin from Sigma Aldrich Chemical Co., St. Louis, Mo., USA; elemental iron from Novartis Pharmaceuticals Corporation, East Hanover, NJ; and deferasirox from Amoun Pharmaceutical Co. SAE—Egypt.
Experimental animals
Adult male albino rats weighing 120–150 g were used in this investigation. They were purchased from Nile Pharmaceutical Co. in Cairo, Egypt. The rats were kept under standard laboratory settings, which included a 12-hour light/dark cycle, 25 ± 2°C, and 60 ± 5% humidity. They were housed in cages with unlimited access to food and water, and were fed pellet diets and sterile water. The typical pelleted rat chow is bought from Cairo, Egypt’s El Gomhorya Company. Every animal procedure approved by the National Center for Radiation Research and Technology’s ethics committee (80A/23).
Experimental design
A total of 96 male albino rats were equally divided randomly into the following 12 groups:
Group 1: Control (C), were eight healthy untreated rats.
Group 2: Hesperidin (H), included eight rats that were orally administered with hesperidin (100 mg/kg b w) four times per week for 4 weeks, according to Aalikhani et al. 18
Group 3: Rutin (R), included eight rats that were orally administered with rutin (60 mg/kg b w) four times per week for 4 weeks, according to Hawula et al. 19
Group 4: H + R, included eight rats that were simultaneously orally administered with hesperidin as in group 2 and rutin as in group 3.
Group 5: Iron (Fe), included eight rats that were intraperitoneally administered elemental iron (100 mg/kg b w) three times per week for 4 weeks to induce the toxicity of iron according to Alikhani et al. 20
Group 6: Fe + IR, included eight rats that were administrated elemental iron as in group 5 and then received a single 5 Gy radiation treatment in order to investigate the role of ionizing radiation in causing iron overload and, ultimately, ferroptosis.
Group 7: Fe + IR + Ref, included eight rats that were administrated elemental iron as in group 5, then exposed to radiation as in group 6, and finally treated with Deferasirox as a reference iron chelator (orally administered 100 mg/kg b w three times per week for 4 weeks) according to Adel et al. 21
Group 8: Fe + H, included eight rats that were administrated elemental iron as in group 5, and then treated with hesperidin as in group 2.
Group 9: Fe + R, included eight rats that were administrated elemental iron as in group 5, and then treated with rutin as in group 3.
Group 10: Fe + IR + H, included eight rats that were administrated elemental iron as in group 5, then exposed to radiation as in group 6, and finally treated with hesperidin as in group 2.
Group 11: Fe + IR + R, included eight rats that were administrated elemental iron as in group 5, then exposed to radiation as in group 6, and finally treated with rutin as in group 3.
Group 12: Fe + IR + H + R, included eight rats that were administrated elemental iron as in group 5, then exposed to radiation as in group 6, and finally treated with hesperidin as in group 2 and rutin as in group 3 at the same time.
Tissues and blood samples
After 24 h following the last treatment, all rats that had been anesthetized and scarified had their heart tissues and blood samples collected. The serum required for biochemical analysis was extracted from blood samples by centrifuging them for 10 min at 4000 rpm after they had coagulated. The heart tissues were removed from the rats, cleaned in freezing water, dried, and separated into three sections. After being cleaned with saline, one portion was preserved in 10% formalin for histological analysis. Alternatively, the second portion was homogenized in phosphate-buffered saline using a glass homogenizer and the resulting homogenates were used to measure the levels of iron, MDA, NO, and GSH as well as the activity of SOD and GPx4. To assess the levels of gene expression of divalent metal transporter (DMT1), nuclear factor-erythroid factor 2-related factor 2 (NRF2), acyl-CoA synthetase long-chain family member 4 (ACSL4), caspase 3, and glutathione peroxidase 4 (GPX4), the final portion of heart tissues was used for RNA extraction.
Serum cardiac and iron metabolism markers
The level of creatine kinase-MB (CK-MB) in sera of the different studied groups was determined using kinetic BioMed-CK-MB kit purchased from EGY-CHEM for lab technology Company (Badr City, Industrial Area Piece 170 250 Fadan In East of Elrubaki, EGYPT). While the Iron levels were measured using a colorimetric iron assay kit purchased from Biovision Company (Biovision Incorporated 155 S. Milpitas Boulevard, Milpitas, CA 95035, USA), and 50 µL of homogenates and 50 µL of sera were used to determine the levels of iron in cardiac tissues and sera, respectively. While serum troponin I, TIBC, ferritin, transferrin, and hepicidin were determined in using My BioSource ELISA rat TIBC, ferritin, transferrin, and Elabscience ELISA rat hepicidin kits, respectively.
Heart antioxidant and oxidative stress markers
The levels of GSH, MDA and NO and the activity of SOD in the heart tissues of the different studied groups were determined in heart tissue using colorimetric kits (Bio-diagnostic kits). While the activity of GPX4 was determined using ELISA kits (My BioSource ELISA rat GPX4 activity).
Levels of gene expression of GPX4, NRF2, DMT1, ACSL4, and caspase3
The RT-qPCR was utilized to assess the expression levels of the GPX4, NRF2, DMT1, ACSL4, and caspase3 genes. First, 30 mg of cardiac tissue was treated with the Gene JET RNA Purification Kit (Thermo Scientific) to extract total RNA. Next, 1 μg of RNA template and reverse transcriptase (EntiLinkTM) were used to create first-strand complementary DNA (cDNA). Using Sequence Detection Software (PE Biosystems, CA), RT-qPCR was carried out in a Step One Plus heat cycler (Applied Biosystems, USA). Table 1 lists the primers that were used for the GAPDH gene with the GPX4, NRF2, DMT1, ACSL4, and caspase3 genes. Briefly, the reaction was performed as follows: 900 nM of each primer, 2 μL of cDNA, and 2×SYBR Green PCR Master Mix (Applied Biosystems) make up a reaction mixture with a total volume of 25 μL. A 95°C starting step was followed by 40 cycles of 20 s at 95°C, 30 s at 60°C, and 20 s at 72°C during the PCR thermal cycling process. Using the 2−ΔΔCT approach as outlined by Livak and Schmittgen, 22 the relative mRNA expression of the genes under study was assessed.
List of primers used for quantitative real-time polymerase chain reaction.

Histopathological examination
Samples of heart tissue were taken from each animal group and preserved in 10% neutral buffered formalin. Following Bancroft et al. 23 instructions, the fixed specimens were trimmed, cleaned, and dehydrated in increasing alcohol grades. They were then clarified in xylene, imbedded in paraffin, sectioned at 4–6 μm thickness, and stained with hematoxylin and eosin. According to Acikel et al., 24 the frequency and severity of the cardiac tissue specimens were divided into the following degrees: (0) Nothing has changed: (1) Moderate: significant myofibrillar degeneration and/or diffuse inflammatory process; (2) Severe: necrosis along with the diffuse inflammatory process; (3) Mild: focal myocyte destruction or modest multifocal degeneration with a low level of inflammation.
Statistical analysis
The statistical analysis was conducted using IBM SPSS software (version 26.0; IBM Corp., Armonk, NY, USA), and the findings were displayed as means ± SE. The data were evaluated using a one-way analysis of variance (ANOVA), and a post hoc test (LSD) was employed for multiple comparisons. A significance level of P < 0.05 was applied.
Results
Hesperidin and rutin could reduce cardiac biomarkers
Our results revealed that the levels of serum troponin I and CK-MB were significantly increased in Fe and Fe + IR groups compared to control group, with P < 0.001. While after hesperidin and/or rutin treatment, the levels of serum troponin I and CK-MB were decreased again; as shown in Figure 1.

Effect of hesperidin and rutin on cardiac markers. (A) Serum troponin I. (B) Serum CK-MB.
Hesperidin and rutin could chelate and reduce iron metabolism markers
For hesperidin, our results revealed that it could significantly decrease cardiac iron (4.6 ± 0.6, 5.4 ± 0.4 µmol/g), serum iron (21.1 ± 1.1, 29.1 ± 1.0 nmol/mL), TIBC (65.3 ± 2.3, 70.2 ± 2.4 μmol/mL), transferrin (37.1 ± 0.9, 42.2 ± 0.9 ng/mL), ferritin (139.5 ± 1.0, 159.5 ± 1.1 ng/mL), hepicidin (88.9 ± 1.5, 103.9 ± 1.5 ng/mL) and DMT1 expression levels (1.3 ± 0.2, 3.3 ± 0.2) in Fe + H, and Fe + IR + H groups; respectively. Also, according to rutin, these biomarkers were significantly decreased as cardiac iron (4.4 ± 0.4, 4.6 ± 0.2 µmol/g), serum iron (20.5 ± 0.8, 28.3 ± 0.9 nmol/mL), TIBC (62.0 ± 1.8, 67.9 ± 1.0 μmol/mL), transferrin (35.3 ± 0.9, 40.3 ± 0.9 ng/mL), ferritin (129.5 ± 2.7, 146.7 ± 1.4 ng/mL), hepicidin (80.0 ± 2.8, 105.1 ± 2.7 ng/mL) and DMT1 expression levels (1.1 ± 0.1, 3.1 ± 0.1) in Fe + R and Fe + IR + R groups; respectively, when compared to cardiac iron (6.6 ± 0.2, 7.7 ± 0.2 µmol/g), serum iron (39.2 ± 0.8, 47.3 ± 0.5 nmol/mL), TIBC (90.8 ± 1.8, 110.7 ± 3.7 μmol/mL), transferrin (60.9 ± 1.5, 65.8 ± 1.6 ng/mL), ferritin (202.0 ± 3.1, 241.2 ± 2.6 ng/mL), hepicidin (125.9 ± 3.1, 149.2 ± 2.6 ng/mL) and DMT1 expression levels (3.9 ± 0.2, 6.7 ± 0.1) in Fe and Fe + IR groups; respectively, as shown in Figure 2. It is noteworthy that both hesperidin and rutin were good iron chelators and there were no significant difference between their effect but when treated as a combination more potent effects were taken when compared with Deferasirox which is an iron chelator and used as a reference in this experiment (Figure 2).
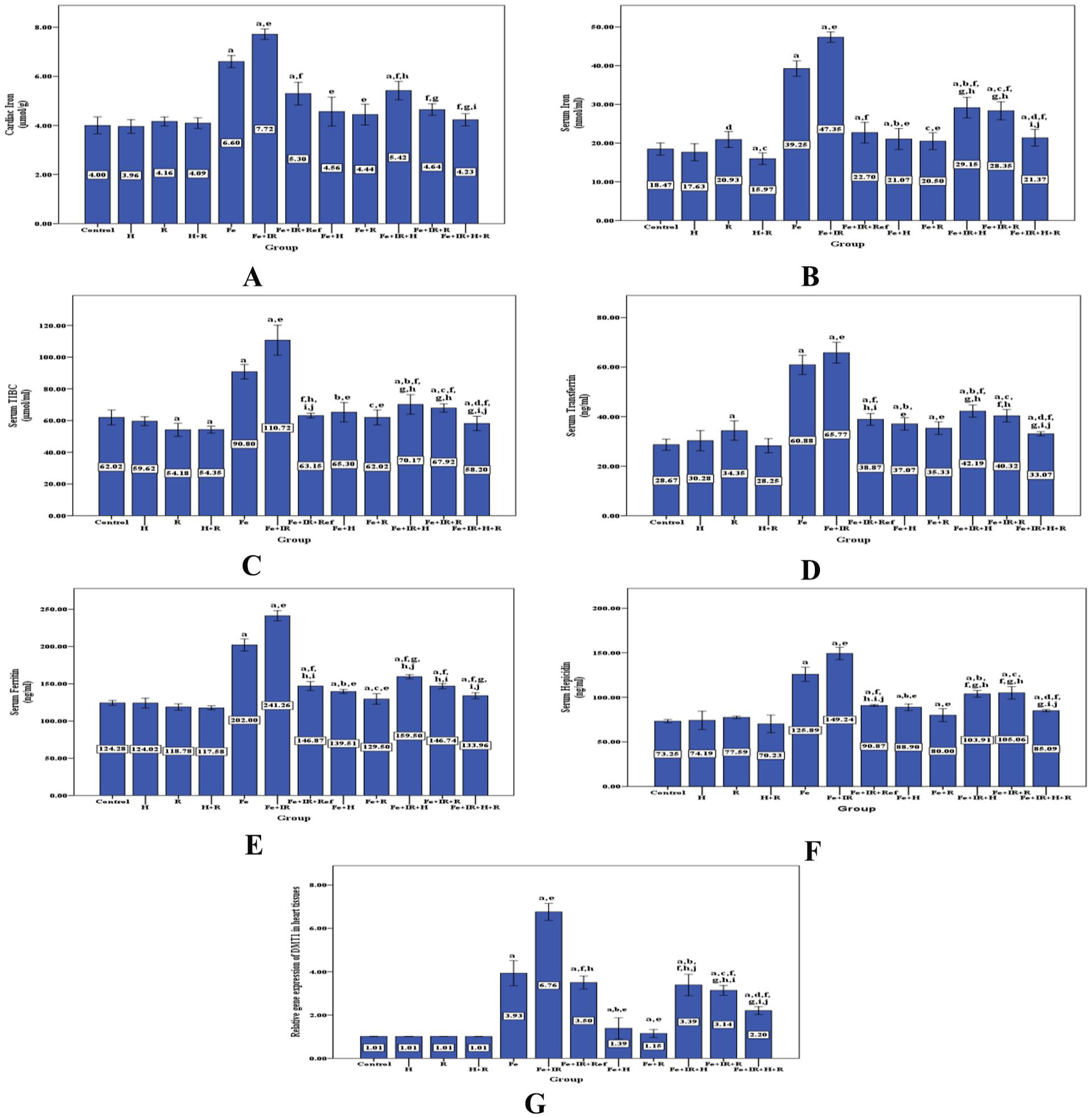
Effect of hesperidin and rutin on iron metabolism markers. (A) Cardiac iron. (B) Serum iron. (C) Serum TIBC. (D) Serum transferrin. (E) Serum ferritin. (F) Serum hepicidin. (G) Relative gene expression of DMT1 in heart tissues.
Hesperidin and rutin act as antioxidant agents
Our results showed that both hesperidin and rutin had antioxidant and free-radical scavenging properties as after the treatment of hesperidin the levels of heart MDA (43.9 ± 4.3, 63.9 ± 4.4 nmol/mg tissue), NO (26.5 ± 2.2, 46.7 ± 2.3 nmol/mg tissue) and ACSL4 expression levels (1.2 ± 0.1, 2.2 ± 0.1) were significantly decreased (Figure 3); while the levels of heart GSH (212.2 ± 2.8, 202.2 ± 2.8 nmol/mg tissue), SOD (66.8 ± 0.9, 55.7 ± 0.9 U/mg tissue), GPx4 (142.2 ± 0.8, 119.0 ± 0.8 U/mg tissue), Nrf2 expression levels (1.0 ± 0.03, 0.9 ± 0.01, 0.7 ± 0.04) and GPx4 expression levels (1.1 ± 0.01, 0.8 ± 0.01) were significantly increased in Fe + H and Fe + IR + H groups; respectively (Figure 4). As well after the treatment of rutin, the levels of MDA (51.3 ± 4.4, 74.9 ± 3.9 nmol/mg tissue), NO (23.7 ± 1.9, 43.9 ± 1.8 nmol/mg tissue) and ACSL4 expression levels (1.4 ± 0.1, 1.9 ± 0.1) were significantly decreased (Figure 3), while the levels of GSH (204.2 ± 2.0, 194.3 ± 2.0 nmol/mg tissue), SOD (62.5 ± 0.6, 51.9 ± 0.7 U/mg tissue), GPX4 (140.7 ± 2.9, 119.3 ± 1.5 U/mg tissue), Nrf2 expression levels (0.9 ± 0.02, 0.7 ± 0.01), and GPx4 expression levels (0.9 ± 0.01, 0.8 ± 0.01) were significantly increased in Fe + R and Fe + IR + R groups (Figure 4); respectively, when compared to Fe and Fe + IR groups. By comparing hesperidin and rutin with Deferasirox as an antioxidant, it was found that no significant change between them, but when administrated as a combination a great effect was obtained which exceeded Deferasirox effect itself.

Effect of hesperidin and rutin on oxidative stress markers in the heart tissues of rats. (A) MDA levels. (B) NO levels. (C) Relative gene expression of ACSL4.

Effect of hesperidin and rutin on antioxidant markers in the heart tissues of rats. (A) GST levels. (B) GPx activity. (C) SOD activity. (D) Relative gene expression of GPx4. (E) Relative gene expression of Nrf2.
Anti-apoptotic effect of hesperidin and rutin
The present study was conducted to investigate the anti-apoptotic effect of hesperidin and rutin by investigating the expression level of caspase-3 in the heart tissues of rats. Our results revealed that the level of expression of caspase-3 was significantly increased in Fe and Fe + IR groups compared to control group, with P < 0.001. While after hesperidin and/or rutin treatment, the level of expression of caspase-3 was decreased again; as shown in Figure 5.
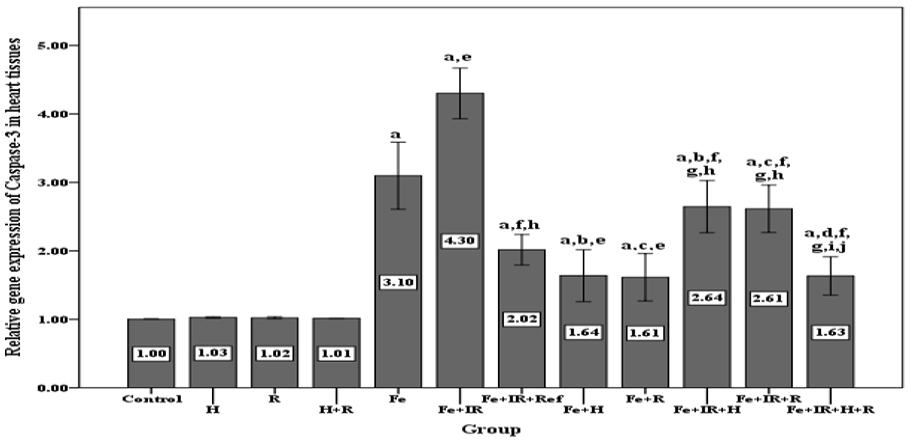
Effect of hesperidin and/or rutin on caspase-3 gene expression levels in the heart tissues of rats.
Histopathological findings
Tissue specimens of heart from control (Figure 6A), hesperidin (Figure 6B), rutin (Figure 6C), hesperidin and rutin treated (Figure 6D) showed normal histological structure of myocardial muscle. Short branches that joined to form a network of muscle fibers were seen. Myocardial fibers exhibited normal arrangement and distinct striations, with no signs of degeneration or necrosis. The muscle fibers seemed to be short branches that joined together to form a network. There was no visible degeneration or necrosis, and the cardiac fibers were organized consistently with distinct striations. There was no change in the degree of muscle injury in the heart (0). The Fe group’s cardiac tissue slice had an abnormal amount of eosinophilic intermuscular oedema. Moderate to substantial myofibrillar degeneration (degree 2) was demonstrated by derangement of cardiac fibers with degree of myocardial muscle injury (Figure 6E). Conversely, the Fe + IR group’s heart tissue portion displayed oedema and hemorrhages between the myocardial muscle bundles, that is, dilatation and congestion of the coronary branch, primarily from lymphocytes and macrophages, with a little infiltration of mononuclear cells (degree 3) (Figure 6F). In contrast, the muscle tissue sections of the animal group Fe + IR + R showed signs of intermuscular oedema together with a low level of leukocytic infiltration. The degree of cardiac muscle injury was found to be either modest multifocal degeneration or mild focal myocyte damage (degree 1) (Figure 6G). Cardiac muscle tissue sections of animals’ groups Fe + H, Fe + IR + H, Fe + R, Fe + IR + R revealed oedema, a dilatation of the cardiac fibers, and a buildup of blood capillaries between the muscles. The degree of cardiac muscle injury was found to be either modest multifocal degeneration or mild focal myocyte damage (degree 1) (Figure 6H–K). On the other hand, animals’ group Fe + IR + H + R muscular tissue sections showed normal histological structure of myocardial muscle (degree 0) (Figure 6L).

Photomicrograph of rat heart tissue of: (A) Control group. (B) H group. (C) R group. (D) H + R group, displaying the cardiac muscle’s normal histological structure (degree 0). (E) Fe group, displaying excessive eosinophilic inter-muscular oedema (degree 2). (F) Fe + IR group, exhibiting bleeding and oedema between the cardiac muscle bundles, coronary branch dilatation and congestion, and a small number of mononuclear cell infiltrations, primarily macrophages and lymphocytes (degree 3). (G) Fe + IR + Ref group, exhibiting minimal levels of leukocytic infiltration and intermuscular oedema (degree 1). (H) Fe + H. (I) Fe + IR + H. (J) Fe + R. (K) Fe + IR + R, groups, displaying intermuscular blood capillary congestion and myocardial fiber derangement with oedema (degree 1). (L) Fe + IR + H + R group, exhibiting normal histological structure of cardiac muscular fibers (degree 0) (H&E X200).
Discussion
Excess iron stored in the body can lead to iron overload and the accumulation of iron can cause damage to the heart specifically. 13 Iron chelation is actually the main treatment for patients with iron overload, including those with β-thalassemia. Deferasirox, deferiprone, and deferrioxamine are the three main iron chelators that have been shown to reduce iron accumulation in heart tissue and, as a result, enhance cardiac function in these patients. 25 However, these medications have side effects that include elevated liver enzymes, nausea, vomiting, and abdominal pain. 26 Therefore, our study aimed to evaluate the therapeutic effects of hesperidin and rutin against iron overload and radiation induced ferroptosis in rats using deferasirox as a reference iron chelator.
Our results showed that the treatment of hesperidin and/or rutin caused a significant decrease in the levels of cardiac and iron metabolism biomarkers in iron loaded and/or ionizing radiation exposed rats, which indicate that both hesperidin and rutin have iron chelating activities (Figures 1 and 2). According to Chen et al., 27 the flavonoids were classified depending on unsaturation degree, chemical structure and carbon ring oxidation into seven groups; anthocyanins, chalcones, flavones, isoflavones, flavanones, flavanols and flavonols. Hesperidin (6-O-α-L-rhamnopyranosyl-β-D-glucopyranoside) is a flavanone glycoside, while rutin (3′, 4′, 5, 7-tetrahydroxyflavone-3-rutinoside) is a complex of disaccharide rutinose and flavonol quercetin. 28 Hesperidin and rutin both exhibit a variety of biological effects, such as anti-inflammatory, anti-cancer, antioxidant, and radioprotective properties. 29 The iron chelation properties of hesperidin and rutin were reported by several studies.29–31 The ability of hesperidin to chelate iron and reduce the iron metabolism biomarkers was similar to that of rutin and Deferasirox as there were no significant differences between them, but when treated as a combination more potent effect obtained which was exceeded the effect of Deferasirox itself (Figure 2). The obtained data were in agreement with the study of Pari et al. 32 who found that hesperidin could reduce the levels of iron in blood in iron-induced hepatic and renal toxicity in rats. Additionally, Gao et al. 33 discovered that rutin could dramatically lower mice’s liver iron levels. Our findings corroborated those of Aziza et al., 34 who demonstrated that in iron overloaded male albino rats; the treatment of rutin could considerably lower the levels of serum iron, TIBC, ferritin and transferrin protein. Hawula et al. 19 also noted that supplementation with rutin might dramatically lower serum and liver iron levels, as well as serum transferrin saturation and hepatic ferritin protein expression. As consistent with our results, DMT1 is the sole intracellular iron transporter that governs intracellular iron metabolism and induces ferroptosis; blocking DMT1 may alter iron intake and thereby inhibiting ferroptosis. 35 Also, Zeng et al. 36 found that iron overload can induce ferroptosis via promoting DMT1 expression.
According to our results, hesperidin and rutin ameliorated the induced-ferroptosis in rats via decreasing oxidative stress and increasing the antioxidant biomarkers (Figures 3 and 4). As consistent with our results Estruel-Amades et al. 37 suggested that hesperidin could act as an antioxidant by reducing the production of reactive oxygen species (ROS) by peritoneal macrophages, and increasing the activities of SOD, GPx and catalase activities in spleen, thymus and liver, preventing the oxidative stress induced by exercise and improve exercise performance. Also, Hassan et al. 38 reported that hesperidin could counteract diclofenac-induced hepatotoxicity in male wistar rats through its antioxidant, anti-inflammatory, and antiapoptotic activities. In addition, Chen et al. 39 and Kaplana et al. 40 reported that the treatment of hesperidin could reduce the lipid peroxidation byproducts in the erythrocyte membrane of human, measured as MDA and thiobarbituric acid-reactive substances (TBARS), respectively. Furthermore, El-Sayed et al. 41 found that hesperidin (200 mg/kg/day) supplementation improved the changes in brain lipid peroxidation caused by acrylonitrile and increased the reduction in GSH, SOD, CAT, GPx, and glutathione-s-transferase (GST) levels caused by acrylonitrile. Regarding to rutin, Iova et al. 42 observed that rutin oral administration could reduce the oxidative stress and enhance the antioxidant status in hyperglycemic periodontitis rats. In addition, Rahmani et al. 17 reported that rutin had numerous pharmacological effects on the heart, liver, and kidneys, mainly through its antioxidant effect by enhancing the activity of enzymes such as SOD, GST, GGT, CAT, and GPx. Furthermore, Abarikwu et al. 43 discovered that in rats with cadmium and ethanol-induced liver damage, rutin restored the levels of MDA, GSH, and GST, and the activities of CAT, GPx, SOD enzymes.
Nuclear factor-erythroid-derived 2-related factor 2 (Nrf2) is an essential transcription factor that regulates the cellular responses to oxidative stress. It does this by activating genes that are dependent on the antioxidant response element (ARE), which prevents the production of ROS. 44 Ferroptosis is regulated by GPx4, the sole enzyme capable of lowering lipid hydroperoxidation in biological membranes. 45 Consequently, our results suggested that hesperidin and rutin inhibiting ferroptosis through Nrf2/GPx4 axis. This finding is consistent with Yuan et al. 46 who reported that kaempferol could suppress ferroptosis kaempferol by boosting the Nrf2/GPx4 signaling pathway, suggesting that the Nrf2/GPx4 signaling pathway may play a role in ferroptosis. Also, Wu et al. 47 reviewed that the activation of Nrf2 signaling is a key molecular mechanism of protection against cardiovascular diseases by natural products. Additionally, Zhu et al. 48 indicated that hesperidin may prevent degeneration in human nucleus pulposus cells by inhibiting ferroptosis through enhancing Nrf2 expression. Moreover, Tabolacci et al. 49 demonstrated that rutin could play a crucial role against UVA-induced skin damage through a activation of the Nrf2/GPx4 signaling pathway.
A member of the Acyl-CoA synthetase long-chain family (ACSLs), Acyl—CoA synthetase long-chain family member 4 (ACSL4) is capable of converting both saturated and unsaturated fatty acids. By interacting long-chain polyunsaturated fatty acids with coenzyme A, ACSL4 produces long-chain fatty acyl-CoA esters, which initiates lipid peroxidation and may serve as a ferroptosis biomarker.50,51 According to our results, hesperidin and rutin ameliorating the induced-ferroptosis rats in part via ACSL4 down expression (Figure 3C). By deactivating the ACSL4 enzyme or downregulating the ACSL4 gene, ferroptosis is avoided and the quantity of substrates needed for lipid peroxidation is decreased.52,53
Lee et al. 54 reported that ferroptosis induces apoptosis. Also, Shokoohi et al. 55 revealed that ROS generation could induce apoptosis. Caspases, are proteolytic enzymes which cleave peptide bonds after aspartate residues, have a crucial in apoptosis. Caspase-3 is executioner caspase which is a common mediator of extrinsic and intrinsic apoptotic pathways. 56 Our results revealed that the expression level of caspase-3 was significantly increased in Fe, and Fe + IR groups compared to the control group, while after hesperidin and/or rutin treatment in Fe + H, Fe + IR + H, Fe + R, Fe + IR + R, Fe + IR + R + H groups, its expression level was decreased again (Figure 5). This is consistent with the findings of Ali et al. 57 who demonstrated that hesperidin and/or rutin treatment could effectively reduce caspase-3 expressions in paclitaxel-administered rats. Moreover, our results were in agreement with Shokoohi et al. 58 who found that the hesperidin treatment could reduce apoptosis through its antioxidant properties. Also, Gur et al. 59 demonstrated that in the testicular tissues of the abamectin-induced testicular toxicity rats, hesperidin administration considerably enhances the expression of the anti-apoptotic gene Bcl-2 and significantly lowers the expression of Bax and Caspase-3. This indicates that hesperidin and rutin exerted their protective role through the inhibition of apoptosis, that is, anti-apoptotic effects.
In light of the established iron chelation, antioxidant, and antiapoptotic properties of rutin and/or hesperidin, we advise their use in clinical trials to treat iron overload in patients, particularly those with β-thalassemia. However, our research had certain drawbacks. First, because of the limited sample size of the study, these findings require confirmation through larger research. Second, while there are many different ways that rutin may lessen ferroptosis, our current study focused only on its function in iron chelation, ROS generation inhibition, and lipid peroxidation. Third, the study only evaluated the anti-ferroptosis characteristics of rutin and did not investigate its effects on particular processes by gene transfection or deletion. Finally, additional methods like immunofluorescence (IF), western blot, FACS, or immunohistochemistry (IHC) for ferroptosis-related markers, especially Nrf2, DMT1, ACSL4, GPX4 and caspase3 proteins, will support our findings and validate them. Histological analysis of all vital organs should be performed, and the outcomes should be contrasted with those of mice that are healthy and in control. To find out how rutin and/or hesperidin prevent ferroptosis, more research is required.
Conclusions
According to the data indicated above, hesperidin, rutin, and their combination—which was the most effective—counteract iron excess and/or ionizing radiation-induced ferroptosis in rat heart tissue by iron chelation, antioxidant, and antiapoptotic effects. Hesperidin, rutin, or their combination is therefore advised by this study to alleviate the adverse effects of medications like Deferasirox.
Footnotes
Author contributions
All authors were involved in conceiving the study and in protocol development. H. Abdullah, F. Moawed and E. Ahmed measured the biochemical and molecular parameters. H. Abdullah, R.A. Haroun and F. Abdel Hamid analyzed the data. F. Abdel Hamid and R.A. Haroun wrote the first draft of the manuscript. All authors revised and approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethicals approval
The current study was approved by the ethics committee of the National Centre for Radiation Research and Technology (NCRRT), Egyptian Atomic Energy Authority, Cairo, Egypt (The approval No. 89A/21).
Animal welfare
The adult male albino rats used in this investigation weighed around 120–150 g. They were purchased from Nile Pharmaceutical Co. in Cairo, Egypt. The animals were kept under conventional laboratory settings, which included a 12 h light/dark cycle, a temperature of 25 ± 2°C, and a humidity of 60% ± 5%. They were housed in cages with free access to food and drinking water, and they were provided pellet diets and sterilized water. The regular pelleted rat chow bought from El Gomhorya Company, Cairo, Egypt, has 65% carbs (corn starch 15% and sucrose 50%), 20.3% proteins (casein 20% and DL-Methionine 0.3%), 5% fat (corn oil 5%), 5% fibers, 3.7% salt mixture, and 1% vitamin mixture. The National Center for Radiation Research and Technology’s (80A/23) ethics committee was followed in all animal procedures.
Availability of data of material
In this published article, all data created or analyzed during this investigation are included.
