Abstract
The aim of the study was to investigate the influence of naringenin (NGN) and its methylated derivatives (50 or 100 mg kg−1) on finasteride-caused depression-like performance in mice to identify the effects on behavior and biomarkers of inflammation in the management of depression. Depression-like behavior was induced by repeated dose of finasteride (100 mg kg−1, subcutaneously) in mice. The effects of the naringenin (50 or 100 mg kg−1) or its methylated derivatives (Ngn-M; 50 or 100 mg kg−1 or Ngn-DM; 50 or 100 mg kg−1) and duloxetine (DXT, 10 mg kg−1) were evaluated for the immobility time in tail suspension and forced swimming tests following finasteride pre-treatment. The levels of brain pro-inflammatory cytokines such as IL-1β and TNF-α were also measured by Enzyme-Linked Immunosorbent Assay to further evaluate the impact of naringenin and its methylated derivatives on inflammation. Pre-treatment with finasteride substantially increased both the immobility time spent in tail suspension and forced swimming tests and brain levels of IL-1β and TNF–α in mice. Doluxetine (DLX) was given at a dose of 10 mg kg−1, and Naringenin or its methylated derivatives were given at doses of 50 and 100 mg kg−1 orally. It reduced immobility time in both tests, restored the preference to sucrose solution, and normalized cytokine levels (p < 0.01) in mice. Similar effects were observed with DTX (10 mg kg−1) as positive control. The increased brain levels of malondialdehyde (MDA) or nitrite were considerably (p < 0.05) decreased while substantially (p < 0.05) increased glutathione (GSH), superoxide dismutase (SOD), and catalase (CAT) levels after finasteride pre-treatment relative to vehicle-control by naringenin or its methylated derivatives (50 or 100 mg kg−1). These findings demonstrated the potential for methylated flavonoids as safe and effective anti-depressive agents.
Introduction
Depression seems to be the most debilitating psychiatric disorder in the world, with high rates of morbidity and mortality. 1 Neuroinflammation in depression has piqued researchers’ interest in the last 20 years. Inflammatory markers are elevated in most of the patients with major depression. Furthermore, cytokines have been shown to have an effect on several pathways involved in the depression’s pathogenesis such as the hippocampal neurogenesis inhibition, synaptic modulation of hypothalamic–pituitary–adrenal activation, and changes in neurotransmission. 2 As a result, inflammation and inflammatory markers assessment can help us better understand depression’s pathophysiology and treatment.
Finasteride is a steroidal molecule that prevents the transformation of deoxy-corticosterone to dihydrodeoxycorticosterone and testosterone to dihydrotestosterone by inhibiting type-II 5-reductase enzyme. This enzyme helps to produce allopregnanolone, an active substance in the synthesis of neurosteroids, which has an anti-depressant and anxiolytic effect.3 Finasteride is primarily employed to treat benign prostatic hyperplasia (BPH) and male androgenetic alopecia (MAA). 4 The neuropsychiatric effects of finasteride lasted for a long time after the medication was discontinued. 5 Psychological changes such as cognitive deficits, anxiety, a lowered emotional threshold, and sensitivity to the consequences of brain injuries are termed as “post-finasteride syndrome” which influences functional impairment. 3 Thus, patients taking finasteride have been reported to have increased anxiety and a depressed mood. 4 The basic mechanisms that cause finasteride treatment to have an effect on human’s brain function are unknown. A recent research in rats found a depression-like behavior following withdrawal for a month after 20 days of finasteride administration with a decreased hippocampal neurogenesis. 5 Romer et al. 5 demonstrated that 7 days of finasteride (100 mg kg−1, s. c.) administration decreased hippocampal neurogenesis in adult rats. Therefore, adult neurogenesis in the hippocampus has been associated with depression-like behavior.4,5 Thus, following the short-term administration of finasteride, which results in depression-like behavior, researchers can assess the effects of anti-depressants of bioactive compounds.
Flavonoids and their glycosides were shown to have effective anti-inflammatory actions on the brain, either by scavenging free radicals or directly modulating major elements of the neurohumoral pathway. 6 Several depression animal models of rats or mice involving behavioral, learned helplessness, and prolonged stress showed that the administration of flavonoids could reverse depression-like behavior. 7
In the literature, naringenin was displayed to have a number of CNS activities, implying that it could have therapeutic potential in brain disorders. 8 Earlier research revealed that naringenin enhanced signaling of insulin from the brain and its cognitive functions, as well as reducing dementia associated with neurodegenerative diseases caused by administrating streptozotocin into the brain. 9 The anti-oxidant and anticholinesterase activities of naringenin on brain’s hippocampus have been linked to a reduction in type-2 diabetes-induced memory loss. 8 Naringenin was able to lower β-amyloid levels and reduces inflammation in the hippocampal regions of the brain. 10 Furthermore, the reduction of pro-inflammatory cytokines as well as the neuroprotective activity of naringenin has been shown beneficial in the treatment of chronic inflammatory disorders such as Alzheimer’s disease. 11 Naringenin suppressed the NF-kappa B signaling pathway to protect against stroke in an experimental model. It was reported that inhibiting NF-κB-mediated neuroinflammation improved cognitive disorders in ischemic brains. In addition, by inhibiting oxidative stress, naringenin showed neuroprotective effects toward 6-hydroxydopamine and iron overload-induced neurotoxic effects. Furthermore, naringenin has been shown to have anti-depressant properties by inhibiting monoamine oxidase, a well-known therapeutic molecule in the management of depressive disorders. 12 The inhibition of NF-κB activation and decreased caspase-3 activation, as well as a decrease in pro-inflammatory marker concentrations, has been attributed to the possible benefits of naringenin in persistent neurological disorders linked to oxidative damage and neuroinflammation. 13
Although metabolic instability with reduced oral bioavailability limits flavonoids’ pharmaceutical potential, it has been proposed that methylation of free hydroxyl groups can considerably improve metabolic stabilization and transport across the membranes, promoting absorption and dramatically enhancing oral bioavailability. 14
Thus, the antidepressant activity of naringenin (Ngn) and its unique methylated derivatives, such as naringenin 7-O-methyl ether (Ngn-M) and naringenin 4′, 7-dimethyl ether (Ngn-DM), was investigated on repeated administration of finasteride for 6 days since no scientific evidence involving the use of methylated derivatives of naringenin against depression is reported so far.
Additionally, the influence of Ngn and Ngn-methylated derivatives (50 or 100 mg kg−1) was investigated on finasteride-induced depression-like behavior in mice (C57Bl/6). Furthermore, the effects of Ngn and Ngn-methylated derivatives (Ngn-M or Ngn-DM) were examined on the levels of interleukin-1β (IL-1β) and tumor necrosis factor (TNF-α) in the brains of mice, with the aim to illustrate the function of these cytokines in the etiology and management of depression.
Material and methods
Sample size calculation and justification
The sample size was established using power analysis; standard deviation was used to measure sample variability; type -1 error at p < 0.05; and two-way or one-way ANOVA with an 80 percent analysis capacity was used. The sample size was calculated using the G power software.
Experimental animals and groups
Ten weeks old, male mice (C57BL/6, n = 168 mice) were maintained in polyacrylic cages at room temperature on a 12/12-h light/dark cycle. The animals received a pellet diet and provided access to water ad libitum for 1 week prior to the start of the experimental procedures. All animal studies were approved by the Animal Ethics Committee of the Institution (JMHC/4300/32-02/20) adhered to the guidelines for research on use and care of laboratory animals.
15
The study was planned into two phases. The first phase comprised administration of finasteride (100 mg kg−1) for inducing depression-like behavior in mice for 30 min, 2 h, 24 h, or 3 or 6 days. The mice (n = 60) were divided into two groups in the first phase—control (10% HPβCD, s. c; n = 30) and finasteride-treated (100 mg kg−1 s. c.; n = 30). The 60 mice were further divided into five groups, namely, 30 min, 2 h, 24 h, 3-days, and 6-days with six mice in each group. On seventh day, control and finasteride pre-treated mice were subjected to behavioral tests to determine the time course of finasteride’s effect for inducing depression-like behavior in mice (Figure 1). Diagrammatic representation of experimental protocol first phase study: C1–C5: Control group, F1–F5: Finasteride-treated groups, OFT: Open field test, FST: Forced swim test, and SPT: Source preference test.
Later, the second phase study was planned based on the outcomes of the first phase study, which included the induction of depression-like behavior with finasteride for 6 days and drug treatment. The induction of depression-like behavior with finasteride administration (100 mg kg−1) for 6 days comprised 96 mice. Finasteride-induced depression-like behavior in mice after 6 days was further divided into the following groups, namely, F1, F2+Ngn (50 mg kg−1), F3+Ngn (100 mg kg−1), F4+Ngn-M (50 mg kg−1), F5+Ngn-M (100 mg kg−1), F6+Ngn-DM (50 mg kg−1), F7+Ngn-DM (100 mg kg−1), and F8+DLX (10 mg kg−1). Each group consisted of 12 mice (12 × 8 = 96). The control (10% HPβCD, s. c) group comprised 12 mice.
The control mice pre-treated with 10% HPβCD, subcutaneously, received 0.9% sodium chloride solution orally. Finasteride (100 mg kg−1, s. c.,) pre-treated mice (F1–F8) received vehicle (0.9% NaCl, p. o.) or drugs (Ngn, Ngn-M, Ngn-DM, 50 or 100 mg/kg, and DLX, 10 mg kg−1, p. o.) administration for 14 days prior to testing and brains collection (Figure 2). Diagrammatic representation of experimental protocol first phase study: F1–F8: Finasteride-treated groups, Ngn: Naringenin, Ngn-M and Ngn-DM: Naringenin methylated derivatives, DLX: Duloxetine, Nacl: Sodium chloride, OFT: open field test, FST: Forced swim test, SPT: Source preference test, MDA: malondialdehyde, GSH: reduced glutathione, CAT: Catalase, SOD: Superoxide dismutase, TNF-α: Tumour necrosis factor alpha, and IL-1β: Interleukin-1β.
Drugs and chemicals
Naringenin (Ngn), naringenin 7-O-methyl ether (Ngn-M), and finasteride were obtained from the Sigma Aldrich, China. Finasteride suspended in 10% w/v HPβCD (hydroxypropyl-β-cyclodextrin) was used in this study. Duloxetine (DTX) was procured from Beijing Biopharmaceutical Ltd. (China). Mouse ELISA KIT for interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF-α) was purchased from Invitrogen Inc. (United States). Naringenin 4′, 7-dimethyl ether (Ngn-DM) was prepared and purified as described earlier. 15
Behavioral tests
Locomotor activity (open-field test)
The individual mice were placed in the corner of a plastic apparatus ((35 cm3 × 25 cm3 × 20 cm3, divided into 12 equal squares), where they remained for 5 min to allow them to acclimatize to their new environment. A video camera was used to record the test session, and the subsequent 5 min were used to record the number of crossings (four-paw crossing the sector) and rearings (raising forepaws).
Sucrose preference test
Sucrose preference was tested because it is a hallmark of anhedonia. Animals were evaluated in the search for their curiosity to find sucrose solution over normal water. Two bottles were presented to the animals during the training session for 24 h. The first was filled with sucrose water (1%), and the second was filled with normal water. During the training period to avoid position preference, the position of the bottles varied. A 2-h testing session was conducted after 18 h of fasting (without food and water). The following formula was used to compute the preference for sucrose. 16
Sucrose preference (%) = Weight of sucrose intake/Weight of sucrose intake + Weight of sucrose intake.
Forced swimming test
The mice were allowed to swim in the FST in a tank (20 cm × 10 cm in height and diameter, respectively) filled with water (25°C) up to a height of 12 cm. A 15-min session was used to assess depression-like behavior in the mice. The mice were taken out from the container after swimming, wiped with towels, and kept warm in their home cages under a lamp. Session recordings were made for later analysis. The mouse was deemed immobile when it remained passively floating in the water, making only the movements required to maintain its nose or head above the water. 14 The mice were permitted to move their forepaws and assist themselves by pushing their paws against the wall of the tank. The assessor was unaware of the treatment conditions because the videotapes were coded.
Tail suspension test
The tail suspension test was performed basically as defined by Steru et al. 17 The mice were hung from the roof of the assembly (30 cm3 × 30 cm3 × 30 cm3) with adhesive tape one cm from the tip of the tail. Only when mice hung motionless and passively were they considered immobile. The duration of inability to move was observed for the entire six-minute duration of the test period.
Biochemical studies
Mice were killed by decapitation, and the brains were removed quickly. However, the other half of the brain was further purified with a mixture of 10% paraformaldehyde and 5% hydrochloric acid (pH 7.2). Brain homogenates were prepared in 0.1 M phosphate-containing buffer (10% w/v, pH 7.2). The homogenates were centrifuged for 20 min at 12,000 × g, and supernatants were collected and employed for biochemical and Enzyme-Linked Immunosorbent Assay (ELISA) analyses.
Assessment of glutathione concentration
The reduced glutathione (GSH) is an extremely powerful antioxidant substance that guards cells from free radical damage by scavenging them. The GSH decreased concentrations were determined according to the method in the homogenous brains’ aliquots of the corresponding groups. Equal volumes of tissue homogenate (0.3 ml) and 15% trichloroacetic acid (TCA) (0.3 ml) were combined and centrifuged for 15 min at 12,000 rpm at 4°C in a cold centrifuge. The supernatant (0.2 ml) was combined with 0.7 mM DTNB reagent (1.8 ml) and phosphate buffer to make a final volume of 2 ml (0.3 M, pH 7.8). The absorbance was then determined employing a spectrophotometer at 412 nm against a blank reagent. Reduced GSH concentrations from the tissues of the brain were quantified in micromoles per gram of the tissue (mol/g tissue).
Determination of superoxide dismutase activity
The SOD activity was measured as mentioned earlier. 18 In short, one mL of supernatant of the brain tissue was added to form 1:10 dilutions by using 9 ml of distilled water. In a spectrophotometer, 0.3 ml of the sample was transferred to a 3 ml of 0.04 M carbohydrate buffer (pH 11), which was then easily combined by reversal, and 0.4 ml of 0.2 mM adrenaline was added. A buffer of 3.0 ml, an adrenaline of 0.2 ml, and water of 0.3 ml are used in the reference cuvette. The improvement in absorbance at 490 nm was measured to determine SOD activity.
Assessment of catalase activity
The activity of catalase was assessed by using Sinha method, 19 based on H2O2 loss (hydrogen peroxide) from the enzyme source (catalase). In brief, each treatment of brain supernatant (one mL) was mixed with 4 ml of phosphate buffer (pH 7.2) and 5 ml of H2O2 solution. A gentle stirring at room temperature mixed the reaction mixture. 1 ml of the reaction mixture was combined with three mL of acetic acid/dichromate reagent. The absorbance was determined at 565 nm using a spectrophotometric method, and the absorbance change was measured at one-minute intervals. The function of catalase (CAT) was represented as μmoles of H2O2 that are broken down every minute by milligram of protein.
Assessment of lipid peroxidation
The MDA (malondialdehyde) content was determined using the technique previously mentioned. 20 MDA is a marker of lipid peroxidase tissue injury. In brief, distilled water (0.4 ml) and 15% TCA (2.0 ml) were transferred to brain homogenates of each 0.4 ml and centrifuged for 15 min at 4000 rpm. The mixture was then placed in a 75°C water bath for 30 min before being cooled at 37°C with 0.2 ml of TBA (thiobarbituric acid) transferred to the supernatant (0.8 ml). The supernatant was cooled and measured at a wavelength of 543 nm with a spectrophotometer. The malondialdehyde content was measured employing a molar extinction coefficient, and the results were expressed in moles of MDA per gram of tissue.
ELISA assay
The animals were killed after the behavioral experiments. The hippocampi were separated, weighed quickly, and placed in a lysis buffer for the radio immunoprecipitation assay (RIPA). The protein concentration was then measured using the bicinchoninic acid (BCA) assay. Enzyme immunoassay was used to measure IL-1β and TNF-α levels. The samples were transferred to each well as per the manufacturer’s protocol, and the plate was incubated at room temperature for 1 h without blocking. The optical density was determined at 365 nm after several washes and proper color growth. The assay’s sensitivity for IL-1β and TNF-α was 6 and 2 pg/ml, respectively.
Statistical analysis
Data were represented as means ± S.E.M and analyzed using two-way or one-way ANOVA followed by Dunnett’s t-test for comparisons among groups. A p < 0.05 value was deemed statistically significant. The normality of the data was assessed by both visually and through normality tests (Shapiro–Wilk test) using the SPSS software.
Results
Effect of Ngn, Ngn-M, Ngn-DM, or DLX on locomotor activity (open-field test) pre-treated with finasteride
Criteria for systematic literature search. Effects of finasteride and Ngn, Ngn-M, or Ngn-DM (50 and 100 mg/kg) on crossings in mice after 30 min, 2 h, 24 h, 3 days, and 6 days. Control rats received 10% HPβCD subcutaneously (s.c) and finasteride (100 mg/kg, s.c) was administered subcutaneously for inducing depression-like behavior for 30 min, 2 h, 24 h, 3 days, and 6 days in the first phase study. In the second phase, the control (10% HPβCD, s.c) and the finasteride pre-treated mice induced with the depression-like behavior for 6 days (F1–F8) were administered with vehicle (0.9% NaCl, p.o) or drugs (Ngn, Ngn-M, Ngn-DM, 50 or 100 mg/kg, and DLX, 10 mg/kg, p.o.) for 14 days prior to testing. Two-way ANOVA followed by Dunnett’s post hoc test (n = 12).
Data are expressed as mean ± S.E.M. #p < 0.05, vs control group, *p < 0.05; **p < 0.01, vs. Finasteride-treated (F1) group. F1—F8: Finasteride-treated groups, Ngn: Naringenin, Ngn-M and Ngn-DM: Naringenin methylated derivatives, DLX: Duloxetine, and NaCl: Sodium chloride.
Effects of finasteride and Ngn, Ngn-M, or Ngn-DM (50 and 100 mg/kg) on rearing in mice after 30 min, 2 h, 24 h, 3 days, and, 6 days. Control rats received 10% HPβCD subcutaneously (s.c), and vehicle (0.9% sodium chloride (NaCl) solution) orally for 30 min, 2 h, 24 h, 3 days, and 6 days. Finasteride (100 mg/kg, s.c) was administered subcutaneously for inducing depression-like behavior for 30 min, 2 h, 24 h 3 days, and 6 days. Control (10% HPβCD, s.c) and finasteride pre-treated mice (F1—F8) were administered with vehicle (0.9% NaCl, p.o) or drugs (Ngn, Ngn-M, Ngn-DM, 50 or 100 mg/kg, and DLX, 10 mg/kg, p.o.) for 14 days prior to testing on 21st day. Two-way ANOVA followed by Dunnett’s post hoc test (n = 12).

Data are expressed as mean ± S.E.M. #p < 0.05, vs control group, *p < 0.05; **p < 0.01, vs. Finasteride-treated (F1) group. F1—F8: Finasteride-treated groups, Ngn: Naringenin, Ngn-M and Ngn-DM: Naringenin methylated derivatives, DLX: Duloxetine, and NaCl: Sodium chloride.
In the second phase of the study, the analysis of the treatment with Ngn, Ngn-M, Ngn-DM, or DLX for 14 days showed significant differences among groups. The post hoc test showed significant increase in crossings (F8, 45 = 34.52; p < 0.01) and rearing (F8, 45 = 24.61; p < 0.01) in open field as compared to finasteride-treated group.
Effect of Ngn, Ngn-M, Ngn-DM, or DLX on sucrose preference test pre-treated with finasteride
Effects of finasteride and Ngn, Ngn-M, or Ngn-DM (50 and 100 mg/kg) on sucrose preference in mice after 30 min, 2 h, 24 h, 3 days, and 6 days. Control rats received 10% HPβCD subcutaneously (s.c), and vehicle (0.9% sodium chloride (NaCl) solution) orally for 30 min, 2 h, 24 h, 3 days, and 6 days. Finasteride (100 mg/kg, s.c) was administered subcutaneously for inducing depression-like behavior for 30 min, 2 h, 24 h, 3 days, and 6 days. Control (10% HPβCD, s.c) and finasteride pre-treated mice (F1—F8) were administered with vehicle (0.9% NaCl, p.o) or drugs (Ngn, Ngn-M, Ngn-DM, 50 or 100 mg/kg, and DLX, 10 mg/kg, p.o.) for 14 days prior to testing on 21st day. Two-way ANOVA followed by Dunnett’s post hoc test (n = 12).

Data are expressed as mean ± S.E.M. #p < 0.05, vs control group, *p < 0.05; **p < 0.01, vs. Finasteride-treated (F1) group. F1—F8: Finasteride-treated groups, Ngn: Naringenin, Ngn-M and Ngn-DM: Naringenin methylated derivatives, DLX: Duloxetine, and NaCl: Sodium chloride.
In the second phase of the study, the analysis of the treatment with Ngn, Ngn-M, Ngn-DM, or DLX for 14 days showed significant differences among groups. The post hoc test showed significant increase in sucrose consumption (F8, 45 =13.87; p < 0.01) compared to finasteride-treated group (Table 3).
Effect of Ngn, Ngn-M, Ngn-DM, or DLX on the duration of immobility in forced swimming test pre-treated with finasteride
The findings demonstrated that a 6-day pre-treatment with finasteride (100 mg kg−1, s. c.) prior to testing revealed significant differences between groups. The post hoc test showed that finasteride (100 mg kg−1) considerably increased the duration of immobility in forced swimming test (FST) compared to control mice (F9, 56 = 7.22; p < 0.01; Figure 3). Nonetheless, no significant effect was noticed in the duration of immobility of FST in 30 min, 2 h, 24 h, or 3-day study after finasteride administration compared to control group. Effects of finasteride on immobility in mice for forced swimming test (FST) and tail suspension test (TST) after 30 min, 2 or 24 h, or 3 or 6 days summarizing the time-course of finasteride’s effects on the FST and TST. Control rats received 10% HPβcd subcutaneously (s.c) for 30 min, 2 h, 24 h, 3 days, and 6 days. Finasteride (100 mg/kg, s. c) was administered subcutaneously for inducing depression-like behavior for 30 min, 2 h, 24 h, and 3 days. Data are expressed as mean ±S.E.M. **p > 0.05, vs finasteride-treated group. Two-way ANOVA followed by Dunnet’s post hoc test (n = 12).
In the second phase of the study, the drug treatments (Ngn, Ngn-M, Ngn-DM, or DLX) for 14 days showed significant differences among groups. The post hoc test showed substantial reduction in the duration of immobility (F8, 45 =14.72; p < 0.01; Figure 4) compared to finasteride-treated group. Effect of Ngn, Ngn-M, Ngn-DM, and DLX on finasteride-induced immobility in mice for forced swimming test (FST). Control (10% HPβCD, s. c) and finasteride pre-treated mice (F1–F8 for 6 days were administered with vehicle [0.9% Nacl, p. o] or drugs [Ngn, Ngn-M, and Ngn-DM, 50 or 100 mg/kg, and DLX, 10 mg/kg, p. o] for 14 days prior to testing on 21st day). Each bar signifies mean ± S.E.M., n = 12. #p < 0.01 compared with control group; *p < 0.05, **p < 0.01 compared with finasteride-treated group. Two-way ANOVA followed by Dunnet’s post hoc test. F1–F8: Finasteride pre-treated groups, Ngn: Naringenin, Ngn-M and Ngn-DM: Naringenin methylated derivatives, and DLX: Duloxetine.
Effect of Ngn, Ngn-M, and Ngn-DM on the duration of immobility in tail suspension test (TST) pre-treated with finasteride
Consequently, pre-treatment with finasteride (100 mg kg−1, s. c.) revealed significant differences between groups. The post hoc test showed that finasteride (100 mg kg−1) substantially increased immobility period in tail suspension test (F9, 56 = 5.72; p < 0.01; Figure 3) in a 6-day study compared to control mice. Nonetheless, no significant effect was noticed in the duration of immobility of TST in 30 min, 2 h, 24 h, or 3-day study after finasteride administration compared to control group.
However, in the second phase of the study, treatment with Ngn, Ngn-M, Ngn-DM, or DLX showed significant differences among groups. The post hoc test showed substantially shortened duration of immobility (F8, 45 = 11.71; p < 0.01; Figure 5) compared to finasteride-treated group. Effect of Ngn, Ngn-M, Ngn-DM, and DLX on finasteride-induced immobility in mic
Naringenin or its methylated derivatives increased antioxidant molecules in the brain levels in finasteride-treated mice
The effects of Ngn or its methylated derivatives (Ngn-M or Ngn-DM) in finasteride-induced depression in mice on GSH, SOD, and catalase (anti-oxidant molecules) levels are shown in Figures 6–8. One-way ANOVA revealed a significant variation between treatments. When comparing finasteride-treated mice to control mice, post hoc analysis using the Dunnett’s t-test revealed that finasteride-treated mice had lower enzymatic levels of GSH (p < 0.01), SOD (p < 0.01), and catalase (p < 0.01) in the brain. However, Ngn, Ngn-M, Ngn-DM (50 or 100 mg kg−1), or DTX (10 mg kg−1) improved considerably the levels of the endogenous anti-oxidant molecules as compared to finasteride-treated mice (F8, 45 = 31.25; p < 0.01). Effect of Ngn, Ngn-M, Ngn-DM, and DLX on finasteride-induced reduced brain levels of glutathione (GSH) in mice. Control rats received 0.9% sodium chloride solution orally. Finasteride pre-treated mice were administered with drugs (Ngn, Ngn-M, Ngn-DM, 50 or 100 mg/kg, and DLX, 10 mg/kg, p. o) for 14 days. Each bar signifies mean ± S.E.M., n = 12. #p < 0.01 compared with control group; *p < 0.05, **p < 0.01, ***p < 0.001 compared with finasteride-treated group. One-way ANOVA followed by Dunnet’s post hoc test. F1–F8: Finasteride pre-treated groups, Ngn: Naringenin, Ngn-M and Ngn-DM: Naringenin methylated derivatives, and DLX: Duloxetine. Effect of Ngn, Ngn-M, Ngn-DM, and DLX on finasteride-induced reduced brain levels of superoxide dismutase (SOD) activity in mice. Control rats received 0.9% sodium chloride solution orally. Finasteride pre-treated mice were administered with drugs (Ngn, Ngn-M, Ngn-DM, 50 or 100 mg/kg, and DLX, 10 mg/kg, p. o) for 14 days. Each bar signifies mean ± S.E.M., n = 12. #p < 0.01 compared with control group; *p < 0.05, **p < 0.01, ***p < 0.001 compared with finasteride-treated group. One-way ANOVA followed by Dunnet’s post hoc test. F1–F8: Finasteride pre-treated groups, Ngn: Naringenin, Ngn-M and Ngn-DM: Naringenin methylated derivatives, and DLX: Duloxetine. Effect of Ngn, Ngn-M, Ngn-DM, and DLX on finasteride-induced reduced brain levels of catalase (CAT) activity in mice. Control rats received 0.9% sodium chloride solution orally. Finasteride pre-treated mice were administered with drugs (Ngn, Ngn-M, Ngn-DM, 50 or 100 mg/kg, and DLX, 10 mg/kg, p. o) for 14 days. Each bar signifies mean ± S.E.M., n = 12. #p < 0.01 compared with control group; *p < 0.05, **p < 0.01, ***p < 0.001 compared with finasteride-treated group. One-way ANOVA followed by Dunnet’s post hoc test. F1–F8: Finasteride pre-treated groups, Ngn: Naringenin, Ngn-M and Ngn-DM: Naringenin methylated derivatives, and DLX: Duloxetine.


Naringenin or its methylated derivatives reduces the brain concentrations of malondialdehyde and nitrite in finasteride-treated mice
Figures 9 and 10 show the effect of Ngn or its methylated derivatives (Ngn-M or Ngn-DM) on finasteride-induced depression with increase in MDA and nitrite concentrations in the brains of mice. The one-way ANOVA showed substantial differences between treatment groups for MDA and nitrite. The post hoc analysis using the Dunnett’s t-test revealed that the brain levels of MDA (p < 0.01) and nitrite (p < 0.01) were increased significantly after finasteride administration relative to control group as shown in Figures 9 and 10. However, Ngn, Ngn-M, Ngn-DM (50 or 100 mg kg−1), or DTX (10 mg kg−1) substantially (F8, 45 = 25.67; p < 0.01) decreased in MDA and nitrite levels when compared to finasteride-treated group. Effect of Ngn, Ngn-M, Ngn-DM, and DLX on finasteride-induced reduced brain levels of malondialdehyde (MDA) concentrations in mice. Control rats received 0.9% sodium chloride solution orally. Finasteride pre-treated mice were administered with drugs (Ngn, Ngn-M, Ngn-DM, 50 or 100 mg/kg, and DLX, 10 mg/kg, p. o) for 14 days. Each bar signifies mean ± S.E.M., n = 12. #p < 0.01 compared with control group; *p < 0.05, **p < 0.01, ***p < 0.001 compared with finasteride-treated group. One-way ANOVA followed by Dunnet’s post hoc test. F1–F8: Finasteride pre-treated groups, Ngn: Naringenin, Ngn-M and Ngn-DM: Naringenin methylated derivatives, and DLX: Duloxetine. Effect of Ngn, Ngn-M, Ngn-DM, and DLX on finasteride-induced reduced brain levels of nitrite concentrations in mice. Control rats received 0.9% sodium chloride solution orally. Finasteride pre-treated mice were administered with drugs (Ngn, Ngn-M, Ngn-DM, 50 or 100 mg/kg, and DLX, 10 mg/kg, p. o) for 14 days. Each bar signifies mean ± S.E.M., n = 12. #p < 0.01 compared with control group; *p < 0.05, **p < 0.01, ***p < 0.001 compared with finasteride-treated group. One-way ANOVA followed by Dunnet’s post hoc test. F1–F8: Finasteride pre-treated groups, Ngn: Naringenin, Ngn-M and Ngn-DM: Naringenin methylated derivatives, and DLX: Duloxetine.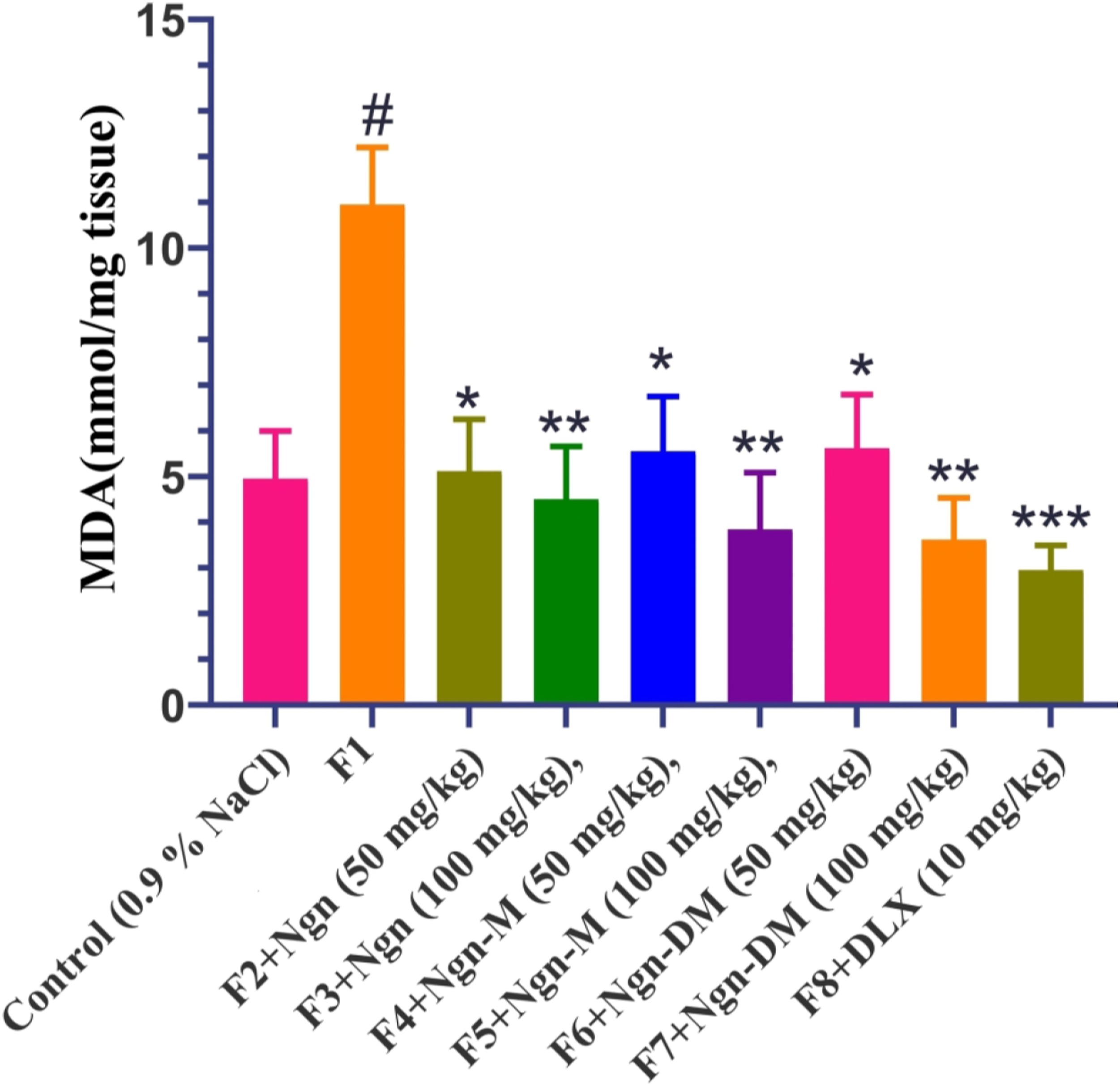

Effect of Ngn, Ngn-M, and Ngn-DM on brain TNF-α level in mice
Pre-treatment with finasteride (100 mg kg−1, s. c.) increased brain TNF-α levels by 25% (p < 0.01) relative to vehicle-treated mice, according to an ELISA study (Figure 11). The post hoc analysis using the Dunnett’s t-test revealed that the treatments with Ngn, Ngn-M, Ngn-DM (50 or 100 mg kg−1), or DTX (10 mg kg−1) substantially reduced brain TNF-α levels (F8, 45 = 27.71; p < 0.01) compared to finasteride-treated group, which is consistent with behavioral findings. Effect of Ngn, Ngn-M, Ngn-DM, and DLX on finasteride-induced reduced brain levels of TNF-α in mice. Control rats received 0.9% sodium chloride solution orally. Finasteride pre-treated mice were administered with drugs (Ngn, Ngn-M, Ngn-DM, 50 or 100 mg/kg, and DLX, 10 mg/kg, p. o) for 14 days. Each bar signifies mean ± S.E.M., n = 12. #p < 0.01 compared with control group; *p < 0.05, **p < 0.01, ***p < 0.001 compared with finasteride-treated group. One-way ANOVA followed by Dunnet’s post hoc test. F1–F8: Finasteride pre-treated groups, Ngn: Naringenin, Ngn-M and Ngn-DM: Naringenin methylated derivatives, and DLX: Duloxetine.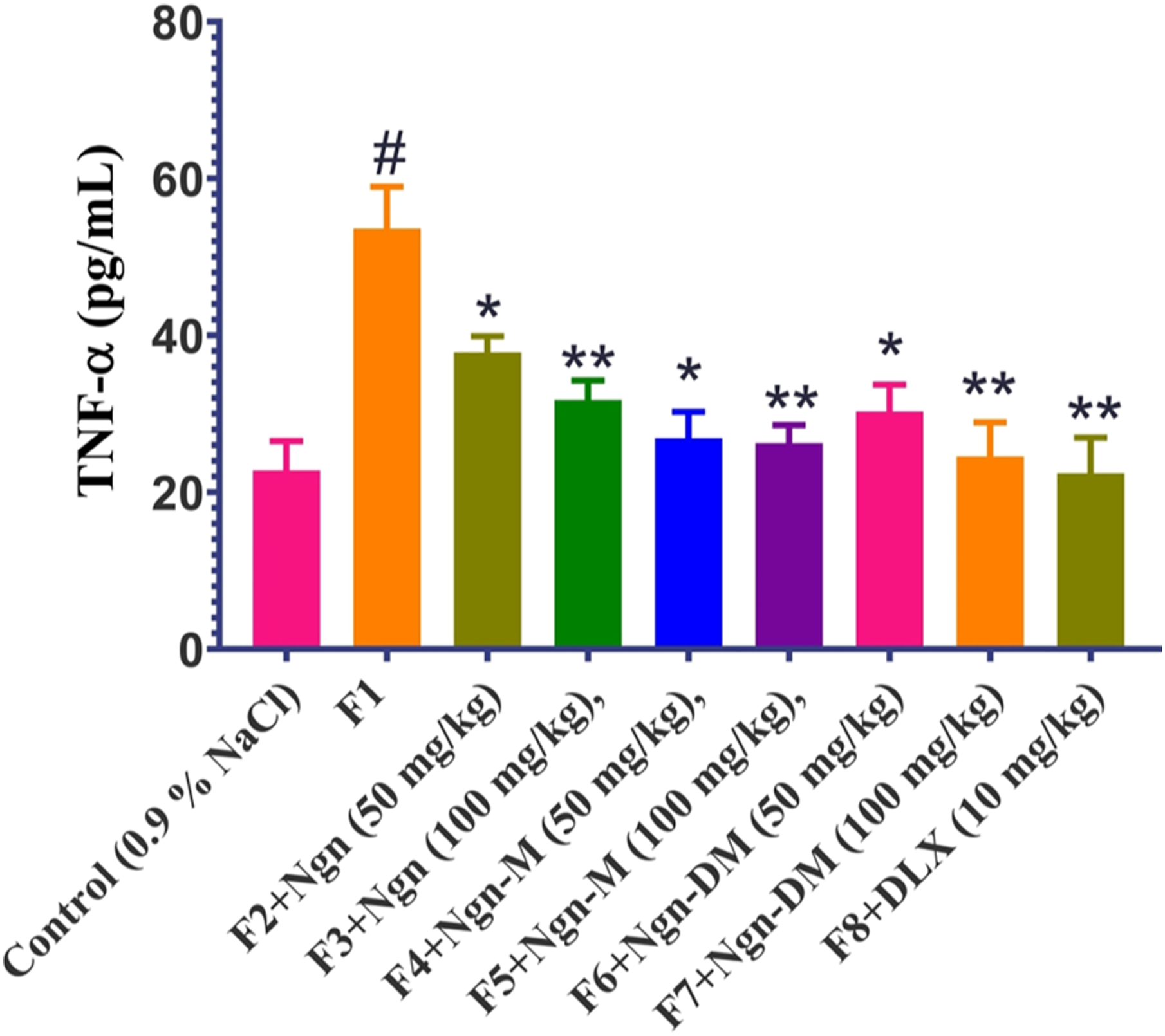
Effect of Ngn, Ngn-M, and Ngn-DM on brain IL-1β level in mice
Figure 12 shows that the pre-treatment with finasteride (100 mg kg−1, s. c.) substantially increased brain IL-1βs levels in mice by about 21% (p < 0.01). This effect was reversed and substantially decreased brain IL-1β levels (F8, 45 = 27.71; p < 0.01) in Ngn, Ngn-M, Ngn-DM (50 or 100 mg kg−1), or DTX (10 mg kg−1) treated groups compared to the finasteride-treated mice according to the post hoc analysis using the Dunnett’s t-test. Effect of Ngn, Ngn-M, Ngn-DM, and DLX n finasteride-induced reduced brain levels of IL-1β in mice. Control rats received 0.9% sodium chloride solution orally. Finasteride pre-treated mice were administered with drugs (Ngn, Ngn-M, Ngn-DM, 50 or 100 mg/kg, and DLX, 10 mg/kg, p. o) for 14 days. Each bar signifies mean ± S.E.M., n = 12. #p < 0.01 compared with control group; *p < 0.05, **p < 0.01, ***p < 0.001 compared with finasteride-treated group. One-way ANOVA followed by Dunnet’s post hoc test. F1–F8: Finasteride pre-treated groups, Ngn: Naringenin, Ngn-M and Ngn-DM: Naringenin methylated derivatives, and DLX: Duloxetine.
Discussion
Inflammatory processes have been linked to severe depression. The acute (24 h or 3-days) treatment of finasteride (100 mg kg−1) had not shown any result in depression-like activity in our research. The study’s key observation was that short-term administration of finasteride for 6 days in first and second phase studies showed significant induction of depression-like behavior in mice. Finasteride had no effect on OFT, SPT, FST, or TST either immediately after 30 min, 2 24 h, or after 3 days (Figure 3 and Tables 1, 2, and 3), respectively.
There was no significant reduction of immobility in the both tests (FST and TST) on 6-days in the control group as compared to 30 min. From Figure 3, it is clear that mice in control group restored to normal behavior after 6-days reaching to their baseline values. The significant increase after 2 h or 1 or 3-days could be due to the perceived anxiety in mice, which was restored to normal behavior after 6 days. Hence, there was no anti-depressant effect noticed due to administration of 10% HPβCD in control mice. Our findings from the first and second phases of behavioral studies for 6-days after finasteride administration show a significant increase in the duration of immobility (Figure 3), implying depression-like activity, which is consistent with a previous study in which acute systemic or intra-hippocampal finasteride administration or intra-amygdalar finasteride injection increased FST immobility in female rats. 21 Frye and Walf, 21 on the other hand, found that finasteride treatment (50 mg kg−1) to female rats induced behavior similar to that of depression in the forced swimming test. It’s likely that the sex of the rats employed in the research played a role in these variations. Furthermore, the lack of acute effects may be attributed to a rise in allopregnanolone levels after the stress induced by swimming. According to our knowledge, the current study is the first to demonstrate that short-term administration of finasteride induces behavior similar to depression in mice. Furthermore, the results of the study demonstrated a significant improvement in the behavioral and biochemical changes caused by short-term finasteride administration with the treatment of methylated derivatives of naringenin.
Finasteride inhibits 5-reductase, an enzyme required for the transformation of progesterone to dihydro-progesterone, and then to tetrahydro-progesterone or allopregnanolone. 22 The study found that giving finasteride intraperitoneally 1 h before immobilization stress reduced allopregnanolone levels in a dose-dependent manner. 23 Furthermore, 1 month after discontinuing finasteride administration, naive rats had lower allopregnanolone levels in the cerebral cortex. 24 Prior studies have shown that administering allopregnanolone directly caused changes in behavior similar to that of depression in animals. 25 The major depression signs may be due to neurosteroid hormone imbalances, where the plasma and CSF levels of allopregnanolone are abnormally low. Dihydrotestosterone (DHT) has been shown to have anti-depressant properties by Frye and Wawrzycki. 24 The administration of finasteride reduces DHT concentrations, 26 and clinical trials have also reported that low testosterone concentrations are related to the signs and symptoms of depression. 27 As a result, our future research will likely focus on the lower levels of neuroactive steroids in certain regions of the brain, that has been observed previously to induce depression-like behavior. The findings of the previous study 28 revealed that repeated administration of finasteride for 7 days reduced hippocampal neurogenesis in rats.
The findings of our research also revealed that finasteride administration caused inflammation and depression-like behavior in mice. Our preliminary findings did not show the finasteride’s effects on the behaviors similar to that of depression in mice employing OFT, SPT, TST, and FST at doses of 25, 50 and 75 mg kg−1 subcutaneously at time points of 30 min, 2 h, 24, or 3 or 6 days. Consequently, finasteride at a dose of 100 mg kg−1 prior to testing considerably increased the period of immobility in OFT, SPT, FST, or TST, and showed significant effect on locomotor function in a 6-day study (Figure 3 and Tables 1, 2, and 3).
In the current study, the duration of immobility in FST or TST and the levels of pro-inflammatory cytokines like IL-1β and TNF-α were considerably elevated following finasteride (100 mg kg−1) pre-treatment suggesting a connection involving inflammatory mechanisms and depression-like behavior in rats. Naringenin or its methylated derivatives at doses of 50 or 100 mg kg−1 and the DTX (10 mg kg−1) substantially reduced immobility and normalized IL-1β and TNF-α levels. Additionally, finasteride-treated mice showed decreased preference to sucrose solution as compared to control group as indicative of depression. The treatments with Ngn, Ngn-M, Ngn-DM, and DLX restored preference to the sucrose solution with increase in the consumption relative to finasteride-treated group in agreement with the previous study. 16 Methylation of flavonoids was found to significantly improve their metabolic stability and transport across the membranes, promoting absorption and favorably influencing their bioavailability. Our findings demonstrated that the effects of high doses of Ngn-M or Ngn-DM were more pronounced in the behavioral and biochemical changes caused by short-term finasteride administration as compared to the high doses of Ngn alone. Nevertheless, it was not possible to demonstrate any metabolic benefit of the non-methylated compound over the methylated compound in the current study because there was no simple metabolic turn-over of Ngn-DM and no measurable quantities of metabolites could be identified in the case of Ngn. Hence, this is considered as the limitation of our current study.
In the last two decades, there has been a plethora of evidence linking inflammation and depression. A variety of inflammatory cytokines have been shown to have altered peripheral and brain concentrations in depressive disorders, and anti-depressants were shown to inhibit the actions of inflammatory cytokines in the brain. 29 Recent meta-analyses have found that IL-1β and TNF-α are remarkable pro-inflammatory markers in the etiology and management of depression. 30 It is well-known that pro-inflammatory cytokines and inflammation that are frequently associated with stress have also been shown to cause alterations in structure and functions of the brain, contributing to the emergence of major depressive disorders. 31 Most evidence demonstrates that pro-inflammatory cytokines, such as TNF-α and IL-1β, are both capable of altering serotonin transporter activity as well as the kynurenine pathway, 32 and that increases TNF-α or IL-1β in the hippocampus leading to serotonin abnormalities and neurotransmitter loss causing neuronal loss. 33 It was shown that IL-1β and TNF-α increased excitotoxicity and reduced the expression of the IL-1β receptor alleviated stress-induced neurogenesis. 34 Thus, inflammation and its mediators often associated with stress, as part of a widespread physiological response or a precursor to the cascade of events that eventually lead to depression, can play a relatively subtle role.
The effects of flavonoids (the Ngn and its methyl derivatives) on inflammation and pro-inflammatory cytokines may assist in the improvement of serotonergic dysfunctions and the modulation of the HPA axis, ultimately preserving the structural and functional activities of the hippocampus. 35 Of late, our findings demonstrated that Ngn or its methylated derivatives have anti-depressant-like effects that are linked to the nitric oxide signaling pathway which are stimulated by IL-1β. Consequently, further investigation of the intricate system and the detailed function of each component are warranted.
Naringenin has strong anti-oxidant and anti-inflammatory activity, 36 which are considered crucial for its anti-depressive and neuroprotective effects. 37 It is essential to elucidate the link between interleukin-1 (IL-1) and the toll-like receptor 4 (TL4)/nuclear factor–kappa B (NF-κB) pathway in inflammation and depression involving the role of naringenin. 37 Ngn-M, for example, has been shown to inhibit the activation of the ERK1/2, JNK, and NF-κB signaling pathways in lipopolysaccharide-activated macrophages in-vitro through down-regulation of TNF-α, IL-6, and IL-1β. 38 Furthermore, methylation (and probably double methylation of Nng-DM) may not completely abolish anti-inflammatory effect of naringenin, though the potency and range of suppressive effects demonstrated by the flavonoids may differ. Furthermore, methylation can offer flavonoids a distinct pharmacological action.
Natural remedies in the management of depression have recently sparked increased interest due to their improved performance and reduced side effects, though the mechanisms remain unknown. As a result, the quest for effective and safe plant-based medicines to replace current conventional drugs is intensifying. 39 This has resulted in the identification of many phytochemicals with anti-depressant properties. 39
Previous studies have also established naringenin’s anti-depressant efficacy by raising 5-hydroxy-tryptamine, norepinephrine, and BDNF levels in the brain. 40 In this report, naringenin or its methylated derivatives was found to be effective in reducing the neurological behavior caused by finasteride administration. Future research on the beneficial effects of naringenin in neurological abnormalities caused by finasteride should be planned to elucidate the role of noradrenergic, dopaminergic, and serotonergic processes, as well as molecular mechanisms involving NF-kB and BDNF levels linked with the administration of finasteride and stress responses.
As a consequence, it has been reported that the high levels of IL-6 and IL-1β exacerbate oxidant-induced blood barrier permeability. 41 Therefore, inhibiting oxidative/nitrergic pathways as well as inflammatory cascades has been suggested as a new therapeutic goal for reducing neuronal injury in prolonged stress-induced neuronal etiologies. 41 Further, there is a growing body of evidence supporting the benefits of anti-oxidants in the treatment of stress-associated anxiety, depression, and cognitive impairment. 42 Future research is needed to assess the effects of finasteride on stress-associated disorders like depression, anxiety, and cognitive dysfunction, as well as to comprehend the underlying neuronal mechanisms.
Conclusion
Our findings demonstrated that repeated administration of finasteride causes behavior similar to that of depression in male mice. Finasteride-treated mice had reduced levels of endogenous anti-oxidant molecules as well as higher levels of MDA and nitrite, indicating elevated oxidative and nitrergic stress. As a result, the naringenin or its methylated derivatives inhibits oxidative and nitrergic stress and the pro-inflammatory cytokines release that can lead to their potential to ameliorate neurobehavioral disorders in mice treated with finasteride. Based on the data presented above, we can deduce that the effects of Ngn or its methylated derivatives on increased IL-1β and TNF-α levels caused by finasteride pre-treatment, and depression-like activity, could be a safe and effective anti-depressant-like actions of naringenin or its methylated derivatives.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors state that the current research project received self-funding from the Jinzhou Mental Health Center, Jinzhou. The study was conducted at the Mental Health Center, Jinzhou.
Ethical approval
Ethical approval for this study was obtained from Animal ethics committee, approval no (PSY/IAC/GCPH_25_01/21).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
