Abstract
Background
Abdominal aortic aneurysm (AAA) is a potentially life-threatening vascular abnormality, that, if ruptured, is almost universally fatal without repair, and is associated with up to 50% mortality even if repaired in hospital. To date, there is no drug therapy that has clinically proven benefit to reduce or prevent expansion of AAA. The aim of this study was to investigate whether Daxx could affect AAA through inhibiting the PI3K/AKT/ID2 signaling pathway mediated by transforming growth factor β-1 (TGFβ1).
Methods
The AAA model was constructed by injecting angiotensin Ⅱ (Ang-Ⅱ) into rats, and the Daxx lentivirus vector was constructed. Hematoxylin and eosin (HE) staining was used to detect the wall thickness of the abdominal aorta in rats. The gene and protein expressions in abdominal aortic tissues were detected utilizing western blot, immunohistochemistry (IHC) and fluorescence quantitative real-time polymerase chain reaction (qRT-PCR). Finally, the concentration of TGF-β1 in abdominal aortic tissue was determined by ELISA.
Results
The abdominal aortic wall thickness was decreased in the Daxx expression group (by HE staining), and Daxx overexpression markedly reduced the protein expression levels of MMP2 and MMP9. Proteins related to the PI3K/AKT/ID2 signaling pathway were highly enhanced in the aneurysm wall of rats, but were reduced following Daxx addition. Moreover, Daxx reduced the damage to elastin (by IHC), and the expression levels of α-SMA and SM22α were up-regulated by Daxx (by qRT-PCR). The concentration of TGF-β1 was correlated with the activation of the PI3K/AKT/ID2 signaling pathway (by ELISA), whereas AKT overexpression weakened the inhibitory effect of Daxx.
Conclusion
Daxx ameliorated several mechanisms that contributed to expansion of AAA suppressing the tissue concentration of TGF-β1, thereby inhibiting the activation of the PI3K/AKT/ID2 signaling pathway. This evidence might form the basis to develop a therapeutic target for AAA.
Keywords
Introduction
Abdominal aortic aneurysm (AAA) is a common, permanent, irreversible, and often progressive local dilatation of the abdominal aorta with a diameter ≥3.0 cm. 1 Although surgical repair is an effective treatment of AAA, 2 it is accompanied by both morbidity and mortality, especially when ruptured. However, for patients with surgical contraindications or small asymptomatic aneurysms the principal therapeutic goal would be to limit or prevent aneurysmal enlargement. To date, there is no drug therapy that has clinically proven benefit to reduce or prevent expansion of AAA or prevent its rupture. Therefore, further investigation of the molecular mechanisms of AAA development and progression could ultimately evolve to identify therapeutic targets.
The pathological changes of AAA are quite complex, including apoptosis of vascular smooth muscle cells (VSMCs), inflammatory cell infiltration, elastin reduction, and excessive deposition of collagen. Specifically, the phenotypic transformation of VSMC, from contractile phenotype to synthetic phenotype, plays a key role in the formation and development of AAA. 3 Recent investigations have indicated that VSMCs could transform into yet another phenotype resembling macrophages. This macrophage-like phenotype might also play an important role in vascular tissue inflammation by producing immunologically active cytokine, expressing macrophage-like antigens. 4 Normally, VSMC shows a contractile phenotype that maintains vasoconstriction and regulation of vascular wall tension principally through expression of a series of contractile proteins such as a-SMA and SM22a. Under pathological stimulation, VSMCs undergo reprogramming to the synthetic phenotype, marked by synthesis of more collagen that migrates to the intima with increased expression of synthetic proteins like OPN, resulting in abnormal deposition of collagen in the aortic vessel wall. Additionally, the activation of matrix metalloproteinases (MMPs) has been confirmed to contribute to AAA expansion, while tissue inhibitor of metalloproteinase (TIMP) was weakened, resulting in destruction of elastin and collagen of extracellular matrix (ECM). 5
The death-domain-associated protein (Daxx) is a highly conserved nuclear protein. It is widely distributed in subcellular regions such as karyoplasm, nucleoli, cytoplasm, and heterochromatin, and functionally plays an important role in apoptosis and transcriptional regulation. 6 Structurally, Daxx is modular with an N-terminal helical bundle capable of interacting with a variety of cytoplasmic and nuclear proteins, and its activity is regulated by posttranslational modifications and ubiquitin-dependent degradation. 7 Daxx has been shown to regulate TGF-β1 by inhibiting transcriptional activity of Smad4 through its C-terminal domain. 8 Increasingly, studies have verified that TGF-β1 is closely related to the expansion of AAA. For example, TGF-β1 signaling and microRNA cross-talk can regulate AAA progression, 9 and the TGF-β1/Smad signaling pathway is involved in collagen expression. 10 However, the mechanism of action between Daxx and AAA is poorly understood. Reportedly, TGF-β1 can affect the phenotypic transformation of VSMC by regulating the PI3K/AKT/ID2 signaling pathway. 11 We hypothesized that Daxx might inhibit AAA expansion by regulating the PI3K/AKT/ID2 signaling pathway.
Materials and methods
Animal models
Animal-based research, the study comprised 50 male SD rats randomly divided equally into 5 groups: normal (Sham), AAA surgery (AAA), AAA + viral vector control (AAA + Vector), AAA + Daxx overexpression (AAA+Daxx), and AAA + Daxx overexpression + AKT overexpression (AAA + Daxx + AKT). The weight and blood pressure of the rats were measured. Rats in the sham group were injected subcutaneously with normal saline, and rats in other groups were injected with Ang-Ⅱ. 12 All animal experiments were approved by local ethics committees of our hospital.
Construction of Daxx, AKT and blank vector
Primer5 software was used to design the primer sequences of Daxx and AKT genes in rats, and the primers were synthesized by Shanghai Sangon Biotech. The Daxx and AKT fragments of rats were obtained by PCR apparatus (HEMA9600, Hema Medical Instrument). pLVX-IRES-puro (632183, Takara) was selected as the expression vector of lentivirus. 13 The PCR product of Daxx/AKT and the vector pLVX-IRES-puro were double digested by EcoRI (ER0271, Thermofisher) and BamHI (ER0051, Thermofisher). The digested vector was connected with the target gene fragments. The blank vector without target gene was also constructed. The ligation products were transformed into DH5α cells. After transformation, the cells were cultured and collected. Virus fluid was extracted from cells. PLVX-IRES-PURO-Daxx and AKT plasmids were extracted and digested by EcoRI + BamHI enzymes. The corresponding bands were observed by 1% agarose gel electrophoresis. According to the image of agarose gel electrophoresis (Supplementary Figure 1), the length of the fragments obtained by enzyme digestion was consistent with the expected Daxx and AKT length, thus indicating that the construction of Daxx and AKT overexpression vectors was successful.
Infusion of lentivirus vectors
Daxx, AKT, and blank virus liquids were diluted with PBS to a titer of 5×109 pfu/mL. Rats in the AAA + Vector group were infused with blank vector fluid. In the AAA + Daxx group, the rats were infused with Daxx virus fluid. In the AAA + Daxx + AKT group, the rats were infused with Daxx virus fluid and AKT virus fluid. 14 After 14 days of infusion, all rats were sacrificed, and their aortic tissues were removed for analysis.
Hematoxylin and eosin (HE) staining
The AAA wall tissue was fixed in 10% formalin buffer for 24 h. After dehydration and permeation, the tissues were cut into sections with a slicer (YD-315, Zhejiang Jinhua Yidi test equipment). then baked at 60°C for 12 h. After deparaffinization and rehydration, the sections were stained with Hematoxylin and eosin solutions, each for 5 min, and dehydrated in graded alcohol (95%∼100%) for 10 min. The sections were soaked in xylene twice, each for 10 min, then sealed with neutral gum. 15 The wall thickness of the abdominal aorta in each group was captured with a microscope imaging system.
Western Blot(WB)
Total protein was extracted from abdominal aortic tissue with RIPA lysis buffer (#P0013B, Beyotime Biotechnology). The supernatant containing protein was mixed with loading buffer, and bathed in boiling water for 5 min. The target protein was isolated by SDS-PAGE gel electrophoresis. The protein was transferred to a nitrocellulose membrane, which was sealed with 5% skim milk for 90 min. The membranes were incubated with primary antibodies α-SMA (14395-1-AP, 1:4000, proteintech), SM22α (10493-1-AP, 1:1000, proteintech), OPN (22952-1-AP, 1:2000, proteintech), MMP2 (10373-2-AP, 1:1000, proteintech), Timp (ab61224, 1:1000, abcam), PI3K (ab227204, 1:2000, abcam), p-PI3K (ab182651, 1:1000, abcam), AKT (10176-2-AP, 1:3000, proteintech), p-AKT (ab81283, 1:6000, abcam), ID2 (PA5-87554, 1:1000, ThermoFisher), and β-actin (66009-1-Ig, 1:5000, proteintech) for 90 min. After incubation, the membranes were washed 3 times with phosphate-buffered saline/0.1% Tween 20 (PBST), each for 15 min. Then the membranes were incubated with secondary antibodies HRP goat anti-mouse IgG (SA00001-1, 1:5000, proteintech), and HRP goat anti-rabbit IgG (SA00001-2, 1:6000, proteintech) for 90 min. After incubation, the membranes were washed 3 times with PBST, each for 10 min. To visualize the bands, the membranes were incubated with Enhanced chemiluminescence solution (K-12045-D50, Advansta) for 1 min, and exposed with X-ray films for 10 min in a cassette.
Immunohistochemical analysis (IHC)
Paraffin sections taken from the rat abdominal aortas were baked at 60°C for 12 h. The sections were deparaffinized and rehydrated, and the antigen were repaired using microwave oven heating. 1% periodic acid was added to the sections for 10 min at room temperature. The sections were washed with PBS three times for 3 min each and incubated with primary antibodies: α-SMA (14395-1-AP, 1:200, proteintech), SM22α (10493-1-AP, 1:200, proteintech), OPN (22952-1-AP, 1:200, proteintech), PI3K (ab227204, 1:200, abcam), AKT (10176-2-AP, 1:200, proteintech), p-PI3K (ab182651, 1:200, abcam), p-AKT (ab81283, 1:200, abcam), ID2 (AP5-87554, 1:200, ThermoFisher), and Elastin (15257-1-AP, 1:200, proteintech) overnight at 4°C. The sections were washed with PBS three times for 5 min each, then incubated with secondary antibodies at 37°C for 30 min and washed in similar manner as washing primary antibodies. DAB solution was used to develop color for 5 min and rinsed with distilled water. The sections were dehydrated by multi-gradient alcohol and sealed with neutral gum, and ultimately examined under a microscope.
Fluorescence quantitative real-time polymerase chain reaction (qRT-PCR)
Total mRNA was extracted from abdominal aortic tissue by Trizol (15596026, ThermoFisher). As templates, the total RNA was reverse transcribed with mRNA reverse transcription kit (CW2569, Kang Weiji, Beijing, China) while cDNA was obtained. The concentration of RNA was measured by the absorbance at 260 nm, and RNA quality was assessed using 260/280 ratio ≥2. Primer sequences of β-actin, AKT, α-SMA, ID2, OPN, PI3K, and SM22α were derived from Primer Bank. cDNA was amplified by adding the fluorescent dye Ultrasybr Mixture (CW2601, Kangwei Shiji, Beijing, China). The fluorescence signals during PCR were monitored in real-time. β-actin was used as an internal reference. qRT-PCR was performed with the SYBR Green RT-PCR Kit (Takara Biotechnology, Dalian, China) using a Light Cycler 480 II system (Roche Diagnostics, Basel, Switzerland). The relative expression of mRNA was determined using the 2-ΔΔCt method by normalizing the expression against that of β-actin to calculate the relative expression amount of gene. The primers sequences of β-actin, AKT, α-SMA, ID2, OPN, PI3K, and SM22α were listed in Supplement Table 1.
Enzyme-linked immunosorbent assay (ELISA)
The plasma sample was treated with TGF-β1 ELISA Kit (CSB-E04727R, Wuhan, Huamei) and all procedures were carried out according to the instructions. The optical density (OD value) of each hole was measured at 450 nm with a multi-functional enzyme labeled analyzer (MB-530, Shenzhen, Huisong). The concentration of TGF-β1 in all serum samples was calculated according to the standard curve.
Statistical analysis
Statistical analysis was performed using SPSS 20.0 (IBM). Normal distribution test was conducted for all continuous variables. After confirmation of variance equality among different groups, statistical differences between 2 independent groups were analyzed using unpaired Student t test, whereas statistical difference among ≥3 groups was analyzed using one-way analysis of variance (ANOVA). The representation of data is average ± standard deviation (X±SD). p value less than 0.05 was considered statistically significant.
Results
Daxx inhibits vascular wall thickening of abdominal aorta
Rupture of an AAA is often a lethal event and, in many instances, the first and only clinical evidence of pathology is aortic expansion.
16
In order to investigate whether Daxx could inhibit abdominal aortic dilatation, HE staining was used to compare the thickness of the abdominal aortic wall from each group (Figure 1(a) and (b)). Compared with the Sham group, the wall thickness of AAA group and AAA + Vector group were increased. However, the wall thickness of the AAA + Daxx group was thinner than that of the AAA + Vector group, indicating that Daxx could significantly inhibit the thickening of the abdominal aortic wall. In contrast, the thickness of the aortic wall in the AAA + Daxx + AKT group was greater than the AAA + Daxx group, suggesting that overexpression of AKT might counteract the inhibitory effect of Daxx. Daxx inhibits vascular wall thickening of abdominal aorta. (a). HE staining detected the thickness of vessel wall. (b). Quantitative analysis of A. Scale bar = 100 μm; the magnification is 40 times; * p <0.05 vs Sham group; # p <0.05 vs AAA + vector group; & p <0.05 vs AAA + Daxx group.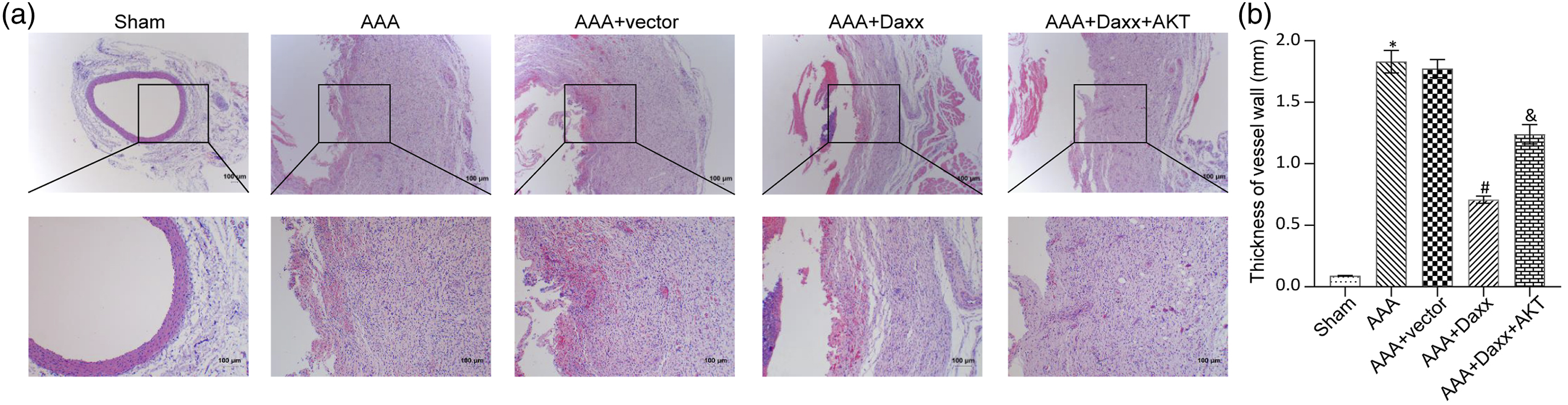
Daxx inhibits the degradation of elastin and collagen
Elastin is an important factor in determining the shape of arteries.
17
Failure of the collagen safety net contributes to the formation of an abdominal aortic aneurysm, and degradation of elastin and collagen is directly related to up-regulation of MMPs activity.
18
To determine whether Daxx could inhibit destruction of elastin and collagen, WB was used to detect tissue expression levels of MMPs and Timp (Figure 2(a) and (b)), and IHC was performed to evaluate expression levels of Elastin (Figure 2(c) and (d)). Compared with the Sham group, the expression levels of MMP2 and MMP9 in the AAA group and the AAA + Vector group were significantly increased, whereas the expression level of Timp and the content of Elastin were decreased. In contradistinction, the expression levels of MMP2 and MMP9 in the AAA + Daxx group were lower than those in the AAA + Vector group, and the expression levels of Timp and Elastin were higher. This provided evidence that Daxx inhibited the degradation of elastin and collagen. The expression levels of MMP2 and MMP9 in the AAA + Daxx + AKT group were higher than those in the AAA + Daxx group, again demonstrating that AKT counteracted the inhibitory effect of Daxx. Daxx inhibits the degradation of elastin and collagen. (a). WB was used to measure the expressions of MMP2, MMP9 and Timp. (b). Quantitative analysis of A. (c). IHC evaluated the expression levels of Elastin. (d). Quantitative analysis of C. Scale bar = 100 μm; the magnification is 400 times; * p <0.05 vs Sham group; # p <0.05 vs AAA + vector group; & p <0.05 vs AAA + Daxx group.
Daxx inhibits the transformation of vascular smooth muscle cells from contractile phenotype to synthetic phenotype
Phenotypic conversion of VSMC is considered a key pathophysiological change in various vascular diseases. In AAA, VSMC changes from contractile to synthetic phenotype.
19
To investigate whether Daxx could inhibit this process, WB (Figure 3(a) and (b)) and IHC (Figure 3(c) and (d)) were used to detect tissue expression of SMC phenotypic marker protein. The relative expression level of genes related to SMC phenotypic marker was measured by qRT-PCR (Figure 3(e)). Compared with the Sham group, the expression levels of contractile phenotype related proteins α-SMA and SM22α in the AAA group and the AAA + Vector group were significantly decreased, whereas the expression levels of synthetic phenotype related proteins OPN were increased. Compared with the AAA + Vector group, the expression levels of α-SMA and SM22α in the AAA + Daxx group were elevated, and the expression levels of OPN were decreased. These results indicated that Daxx inhibited transformation of vascular smooth muscle cells from contractile to synthetic phenotype. The content of α-SMA and SM22α in the AAA + Daxx + AKT group was lower than the AAA + Daxx group, and the content of OPN was higher. Again, it appeared that AKT impaired the inhibitory effect of Daxx. Daxx inhibits the transformation of vascular smooth muscle cells from contractile phenotype to synthetic phenotype. (a). The expressions of α-SMA, SM22α and OPN were detected by WB. (b). Quantitative analysis of A. (c). IHC examined the expression levels of α-SMA, SM22α and OPN. (d). Quantitative analysis of C. (e). The relative expressions of α-SMA, SM22α and OPN were evaluated by qRT-PCR. Scale bar = 100 μm; The magnification is 400 times; * p <0.05 vs Sham group; # p <0.05 vs AAA + vector group; & p <0.05 vs AAA + Daxx group.
Daxx inhibits the PI3K/AKT/ID2 signaling pathway
The PI3K/AKT/ID2 signaling pathway has been reported to be extremely active in AAA.
11
To investigate whether Daxx inhibits the PI3K/AKT/ID2 signaling pathway, we detected protein expression levels of PI3K/AKT/ID2 signaling pathway by WB (Figure 4(a) and (b)) and IHC (Figure 4(c) and (d)), and the gene expression levels of PI3K/AKT/ID2 signaling pathway through qRT-PCR (Figure 4(e)). Compared with the Sham group, the expression levels of PI3K, AKT, p-PI3K, p-AKT, and ID2 were significantly increased in the AAA group and the AAA + Vector group. Compared with the AAA + vector group, the expression levels of PI3K, AKT, p-PI3K, p-AKT, and ID2 were decreased in the AAA + Daxx group, supporting the concept that Daxx reduced overactivation of the PI3K/AKT/ID2 signaling pathway. The expression levels of PI3K, AKT, p-PI3K, p-AKT, and ID2 in the AAA + Daxx + AKT group were slightly higher than the AAA + Daxx group (Figure 5). Daxx inhibits the PI3K/AKT/ID2 signaling pathway. (a). WB was performed to detect expressions of AKT, p-AKT, PI3K, p-PI3K and ID2. (b). Quantitative analysis of A. (c). IHC examined the expression levels of AKT, p-AKT, PI3K, p-PI3K and ID2. (d). Quantitative analysis of C. (e). The relative expressions of AKT, PI3K and ID2 were evaluated by qRT-PCR. Scale bar = 100 μm; the magnification is 400 times; * p <0.05 vs Sham group; # p <0.05 vs AAA + vector group; & p <0.05 vs AAA + Daxx group. Daxx inhibits the PI3K/AKT signaling pathway by regulating TGF-β1. ELISA detected the concentration of TGF-β1. * p <0.05 vs Sham group; # p <0.05 vs AAA + vector group; & p <0.05 vs AAA + Daxx group.

Daxx inhibits the PI3K/AKT signaling pathway by regulating TGF-β1
TGF-β1/Smad and PI3K/AKT signaling pathways often interact with each other. 20 To investigate whether Daxx, TGF-β1 and PI3K/AKT signaling pathways were interactive, the plasma TGF-β1 expression level in each group was measured by ELISA. Compared with the Sham group, the expression level of TGF-β1 in the AAA group and the AAA + Vector group was increased, whereas the expression level of TGF-β1 in the AAA + Daxx group was decreased compared with the AAA + Vector group. The expression level of TGF-β1 in the AAA + Daxx + AKT group was slightly higher than the AAA + Daxx group. These results were consistent with proteins related to the PI3K/AKT signaling pathway. Therefore, we concluded that Daxx may play a role in inhibiting the PI3K/AKT signaling pathway by regulating TGF-β1.
Discussion
AAA is a permanent, pathological dilatation of the abdominal aorta, which is asymptomatic in its early stages but results in extremely high mortality once it ruptures. 21 Although surgical treatment is effective, not all patients are surgical candidates. Therefore, we sought to investigate the pathobiology of AAA, ultimately to identify a pharmacological approach to arrest the expansion of or even prevent AAA. To investigate whether Daxx could inhibit the pathological aortic VSMC switch from contractile to synthetic phenotype that characterizes AAA, we constructed animal models and sequentially identified changes of related indicators at tissue and molecular levels. At the conclusion of our investigation, we determined that Daxx inhibited dilatation of AAA through regulation of the TGF-β1-mediated PI3K/AKT/ID2 signaling pathway.
Dense perianeurysmal fibrosis and thickening of the AAA wall results from a relatively large amount of collagen that migrates from the tunica media to the aortic intima. 22 Comparison of aortic wall thickness in each group, confirmed the Ang-II induced abdominal aortic wall thickening, Importantly, overexpression of Daxx inhibited this increased thickness, a clear indicator of treatment efficacy. Similarly, IMD treatment reduced the incidence of AAA and the maximum aortic diameter in Ang-Ⅱ-induced AAA rat models. 23 In healthy aorta, ECM provides a physical scaffold for the cellular composition of the arterial wall, which is continuously remodeled to maintain its integrity. Its main components are collagen and elastin. 24 In AAA, ECM is markedly decomposed by MMP compromising the structure of the aorta, which leads to dilatation and aneurysm formation. 25 Timp is known to significantly inhibit the activity of MMP2 and MMP9, 26 and in this study, MMP2 and MMP9 were highly expressed in AAA, and Timp and Elastin expressions were decreased. Daxx was demonstrated to inhibit the expression of MMP2 and MMP9, thus protecting elastin and collagen. Other substances, such as interleukin-12 and interleukin-23, have also been shown to slow AAA production by inhibiting the expression of MMPs. 27
The VSMC is the principal cell in the aortic wall, which not only plays a key role in maintaining vascular structure but also performs a variety of functions. 28 VSMC differentiates into different phenotypic states during development, expressing typical contractile markers such as α-SMA and SM22. In mature normal blood vessels, VSMC is characterized by a contractile phenotype. In atherosclerosis, the expression of SMC contractile markers is down-regulated while the expression of OPN is up-regulated. 29 In this study, the expression of SM22α was decreased and the expression of OPN was increased in AAA. After adding Daxx, the expression levels of α-SMA and SM22α increased, and the expression levels of OPN decreased, indicating that Daxx inhibited the transformation of vascular smooth muscle cells from contractile type to synthetic type. Similarly, prior research suggested that Daxx expression inhibited Ang-Ⅱinduced proliferation and migration of VSMC, 6 consistent with our findings.
The PI3K/AKT/ID2 signaling pathway can regulate the phenotypic differentiation of cells. When PI3K and AKT are phosphorylated, the PI3K/AKT signaling pathway is activated. 30 When DJ-1 (a homodimeric protein) is used to intervene in cardiovascular diseases, both Daxx and the PI3K/AKT signaling pathway can be used as DJ-1 targets to jointly treat the pathophysiology of cardiovascular diseases. 31 In this study, mRNA and protein expressions related to the PI3K/AKT/ID2 signaling pathway were significantly up-regulated in AAA, and the PI3K/AKT/ID2 signaling pathway was highly activated, but Daxx could significantly reduce activation of the PI3K/AKT/ID2 signaling pathway. This result further supported the evidence of interaction between Daxx and the PI3K/AKT/ID2 signaling pathway.
TGF-β1 is a bifunctional regulator that inhibits or stimulates cell proliferation, and plays a key role in controlling embryogenesis, inflammation and tissue repair, as well as maintaining homeostasis in adult tissues. 32 In the study of thoracic aortic aneurysms, the content of TGF-β1 in diseased tissues was much higher than that in normal tissues. 33 Similarly, we found the concentration of TGF-β1 in AAA was increased, and Daxx inhibited the increase of TGF-β1 concentration in AAA. These results are consistent with previous studies and similar to changes of proteins related to the PI3K/AKT/ID2 signaling pathway. Taken together, we have provided evidence that Daxx regulated the concentration of TGF-β1 and thereby inhibited the PI3K/AKT/ID2 signaling pathway.
Our study has several limitations. First, the Daxx knock down group was not included in the investigation, and it remains to be explored whether the experimental results would remain consistent with the conclusion of this study, if the tissue content of Daxx was very low. Furthermore, TGF-β1 not only mediates the PI3K/AKT/ID2 signaling pathway, but also regulates the specific gravity of ECM in tissues, 34 suggesting that Daxx may also inhibit AAA expansion by affecting other TGF-β1-mediated pathways. Additionally, the sample size was based on empirical considerations, without formal calculation, which is another limitation of this study.
Conclusion
In this study, we found that Daxx inhibited thickening of the abdominal aortic wall, protected elastin and collagen from damage, reduced transformation of vascular smooth muscle cells from contractile type to synthetic type, and down-regulated the activation of TGF-β1-mediated PI3K/AKT/ID2 signaling pathway. Therefore, Daxx inhibited the expansion of AAA through inhibiting the activation of the TGF-β1-mediated PI3K/AKT/ID2 signaling pathway. Finally, the PI3K/AKT/ID2 signaling pathway regulated by TGF-β1 could reasonably serve as a potential therapeutic target for the treatment of AAA.
Supplemental Material
sj-pdf-1-eji-10.1177_1721727X221091532 - Supplemental material for Daxx ameliorates abdominal aortic aneurysm through inhibiting the TGF-β1-mediated PI3K/AKT/ID2 signaling pathway
Supplemental material, sj-pdf-1-eji-10.1177_1721727X221091532 for Daxx ameliorates abdominal aortic aneurysm through inhibiting the TGF-β1-mediated PI3K/AKT/ID2 signaling pathway by Jie Chen, Guo-zuo Xiong, Dong-yang Luo, Qing-qing Zou, Yang-yi-jing Wnag and Guo-shan Bi in European Journal of Inflammation
Footnotes
Acknowledgments
We would like to give our sincere gratitude to the reviewers for their constructive comments.
Authors’ Contribution
Conception and study design: GSB, JC, GZX; Data acquisition: DYL, QQZ; Data analysis: JC, YYJW; Manuscript drafting: JC, GZX; Manuscript revising: GSB. All authors read and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by a grant from Project of Hunan Provincial Health Commission (grant number: B2019111); Major guidance project of Hunan Provincial Health Commission (grant number: 202104010169); Hunan Provincial Natural Science Foundation of Youth Project (grant number: 2020JJ5504); General guidance project of Hunan Provincial Health Commission (grant number: 20201949); Hengyang Science and Technology Bureau Guidance Project (grant number: 2019jh010989).
Ethics approval
*All experimental protocols were approved by the Animal Ethics Committee of the laboratory animal center of University of South China. (APPROVAL NUMBER/ID: No20190130c0800510).
Animal welfare
*The animals used were treated in accordance with the Guide for the Care and Use of Laboratory Animals, published by the National Institutes of Health (NIH).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
