Abstract
Background
Systemic inflammation can significantly impact gliomas’ onset, progression, and prognosis. Glioblastoma multiforme (GBM) represents the glioma subtype characterized by the most profound inflammatory and immunosuppressive states. Consequently, various blood-borne biomarkers have been scrutinized concerning their prognostic value in GBM patients.
Objective
We sought to investigate whether the recently introduced Global Immune-Nutrition-Inflammation Index (GINI) holds prognostic significance for GBM patients treated with the standard Stupp protocol.
Methods
We retrospectively analyzed the data from a cohort of newly diagnosed GBM patients receiving the standard Stupp regimen using the propensity score-matching methodology. The GINI was computed using the original formula: GINI = [(C-reactive protein × Monocytes × Platelets × Neutrophils) ÷ (Albumin × Lymphocytes)]. We employed receiver operating characteristic (ROC) curve analysis to identify the optimal cutoff values for GINI, which could help distinguish between different survival outcomes. The primary and secondary objectives were the differences in overall survival (OS) and progression-free survival (PFS) between the GINI groups.
Results
The optimal GINI cutoff value was 1350. Out of 294 eligible patients, 211 were PSM-matched: GINI<1350 (
Conclusion
High pretreatment GINI values are robustly and independently associated with inferior PFS and OS outcomes in selected GBM patients who receive standard Stupp protocol. These findings suggest that if further confirmed, the novel GINI could serve as a valuable biological marker for the prognostic stratification of GBM patients.
Introduction
Glioblastoma multiforme (GBM) represents the predominant form of malignant primary brain tumors, constituting approximately 50% of all malignant neoplasms within this anatomical region. 1 GBM is characterized by its highly aggressive nature, with nearly inevitable recurrences despite the standard of care, which involves maximum-safe surgery followed by concurrent radiotherapy (RT) and temozolomide (TMZ) alongside adjuvant TMZ, commonly referred to as the Stupp protocol. Reflecting the pernicious characteristics of the tumor, median overall survival (OS) is typically less than 18 months.2,3 Even in well-selected patients in benchmark clinical trials, 4 the 5-year OS remains below 10%. Regrettably, despite substantial advancements in diagnostic imaging, surgical procedures, RT techniques, chemotherapy, and supportive care, survival rates have not surpassed those of the standard Stupp regimen. 5 The combination of adjuvant tumor-treating fields (TTF) treatment during adjuvant TMZ has emerged as an innovative therapeutic option for newly diagnosed GBMs. While this approach has been approved for routine use, the median OS rate for patients has only seen a marginal increase from 15 to 18 months to 20.9 months. 6 It is regrettable that in several nations, TTF therapy may not be accessible.
Given the inevitability of disease recurrences and unfavorable survival outcomes, stratifying GBM patients into prognostic cohorts is crucial for tailoring personalized treatment regimens and meeting individual requirements. These endeavors may facilitate the recommendation of treatment intensification for appropriately selected patients, thereby enhancing their prognosis while avoiding futile and aggressive interventions and their attendant toxicities for others. For newly diagnosed GBMs, in addition to traditional variables such as age, neurological functions, intracranial pressure status, and recursive partitioning analysis (RPA) group, the status of novel genetic and molecular markers like the isocitrate dehydrogenase (IDH)-1/2 mutation, 1p/19q codeletion, and O6-methylguanine-DNA methyl-transferase (MGMT) gene promoter methylation play a crucial prognostic role. Despite their successful patient stratification capacities, the accessibility difficulties for the genetic markers because of their prohibitive cost render them hard to apply for patients of the low-income countries. The extent of tumor resection, use of steroids, type of chemo-RT, and preferred adjuvant chemotherapy regimens are also paramount in determining the prognosis of GBMs.7,8 Nevertheless, there are unexplained disparities in the survival outcomes of individuals with GBM who exhibit similar clinical, pathological, genetic, molecular, and treatment characteristics. The outcomes of the benchmark randomized phase III Organization for Research and Treatment of Cancer/National Cancer Institute of Canada (EORTC-NCIC) trial are a compelling illustration of the substantial variations in survival outcomes. 4 The long-term results of this study revealed that patients with MGMT-methylation who underwent the Stupp protocol had a median OS of 23.4 months (range: 18.6–32.8 months). However, the difference in OS between the lower and upper ends of the range was 14.2 months, suggesting that some patients experienced a 76.3% longer survival despite having the same MGMT status as others. Therefore, it’s essential to identify new variables with more potent prognostic abilities to enhance the accuracy of predicting outcomes for GBM patients, irrespective of the even distribution of other factors.
Emerging data indicates that systemic inflammation may have a substantial influence on the genesis, progression, and prognosis of gliomas. 9 GBM, compared to other gliomas, exhibits a remarkably higher ability to cultivate a potent inflammatory and immune-suppressed environment, fostering a highly aggressive, treatment-resistant disease phenotype that can easily evade immune surveillance.9,10 Several blood-based markers of systemic inflammation, notably serum protein levels and peripheral blood cell counts, including C-reactive protein, albumin, platelets, monocytes, neutrophils, and lymphocytes, have been investigated for their prognostic utility in GBM patients. Previous studies and meta-analyses have consistently demonstrated a solid and unmistakable association between these biomarkers, individually or in various combinations, and the survival outcomes of GBM patients.11–21
The Global Immune-Nutrition-Inflammation Index (GINI) is a recently developed nearly all-inclusive immunological, inflammatory, and nutritional biomarker that incorporates cellular and biochemical components of these processes, specifically pretreatment C-reactive protein (CRP), albumin, monocyte, platelet, neutrophil, and lymphocyte measurements. 22 These parameters are readily accessible in routine complete blood count and biochemistry tests without additional cost. Moreover, they can be easily and objectively reproduced in any laboratory and are not prone to subjectivity, such as performance or functional status. In the first GINI study, Topkan and colleagues examined 802 newly diagnosed stage IIIC non-small cell lung cancer (NSCLC) patients who received definitive concurrent chemo-RT. They found that patients with a GINI≥1562 had significantly shorter median survival times than those with a GINI<1562. 22 Since GBM is the glioma subtype inducing the most profound inflammatory response and immune suppression, we postulated that the novel GINI might be a valuable tool for more accurately predicting GBM patients' treatment outcomes. Hence, current propensity score-matching (PSM) analysis aimed to explore the likely prognostic relevance of the novel GINI in newly diagnosed GBM patients treated with the standard Stupp regimen.
Patients and methods
Study population and ethics
A retrospective review of the medical records of all newly diagnosed GBM patients who received the Stupp protocol at the Department of Radiation Oncology, Baskent University Medical Faculty, over the period spanning from February 2007 to December 2022, was conducted. The study enrolled participants who met the following criteria: age between 18 and 80 years, Eastern Cooperative Oncology Group (ECOG) performance status of 0-1, histologically confirmed diagnosis of GBM, no previous chemotherapy or cranial RT, availability of preoperative and postoperative gadolinium-enhanced magnetic resonance imaging (MRI) scans, as well as availability of information on chemotherapy and RT. In addition, participants were required to have baseline complete blood count and biochemistry test results to confirm satisfactory hematologic, renal, and hepatic functions, the absence of active infection, and a history of prior immunosuppressive diseases. The analysis excluded patients who did not receive concurrent or adjuvant TMZ and those who underwent hypofractionated RT regimes or whole-brain RT. This methodology aimed to ensure that the study results were derived from a uniform patient cohort, thus minimizing the potential confounding effects of these treatments on the investigated outcomes.
The present retrospective study followed the guidelines outlined in the Helsinki Declaration and its subsequent revisions. Before collecting patient data, the Institutional Review Board of Baskent University reviewed and approved the study design (Project No. KA19/167). Each eligible patient provided their signed informed consent before commencing the recommended treatment. This consent allowed for examining blood tests and pathological specimens and disseminating research results through academic publications.
Concurrent and adjuvant treatment
Upon initial assessment, all patients underwent a comprehensive evaluation. If deemed feasible, a maximal safe resection was performed following the established institutional protocols for treating GBM patients. After the surgical procedure or biopsy, a cumulative dose of 60 Gy (2.0 Gy/day in 30 fractions) of partial brain RT was delivered using 3-dimensional conformal RT (3D-CRT) or intensity-modulated RT (IMRT) techniques. Per our corporate care standards, the implementation of treatment plans included co-registered CT and contrast-enhanced MRI fusion images, regardless of the RT technique employed. The concurrent treatment phase was commenced 2 weeks after a burr hole stereotactic biopsy and 4 to 6 weeks after the open surgery procedures, contingent upon the patient’s recovery and the status of wound healing. All patients received concurrent TMZ (75 mg/m2/day, 7 days per week, for 6 weeks) during the RT course. In addition, patients were administered trimethoprim-sulfamethoxazole prophylaxis to mitigate the risk of
Measurement of GINI
The pretreatment GINI was calculated according to the original formula published by Topkan and colleagues 22 : GINI = [(C-reactive protein × Monocytes × Platelets × Neutrophils) ÷ (Albumin × Lymphocytes)], using the C-reactive protein, monocyte, platelet, neutrophil, albumin, and lymphocyte measures obtained immediately before the first fraction of concurrent RT and the first dose of TMZ. The CRP and albumin measurements were carried out using the Abbott Architect c8000 Biochemistry Autoanalyzer, while blood cell counts were determined using the RUBY CELL-DYN Ruby version 2.2, following the manufacturer’s instructions for both.
Response assessment
We conducted brain MRI scans at 2- and 3-month intervals during the first and second follow-up years. Subsequently, we regularly scanned patients at 6-month intervals or more frequently based on their health condition until radiological evidence of disease progression or the last follow-up or death. The Response Assessment in Neuro-Oncology (RANO) Working Group Report was utilized to assess and record responses using serial MRI scans. 23 The records represented the best response achieved following RT and concurrent TMZ completion.
Statistical analyses
This retrospective cohort analysis investigated the potential relationship between pretreatment GINI values and survival outcomes. The primary and secondary endpoints were OS and progression-free survival (PFS). Survival times were operationally defined as the duration between the onset of concurrent treatment and either the date of death from any cause or the final visit for OS and the duration between the commencement of concurrent therapy and the date of the initial observation of disease progression or death or the final visit for PFS.
Quantitative variables were delineated using medians and ranges, while categorical variables were characterized through percentage frequency distributions. Where necessary, the study participants were stratified into relevant groups to facilitate intergroup comparisons. Categorical data was employed to classify patients into subgroups based on factors such as resection type. Receiver operating characteristic (ROC) curve analysis was used to assess continuous variables, such as GINI, to determine the optimal cutoff values for stratifying the study population into two groups with significantly different OS and PFS outcomes. The event of death (for OS) and disease progression/death (for PFS) were used as the endpoints for ROC curve analysis to find the ideal cutoff. We used the Youden (J) index to determine the optimal cutoff value, defined as the point where the J-index [(sensitivity + specificity) - 1] reaches its highest value between the curve and the neutral line in the ROC curve analysis graph. The frequency distributions of the groups under investigation were assessed and compared using the Chi-square test, Pearson’s exact test, and Spearman’s correlation estimations. Survival outcomes were estimated using the Kaplan-Meier method, and comparisons between different groups were made using two-sided log-rank testing. The Cox proportional hazards model was used for multivariate comparisons, incorporating only the factors indicating univariate significance. Any two-tailed
We employed the Bonferroni correction to minimize false-positive results when conducting subgroup analyses involving three or more subcategories, such as conceivable interactions between the survival outcomes and resection types or GINI groups. This approach ensured that the probability of erroneously rejecting the null hypothesis was at an acceptable threshold. We used PSM analysis, a quasi-experimental approach, to create an artificial control group by pairing each treated unit with a non-treated unit that had comparable features. This process of matching is an established statistical methodology that allows the researcher to assess the influence of an intervention. Therefore, we computed propensity scores for the GINI groups to address any potential confounding factors and ensure that the GINI groups were adequately balanced and similar. To accomplish this objective, we used the closest-neighbor matching method in conjunction with logistic regression. Using a caliper of 0.2 and without replacement, we were able to create matched groups with a 1:1 ratio. Age, gender, performance score, RPA group, resection type, and adjuvant TMZ cycles were included in the matching criteria.
Results
Pretreatment and treatment characteristics for the entire study cohort and propensity score matched patients per Global Immune-Nutrition-Inflammation Index group.

Abbreviations: GINI: Global Immune-Nutrition-Inflammation Index; PSM: Propensity score-matched; KPS: Karnofsky performance score; RTOG RPA: Radiation Therapy Oncology Group recursive partitioning analysis; GTR: Gross total resection; STR: Subtotal resection; IDH: Isocitrate dehydrogenase; CRP: C-reactive protein.
Comparative treatment outcomes in propensity score matched Global Immune-Nutrition-Inflammation Index groups.

Abbreviations: GINI: Global Immune-Nutrition-Inflammation Index; 3D-CRT: 3-dimensional conformal radiotherapy; IMRT: Intensity-modulated radiotherapy; BEVIRI: bevacizumab plus irinotecan; RO: reoperation; SRS/SRT: stereotactic radiosurgery/stereotactic radiotherapy; mo.: Months; CI: Confidence interval.
Before proceeding with the PSM analysis, an ROC curve analysis was conducted to identify a potential GINI cutoff that could significantly impact the treatment outcomes. The results showed statistical significance at 1347 for PFS [area under the curve (AUC): 88.1%; sensitivity: 79.1%; specificity: 73.9%; Youden index: 0.530] and at 1350 (AUC: 85.7%; sensitivity: 75.7%; specificity: 74.1%; Youden index: 0.498) for OS (Figure 1). Given the proximity of the two cutoffs, we confidently determined a unified threshold of 1350 points to ease subsequent analysis. Our decision was based on careful consideration and rigorous analysis, which showed that this cutoff did not result in any discernible patient migration between the two distinct GINI cohorts. Consequently, the patients were divided into two unique cohorts based on their GINI scores: <1350 and ≥1350. Then, the PSM analysis revealed 211 matching patients, with 95 and 116 individuals assigned to the GINI <1350 and GINI ≥1350 cohorts, respectively. Therefore, all subsequent data presented in this study represents the outcomes of the PSM cohorts (Figure 2). The results of receiver operating characteristic curve analyses depicting the relationship between the pretreatment Global Immune-Nutrition-Inflammation Index measures and the survival status after the standard Stupp protocol: (a) progression-free survival; (b) overall survival. Comparative survival results between the Global Immune-Nutrition-Inflammation Index (GINI) groups after the standard Stupp protocol. (a) Progression-free survival. (b) Overall survival (dark blue: GINI <1350; red: GINI ≥1350).
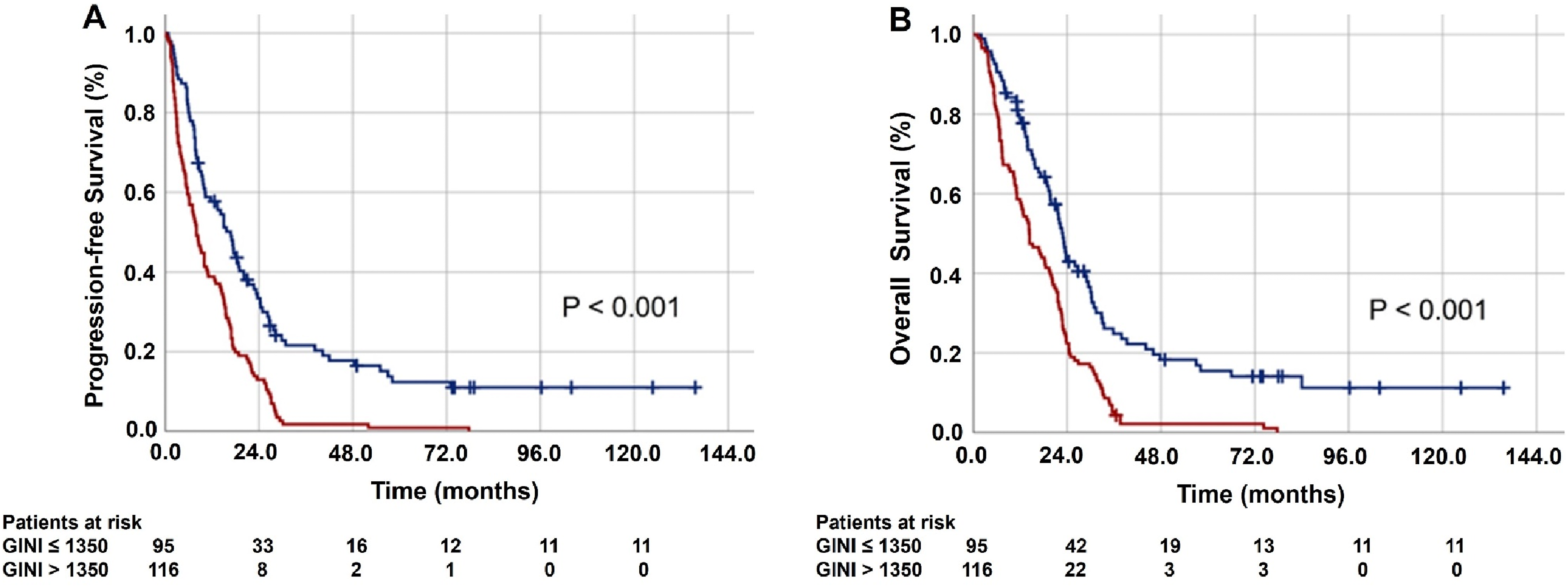
The results of the comparative analyses between the two GINI score groups revealed that patients with a GINI score of ≥1350 experienced significantly shorter median PFS (8.0 vs 16.8 months;
Results of univariate and multivariate analysis.

Note: To prevent biased results CRP, albumin, neutrophils, monocytes, platelets, and lymphocytes counts were not included in the multivariate analysis as they were constituents of the GINI.
Abbreviations: PFS: Progression-free survival; HR: Hazard ratio; OS: Overall survival; KPS: Karnofsky performance score; RTOG RPA: Radiation Therapy Oncology Group recursive partitioning analysis; GTR: Gross total resection; STR: Subtotal resection; 3D-CRT: 3-dimensional conformal radiotherapy; IMRT: Intensity-modulated radiotherapy; GINI: Global Immune-Nutrition-Inflammation Index.
Discussion
GBM is universally recognized as the glioma subtype associated with the poorest prognosis, even when subjected to the most efficacious treatment regimen, commonly referred to as the Stupp protocol.
4
Nevertheless, survival outcomes among patients with similar demographics, disease characteristics, and treatment modalities significantly vary, thereby underscoring the compulsory need for developing advanced prognostic markers that surpass the existing ones regarding efficacy and precision. Hence, the primary objective of this study was to assess the prognostic utility of the novel GINI in stratifying GBM patients into distinct prognostic groups based on PFS and OS outcomes. Our findings indicate that a high baseline GINI (≥1350) represents a novel biomarker significantly associated with inferior median and long-term PFS (
The conventional prognostic factors for newly diagnosed GBM patients typically encompass age, extent of tumor resection, neurological functions, intracranial pressure status, RTOG RPA group, administration of steroids, selected concurrent and adjuvant chemotherapy, and the use of salvage therapies. More recently, this list has also included molecular markers such as the IDH-1/2 mutation, 1p/19q codeletion, and MGMT gene promoter methylation.7,8 Several combinations of these factors have proven to be effective in predicting the prognosis of these patients. However, the vast majority of studies have consistently incorporated various clinical characteristics, some of which may have been subjectively assessed, such as neurological functions, work capacity, performance score, and alterations in personality.24,25 Additionally, the findings of Oszvald et al.
26
and Chaichana et al.
27
have suggested that the significance of age may be less substantial than indicated by the RTOG RPA. Our research confirms age ≥50 years, KPS 70–80, RTOG RPA class V, STR and biopsy only, and lack of salvage therapy as poor prognostic factors for PFS (
Our research revealed notable findings about the prognostic significance of GINI measurements before therapy in these individuals. Specifically, a GINI ≥1350 was strongly linked to significantly shorter median PFS (8.0 vs 16.8 months;
Discussing these novel discoveries with a conventional proof-based approach is challenging because there are no similar GBM studies. Nevertheless, they perfectly align with a previously published first-of-its-kind study on stage IIIC NSCLC patients treated with definitive concurrent chemo-RT.
22
In that study, Topkan et al. demonstrated that the newly created GINI efficiently stratified patients into two prognostic groups, with GINI >1532 representing the poor prognostic group. Additionally, we can confidently redefine the GINI as the product of the CRP-to-albumin ratio (CAR) and the pan-immune-inflammation value (PIV). This redefinition allows us to analyze the biochemical and cellular components separately. Two previous studies conducted by Topkan et al. investigated the influence of CAR and PIV indices separately on the outcomes of GBM patients who underwent treatment with the standard Stupp protocol.16,20 In the first study, the results of the multivariate analysis revealed that a presenting high-CAR value was significantly and independently associated with inferior PFS (
The effective treatment of a patient relies on evidence-based research results about prognostic variables. Therefore, it is crucial to ascertain if GINI can serve as a dependable biological indicator for forecasting the prognosis of GBM patients. Prognostic indicators in oncology are clinical markers that aid in predicting the future prognosis of a patient, whether they get treatment or not. 28 These assessments may classify patients into different risk categories and provide information for developing personalized treatment plans depending on the level of risk. However, a prognostic factor should preferably have specific characteristics suitable for academic research. These characteristics include being readily attainable, measured objectively, reproducible, relevant to all patients irrespective of their clinical conditions, and, ideally, cost-effective. Additionally, a prognostic factor should inform clinicians about multiple parameters related to patient and disease characteristics in a single measure that renders it resistant to the influence of intrinsic or extrinsic factors. Given that the novel GINI meets all the criteria for an optimal prognostic factor and displays robust and independent discriminatory ability in predicting patient outcomes, it may be considered a dependable biomarker for the prognostic categorization of GBM patients in everyday oncological practice.
The present study has certain limitations. First, it was a retrospective study conducted at a single center, and the participants were a highly selective group of newly diagnosed GBM patients who received treatment according to a standardized protocol, which inherently introduces potential selection biases that are challenging to predict. Consequently, these findings may not be generalizable to all GBM patients, like those with a baseline KPS ≤60. Second, due to the novelty of the GINI in the GBM literature and the absence of prior investigations to gauge its potential impact on GBM patients' survival outcomes, we could not perform a sample size calculation in this study. As a result, we lacked the necessary information to perform such calculations. However, future studies can use our current findings as a foundation to determine sample size and conduct statistical power analysis, potentially leading to more reliable results. Third, because concurrent and adjuvant TMZ was uniformly administered to all patients, all of whom had KPS ≥70 performance status and received conventionally fractionated RT, our results cannot be generalized to all GBM patients, like those who could not tolerate TMZ, had poorer performance scores, or were undergoing alternative fractionation schemes with different total doses. Fourth, the GINI cutoff value established in this study was predicated on a solitary measurement taken before the commencement of therapy. Consequently, the current cutoff may not be the most precise in categorizing GBM patients into two distinct cohorts with varying clinical outcomes, as all six GINI components are susceptible to significant fluctuations during concurrent and adjuvant treatment phases. Further studies are required to examine the impact of GINI values before, during, and after the treatment in order to identify the best-fit cutoff value(s) that correlate with the outcomes. And fifth, although we have endeavored to minimize heterogeneity in our patient population and potential biases through a PSM analysis that considers patient, tumor, and treatment variables, it is crucial to acknowledge the retrospective nature of our study. Therefore, it is critical to consider our present findings as hypothesis-generating unless they are confirmed by future investigations on larger scales that are specifically tailored to overcome these impediments. However, despite the noted impediments, if our findings are backed by further research, they may efficiently aid in such patients' prognostic stratification and selection of the best-fit individualized therapies.
Conclusions
In conclusion, our findings suggest that a high pretreatment GINI (a nearly all-in-one systemic immune, inflammation, and nutritional biomarker) is robustly associated with poorer survival outcomes in newly diagnosed GBM patients who undergo standard Stupp protocol. If further confirmed through rigorous testing, the novel GINI holds promise for patient stratification and personalized therapeutic decisions.
Footnotes
Author contributions
E.T., N.K.D, S.S., D.O., A.A.B, H.M., B.P., and U.S.; methodology, A, E.T., N.K.D, S.S., D.O., A.A.B, H.M., B.P., and U.S.; software, , E.T., N.K.D, S.S., D.O., A.A.B, H.M., B.P., and U.S.; validation, , E.T., N.K.D, S.S., D.O., A.A.B, H.M., B.P., and U.S.; formal analysis, , E.T., N.K.D, S.S., D.O., A.A.B, H.M., B.P., and U.S.; investigation, , E.T., N.K.D, S.S., D.O., A.A.B, H.M., B.P., and U.S.; resources A, E.T., N.K.D, S.S., D.O., A.A.B, H.M., B.P., and U.S.; data curation, A.K., E.T., D.O., E.E.O., B.P., and U.S.; writing—original draft preparation, E.T., N.K.D, S.S., D.O., A.A.B, H.M., B.P., and U.S.; writing—review and editing, E.T., N.K.D, S.S., D.O., A.A.B, H.M., B.P., and U.S.; visualization, E.T., N.K.D, S.S., D.O., A.A.B, H.M., B.P., and U.S.; supervision, E.T., N.K.D, S.S., D.O., A.A.B, H.M., B.P., and U.S.; project administration, E.T., N.K.D, S.S., D.O., A.A.B, H.M., B.P., and U.S. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Trial registration
This is a retrospective study.
Ethical statement
Data availability statement
The datasets used and/or analyzed during the current investigation are available from the Baskent University Department of Radiation Oncology Institutional Data Access:
