Abstract
Background
Nickel (Ni), commonly-used heavy metals in industrial activities, can lead to embryo and organ toxicity, especially cardiovascular damage. Geraniol (GER) has various beneficial effects such as anti-oxidant, anti-inflammatory, anti-tumor, anti-ulcer, anti-microbial, and neuroprotective activities.
Objective
The objective of this study was to investigate the effect of GER on Ni-induced embryotoxicity and cardiotoxicity in rats.
Methods
40 mother Wistar rats were randomly divided into five groups: Control, GER 250, Ni, Ni + GER 100, and Ni + GER 250. On the 20th day of pregnancy, the animals were sacrificed and fetuses along with blood and tissue samples were collocated for morphological, serological, biochemical, and histopathologic analysis.
Results
Morphological assessments revealed GER’s capacity to mitigate the incomplete ossification of fetal skeletons, indicating a potential safeguarding against the impact of Ni-induced embryotoxicity. Serological and biochemical analyses further affirm GER’s role, with noteworthy reductions in cardiac injury markers, such as CRP, CKMB, CPK, LDH, and troponin, in response to GER administration, thereby suggesting its cardioprotective potential. Moreover, treatment with GER 250 could significantly reduce the level of MDA and increase the level of TAC compared to the Ni group. Histopathological examinations corroborated these findings, underscoring GER’s ability to counteract cardiac injury and diminish structural damage in affected tissue.
Conclusions
These multidimensional analyses indicate the protective prowess of GER against Ni-induced embryotoxic and cardiotoxic effects, shedding light on its potential therapeutic significance in combating adverse impacts stemming from Ni exposure.
Introduction
Nickel (Ni) is one of the most commonly-used heavy metals in modern industry activities, including coinage, catalysis, casting and plating. 1 The Ni-related environmental pollution is one of the major consequences of high utilization of Ni-containing products. 2 The main root of exposure to Ni, in humans, is through oral and respiratory system. 3 Nevertheless, cardiac intervention stents, particularly those composed of cobalt chromium, contain a substantial amount of Ni (up to 35%), contributing significantly to human exposure through this route. 4
After entering the body, this heavy metal can pile up in various organs such as liver, lungs, heart, bones, fat tissue, peripheral nerves and brain 5 and consequently lead to organ toxicity. For example, Ni nanoparticles are responsible for various adverse events, including cancer, skin allergies, atherosclerosis, lung fibrosis, hepatotoxicity, and harmful impacts on fetal development and female reproductive system.6–9 Among the aforementioned toxicities, cardiovascular damage is of a great importance. In the study of Cuevas et al. (2010), it has been affirmed that Ni hydroxide nanoparticles could result in acute endothelial disruption along with alterations in the vaso-constriction and vaso-relaxation. 10 Moreover, acute heart rate alterations and elevated mortality rate in the cases of myocardial infarction have been reported following exposure to Ni.11,12 The underlying mechanism for Ni-induced cardiotoxicity is not completely understood. It could be related to the over-production of reactive oxygen species (ROS) or the impairment of antioxidant system.13–15
Ni could also cause impairment in the fetal development and female reproductive system. Several studies have reported these toxicities in different animal models.16–20 Saini et al. (2013) showed that Ni exposure could lead to severe skeletal anomalies, fetal malformations, and higher mortality rates in the Swiss Albino mice embryos. 21 This toxic effect might be due to the oxidative stress, at least to some extent. Interestingly, in Ku et al (2015) study, the use of vitamin C (an antioxidant) as a pretreatment in the embryos of zebrafish treated with NiSO4 and buprofezin could significantly lower the mortality and the elevated levels of ROS. 22 Altogether, prior investigations underscore the fact that Ni-induced toxic effects can be treat or prevented using antioxidants.22–24
Geraniol (GER), found in different plant resources, including citronella, oranges, red roses, aromatic vegetable oils, and lemon ginger, is an acyclic monoterpene, which not only exhibits anti-inflammatory and antioxidant properties, but has also been reported to have anti-ulcer, antimicrobial, anti-tumor and neuroprotective activities.25,26 Importantly, it could restore the ferric nitrilotriacetate (Fe-NTA)-induced reduction in the activity of antioxidant enzymes, including glutathione peroxidase, glutathione S-transferase and catalase. 27 In the study of Younis et al. (2021), GER demonstrated a protective effect against cardiotoxicity due to doxorubicin 28 . This capacity may be due activation of nuclear factor erythroid 2-related factor 2 (Nrf2), which may consequently enhance Nrf2-related antioxidative signaling pathway, reduce apoptosis and subdue the inflammation. 28 Moreover, GER effectively attenuates inflammation by inhibiting the nuclear translocation of NF-κB, a pivotal transcription factor orchestrating the inflammatory response. Notably, GER has also demonstrated a significant decrease in pro-inflammatory cytokine levels, including TNF-α, IL-1β, and IL-18, in rodent models of doxorubicin-induced cardiotoxicity. 28 This finding suggests a broader anti-inflammatory effect extending beyond the modulation of NF-κB signaling. In addition to its anti-inflammatory properties, GER has been shown to exert a protective effect against apoptosis in the heart. Treatment with GER has been observed to reduce the expression of Bcl-2, a protein that promotes cell survival, while simultaneously inhibiting the expression of Bax, a protein that triggers cell death. Furthermore, GER treatment has been associated with decreased levels of cleaved caspase-3 and 9, key executioners of apoptosis, in the heart tissues of animals exposed to doxorubicin. 28
To date, no study has investigated the protective effects of GER against Ni-induced embryotoxicity and cardiotoxicity. This is a significant gap in our understanding, considering the widespread exposure to Ni and its documented detrimental effects on human health. Ni toxicity poses a substantial threat to both developing embryos and the cardiovascular system, highlighting the urgent need for effective protective strategies. The objective of current paper was to investigate the effect of GER on Ni-induced embryotoxicity and cardiotoxicity in rats.
Material and methods
Chemicals
Nickel oxide powder was provided by the department of pharmacology, Babol branch, Islamic Azad university, Babol, Iran. GER, which was in liquid form and soluble in corn oil, Alizarin Red and Alcian Blue Powders were obtained from Sigma-Aldrich (St Louis, MO, USA). For proper anesthesia in rats, 10% ketamine (Bremer Pharma GMBH, Germany) and xylazine 2% (Alfasan Co, Netherlands) were used. From Teb Pajohan Razi Company, Iran, we purchased MDA and TAC kits.
Animal experimental design
Forty female Wistar rats, aged 3–4 weeks and weighing 180 ± 20 g, were procured from the laboratory animal facility at Babol University of Medical Sciences. These animals were maintained under a 12-h light/12-h dark cycle and were housed in a controlled environment with regulated temperatures (22 °C–24°C) and humidity (50 ± 5%). The rats were accommodated in cages containing LSB Aspen woodchip bedding and were provided ad libitum access to food and tap water throughout the entire experimental duration. In order to induce pregnancy in the female rats, they were paired with male rats in a ratio of one male to two females. Pregnancy onset was determined upon observation of the vaginal plaque, marking day zero of pregnancy. All experimental procedures were conducted in compliance with the guidelines and regulations established by the Research Ethics Committee of Islamic Azad University-Babol Branch (Code: IR.IAU.BABOL.REC.1400.033).
Study rats were randomly divided into five groups as follows: 1. Control: received saline by gavage until the 16th day of pregnancy. This group was also received water as intraperitoneal (IP) injections on days 8,12, and 16 of pregnancy. 2. GER 250: received 250 mg/kg of GER by gavage until the 16th day.
29
3. Ni: received 25 mg/kg of Ni by IP injections on days 8, 12, and 16.
30
4. Ni + GER 100: received 100 mg/kg of GER by gavage until the 16th day, along with the same treatment like the Ni group.30,31 5. Ni + GER 250: received 250 mg/kg of GER by gavage until the 16th day, along with the same treatment like the Ni group.29,30
Sample collection and preparation
On the 20th day of gestation, the mother rats were anesthetized with the IP injection of 100 μL ketamine (100 mg/kg) and xylazine (10 mg/kg) combination, 32 weighed, killed (using decapitation technique), and 5 mL blood samples were promptly obtained directly from the ventricles and transferred into five-mL microtubes to undergo subsequent serum separation through being centrifuged for a duration of 15 min at 3000 r/min, and being utilized then for serological and cardiac markers assessment. Furthermore, their hearts were extracted for hematoxylin-eosin (H&E) staining and biochemical examination.
A cesarean section was carried out alongside a hysterectomy and bilateral oophorectomy, as detailed in another source. 33 The gravid uterus was extracted, and all fetuses together with their corresponding placentas were individually extracted. The count of living and deceased fetuses was recorded within each group. Additionally, the fetal body weight and the percentage change relative to the control group were assessed. Ultimately, decapitation technique was employed for the euthanasia of the fetuses, 34 after which samples were collected for further examinations.
Fetal observations
The number of viable fetuses per mother was tallied, their weights measured, and they were scrutinized for any morphological irregularities. One-quarter of the fetuses were preserved in 95% alcohol for the purpose of dual staining (using Alizarin Red S and Alcian Blue) to observe skeletal abnormalities. 35 The remaining fetuses were preserved in Bouin’s fixative to facilitate examination of soft tissue, 36 their heart was harvested for H&E staining and biochemical examination, or they were subjected to radiography examination. An X-ray mobile radiograph with a voltage of 50 kV and 1.3 mA was used to image and prepare the radiographs of the fetuses. All morphological analyses were carried out by a blinded skilled veterinary pathologist (Seyed Mohammad Hosseini). The reduction of bone ossification was graded from 0 to 3 (0 was none, and three was the maximum score). Eventually, the Kruskal-Wallis and Mann-Whitney U tests were performed for morphological assessments (non-parametric).25,37
Serological and cardiac markers
The activity of C-reactive protein (CRP, μg/dL), Creatinine Kinase-MB (CK-MB, IU/L), Lactate dehydrogenase (LDH, IU/L), Creatine phosphokinase (CPK, IU/L), and troponin (ng/mL) in serum samples was measured using commercial ELISA kits (Pars Azmun, Karaj, Iran).
Biochemical analysis
Malondialdehyde (MDA)
The assessment of lipid peroxidation levels in the heart tissue samples was conducted using the Malondialdehyde (MDA) assay. This involved employing a commercial ELISA kit (Teb Pazhouhan Razi, Tehran, Iran) to measure MDA levels through a spectrophotometric method outlined elsewhere. 38 The OD of the samples was measured at 540 nm, using an absorbance microplate reader (ELx808, BioTek, Winooski, VT, United States).
Total antioxidant capacity (TAC)
ELISA kit (Teb Pazhouhan Razi, Tehran, Iran) was used for measuring total anti-oxidant capacity (TAC). Detailed descriptions of the technique were published by Miller et al. (1993). 39 Afterwards, in order to measure the OD of the samples at 420 nm, an absorbance microplate reader (ELx808, BioTek, Winooski, VT, United States) was used.
Histopathological analysis
The maternal and fetal cardiac tissue samples were preserved in a 10% formalin solution, subsequently subjected to processing utilizing a tissue processing apparatus (involving dewatering, clearing, and staining), and finally embedded in paraffin blocks. These blocks were then sliced into 5 µm thick layers and stained with H&E, with an average of four sections applied to each slice. Histopathological analysis was conducted using digital light microscopy to assess for hyperemia, necrosis, and hyalinization in tissue sections. A blinded, experienced veterinary pathologist (Seyed Mohammad Hosseini) performed the analyses at magnifications of×40, ×100, and ×400. Hyperemia was scored on a scale of 0 to 3, with 0 representing no hyperemia and three indicating the most severe hyperemia. Hyalinization was evaluated and graded as mild, moderate, or severe, based on the percentage of affected tissue: (−) normal histology, (+) mild (less than 25%), (++) moderate (25%–50%), and (+++) severe (greater than 50%).40,41 Non-parametric statistical analyses, specifically the Kruskal-Wallis and Mann-Whitney U tests, were employed to compare histopathological findings between groups.25,37
Statistical analysis
All data are presented as means and standard deviations (SD). One-way analysis of variance (ANOVA) was employed to evaluate differences between study groups, followed by Tukey’s post hoc test for pairwise comparisons. Non-parametric Kruskal-Wallis and Mann-Whitney U tests were used to analyze data for ossification and histopathological assessments. 25 Statistical significance was set at a p-value of less than 0.05. All statistical analyses were performed using GraphPad Prism 9.
Results
The effect of GER on maternal body weight (BW)
Effect of Ni and geraniol administration on the maternal and embryonic parameters.
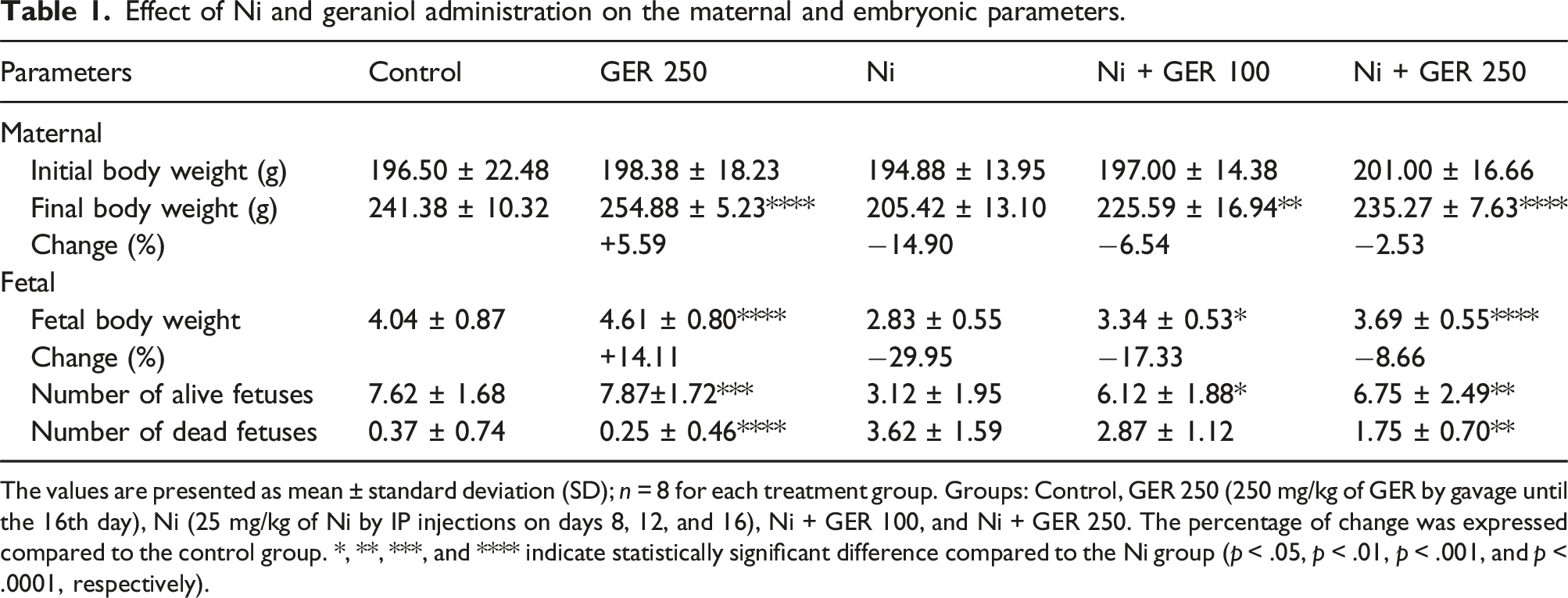
The values are presented as mean ± standard deviation (SD); n = 8 for each treatment group. Groups: Control, GER 250 (250 mg/kg of GER by gavage until the 16th day), Ni (25 mg/kg of Ni by IP injections on days 8, 12, and 16), Ni + GER 100, and Ni + GER 250. The percentage of change was expressed compared to the control group. *, **, ***, and **** indicate statistically significant difference compared to the Ni group (p < .05, p < .01, p < .001, and p < .0001, respectively).
The effect of GER on fetal BW, the number of alive, and dead fetuses
In order to determine how gestational exposure to Ni and different doses of GER affect fetal BW, we measured fetal BW and also calculated the percentage of change compared to the control group. The Ni group had the lowest fetal BW, significantly reduced compared to the control group (p-value <.0001) (Table 1). The Ni + GER 100 and Ni + GER 250 groups had significantly higher BWs compared to the Ni group (p < .05 and p < .0001, respectively).
In order to determine the potential protective effects of GER supplementation on Ni-induced embryotoxicity, we measured the number of alive fetuses and the number of stillbirth in each group. The lowest the number of alive fetuses was observed in the Ni group, significantly reduced compared to the control group (p-value <.001) (Table 1). The Ni + GER 100 and Ni + GER 250 groups had significantly higher numbers of alive fetuses compared to the Ni group (p < .05 and p < .01, respectively). Moreover, Ni treatment significantly increased stillbirth, compared to the control group (p-value <.0001) (Table 1). The Ni + GER 100 and Ni + GER 250 groups had lower numbers of dead fetuses compared to the Ni group, however, the difference was only significant in the Ni + GER 250 group (p < .01).
The effect of GER on the fetal morphometric analysis
Effect of Ni and geraniol administration on the fetal morphometric parameters.

The values are presented as mean ± standard deviation (SD); n = 8 for each treatment group. Groups: Control, GER 250 (250 mg/kg of GER by gavage until the 16th day), Ni (25 mg/kg of Ni by IP injections on days 8, 12, and 16), Ni + GER 100, and Ni + GER 250. *, **, ***, and **** indicate statistically significant difference compared to the Ni group (p < .05, p < .01, p < .001 and p < .0001, respectively). $$ and $$$$ Indicate statistically significant difference between the treatment groups (p < .01 and p < .0001, respectively).
The effect of GER on the fetal bone ossification
In order to evaluate the effects of gestational exposure to Ni and different doses of GER on fetal bone ossification, utilizing the Alizarin red S and Alcian blue double-staining and radiography, a comparative evaluation was made between the study groups based on the reduction of fetal bone-ossification (Figures 1 and 2). The Ni group had the highest reduction of bone-ossification, significantly higher compared to the control group (p-value <.0001), except for the Nasal cartilage which had no significant difference among all the study groups (Table 3). The Ni + GER 100 and Ni + GER 250 groups had significantly lower reduced bone-ossifications compared to the Ni group. Importantly, the Ni + GER 250 group had a significantly-enhanced ossification level compared to Ni + GER 100 group (Table 3). Effect of Ni and geraniol administration on the fetal skeletal anomalies (Alizarin red S and Alcian blue double staining; lateral and posterior views) in the study groups. Groups: Control, GER 250 (250 mg/kg of GER by gavage until the 16th day), Ni (25 mg/kg of Ni by IP injections on days 8, 12, and 16), Ni + GER 100, and Ni + GER 250. Nasal cartilage (NC), Nasal bone (N), Occipital bone (O), Maxilla (MX), Mandible (MB), Frontal bone (F), Parietal bone (P), Cervical vertebra (CV), Coccyx (CX), Femur (Fe), Scapula (S), Humerus (H), Radius (R), Ulna (U), Tibia (T), Fibula (Fi), Ribs (Ri). Effect of Ni and geraniol administration on the fetal skeletal anomalies (radiography; lateral views) in the study groups. Groups: Control, GER 250 (250 mg/kg of GER by gavage until the 16th day), Ni (25 mg/kg of Ni by IP injections on days 8, 12, and 16), Ni + GER 100, and Ni + GER 250. Control and GER 250 groups demonstrated normal conditions. Atrophy, death, malformation and reduction of morphometric parameters were detected in other groups. Effect of Ni and geraniol administration on the reduction of fetal bone-ossification of rats. Values are expressed as means ± standard error (SE); n = 28 for each group. Groups: Control, GER 250 (250 mg/kg of GER by gavage until the 16th day), Ni (25 mg/kg of Ni by IP injections on days 8, 12, and 16), Ni + GER 100, and Ni + GER 250. # Asymptotic significance differences of liver and kidney lesions between groups treated observed (p < .05; Kruskal–Wallis test). *, **, ***, and **** indicate statistically significant difference compared to the Ni group (p < .05, p < .01, p < .001, and p < .0001, respectively; Mann–Whitney U test).$$$, $$$, and, $$$$ indicate statistically significant difference between the treatment groups (p < .05, p < .01, p < .001, and p < .0001, respectively; Mann–Whitney U test).
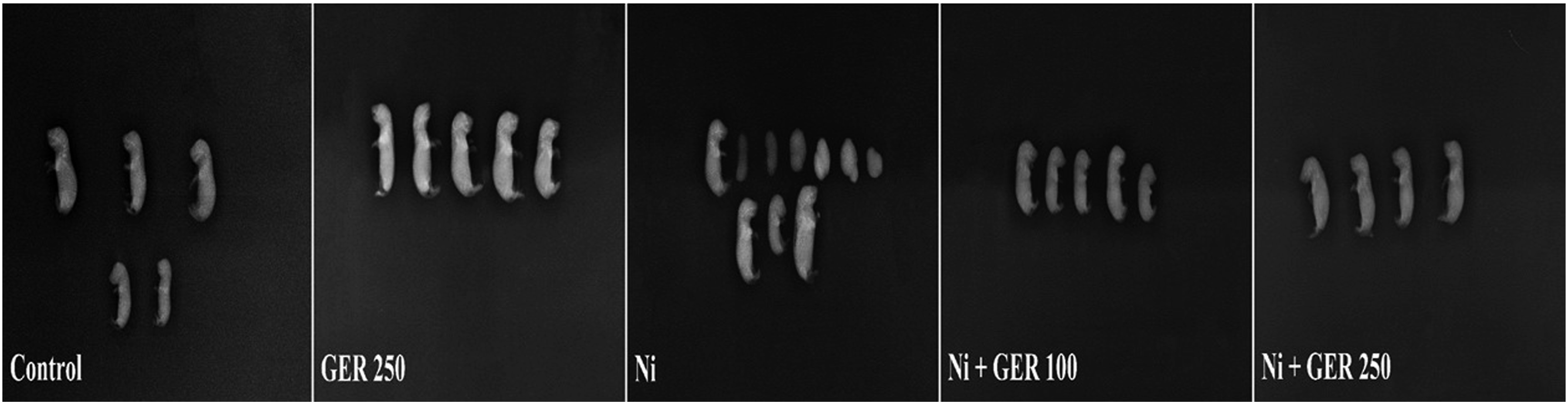

The effect of GER on embryonal somites and organogenesis
Ni is known to be a toxic metal that can have detrimental effects on embryonic development, including somite formation and organogenesis. Hence, the number of embryonal somites were counted in each group. The number of counted somites in Ni group was significantly reduced compared to the control group (Figure 3). The Ni + GER 100 and Ni + GER 250 groups had significantly higher numbers of somites compared to the Ni group (p < .0001). The Ni + GER 250 group had significantly higher numbers of somites compared to the Ni + GER 100 group (p-value <.05). While the control and GER 250 groups demonstrated no delay in the fetal organogenesis, the Ni group showed retardation of 120 h. In the Ni + GER 100 and Ni + GER 250 groups, the delay was 72 and 36 h, respectively. The neural structure was complete in all groups except the Ni group (which was relative). The neural tube was closed in all groups. The process of organogenesis was not completed only in the Ni group (Figure 4). Effect of Ni and geraniol administration on the number of somites and organogenesis parameters in the study groups. Groups: Control, GER 250 (250 mg/kg of GER by gavage until the 16th day), Ni (25 mg/kg of Ni by IP injections on days 8, 12, and 16), Ni + GER 100, and Ni + GER 250. Effect of Ni and geraniol administration on organogenesis and embryonal development in the study groups. Groups: Control, GER 250 (250 mg/kg of GER by gavage until the 16th day), Ni (25 mg/kg of Ni by IP injections on days 8, 12, and 16), Ni + GER 100, and Ni + GER 250.

The effect of GER on serological markers
To elucidate the mechanisms underlying the observed morphological and bone ossification changes, serum levels of phosphorus and calcium, key indicators of bone metabolism, were analyzed. Additionally, serum levels of inflammatory markers (C-reactive protein and lactate dehydrogenase) and cardiac muscle damage biomarkers (creatine kinase-MB, creatine phosphokinase, and troponin) were assessed in each experimental group to evaluate potential inflammatory and cardiac responses.
The highest phosphorus level was observed in the Ni group, significantly increased compared to the control group (p-value <.0001) (Figure 5(a)). The Ni + GER 100 and Ni + GER 250 groups had significantly lower phosphorus levels compared to the Ni group (p-value <.0001). Effect of Ni and geraniol administration on Phosphorus (A) and Ca (B) levels in the study groups. Groups: Control, GER 250 (250 mg/kg of GER by gavage until the 16th day), Ni (25 mg/kg of Ni by IP injections on days 8, 12, and 16), Ni + GER 100, and Ni + GER 250. Values are expressed as mean ± standard deviation (SD). **** indicates statistically significant difference compared to the Ni group (p < .0001).
The highest calcium (Ca) level was observed in the Ni group (Figure 5(b)). The Ni + GER 100 and Ni + GER 250 groups had lower Ca levels compared to the Ni group. However, no significant difference was observed among all the study groups.
The highest C-reactive protein (CRP) level was observed in the Ni group, significantly increased compared to the control group (p-value <.0001) (Figure 6(a)). The Ni + GER 100 and Ni + GER 250 groups had significantly lower CRP levels compared to the Ni group (p-value <.0001). The Ni + GER 250 group had a significantly lower CRP level compared to the Ni + GER 100 group (p-value <.01). Effect of Ni and geraniol administration on CRP (A), CK-MB (B), CPK (C), LDH (D), and troponin (E) levels in the study groups. Groups: Control, GER 250 (250 mg/kg of GER by gavage until the 16th day), Ni (25 mg/kg of Ni by IP injections on days 8, 12, and 16), Ni + GER 100, and Ni + GER 250. Values are expressed as mean ± standard deviation (SD). *, ***, and **** indicate statistically significant difference compared to the Ni group (p < .05, p < .001, and p < .0001, respectively). $$, $$$, and $$$$ indicate statistically significant difference between the treatment groups (p < .01, p < .001, and p < .0001, respectively).
The highest Creatinine Kinase-MB (CK-MB) level was observed in the Ni group, significantly increased compared to the control group (p-value <.0001) (Figure 6(b)). The Ni + GER 100 and Ni + GER 250 groups had significantly lower CK-MB levels compared to the Ni group (p-value <.0001). The Ni + GER 250 group had a significantly lower CK-MB levels compared to the Ni + GER 100 group (p-value <.0001).
The highest Creatine phosphokinase (CPK) level was observed in the Ni group, significantly increased compared to the control group (p-value <.0001) (Figure 6(c)). The Ni + GER 100 and Ni + GER 250 groups had significantly lower CPK levels compared to the Ni group (p < .001 and p < .0001, respectively). The Ni + GER 250 group had a significantly lower CPK level compared to the Ni + GER 100 group (p-value <.0001).
The highest Lactate dehydrogenase (LDH) level was observed in the Ni group, significantly increased compared to the control group (p-value <.0001) (Figure 6(d)). The Ni + GER 100 and Ni + GER 250 groups had significantly lower LDH levels compared to the Ni group (p < .001 and p < .0001, respectively). The Ni + GER 250 group had a significantly lower CPK level compared to the Ni + GER 100 group (p-value <.001).
The highest troponin level was observed in the Ni group, significantly increased compared to the control group (p-value <.0001) (Figure 6(e)). The Ni + GER 100 and Ni + GER 250 groups had significantly lower troponin levels compared to the Ni group (p < .05 and p < .001, respectively).
The effect of GER on biochemical markers
To assess the degree of oxidative stress and inflammation in both maternal and fetal heart tissues, we determined the levels of malondialdehyde, a marker of lipid peroxidation, and total antioxidant capacity, a measure of the tissue’s ability to neutralize free radicals.
Malondialdehyde (MDA)
In the mothers, the highest MDA level was observed in the Ni group, significantly increased compared to the control group (p-value <.0001) (Figure 7(a)). Although the Ni + GER 100 and Ni + GER 250 groups had slightly lower MDA levels compared to the Ni group, the differences were not significant. Effect of Ni and geraniol administration on maternal and fetal MDA (A and B) and TAC (C and D) levels in the study groups. Groups: Control, GER 250 (250 mg/kg of GER by gavage until the 16th day), Ni (25 mg/kg of Ni by IP injections on days 8, 12, and 16), Ni + GER 100, and Ni + GER 250. Values are expressed as mean ± standard deviation (SD). **, ***, and **** indicate statistically significant difference compared to the Ni group (p < .01, p < .001, and p < .0001, respectively). $$$$ indicates statistically significant difference between the treatment groups (p < .0001).
In the fetuses, the highest MDA level was observed in the Ni group, significantly increased compared to the control group (p-value <.0001) (Figure 7(b)). Although the Ni + GER 100 group had slightly lower MDA level compared to the Ni group, the difference was not significant. Importantly, the Ni + GER 250 had a significantly lower MDA level compared to the Ni group (p-value <.01).
Total antioxidant capacity (TAC)
In the mothers, the lowest TAC level was observed in the Ni group, significantly reduced compared to the control group (p-value <.0001) (Figure 7(c)). The Ni + GER 100 and Ni + GER 250 groups had significantly higher TAC levels compared to the Ni group (p-value <.0001). The Ni + GER 250 group had a significantly higher TAC level compared to the Ni + GER 100 group (p-value <.0001).
In the fetuses, the lowest TAC level was observed in the Ni group, significantly reduced compared to the control group (p-value <.0001) (Figure 7(d)). Although the Ni + GER 100 group had slightly higher TAC level compared to the Ni group, the difference was not significant. Importantly, the Ni + GER 250 group had a significantly higher TAC level compared to the Ni group (p-value <.001).
The effect of GER on cardiac histopathologic alterations
To evaluate the extent of tissue damage induced by nickel exposure during pregnancy on both maternal and fetal hearts, we quantified the levels of various biomarkers indicative of cardiac tissue injury in both maternal and fetal heart tissues.
In the histopathological analysis of the maternal cardiac tissues, hyperemia and hyalinization were detected. The Ni group had the highest levels of hyperemia and hyalinization, significantly higher compared to the control group (p-value <.0001) (Figure 8 and Table 4). Although the Ni + GER 100 group had lower level of hyperemia compared to the Ni group, the difference was not significant. Importantly, the Ni + GER 250 group had a significantly lower hyperemia compared to the Ni group (p-value <.05). Furthermore, the Ni + GER 100 and Ni + GER 250 groups had a significantly lower hyalinization levels compared to the Ni group (p < .05 and p < .001, respectively). In none of the sections necrosis was observed. Effect of Ni and geraniol administration on the histopathologic changes of maternal heart in the study groups. Groups: Control, GER 250 (250 mg/kg of GER by gavage until the 16th day), Ni (25 mg/kg of Ni by IP injections on days 8, 12, and 16), Ni + GER 100, and Ni + GER 250. Within the control and GER 250 groups, normal conditions were observed. In the Ni group, conspicuous signs of intense hyperemia (right-sided arrow) and hyalinization (left-sided arrow) were identified. Likewise, in the Ni + GER 100 group, hyperemia (right-sided arrow) and hyalinization (left-sided arrow) were observed, albeit presenting with a mild intensity. Contrastingly, the Ni + GER 250 group exhibited mild hyperemia, without any indication of observed hyalinization. Hematoxylin and Eosin (H&E) staining. ×100 magnification. Effect of Ni and geraniol administration on the histopathologic alterations of maternal and fetal heart of rats. Values are expressed as means ± standard error (SE); nM = 10 and nF = 28 for each group. Groups: Control, GER 250 (250 mg/kg of GER by gavage until the 16th day), Ni (25 mg/kg of Ni by IP injections on days 8, 12, and 16), Ni + GER 100, and Ni + GER 250. #Asymptotic significance differences of liver and kidney lesions between groups treated observed (p < .05; Kruskal–Wallis test). *, **, ***, and **** indicate statistically significant difference compared to the Ni group (p < .05, p < .01, p < .001, and p < .0001, respectively; Mann–Whitney U test).$ and, $$$$ indicate statistically significant difference between the treatment groups (p < .05 and p < .0001, respectively; Mann–Whitney U test).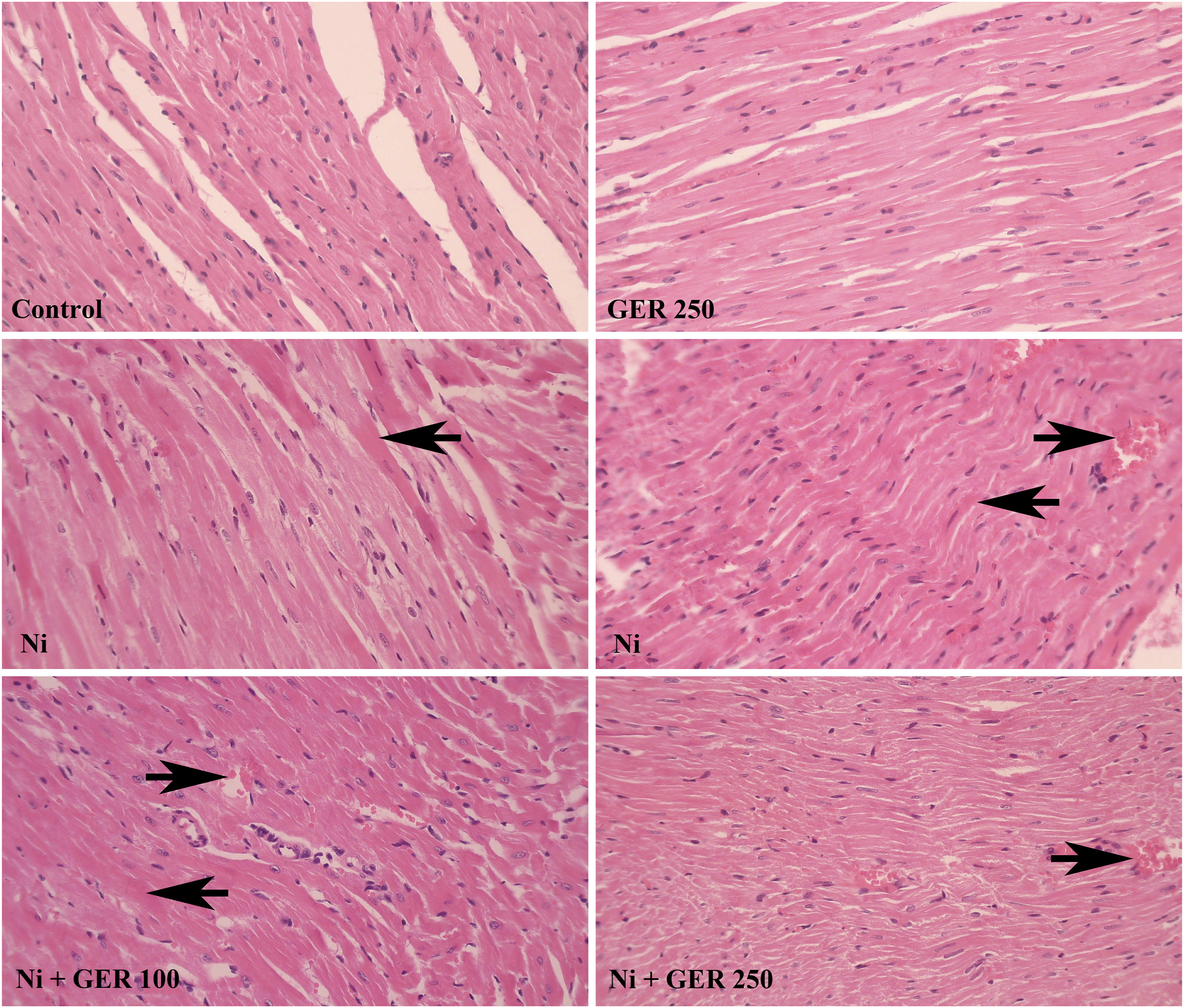
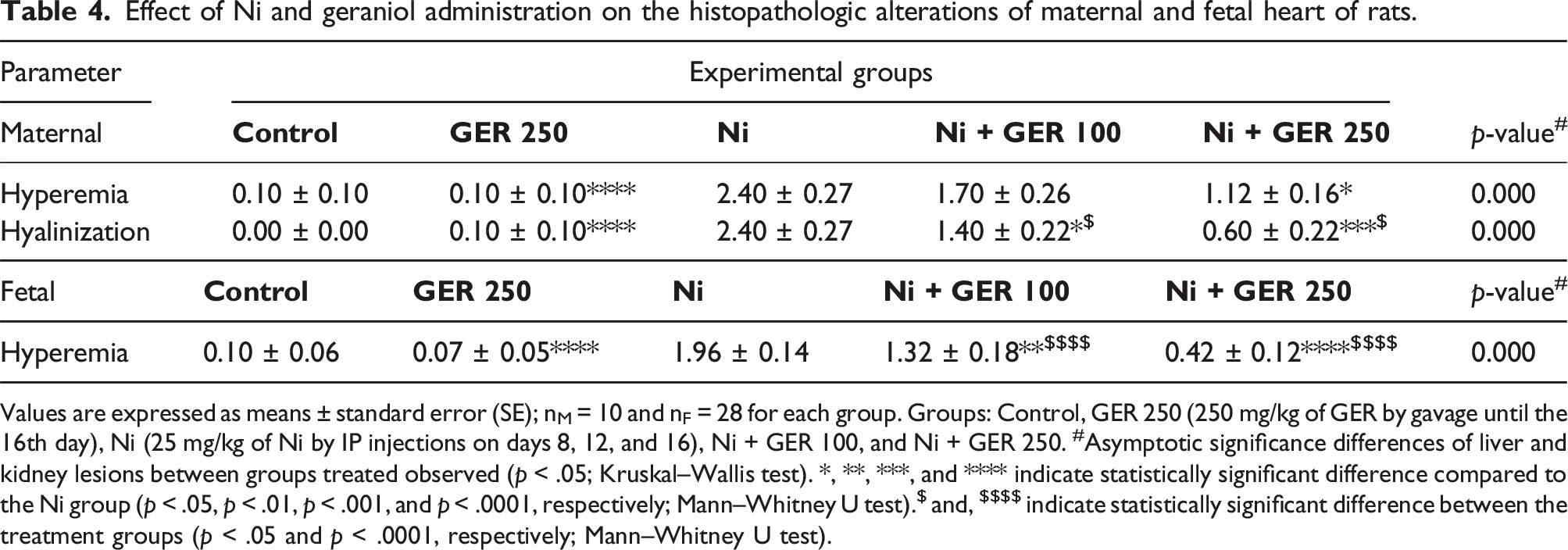
In the histopathology analysis of the fetal cardiac tissues, hyperemia was detected. The Ni group had the highest level of hyperemia, significantly higher compared to the control group (p-value <.0001) (Figure 9 and Table 4). The Ni + GER 100 and Ni + GER 250 groups had significantly lower hyperemia levels compared to the Ni group (p < .01 and p < .0001, respectively). Importantly, the Ni + GER 250 group had a significantly lower level of hyperemia compared to the Ni + GER 100 group (p-value <.0001). Effect of Ni and geraniol administration on the histopathologic changes of fetal heart in the study groups. Groups: Control, GER 250 (250 mg/kg of GER by gavage until the 16th day), Ni (25 mg/kg of Ni by IP injections on days 8, 12, and 16), Ni + GER 100, and Ni + GER 250. The control and GER 250 groups exhibited normal conditions. Within the Ni group, a pronounced presence of hyperemia (right-sided arrow) was noted. Similarly, the Ni + GER 100 group also displayed hyperemia (right-sided arrow), albeit of a milder magnitude. Conversely, the Ni + GER 250 group displayed no discernible signs of hyperemia. Hematoxylin and Eosin (H&E) staining. ×100 magnification.
Discussion
The acyclic monoterpene GER, chemically identified as (2E)-3,7-dimethylocta-2,6-dien-1-ol, is commonly present within the essential oils of numerous plant species. Significant quantities of GER are typically found in lemongrass, rose, lavender, and various other fragrant botanicals. Research has demonstrated the diverse pharmacological attributes of GER, encompassing its antioxidant, anti-inflammatory, antimicrobial, and antitumor properties. 42 These effects are attributed to its ability to modulate multiple signaling pathways across a spectrum of biological processes.43,44
Instances of growth restriction often arise as a result of chemical exposure, yielding deleterious consequences. 45 Assessment of body weight stands as a paramount and exceptionally sensitive determinant of drug-induced side effects. 46 Our findings underscore the noteworthy capacity of GER treatment to effectively forestall body weight diminishment in both maternal and fetal rats, a pattern consistent with prior investigations delineating GER’s mitigative prowess against metal-induced weight loss.31,47 Numerous rationales may elucidate this observed phenomenon. Firstly, GER engenders an augmentation of mucus-protective elements, manifested through escalated sulfhydryl compounds, bolstered antioxidant defenses, and enhanced mucus secretion. 48 Secondly, it stimulates appetite and augments food consumption owing to its aromatic and gustatory attributes. 49 Furthermore, GER manifests antioxidant and anti-inflammatory attributes, inherently capable of alleviating the untoward impacts of toxic agents. 31
Significantly, the diminished fetal weight coincided with incomplete ossification of the fetal skeleton, potentially constituting a contributory factor to growth retardation and the resultant decrease in fetal weight. An analogous association linking fetal weight reduction and delayed skeletal ossification has been documented in prior studies.50–52 Importantly, our results indicated that Ni severely reduced the number of somites and not only delayed fetal organogenesis but also resulted in an incomplete organogenesis and the lack of ossification in rats. These findings were also consistent with previous studies.21,53,54
Our findings suggest that GER exhibited the capacity to advance ossification, paralleling its impact on body weight. Otto et al. (2022) 55 documented the significant role of geranyl-geraniol in augmenting the migratory behavior of angiogenic cells and in averting bisphosphonate-induced osteonecrosis. Geranyl-geraniol have important roles in the intracellular proteins being localized in the membranous layer, especially small GTP-binding proteins, and its depletion contributes to the modulation of various signaling pathways, such as the prevention of bone resorption and the inhibition of osteoclast formation. Disruption of these pivotal pathways results in reduced cell migration, altered cell metabolism, ultimately culminating in apoptosis. 56 Disruption of mechanisms related to bone resorption and osteoclast inhibition has far-reaching consequences, particularly in the context of embryonic development. This disruption leads to reduced cell migration, a fundamental process vital for the appropriate organization of cells during the formation of different tissues, including bone. Moreover, altered cell metabolism arising from these disruptions can induce a cascade of effects that can impair the intricate balance required for healthy skeletal development in embryos. 56 The observed alterations in serum phosphorus and calcium levels provide further evidence for the impact of nickel on bone resorption and osteoclast function. Our study demonstrates a significant increase in serum phosphorus levels in Ni-exposed mothers, while calcium levels were insignificantly high. This finding suggests that Ni may influence skeletal homeostasis in both the mother and fetus, potentially by affecting bone resorption processes. However, to precisely quantify the relative contributions of maternal and fetal skeletal systems to these changes, future studies employing more sophisticated and resource-intensive methodologies are required.
Moreover, our results have indicated that Ni treatment elicits a substantial increase in stillbirth rates and a decrease in the number of live fetuses, consistent with earlier research.21,54 The observed increase in stillbirth likely stems from a complex interplay of factors, each contributing to the compromised development and survival of the fetuses. These factors include disruptions in organogenesis, a significant reduction in somite formation, dysregulation of skeletal homeostasis, elevations in inflammatory markers, and increased cardiac damage markers. Furthermore, histological examinations reveal structural damage to cardiac tissues, further supporting the detrimental effects of Ni exposure on fetal development. Alternatively, the observed increase in stillbirths may be attributed to maternal factors, specifically severe weight loss and the maternal toxicity. This maternal toxicity could compromise the mother’s ability to support fetal development and viability, ultimately contributing to the observed increase in stillbirths.
Nevertheless, treatment with GER at 250 dosage exhibited an enhancement in the quantity of live fetuses and demonstrated a protective effect against Ni-induced embryotoxic and teratogenic outcomes. The adverse impacts of Ni have been observed in mouse embryos during transit through the oviduct 57 and in the course of organogenesis, 53 followed by subsequent cytotoxic and teratogenic manifestations post-implantation. Additionally, studies have revealed the presence of soluble NiCl2 within the cytoplasm and nucleus, with the hypothesis, proposed by Chen et al. in 2005, 58 that Ni ions may utilize the divalent metal transporter-I (DMT-I) for cellular entry. An excessive presence of Ni ions could, therefore, displace other essential metal ions which are necessary for enzymes' structural form and activity, resulting in the enzymes being inactivated, thereby contributing to the embryotoxic and fetotoxic effects induced by Ni. 21
The incomplete ossification observed in various bones of the fetal skeleton may arise from disruptions in calcium metabolism, diminished calcium and magnesium ion levels, or alterations in calcitonin levels within the fetus, ultimately leading to impaired bone growth. This was elucidated by Yamaguchi and Ehara (1996), 59 who, through in vitro studies, demonstrated that osteoblasts exposed to Ni exhibited a notable decrease in alkaline phosphatase activity. Notably, our findings did not indicate a substantial elevation in serum Ca levels in rats treated with Ni; however, there was a significant increase in phosphorus levels. Treatment with GER, however, was able to mitigate the heightened levels of phosphorus and Ca.
Analysis of cardiac and serological biomarkers revealed noteworthy outcomes, signifying a substantial elevation in CRP, CKMB, CPK, LDH, and troponin levels within the Ni group. These findings point towards pronounced myocardial injury, evidenced by heightened levels of multiple cardiac enzyme markers and histological observations of cardiac tissue damage. Notably, concurrent administration of GER markedly mitigated this detrimental impact, particularly at the dosage of 250 mg/kg. Similarly, Younis et al. (2021) 28 have documented that treatment with GER, particularly at a higher dosage of 200 mg/kg, significantly ameliorated escalated levels of serum cardiac injury markers (CK, CK-MB, LDH, AST and ALT) observed in the doxorubicin group. In a separate study, GER demonstrated a significant reduction in altered cardiac enzymes (CKMB, CPK, troponin T, and I), which had been markedly increased due to isoproterenol. 60 Notably, GER may activate nuclear factor erythroid 2-related factor 2 (Nrf2), subsequently enhancing Nrf2-dependent antioxidative signaling, curtailing apoptosis, and dampening the inflammatory response. 28
The increased serum troponin levels observed after nickel administration during pregnancy indicate cardiac muscle damage and myocardial infarction. 61 GER’s ability to prevent this troponin elevation suggests a protective mechanism. Previous research has established the anti-inflammatory properties of GER, demonstrating its ability to reduce nuclear translocation of NF-κB, a key transcription factor involved in inflammation, and decrease IκBα phosphorylation and degradation within the myocardium. Additionally, GER has been shown to significantly decrease the levels of pro-inflammatory cytokines, such as TNF-α, IL-1β, and IL-18, in doxorubicin-treated rats, suggesting a broader anti-inflammatory effect. 28 Furthermore, GER treatment has been observed to reduce Bcl-2 expression, inhibit Bax expression, and decrease the levels of cleaved caspase-3 and nine in heart tissues of doxorubicin-challenged animals. 28 These findings suggest a potential mechanism by which GER could reduce troponin levels and prevent myocardial infarction, likely through its ability to mitigate inflammation and regulate apoptotic pathways.
MDA functions as a measurable marker resulting from the breakdown of unsaturated fatty acids into free radicals. This study showed a notable increase in MDA levels within the Ni group, in line with prior research. 62 However, the MDA levels experienced a substantial decrease and mitigation following the administration of GER (250 mg/kg) to the fetal subjects. Importantly, the concurrent use of GER elicited a modest reduction in MDA levels in the maternal rats. Furthermore, a decline in TAC levels was evident in the Ni group when compared to the control, a shift that was counteracted by the introduction of GER in the treatment groups for both maternal and fetal subjects. In a separate investigation, the application of doxorubicin resulted in a marked elevation in MDA levels within kidney tissue, a response that was significantly and dose-dependently attenuated by pre-treatment with GER. 63 Furthermore, it has been established that GER substantially diminishes MDA levels in methotrexate-induced kidney injury. 64
Treatment with Ni resulted in notable histopathologic alterations (hyperemia and hyalinization) in both mother and fetus hearts. Treatment with 250 mg/kg of GER significantly reduced hyperemia and hyalinization observed in maternal and fetal hearts. Importantly, treatment with 250 mg/kg of GER had a significantly better protective effect against cardiac lesions compared to 100 mg/kg of GER. In a study, rats treated with GER (100 and 200 mg/kg) markedly prevented doxorubicin-induced pathological alterations. 28 Furthermore, in another study, rats pretreated with GER showed significantly diminished myocardial necrosis as well as reduced edema and immune cells infiltration compared to animals in the isoproterenol -treated group. 60
This investigation offers preliminary evidence supporting the protective effects of GER against Ni-induced embryotoxicity and cardiotoxicity in a rat model. The study’s comprehensive methodology, incorporating morphological, serological, biochemical, and histopathological assessments, provides a broad perspective on the impact of Ni and GER on maternal and fetal health. Notably, the study’s strength lies in its well-structured experimental design, which utilizes a sufficient sample size and multiple endpoints to ensure robust evaluation. However, the study’s limitations include the absence of molecular-level analysis due to financial constraints. The lack of PCR, gene expression analysis, Western blotting, or immunohistochemical staining prevents a deeper exploration of the mechanisms underlying both Ni toxicity and GER’s protective effects. Moreover, it is important to acknowledge that this study is conducted on an animal model, and its findings may not be directly translatable to human populations. Future research should prioritize elucidating the molecular mechanisms involved in geraniol’s protective effect, potentially employing techniques such as gene expression analysis and Western blotting. Additionally, human clinical trials are essential to assess the safety and efficacy of GER in safeguarding against Ni-induced toxicity in humans.
Conclusions
This study provides compelling evidence for the protective efficacy of geraniol (GER) against nickel (Ni)-induced embryotoxicity and cardiotoxicity in a rat model. Our findings demonstrate that GER significantly mitigates Ni-induced developmental abnormalities, as evidenced by the improved ossification of fetal skeletons in the GER-treated group. Furthermore, serological and biochemical analyses revealed that GER administration effectively reduces both inflammatory markers and indicators of cardiac injury, including CRP, CKMB, CPK, LDH, and troponin, suggesting a potent cardioprotective effect. This cardioprotective effect is further supported by the observation that GER significantly enhances the antioxidant capacity in the hearts of both mothers and their offspring. Histopathological examination corroborates these findings, demonstrating that GER treatment significantly reduces cardiac tissue damage and structural abnormalities in Ni-exposed animals. While our findings are encouraging, further investigation is warranted to translate these preclinical results into clinical practice.
Footnotes
Authors’ note
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Author contributions
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Data Availability Statement
The data that supports the findings of this study are available from the corresponding author upon reasonable request.
