Abstract
Introduction
Although the novel coronavirus disease 2019 (COVID-19) is endemic worldwide, mortality rates vary considerably across countries and may be related partly to the availability and efficacy of medical treatment. The mortality rate in Japan is 152.9 per million population. 1 The efficacy of dexamethasone (DEXA) in treating patients with COVID-19 has been demonstrated in a meta-analysis, with a reduction in mortality compared with that of standard therapy for patients with moderate to severe disease. 2 In that study, the efficacy of DEXA was largely influenced by a large randomized controlled trial (RCT) conducted in the UK. 3 The published protocol for the RCT did not define the number of days from the onset of symptoms to the start of DEXA administration, and the DEXA-treated group was included at a median of 8 days from the onset of symptoms. Neither meta-analyses nor individual RCTs have established that DEXA administration within 7 days of symptom onset is not effective in reducing mortality.2,3 Furthermore, there is no consensus on the type and dosage of steroids, which has been debated in recent RCTs and meta-analyses.4–6
Current recommendations in Japan for patients with COVID-19 with moderate to severe disease are 6 mg of DEXA to be administered once daily for 10 days; many of these patients will have treatment initiated within 7 days of symptom onset. 7 Results of the RCT implying that DEXA administration within 7 days of symptom onset is ineffective may be related to the inhibition of the immune response by the steroid and thus delayed viral clearance. 8 A meta-analysis reported that corticosteroid use may increase mortality in hospitalized patients with COVID-19 not receiving oxygen, supporting the abovementioned hypothesis. 9 However, early corticosteroid treatment was found to be associated with an increased 90-day mortality rate, independent of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) RNA clearance. 10 To reiterate, the strategy of steroid administration for COVID-19 remains controversial and only a few studies have reported an association between the time from onset of disease to initiation of steroid administration and mortality. In the present study, we conducted a multicenter, retrospective, observational study using data from a large Japanese registry of COVID-19 to clarify the association between time from COVID-19 onset to steroid initiation and mortality and to examine the factors underlying these results.
Methods
Study design and settings
This was a multicenter, retrospective, observational study of patients enrolled in the Japanese COVID-19 Registry (UMIN000039873) from January 1, 2020, to April 30, 2021. The registry was approved by the National Center for Global Health and Medicine Ethics Review Committee (NCGM-G-004232-00) and our study was approved by the Ethics Committee of the Hyogo Prefectural Tamba Medical Center (approval number: Tan-I number 1028). Study data were collected and managed using REDCap (Research Electronic Data Capture), a secure, web-based data capture application hosted at the JCRAC Data Center of the National Center for Global Health and Medicine. 11 Consent for participation in this Japanese COVID-19 Registry was obtained through an opt-out approach. Owing to the retrospective nature of the study, written informed consent was not required.
Participants
Patients had been enrolled in healthcare facilities that voluntarily participated in Japan’s COVID-19 Registry (COVIREGI-JP). Eligible patients had been confirmed positive for SARS-CoV-2 infection using polymerase chain reaction, loop-mediated isothermal amplification, or antigen tests and had been treated for COVID-19 with systemic administration of DEXA or steroids considered equivalent to 6 mg DEXA (methylprednisolone [mPSL] equivalent 1 mg/kg/day, mPSL equivalent 0.5 mg/kg/day, or mPSL equivalent 30–40 mg/day). Patients who had received steroids were only included if the steroids had been used for 7–14 days.
Patients were excluded if they were <18 years of age, pregnant, had a history of chronic obstructive pulmonary disease or a history of steroid or immunosuppressive drug use before the onset of symptoms, had been transferred to another hospital, or their symptom onset date or outcomes were unknown.
Outcomes
The primary outcome was mortality during the study period, comparing the timing of steroid initiation. Differences in dyspnea, oxygenation (nasal cannula, mask, reservoir mask, and high flow nasal cannula), noninvasive mechanical ventilation (bilevel positive airway pressure and continuous positive airway pressure), invasive mechanical ventilation, and extracorporeal membrane ventilation on admission or during hospitalization were used as secondary clinical outcomes.
Specific patient data collected were as follows: age, sex, body mass index (BMI), and underlying diseases reported as risk factors for severe disease (diabetes, hypertension, myocardial infarction, congestive heart failure, cerebrovascular disease, chronic lung disease, malignant tumor, liver cirrhosis, and chronic kidney disease). 7 Data on the most recent biomarkers from the date of admission (white blood cell count, lymphocyte count, hemoglobin, platelets, albumin, lactase dehydrogenase [LDH], D-dimer, and C-reactive protein [CRP]) 7 and concomitant medications (favipiravir, lopinavir/ritonavir, interferon, remdesivir, ciclesonide, hydroxychloroquine, tocilizumab, nafamostat, baricitinib, and ivermectin) were extracted. Data on the number of days from the onset of symptoms to the start of steroid administration and the number of days of steroid administration were also extracted.
Outcomes
The endpoints were 60-day mortality, tracheal intubation avoidance rate, duration of ventilation and hospitalization, time from symptom onset to tracheal intubation, and time from tracheal intubation to death.
Statistical analysis
Descriptive statistics were calculated based on patient demographic and clinical factors (age, sex, BMI, underlying disease, most recent biomarker from the date of admission, concomitant medications, days from the onset of symptoms to the start of steroid administration, and duration of steroid administration).
Patients were categorized based on survival at discharge, and demographic and clinical factors were compared between the two groups using t-tests for continuous factors and chi-square tests for categorical factors. Patients were also categorized based on early administration of steroid (≤7 days from onset of symptoms to the start of steroid) and delayed administration (≥8 days from onset of symptoms to the start of steroid administration) and compared using the Fisher’s exact test (categorical variables) or the Mann–Whitney U test (continuous variables).
Multivariate logistic regression analysis was used to assess the probability of death for all patients using dichotomized age (≥65 years), male sex, BMI (≥30 kg/m2), individual underlying diseases (diabetes, hypertension, myocardial infarction, congestive heart failure, cerebrovascular disease, chronic lung disease, malignant tumor, and renal disease), time from the onset of symptoms to the start of steroid administration (≥8 days), and the duration of steroid administration (≤10 days). Duration of administration (≤10 days), concomitant medications (remdesivir, tocilizumab, and baricitinib), and laboratory values (hemoglobin [Hb] <14.0 g/dL, albumin [Alb] <3.4 g/dL, alanine aminotransferase [ALT] ≥47 U/L, LDH ≥460 U/L, creatine kinase [CK] ≥200 U/L, and CRP ≥9 mg/dL) were also used as explanatory variables.
After initial analyses, the history of liver cirrhosis was excluded owing to the small number of patients; however, its exclusion did not affect the results. Laboratory values for white blood cell counts, platelet counts, and D-dimer counts were also excluded owing to their high numbers of outliers. Concomitant medications were limited to the three drugs (remdesivir, tocilizumab, and baricitinib) currently in standard use in Japan. Cutoff values for laboratory findings were based on previous reports on markers of severity of illness or overall medians (Hb <14.0 g/dL, Alb <3.4 g/dL, 12 ALT ≥47 U/L, 13 LDH ≥460 U/L, 14 CK ≥200 U/L, 15 and CRP ≥9 mg/dL 16 ). All statistical analyses were performed using the statistical software R 4.0.5, 17 with a significance level of p < .05.
Results
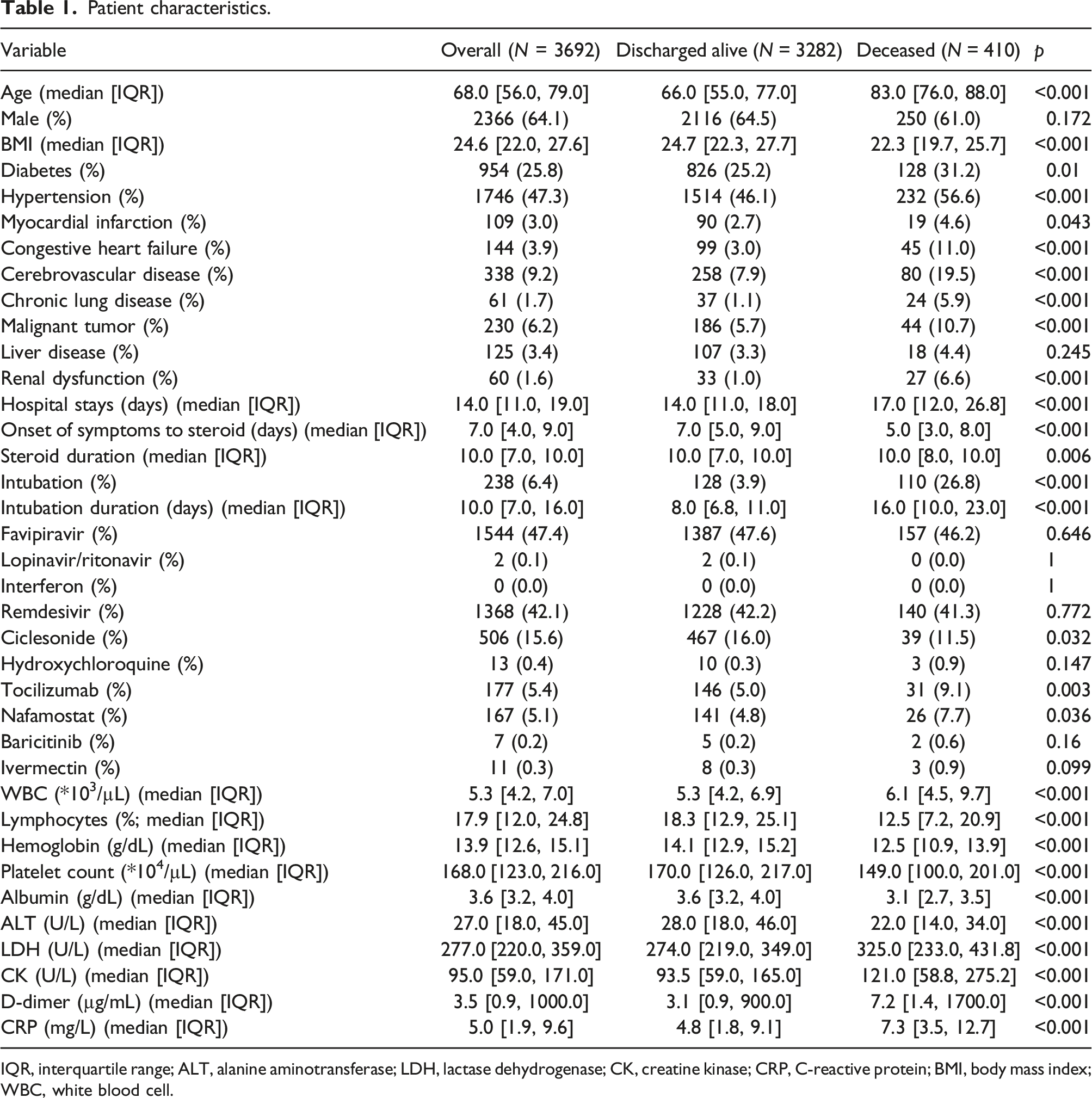
Figure 1 shows the flowchart of patient selection for this study. In total, 37,183 patients were enrolled in the registry during the study period. Of these, 5004 had been treated with steroids equivalent to 6 mg/day of DEXA. After applying the exclusion criteria, 3692 patients were included in the analysis. Figure 2 shows the reasons for exclusion and the number of excluded patients. Patient backgrounds are shown in Table 1. Sex of the included patients was predominantly male (64.1%), the median age was 68 (interquartile range 56–79) years, and the median BMI was 24.6 (interquartile range 22.0–27.6) kg/m2. The most prevalent underlying diseases included hypertension (47.3%) and diabetes (25.8%). The median hospital stay was 14 (interquartile range 11–19) days, 6.4% of patients had undergone tracheal intubation, and the median time from the onset of symptoms to the start of steroid administration was 7 (interquartile range 4–9) days. The overall mortality rate was 11.1%. There were significant differences between discharged and deceased patients in known predictors of severity, such as median age (66 vs 83 years, respectively, p < .001), BMI (24.7 kg/m2 vs 22.3 kg/m2, respectively, p < .001), underlying disease, and laboratory values. There was also a significant difference in the time from symptom onset to steroid administration (7.0 vs 5.0 days, respectively, p < .001). Flowchart depicting the patient selection. The reasons for exclusion and number of excluded patients. Patient characteristics. IQR, interquartile range; ALT, alanine aminotransferase; LDH, lactase dehydrogenase; CK, creatine kinase; CRP, C-reactive protein; BMI, body mass index; WBC, white blood cell.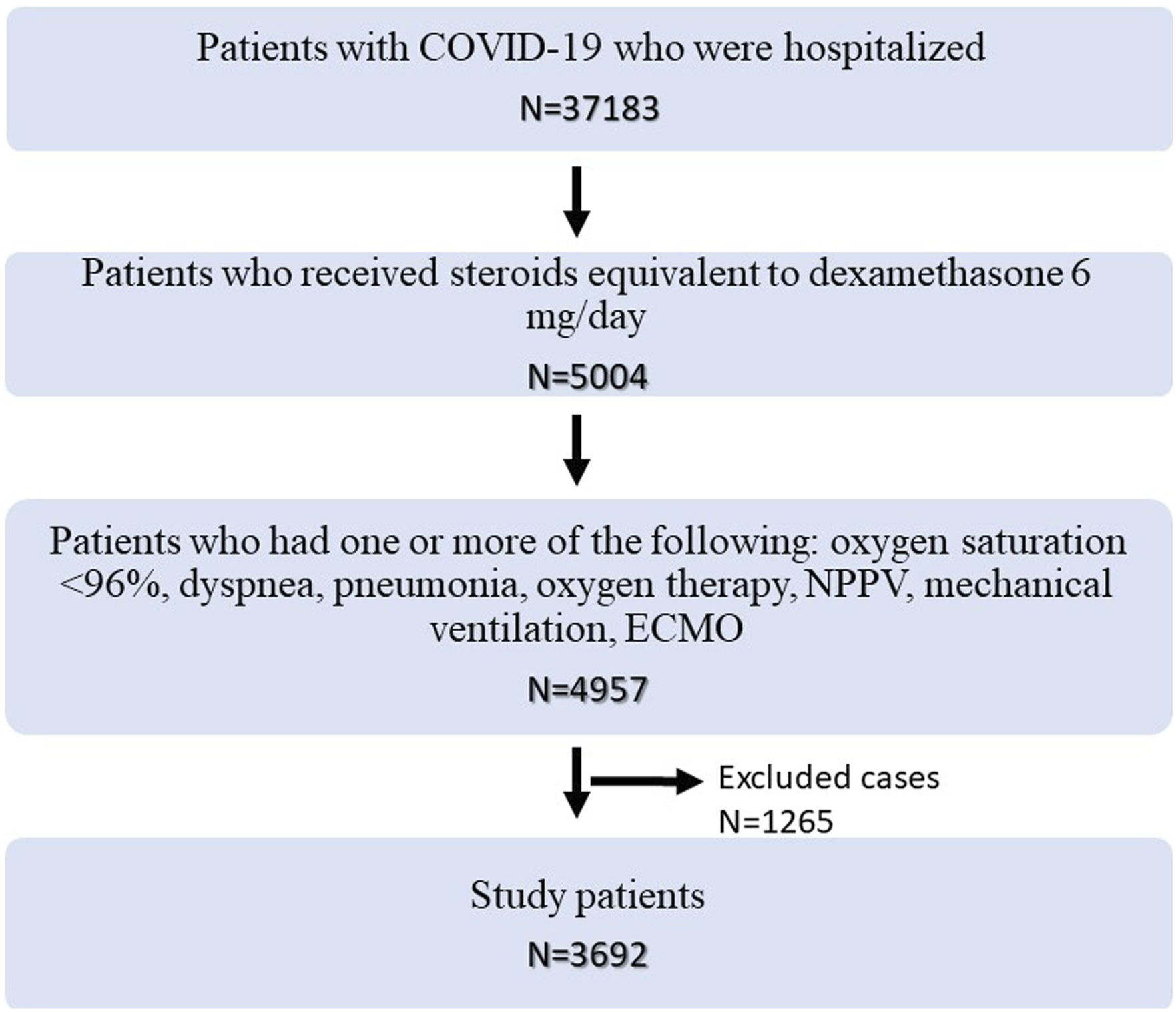

Patients may have been excluded for multiple reasons.
Comparison of demographic and clinical factors of patients with early steroid administration (≤7 days from the onset of illness) and those with delayed steroid administration (≥8 days from the onset of illness).

Administration, Administration of dexamethasone (DEXA) or other steroid treatment; IQR, interquartile range; ALT, alanine aminotransferase; LDH, lactase dehydrogenase, CK; creatine kinase; CRP, C-reactive protein; BMI, body mass index; WBC, white blood cell.
Multivariate analysis of risk factors for death in patients with COVID-19.
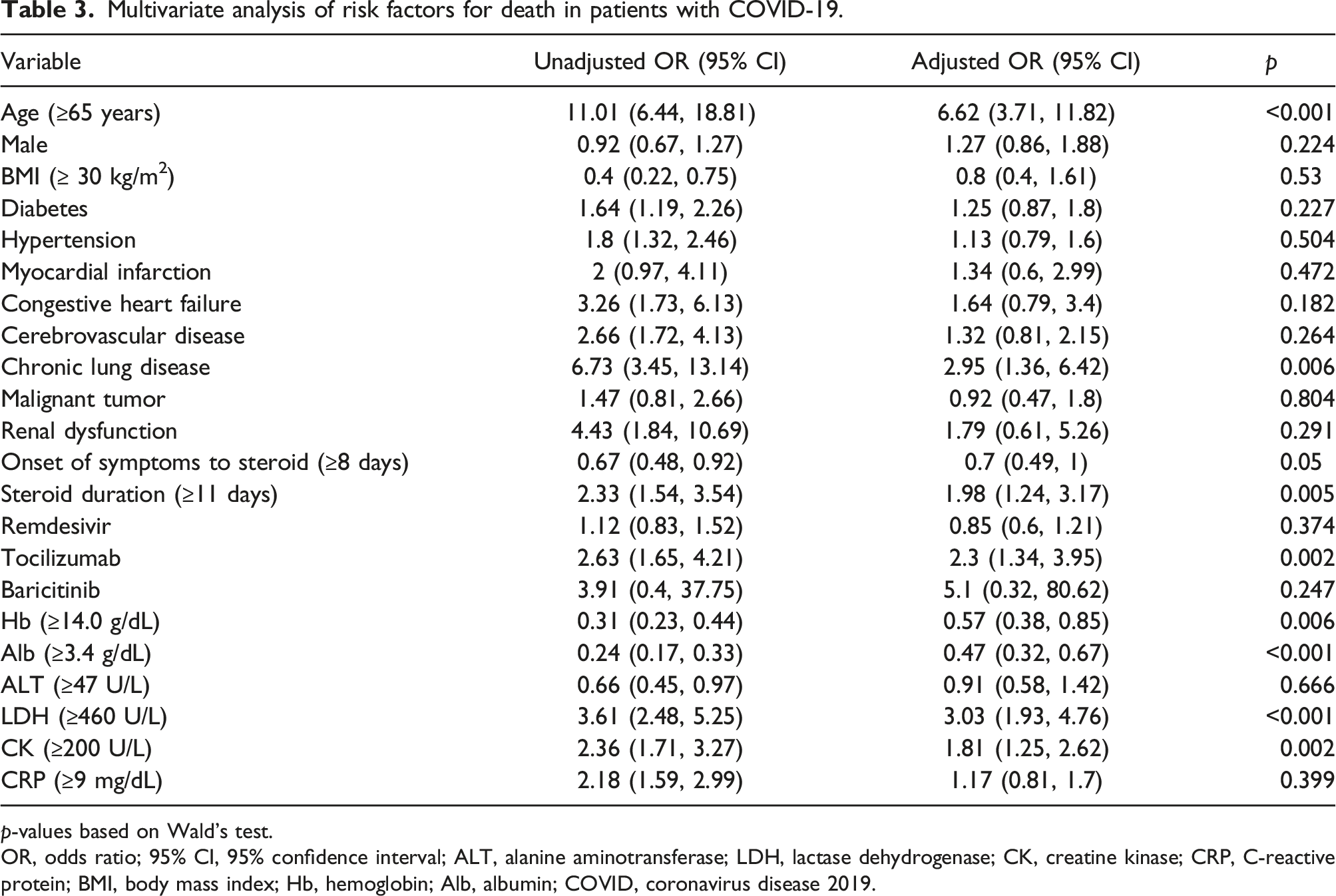
p-values based on Wald’s test.
OR, odds ratio; 95% CI, 95% confidence interval; ALT, alanine aminotransferase; LDH, lactase dehydrogenase; CK, creatine kinase; CRP, C-reactive protein; BMI, body mass index; Hb, hemoglobin; Alb, albumin; COVID, coronavirus disease 2019.
Discussion
In the present study, the number of days from the onset of COVID-19 symptoms to the start of steroid administration was associated with mortality from any cause. Furthermore, multivariate analysis showed that the number of days before the start of steroid administration was independent of severity risk factors, such as age, BMI, and clinical markers. Patients with COVID-19 are classified into three stages. 18 In stage I (early infection), 18 the virus settles in the respiratory tract via the angiotensin-converting enzyme 2 receptors on human cells, followed by viral replication, which causes non-specific cold-like symptoms. In stage II (pulmonary phase), 18 pulmonary involvement is established. Viral replication and localized pulmonary inflammation become sustained, and patients develop viral pneumonia. Dyspnea and hypoxia may appear. Gradually, the inflammatory response is central to the disease, and stage III (hyperinflammation phase) results in a systemic inflammatory response syndrome. 18 In the most severe cases of stage III, there is excessive inflammation characterized by hypercytokinemia similar to acute respiratory distress syndrome, with elevated levels of biomarkers and proinflammatory cytokines, such as interleukin-1B, interleukin-6, interleukin-12, interferon-γ, interferon-γ-inducible protein-10, and monocyte chemoattractant protein-1.18,19 Steroids suppress inflammation by inhibiting the excessive production of inflammatory cytokines and excessive lymphocyte activation, which can cause severe lung injury. 20 Therefore, they are expected to be effective in patients with severe COVID-19.
Several RCTs have examined the benefits of steroid administration for COVID-19. 3 The results of meta-analyses of these trials demonstrated the benefit of steroids against mortality and led to recommendations for steroid use. 2 However, a sub-group analysis of the same meta-analysis yielded a non-significant OR of 0.63 (95% CI 0.39–1.04) for steroid administration to patients within 7 days of onset of symptoms to death, indicating a lack of efficacy. 2 Similar to the results of the aforementioned sub-group analysis, the mortality rate in the present study was significantly higher among patients who received early steroids. 2
There are multiple possible reasons for the lack of a proven benefit of steroids when initiated <7 days after the onset of symptoms. Immunological factors play a central role in the pathogenesis of the disease after 1 week of symptom onset,3,21 SARS-CoV-2 replication peaks within 1 week of onset of symptoms, 22 and steroids suppress immunity and inhibit the clearance of the pathogen simultaneously with inflammation in the lungs, which is believed to have a negative effect on pathogenesis. 8 However, some reports deny the association between steroid administration and delayed viral clearance and between delayed clearance and severity of illness.23–25 In addition, increased mortality has been reported with steroid administration for novel influenza pneumonia and increased secondary bacterial pneumonia, and prolonged intensive care unit stays have been proposed as a cause. 26 A meta-analysis of RCTs reported that steroid administration also increased mortality in patients with COVID-19 in the group of patients who require no oxygen administration. 27 In the present study, we also considered the possibility that early steroid administration may have delayed viral clearance, resulting in worsening and prolongation of disease and, thus, increased mortality. However, there were no significant differences in the rate or duration of tracheal intubation, length of hospitalization, or time from intubation to death between patients with early versus late steroid administration. Furthermore, patients with early initiation of steroid administration had earlier tracheal intubation after symptom onset. These findings suggest the existence of a subset of patients with rapidly progressive and severe disease and higher mortality within the first week after the onset of symptoms, as opposed to adverse effects of steroids worsening disease and increasing secondary infections owing to delayed clearance. An earlier study also found that patients with a short interval from the onset of symptoms to hypoxemia were more likely to experience severe disease. 28 Future studies of this high-mortality-risk subset may increase mortality rates for patients experiencing severe COVID-19-related pneumonia.
There are several limitations to our study. First, this is a retrospective study using registry data. Because of the long enrollment period, we could not consider differences in viral strains or vaccination types. Second, the types and doses of steroids were not uniformly noted in the patient records. Third, the outcomes defined in the Methods are generic, and comparisons of clinical benefit between early or later onset of steroid treatment are not considered primary outcomes. Fourth, we selected patients who had received steroids equivalent to 6 mg DEXA only when steroids were used for 7−14 days; therefore, patients who died within 7 days from the onset of symptoms were excluded. Fifth, we could not evaluate the severity of factors not included in the patient records, such as the PaO2/FiO2 ratio or sequential organ failure assessment scores at the time of steroid administration or tracheal intubation. 29 Sixth, we did not have data on reasons for intubation or the use of Do-Not-Attempt-Resuscitation orders, and the actual causes of death were unknown; thus, whether the deaths were potentially related to steroid use remained unknown. Seventh, we did not have data on the specific steroid types used. Finally, sample size and power calculations were not performed in this study.
Conclusion
This large, retrospective, observational study showed that in patients with COVID-19 receiving steroids equivalent to 6 mg/day of DEXA, compared to later initiation of steroid administration, initiation of steroid treatment within 7 days of symptom onset was an independent risk factor for death. There were no significant differences in patient intubation rates, duration of intubation, length of hospital stay, or time from intubation to death between the early and delayed steroid initiation groups, suggesting the existence of a subset of patients with severe disease and high mortality within 1 week after symptom onset or worsening disease owing to early steroid administration or both. Further studies to identify this subset of patients with high mortality may contribute to mortality improvement.
Footnotes
Acknowledgments
Author contribution
RS conceptualized, designed, and coordinated the study and drafted the manuscript. TK conceptualized and designed the study and drafted, corrected, and restructured the manuscript. AM, NM, and TA analyzed and interpreted the data. RS and TK collected and entered the data. NO and HN revised and restructured the manuscript. All authors were involved in data interpretation and review of the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
COVIREGI-JP was funded by the “Research on Emerging and Reemerging Infectious Diseases and Immunization” program through the Ministry of Health, Labour and Welfare, grant number 19HA1003. Our study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical Statement
Data Availability Statement
![]() are available upon reasonable request. Data on an individual level will be shared, with limitations, to participating healthcare facilities through applications to COVIREGI-JP.
30
are available upon reasonable request. Data on an individual level will be shared, with limitations, to participating healthcare facilities through applications to COVIREGI-JP.
30
