Abstract
Objectives: T helper 17 (Th17) cells are involved in the inflammatory response of atherosclerosis. However, their heterogeneity in the atherosclerotic aorta remains elusive. This study was designed to identify aortic Th17 subsets. Methods: The surface markers and transcription factors of aortic interleukin-17A (IL-17A)-expressing T cells were determined by flow cytometry in an ApoE-deficient mouse atherosclerotic model. Viable aortic IL-17A-expressing T cell subsets were isolated by flow cytometry on the basis of surface markers, followed by characterizing their transcription factors by either flow cytometry or real-time RT-PCR. The effect of aortic IL-17A-expressing T cell subsets on aortic endothelial cells was determined in vitro. Results: C-X-C Motif Chemokine Receptor 3 (CXCR3), interleukin-17 receptor E (IL-17RE), CD200, and C-C Motif Chemokine Receptor 4 (CCR4) marked three subsets of aortic IL-17A-expressing T cells: CXCR3+IL-17RElowCD200+CCR4- T cells expressing T-box protein expressed in T cells (T-bet) and interferon-gamma (IFN-γ), CXCR3+IL-17RElowCD200+CCR4+ T cells expressing T-bet but fewer IFN-γ, and CXCR3−IL-17REhighCD200+CCR4+ T cells expressing very low T-bet and no IFN-γ. Based on these markers, viable aortic Th17 cells, Th17.1 cells, and transitional Th17.1 cells were identified. Both Th17.1 cells and transitional Th17.1 cells were more proliferative than Th17 cells. Compared with Th17 cells, Th17.1 cells plus transitional Th17.1 cells induced higher expression of C-X-C motif chemokine ligand 1 (CXCL1), C-C motif chemokine ligand 2 (CCL2), C-X-C motif chemokine 5 (CXCL5), and granulocyte-macrophage colony-stimulating factor (GM-CSF) in aortic endothelial cells. Conclusion: IL-17A-expressing CD4+ T cells were heterogeneous in atherosclerotic aortas.
Introduction
Atherosclerosis is an inflammatory disease involving multiple cell types in arteries including the aorta. 1 Immune cells such as macrophages, dendritic cells, T cells, and B cells contribute to atherogenesis and influence plaque stabilization. 2 T cells participate in the formation of atherosclerotic lesions. Under the cues from antigen-presenting cells and microenvironmental factors, CD4+ T cells differentiate into various effector subsets, including T helper 1 (Th1), T helper 2 (Th2), regulatory T cells (Tregs), and T helper 17 (Th17) cells. Th17 cells are particularly deeply involved in atherogenesis by producing interleukin-17A (IL-17A), interleukin-17F (IL-17F), interleukin-22 (IL-22), and other cytokines. 3 However, the role of Th17 cells in atherogenesis is under debate. Th17 cells have been shown to boost inflammatory response and foster atherosclerosis progression, although controversies remain,4–6 whereas other reports suggest a plaque-stabilizing effect of IL-17A via collagen synthesis. 7 However, accumulative evidence suggests that Th17 cells are heterogeneous, comprising subpopulations with phenotypic and functional discrepancies. 8 Besides, Th17 cells undergo polarization toward Th1, Th2, Th9, Th22, and Tregs. 9 To our knowledge, the heterogeneity of Th17 cells in the atherosclerotic aorta remains elusive.
Most Th17 studies rely on detecting intracellular IL-17A by flow cytometry, making isolating viable Th17 cells impractical because cells need to be fixed and permeabilized. Recent progress in identifying Th subsets according to surface markers such as CXCR3, CCR4, and CCR6 has made enrichment of viable Th17 cells from organs and tissues possible.10–14 Moreover, IL-17 receptor E (IL-17RE) is highly expressed on the surface of Th17 cells under different circumstances.15–17 CD200, which is an inhibitory immune checkpoint, is also found to be a novel Th17 surface marker. 18 Therefore, Th17 cells could be recognized and enriched by discerning the expression profiles of these surface markers.
In the current study, we revealed Th17 heterogeneity by recognizing viable Th17 cells and Th1-like Th17 cells (Th17.1 cells) in the aortas of atherosclerotic ApoE-/- mice. Based on the expression of IL-17RE, CD200, CXCR3, and CCR4, aortic IFN-γ-expressing Th17.1 cells were separated from aortic Th17 cells. These Th17.1 cells were more potent than Th17 cells in inducing the expression of pro-inflammatory chemokines and cytokines in aortic endothelial cells. Therefore, for the first time, the presence of pathogenic aortic Th17.1 cells in atherosclerosis was confirmed.
Materials and methods
Murine atherosclerosis model
This confirmatory animal study was approved by the Wuhan University Animal Care and Use Committee. The experimental procedures were conducted according to the Wuhan University Guidelines for the Use of Animals and lasted for 8 months. Eight-week-old male apolipoprotein E-deficient mice (ApoE-/-, C57BL/6J background) were purchased from Beijing Biocytogen Co., Ltd. The mice were fed a high-fat chow with 0.2% cholesterol and 21% fat by weight for 12–18 weeks. Three age- and sex-matched wild-type C57BL/6J mice were fed with a normal chow diet for 16 weeks.
Oil red O staining
The mice were euthanized by CO2 inhalation. The aortas were taken, fixed in 4% formaldehyde, and then immersed in 5% oil red O (Beyotime Biotech) for half an hour. After three washes in deionized water, atherosclerotic lesions, that is, oil red O-positive areas along the aorta, were recorded on a Leica DM500 fluorescence microscope (Leica).
Aorta processing and aortic cell collection
Isolation of aortic cells was achieved according to published methods with minor modifications.19,20 Briefly, mice were anesthetized with 2% halothane-oxygen, followed by perfusion with 3 mL of phosphate-buffered saline (PBS) via cardiac puncture to remove circulating blood cells. Each aorta with branches was carefully harvested and minced into fragments of about 1 mm in length. The tissue fragments were then incubated in 100 μL of digestion buffer (RPMI1640 medium containing 10% fetal calf serum [FCS], 200 U/ml collagenase XI, 500 U/ml collagenase I, 50 U/ml hyaluronidase I-s, and 100 U/ml DNase I) for half an hour in a 37oC water bath with gentle pipetting every 5 min. The digested tissues and the digestion buffer were filtered through a 40-µm cell strainer, followed by centrifugation at 250×g for 5 min. The cell pellet was suspended in 0.5 mL of ice-cold PBS for the following experiments. In some experiments, 15 aortas were pooled and digested with proportionally increased reagents. The reagents were purchased from Sigma-Aldrich. In some experiments, the cells were stimulated with 20 ng/mL phorbol ester (PMA), 1 µg/ml ionomycin, and 10 μg/mL brefeldin A (all from Beyotime Biotech) for 4 h at 37oC before further analysis.
Blood leukocyte collection
Peripheral blood was collected from the tail vein and mixed with 1 mL of 2 mM ethylenediaminetetraacetic acid-PBS. After centrifugation at 200×g for 5 min, red blood cells were lysed by suspending the cell pellet in 0.5 mL of RBC lysis buffer (Beyotime Biotech) for 5 min at room temperature. Cells were washed with 1 mL of PBS once and subjected to the same staining procedures as for aortic cells.
Flow cytometry analysis
Flow cytometry antibodies.

Real-time reverse transcription and polymerase chain reaction (RT-PCR)
Primer sequences.

Isolation of aortic endothelial cells
Aortic cells were collected as described above. These cells were then incubated with 200 µl of PBS containing 2 µg/ml FITC-conjugated anti-mouse CD31 antibody (BioLegend) for 20 min on ice. After three washes with PBS, CD31+ AECs were sorted by the BD FACSAria cell sorter.
In vitro culture
Sorted T cells and aortic endothelial cells were suspended at the cell density of 5×104/ml in the culture medium (RPMI1640 medium containing 10% FCS). For intracellular cytokine staining, T cells were treated with 20 ng/mL phorbol ester (PMA), 1 µg/ml ionomycin, and 10 μg/mL brefeldin A for 4 h at 37oC.
To evaluate the effect of T cells on AECs, T cells were stimulated with 20 ng/mL PMA and 1 µg/ml ionomycin for 4 h and washed with PBS once. After that, 3000 T cells and 1000 AECs were mixed in 50 µl of culture medium and seeded in each well of a 96-well V-shaped-bottom microplate. The cells were centrifuged at 200×g for 5 min and incubated for 24 h at 37oC in an incubator. At the end of the co-culture, cells were stained with 2 µg/ml APC anti-mouse CD45 antibody and biotinylated anti-mouse CD144 antibody for 20 min on ice. Cells were washed with 0.5 mL of PBS once and incubated with 2 µg/ml FITC Streptavidin for 20 min on ice. Cells were suspended in PBS and loaded onto the BD FACSAria cell sorter to sort CD144+ AECs.
Statistical analysis
Every experiment was independently carried out 2 or 3 times. The data were shown as mean ± standard deviation. The Student’s t-test or one-way ANOVA with post-hoc Tukey HSD test was used to compare the differences. A
Results
The phenotypes of aortic IL-17A-expressing T cell subsets based on surface and intracellular markers
We fed ApoE-/- mice with a high-fat diet for 16 weeks and observed atherosclerotic lesions in their aortas (Supplementary Figure 1). To characterize the phenotype of IL-17A-expressing T cells, mouse aortas were collected and dissociated with enzymes. The resultant single cells were stimulated with PMA plus ionomycin in the presence of brefeldin A for 4 h. The cells were then stained with fluorochrome-conjugated antibodies against CD3, IL-17RE, CXCR3, CD200, and CCR4, followed by intracellular staining with fluorochrome-conjugated antibodies against IL-17A, RORγt, T-bet, or IFN-γ, respectively. The whole workflow is demonstrated in Supplementary Figure 2(A). As indicated in Figure 1(A), single cells were first gated according to the forward scatter-A and forward scatter-H. Within these single cells, a CD3+IL-17A+ population was observed, suggesting the presence of IL-17A-expressing T cells. These IL-17A-expressing T cells were CD4+ T cells (Supplementary Figure 3). According to the surface staining of CXCR3 and IL-17RE, IL-17A-expressing T cells were divided into two subsets: CXCR3+IL-17RElow T cells and CXCR3−IL-17REhigh T cells. The former constituted about 15% of IL-17A-expressing T cells while the latter accounted for over 80% of IL-17A-expressing T cells (Figure 1(B)). Further analysis revealed that CXCR3+IL-17RElow T cells contained two subsets: CD200+CCR4- cells and CD200+CCR4+ T cells, as shown in Figure 1(C) and 1D. CXCR3−IL-17REhigh T cells were predominantly CD200+CCR4+ (Figure 1(C)). Therefore, three subsets were present in aortic IL-17A-expressing T cells: CXCR3+IL-17RElowCD200+CCR4- T cells (hereinafter R3+RElow200+R4-), CXCR3+IL-17RElowCD200+CCR4+ T cells (R3+RElow200+R4+), and CXCR3−IL-17REhighCD200+CCR4+ T cells (hereinafter R3−REhigh). Phenotype of aortic IL-17A-expressing T cells. (A) Representative dot plots showing the gating strategy for aortic IL-17A-expressing T cell subsets based on CXCR3 and IL-17RE. (B) The proportions of R3+RElow T cells and R3−REhigh T cells in total aortic IL-17A-expressing T cells. (C) Representative dot plots showing the expression of CD200 and CCR4 in R3+RElow T cells and R3−REhigh T cells, respectively. (D) The proportions of 200+R4- and 200+R4+ cells in R3+RElow T cells, respectively. (E) Representative histograms showing RORγt expression in indicated subsets. (F) Representative histograms showing T-bet protein in indicated subsets. (G) The mean fluorescence intensities (MFI) of T-bet in indicated subsets. (H) Representative dot plots showing intracellular staining of IFN-γ in indicated subsets. (I) The proportions of IFN-γ-expressing cells in indicated subsets. 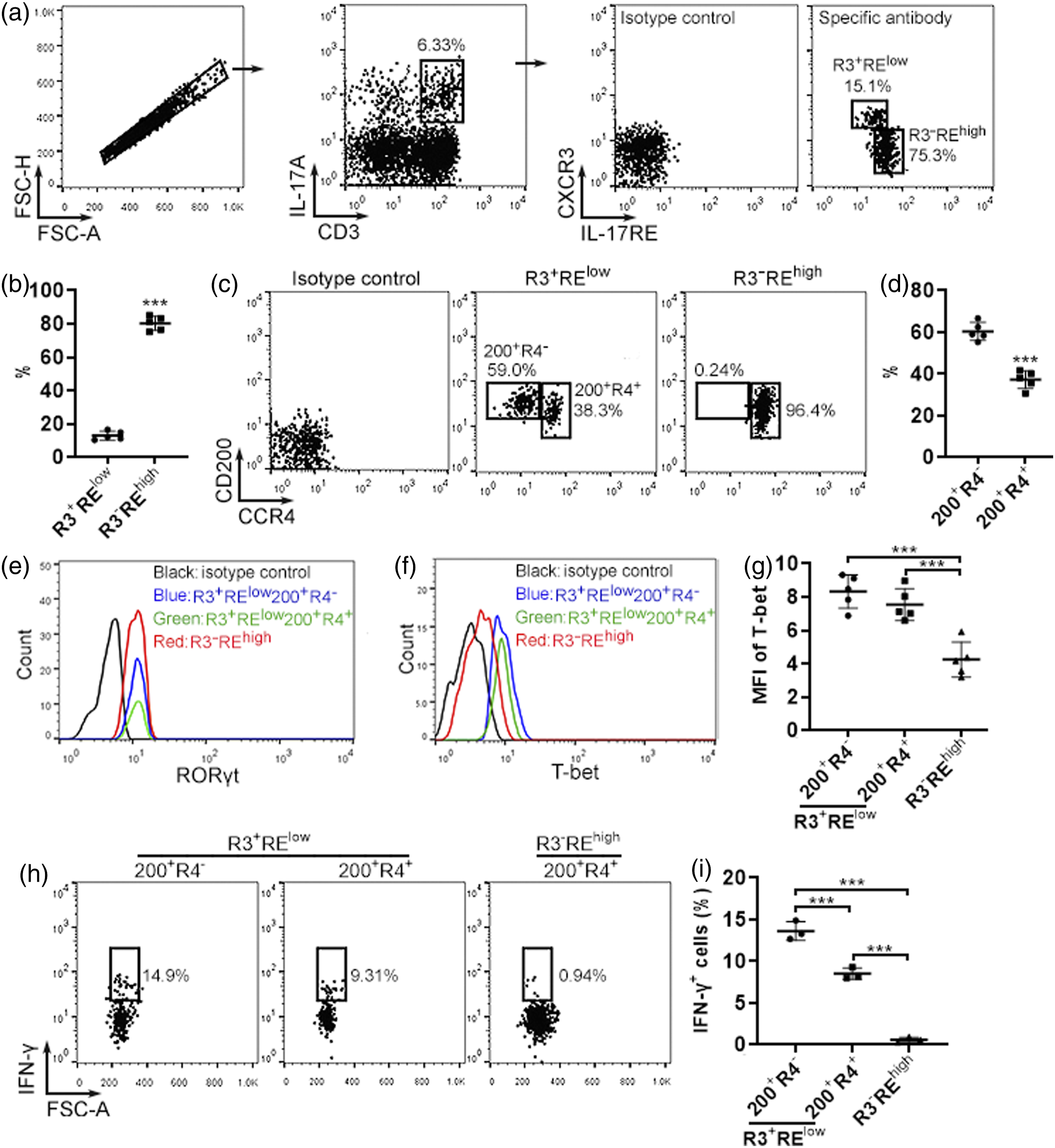
Evaluation of T-bet and RORγt revealed equivalent RORγt expression in the three subsets (Figure 1(E)). However, R3+RElow200+R4- T cells and R3+RElow200+R4+ T cells expressed higher T-bet than R3−REhigh T cells (Figures 1(F) and (G)). Furthermore, substantial IFN-γ expression was found in R3+RElow200+R4- T cells, while R3+RElow200+R4+ T cells expressed fewer IFN-γ and R3−REhigh did not express IFN-γ (Figures 1(H) and (i)). Therefore, R3+RElow200+R4- T cells were Th17.1 cells. R3+RElow200+R4+ T cells seemed to be transitional Th17.1 cells between the Th17 stage and Th17.1 stage. R3−REhigh T cells were Th17 cells.
To explore the presence of these subsets in the blood, we stained circulating leukocytes with the same antibodies against relevant surface and intracellular markers. As shown in Supplementary Figure 4(A), about 2% of CD3+IL-17A+ T cells were found in circulating leukocytes. Interestingly, these cells were all R3−REhigh (Supplementary Figure 4(B)) with uniform RORγt expression (Supplementary Figure 4(C)). No substantial IFN-γ expression was detected in circulating IL-17A-expressing T cells (Supplementary Figure 4(D)). Therefore, these cells were all Th17 cells. Th17.1 and transitional Th17.1 cells were absent in the blood and were likely generated only in the atherosclerotic aorta.
Temporal changes in the frequencies of IL-17A-expressing T cell subsets
To investigate the persistence of the above-mentioned T cell subsets in the atherosclerotic aorta, we enriched aortic cells 12 weeks, 16 weeks, and 18 weeks after the high-fat diet treatment. The same staining as described above was performed to characterize the phenotype of aortic IL-17A-expressing T cells at different time points. As indicated in Figures 2(A) and (C), at week 12, the frequency of aortic IL-17A-expressing T cells was around 3.5%, and this frequency increased to over 6% at week 16 and week 18. At week 12, aortic IL-17A-expressing T cells were all R3−REhigh (Figures 2(A) and (D)). At week 16 and week 18, R3+RElow cells emerged in aortic IL-17A-expressing T cells and increased over time (Figures 2(A) and (D)). Furthermore, at week 12, almost all aortic IL-17A-expressing T cells were 200+R4+. At week 16 and week 18, R3+RElow T cells contained the 200+R4- subset and 200+R4+ subset. The frequency of the 200+R4- subset increased from week 16 to week 18, while the frequency of the 200+R4+ subset decreased in the same period (Figures 2(B) and (E)). R3−REhigh T cells were constantly 200+R4+ through the period tested (Figures 2(B) and (E)). Temporal changes of IL-17A-expressing T cell subsets. (A) Representative dot plots showing the gating strategy for aortic IL-17A-expressing T cell subsets based on CXCR3 and IL-17RE from week 12 to week 18. (B) Representative dot plots showing the expression of CD200 and CCR4 in R3+RElow T cells and R3−REhigh T cells. (C) The proportions of aortic IL-17A-expressing T cells in total aortic cells. (D) The proportions of R3+RElow T cells and R3−REhigh T cells in aortic IL-17A-expressing T cells. (E) The proportions of 200+R4- and 200+R4+ cells in R3+RElow T cells at each time point. 
Identification of viable aortic Th17 and Th17.1 cells based on surface markers
Although the above data uncovered aortic IL-17A-expressing T cell subsets, these cells were fixed and permeabilized for intracellular IL-17A staining, making enrichment of viable Th17 and Th17.1 cells impossible. Next, we tried to identify viable Th17 and Th17.1 cells based on surface marker expression. The whole workflow is illustrated in Supplementary Figure 2(B). As indicated in Figure 3(A), CD3+CD4+ T cells were first recognized in aortic cells, and an IL-17RE+CD200+ population (hereinafter RE+200+) was found in CD3+CD4+ T cells. The rest CD3+CD4+ T cells were IL-17RE− (hereinafter RE−). Real-time RT-PCR revealed that compared with total CD4+ T cells, RE+200+ cells expressed higher Recognition of viable aortic Th17 and Th17.1 cells. (A) Representative dot plots showing the gating strategy for aortic CD3+CD4+RE+CD200+ T cells. (B) mRNA levels of indicated transcription factors in total CD4+ T cells (CD4), RE+200+ T cells, and RE− T cells. Un: under detection threshold. (C) Representative dot plots showing the gating strategy for R3+R4- cells, R3+R4+ cells, and R3-R4+ cells in aortic RE+200+ T cells. (D) Representative histograms showing RORγt and T-bet proteins in R3+R4- cells, R3+R4+ cells, and R3-R4+ cells. (E) The mean fluorescence intensities (MFI) of RORγt and T-bet in the subsets. Expression of indicated cytokines in R3+R4- cells, R3+R4+ cells, and R3-R4+ cells. (A, C, and E) Representative dot plots showing the expression of IL-17A (A), IFN-γ (C), and TNF-α (E) in the subsets. (B, D, and F) The proportions of IL-17A-expressing (B), IFN-γ-expressing (D), and TNF-α-expressing (F) cells in each subset, respectively. 

Distinct proliferation status of aortic Th17 and Th17.1 cells
Ki67 staining of the above R3+R4- T cells (Th17.1), R3+R4+ T cells (transitional Th17.1), and R3-R4+ T cells (Th17) indicated that the former two subsets had equivalent Ki67 expression, whereas Th17 cells expressed fewer Ki67 (Figures 5(A) and (B)). We then assessed mRNA levels of genes encoding cyclin-dependent kinase inhibitors which control cell proliferation. As indicated in Figure 5(C), the three subsets expressed comparable levels of Proliferation of Th17.1 cells (R3+R4- cells), transitional Th17.1 cells (R3+R4+ cells), and Th17 cells (R3-R4+ cells). (A) Representative dot plots showing Ki67 staining of indicated subsets. (B) The proportions of Ki67+ cells in indicated subsets. tTh17.1: Transitional Th17.1 cells. (C) mRNA levels of indicated CDK inhibitors in the subsets. 
The effects of aortic Th17 and Th17.1 cells on AECs
Th17 cells modulate chemokine expression in endothelial cells to facilitate leukocyte recruitment.21,22 To check the effects of aortic Th17 and Th17.1 cells on AECs, RE+CD200+R3-R4+ cells (Th17 cells) and RE+CD200+R3+ cells (i.e., combined Th17.1 and transitional Th17.1 cells, termed cTh17.1 cells hereinafter) were sorted from 15 aortas and stimulated with PMA and ionomycin. Due to their low numbers, functionally related Th17.1 cells and transitional Th17.1 cells were sorted together to prepare enough cells for the following co-culture assay. Meanwhile, mouse CD31+ AECs were enriched by flow cytometry (Figure 6(A)). Stimulated Th17 cells and cTh17.1 cells were then co-cultured with AECs for 24 h at the ratio of 3:1, respectively. After that, cells were stained with a fluorochrome-conjugated anti-CD45 antibody and an anti-CD144 (VE-Cadherin) antibody. CD144+ AECs were sorted from the cell mixture (Figure 6(B)) and subjected to real-time RT-PCR to analyze chemokine and cytokine mRNAs. As indicated in Figures 6(C) to (G), Th17 cells increased the mRNAs of Effects of Th17 and combined Th17.1 cells on AECs. (A) Representative dot plots showing CD31+ AECs before sorting. (B) Representative dot plots showing CD144+ AECs and CD45+ T cells after co-culture. (C to G) mRNA levels of indicated chemokines and GM-CSF in AECs after co-culture with Th17 or combined Th17.1 cells. Alone: AECs cultured alone. Th17: AECs co-cultured with Th17 cells. cTh17.1: AECs co-cultured with combined Th17.1 cells. 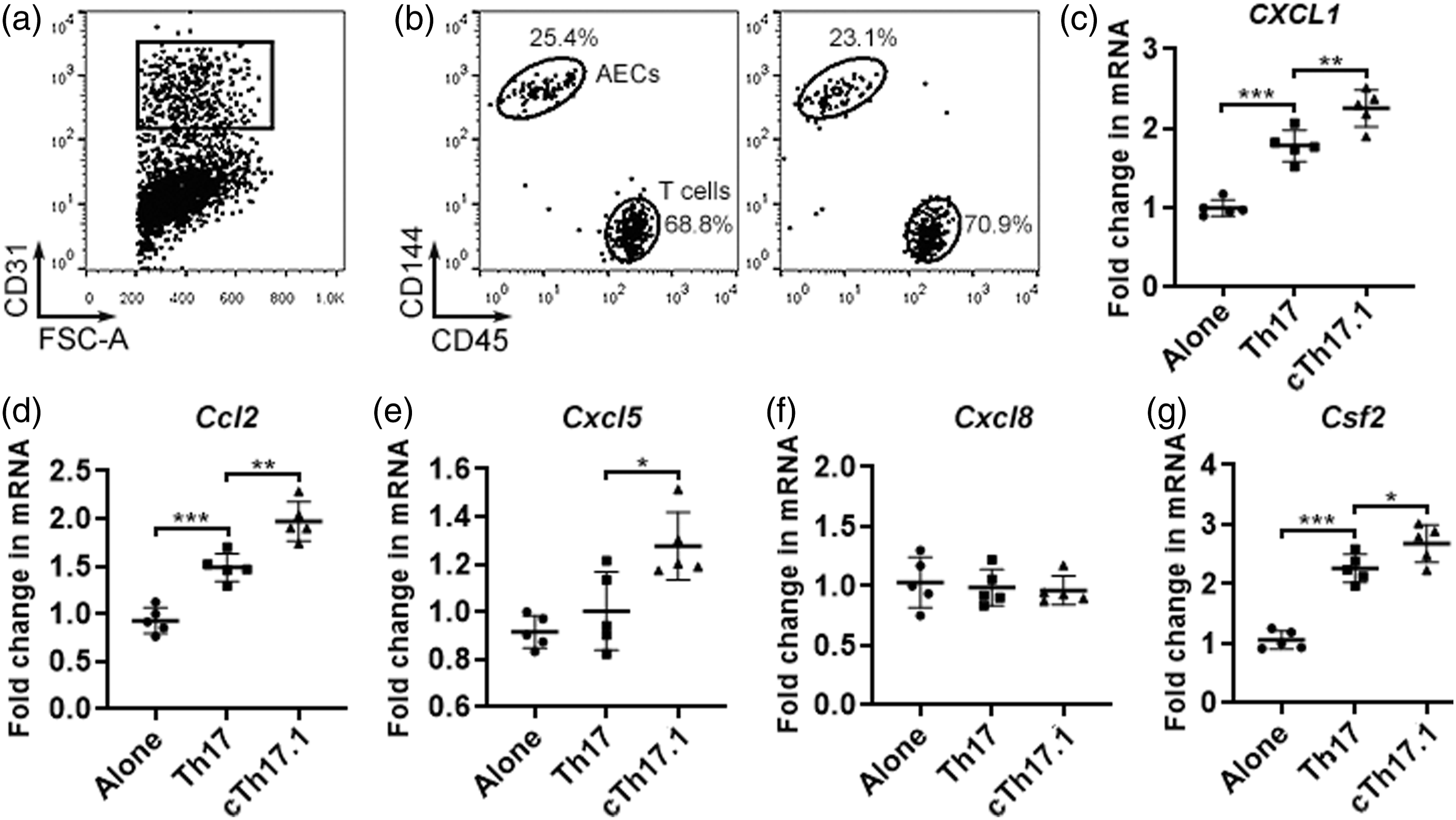
Discussion
The present study characterized the phenotype of IL-17A+CD4+ T cells in the aortas of atherosclerotic mice. The logic of this study is to reveal the significant expression of CD200, IL-17RE, CXCR3, and CCR4 on the surface of aortic IL-17A+CD4+ T cells in the first place, and then distinguish viable Th17 cells according to the unique expression pattern of these surface markers. We found that aortic IL-17A+CD4+ T cells were RE+200+. However, the expression of CXCR3 and CCR4 was differential on aortic IL-17A+CD4+ T cells and indicated different subsets. CXCR3 is expressed by Th1 cells but not by Th17 cells. 23 However, Th17.1 cells, which might be derived from Th17 cells, up-regulate CXCR3 on their surface and acquire other Th1 features such as the expression of T-bet and IFN-γ.23,24 CCR4 is considered to be expressed by Th17 cells but not by Th1 cells. 25 Th17.1 cells lose CCR4 on their surface, demonstrating another Th1 feature. 26 Therefore, our findings are consistent with previous studies, implying that Th17.1 cells in atherosclerotic aortas might possess a similar phenotype to Th17.1 cells found in other pathological conditions.
Notably, aortic IL-17A+CD4+RE+200+R3+R4- T cells, that is, Th17.1 cells, expressed higher T-bet and IFN-γ than IL-17A+CD4+RE+200+R3-R4+ T cells (Th17 cells), further confirming the identity of Th17.1 cells. However, the factors responsible for the generation of aortic Th17.1 cells remain unknown. Several cytokines or inflammatory mediators, including IL-1β, IL-23, prostaglandin E2 (PGE2), and TNF-α, may induce the polarization of Th17 cells toward Th17.1 cells. 23 Interestingly, these factors are present in atherosclerotic lesions,27-31 perhaps constituting a microenvironment favorable for Th17.1 differentiation.
Based on the phenotype of aortic IL-17A+CD4+ T cells, we tried to discriminate viable aortic Th17 and Th17.1 cells by detecting the above-mentioned surface markers and avoiding intracellular cytokine staining. We successfully discerned three CD4+ T subpopulations featuring Th17 and Th17.1 cells, respectively. Of note, both CD4+RE+200+R3+R4- T cells and CD4+RE+200+R3+R4+ T cells expressed relatively high T-bet, suggesting their Th17.1 identity. However, the former produced more IFN-γ and TNF-α than the latter, implying that the former subset might be mature Th17.1 cells, whereas the latter subset was transitional Th17.1 cells. In the future, the two subsets should be subjected to transcriptomic profiling to unveil the molecular mechanisms underlying their functions. Meanwhile, CD4+RE+200+R3-R4+ cells were likely classical Th17 cells.
Another interesting finding is that Th17.1 cells seemed to be more proliferative than Th17 cells. This is consistent with previous research showing faster proliferation of Th1 cells and Th17.1 cells relative to Th17 cells. 32 However, Th17.1 cells were still a minor subset in IL-17A-expressing T cells regardless of their faster expansion. This raises the possibility that Th17.1 cells might completely lose IL-17A expression and become Th1 cells. Indeed, the transition from Th17 cells to IL-17−IFN-γ+ Th1-like cells (sometimes termed ex-Th17 or nonclassical Th1) has been reported.33,34 In the future, the lineage tracing study using IL-17A-EGFP and IFN-γ-RFP transgenic mice could reveal the fate of Th17 and Th17.1 cells in atherosclerosis.
Aortic Th17 cells expressed more p16 INK4A (encoded by
Th17 cells can boost inflammatory response and foster atherosclerosis progression. 4 Th17.1 cells play crucial roles in the pathogenesis of inflammatory bowel disease, multiple sclerosis, and rheumatoid arthritis. 41 According to our data, aortic Th17.1 cells and transitional Th17.1 cells produce IFN-γ and TNF-α. IFN-γ is localized in atherosclerotic lesions to recruit inflammatory cells by inducing the expression of adhesion molecules in ECs during the early stages of atherosclerosis.42,43 IFN-γ also induces CCL2 expression in atherosclerotic lesions.44,45 Moreover, IFN-γ contributes to foam cell formation and reduces plaque stability. 43 TNF-α promotes inflammatory cell recruitment through up-regulating adhesion molecules, CXCL1, CCL2, and GM-CSF in endothelium.46–49 Therefore, aortic Th17.1 cells could up-regulate chemokines and adhesion molecules to exacerbate aortic inflammation. Our research indicated that aortic Th17.1 cells plus transitional Th17.1 cells were more capable of inducing CXCL1, CCL2, CXCL5, and GM-CSF expression in AECs than Th17 cells, implying that Th17.1 cells and transitional Th17.1 cells could be more pathogenic than Th17 cells in recruiting inflammatory cells. At the early stage of atherosclerosis, Th17.1 cells are absent or minor in the atherosclerotic aorta (as evidenced in Figure 2), so Th17 cells play a significant role in atherosclerosis progression. However, over time, Th17.1 cells accumulate in the aorta and possibly co-exist with Th17 cells in atherosclerotic lesions. At the late stage, aortic Th17.1 cells might work together with Th17 cells to exacerbate aortic inflammation and promote the formation of atherosclerotic lesions.
The limitations of this study include the following: (1) The origin and the fate of aortic Th17, Th17.1, and transitional Th17.1 cells remain unclear. Future lineage tracing studies using genetically modified mouse strains might answer these questions. (2) The locations of the three subsets are not determined. Whether they are in atherosclerotic lesions and their potential interactions with other cell types should be investigated in aorta sections in the future. (3) Due to the limited numbers of these subsets, their pathogenicity has not been evaluated in vivo. In vitro expansion and adoptive transfer of aortic Th17 and Th17.1 cells could be conducted to determine their pathogenicity in the future.
Conclusion
According to the present findings, our study reveals the presence of aortic Th17.1 cells and tested their effect on the AECs. The data exhibit the presence of Th17, Th17.1, and transitional Th17.1 cells in the atherosclerotic aorta. Th17.1 cells and transitional Th17.1 cells were more proliferative than Th17 cells. Compared with Th17 cells, Th17.1 and transitional Th17.1 cells induced higher expression of pro-inflammatory chemokines and GM-CSF in AECs. Our study sheds light on the heterogeneity of aortic Th17 subsets in atherosclerosis.
Supplemental Material
Supplemental Material - Identification of heterogeneous subsets of aortic interleukin-17A-expressing CD4+ T cells in atherosclerotic mice
Supplemental Material for Identification of heterogeneous subsets of aortic interleukin-17A-expressing CD4+ T cells in atherosclerotic mice by Guizhen Lin, Lei Zhang, Zheng Yan, Wei Jiang, Beibei Wu, Dongsheng Li and Xiaofang Xiong in International Journal of Immunopathology and Pharmacology
Footnotes
Author’s contributions
All authors contributed to the study. Research design and manuscript writing were performed by Xiaofang Xiong. The animal model was established by Zheng Yan and Wei Jiang. Flow cytometry analysis was conducted by Guizhen Lin and Lei Zhang. Real-time RT-PCR was performed by Beibei Wu. In vitro cell culture was carried out by Dongsheng Li.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Program of the Wuhan Municipal Health and Family Planning Commission Foundation (Grant # WX15A03).
Data availability
The data that support the findings of this study are available from the corresponding author, Dr. Xiaofang Xiong, upon reasonable request.
Ethics approval
Ethical approval for this study was obtained from the Wuhan University Animal Care and Use Committee (APPROVAL ID: 202,025,734).
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
