Abstract
Introduction
Patients with bipolar disorder (BD) exhibit an inflamed condition that is associated with metabolic disturbance and cognitive impairment. Whether inflammation, represented by C-reactive protein (CRP), is causally associated with BD and influences treatment outcome has not been established.
Methods
We examined whether CRP is a causal factor for the risk of BD in drug-naïve, depressed BD patients and investigated whether polymorphisms in
Results
Our results showed that BD patients had significantly higher CRP levels and worse cognitive function than the controls, while the frequencies of
Conclusion
Our study demonstrated the clinical utility of the application of functional genetics in clarifying the interactions among CRP, life event stress, and BD and suggested the important roles of
Keywords
Introduction
Bipolar disorder (BD) is a mental disorder with a high prevalence of metabolic disturbances and cognitive impairment. An increasing amount of evidence suggests that inflammatory and immune alterations may be involved in the pathogenesis of BD, while inflammation is also associated with metabolic disturbances and cognitive impairment. In addition, the condition of greater inflammation observed in BD might explain why the estimated risk of dementia is higher for BD than for major depression.1,2 Proinflammatory cytokines may be potential mediators of interactions between the immune-neuroendocrine system and pathways related to mood and energy, contributing to alterations in brain function in cases of BD.3,4 Additionally, cognitive impairments have been documented in cases of BD,5,6 especially in continuous performance and frontal executive tasks. 7 These cognitive impairments may be long-lasting even in stable euthymic BD patients, 8 which suggests an association with inflammatory status. Moreover, comorbidities with BD, metabolic abnormalities, and cognitive deficits contribute to increased long-term costs to the national health care system.9,10 Therefore, to understand BD better and to help alleviate the burden of long-term costs to the national health care system from BD, it is important to study whether inflammation is causally associated with BD and treatment outcome, focusing on cognitive function. In addition, valproate (VPA), a mood stabilizer, is usually widely used in treating BD, and it could be used at different stages of BD, including depressive, manic, and maintenance phases. 11 Among mood stabilizers, the trend in VPA use gradually increased and amounts of prescription were listed top one among patients newly diagnosed with BD in Taiwan. 12 Previous reports suggest that VPA has neuroprotective effects.13-15 Valproate increases levels of brain-derived neurotrophic factor (BDNF) mRNA and increases the activity of BDNF promoter IV in rat cortical neurons. 14 Valproate also protects dopaminergic neurons in midbrain neurons by stimulating the release of neurotrophic factors from astrocytes. 15 Therefore, we focused on the treatment outcome of VPA in cases of BD.
C-reactive protein (CRP), which is synthesized by hepatocytes, is an acute-phase protein arising in response to proinflammatory cytokines, endogenous signals of innate immunity, and tissue damage. C-reactive protein levels have been a biomarker for the risk of cardiovascular diseases and metabolic disturbances.16,17 In addition, previous studies have revealed significantly higher CRP levels in patients with BD than in healthy controls, but this was not the case for major depressive disorder (MDD) patients.1,18 An elevated CRP level is also an important risk factor for the onset of manic symptoms in depressed patients during 2 years of follow-up. 19 Furthermore, CRP levels in individuals with BD are significantly associated with disease severity 20 and could be a marker of mood state.1,21,22 However, whether elevated CRP levels are causally associated with BD has not yet been established because of study design issues and confounding factors, such as mood stage and use of medications.
Helpful methods for the clinical use of measured variation in genes of known function as instrumental variables have been suggested to examine the causal effect of modifiable exposure on disease in observational studies.23,24 Causal relationships between CRP and BD could not be determined in clinical practice since CRP levels vary across mood states, levels of medication use, and study design in the context of BD patients.
22
Thus, Mendelian randomization analysis provides a more reliable indication of the likely causal role of CRP in the development of BD. According to previous studies, some single nucleotide polymorphisms (SNPs) of the
Materials and methods
Subjects
The Institutional Review Board for the Protection of Human Subjects at National Cheng Kung University Hospital approved the research protocol (IRB No. A-ER-104-031). All participants were recruited from outpatient settings at the National Cheng Kung University Hospital and provided written informed consent regarding their willingness to participate in the research (from March 2015 to August 2019). We also recruited controls from the community after individuals with mental illnesses were excluded by a senior psychiatrist using the Chinese version of the Mini International Neuropsychiatry Interview. All BD patients were initially evaluated via interview by an attending psychiatrist using the Chinese version of the Modified Schedule of Affective Disorder and Schizophrenia–Life Time, which has good interrater reliability, to determine diagnoses from the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5). To avoid possible confounding factors affecting the results, all BD patients exhibited BD II, indicating exclusion of BD I. The BD patients also met the following inclusion criteria: (i) diagnosis of BD II using the 2-days minimum for hypomania, 28 (ii) major depressive status at the time of study entry, with a 17-item Hamilton Depression Rating Scale score >15, (iii) drug-naïve and at first diagnosis with no history of treatment with mood stabilizers or antipsychotics, and (iv) no history of psychiatric or neurological disorder or alcohol or substance abuse. In addition, subjects meeting the following criteria are not prescribed to receive valproate treatment according to the guidance Taiwan Food and Drug Administration: (i) during pregnancy for the treatment of bipolar disorder, and (ii) women of childbearing potential unless they have birth control (contraception).
To avoid confounding factors interfering with plasma CRP level, all subjects meeting the following criteria were excluded through chart reviews and patient-reported questionnaires: (i) a serious surgical condition or physical illness, including all chronic diseases, heart disease, stroke, kidney dialysis, transplant, etc., (ii) patients who were pregnant or breastfeeding,29,30 (iii) a DSM-5 diagnosis of substance abuse within the past 3 months, 31 (iv) previous use of any psychotropic agent, 32 (v) consumption of any anti-inflammatory medication within the past 3 months, having autoimmune disorder, or having an infectious status (plasma CRP >7 mg/L), and (vi) having an organic mental disease, mental disability or dementia.33,34
All of the control subjects underwent cognitive function tests. Among BD patients, 54 BD subjects underwent cognitive function tests after receiving VPA treatment for 12 weeks. BD patients were permitted to treat depressive symptoms (fluoxetine, up to 20 mg/day), and lorazepam (<8 mg) was used for night-time sedation and to treat agitation and insomnia during the study, the dosage of which was adjusted according to clinical manifestation and the patient’s tolerance.
Measurements
Fasting blood samples were collected between 8:00 a.m. and 10:00 a.m. 10 milliliters of whole blood was withdrawn from the antecubital vein of each patient. Plasma, which was isolated from whole blood after centrifugation at 3000
Genomic DNA was extracted from each blood sample using a QIAamp DNA blood kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. The quality of the extracted genomic DNA was checked by agarose gel electrophoresis analysis. The DNA was stored at −80°C until use. The SNPs of the
The serum trough concentration of VPA was assessed using the homogeneous enzyme immunoassay method and measured at the Union Clinical Laboratory (Taipei, Taiwan). The limit of detection was 3 μg/mL.
Cognitive function
Finger-tapping test
The Finger-tapping test (FTT) consists of tapping with the index finger on a computer mouse as many times as possible within 10 s. The test was repeated three consecutive times and performed randomly across subjects, and the order was kept constant for each subject at each session. The average number of taps was then calculated. 35
Continuous performance test
The Continuous Performance Test (CPT) is a psychological test for humans that primarily measures attention.36,37 The critical stimulus may be defined either as a particular single stimulus out of the available set (X task: subjects were asked to respond to number “9”) or a particular sequence of two stimuli out of the available set (AX task: subjects were asked to respond whenever the number “9” was preceded by the number “1”). Only the AX task was used in the present study. Each test session began with 2 min of practice (repeated if necessary) to ensure that subjects knew how to press the button correctly. During the test, numbers from 0 to 9 were randomly presented for 50 milliseconds each at a rate of one per second. Each subject underwent two sessions, including the nonmasked task and the 25% masked task. During the masked session, a pattern of snow was used to toggle the background and foreground so that the image was visually distorted. Masked CPT is more sensitive in detecting cognitive deficits. Subject responses were recorded automatically on a diskette using the CPT machine (Sunrise Systems, version 2.20, Pembroke, MA, USA). 38
Life event scale
The Recent Life Changes Questionnaire was developed and modified from the Schedule of Recent Experience, which was used to collect information concerning subjects’ recent life changes. 39 In this study, we used the Taiwanese version life event scale (LES), which contains 39 items regarding representative life change events in the past 12 months, and the level of perceived stress brought about by recent life-changing events was recorded. 40
Statistical analysis
Statistical analysis was performed using the Statistical Package for Social Sciences 12.0 (SPSS Inc, Chicago, IL, USA). Categorical variables were expressed as numbers and percentages, while continuous variables were expressed as the means ± standard deviations (SD) unless otherwise specified. Categorical variables were assessed using chi-square tests, while continuous variables were assessed using t tests. Hardy-Weinberg equilibrium was assessed by using the χ2 goodness-of-fit test. Since plasma CRP level was not normally distributed, the distribution of CRP levels is shown as median, third quartile (Q3), and first quartile (Q1). Statistical analysis of plasma CRP levels was assessed by the Mann–Whitney U test. The level of significance was set at 0.05 for two-sided tests.
In the Mendelian randomization analysis, we first examined the gene-plasma CRP association in the control subjects by multiple linear regression analyses with adjustment for age, sex, body mass index (BMI), and smoking status. Each genotype was coded by the number of copies of the minor allele under the additive model and analyzed as a continuous variable beta coefficient value (BCRP/per allele), which indicated the per allele increase in plasma CRP level in general. Second, we examined the risk estimate for 
Results
Demographic characteristics of drug-naïve BD patients and control subjects.
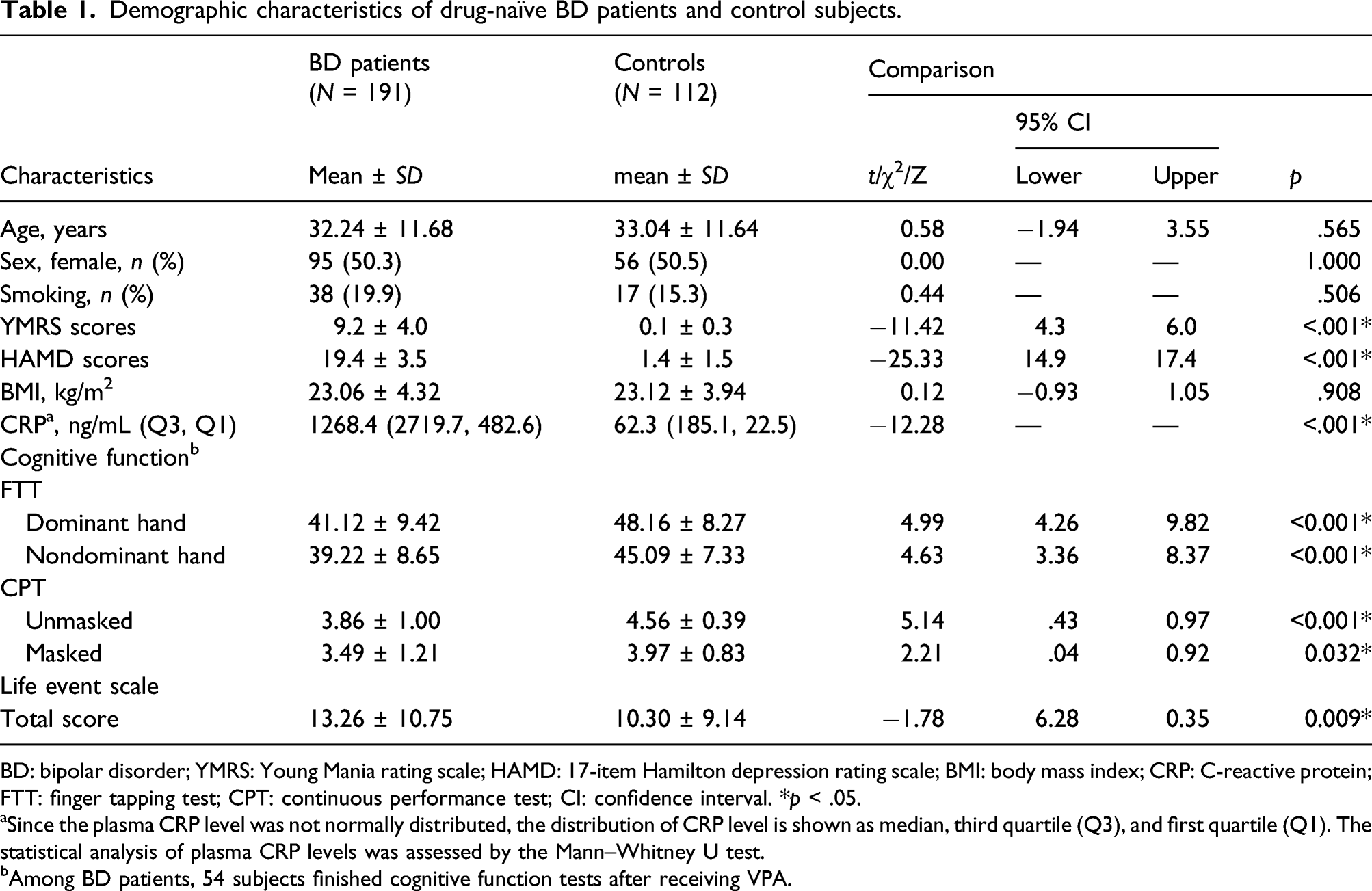
BD: bipolar disorder; YMRS: Young Mania rating scale; HAMD: 17-item Hamilton depression rating scale; BMI: body mass index; CRP: C-reactive protein; FTT: finger tapping test; CPT: continuous performance test; CI: confidence interval. *
aSince the plasma CRP level was not normally distributed, the distribution of CRP level is shown as median, third quartile (Q3), and first quartile (Q1). The statistical analysis of plasma CRP levels was assessed by the Mann–Whitney U test.
bAmong BD patients, 54 subjects finished cognitive function tests after receiving VPA.
Median plasma CRP levels were 1268.4 ng/mL and 62.3 ng/mL in BD patients and control subjects ( Differences in plasma CRP levels between drug-naïve BD patients and controls. Between-group differences in plasma CRP levels were assessed by ANCOVA after adjustment for age, sex, and BMI. (a) Plasma CRP levels in drug-naïve BD patients and controls. (b) To (e) Plasma CRP levels in drug-naïve BD patients and controls separately by genotype. *
The estimates of

CRP: C-reactive protein; SNP: single-nucleotide polymorphism; BD: bipolar disorder; CI: confidence interval; SE: standard error. *
a
Cognitive function in different genotypes of rs1800947 in BD patients.
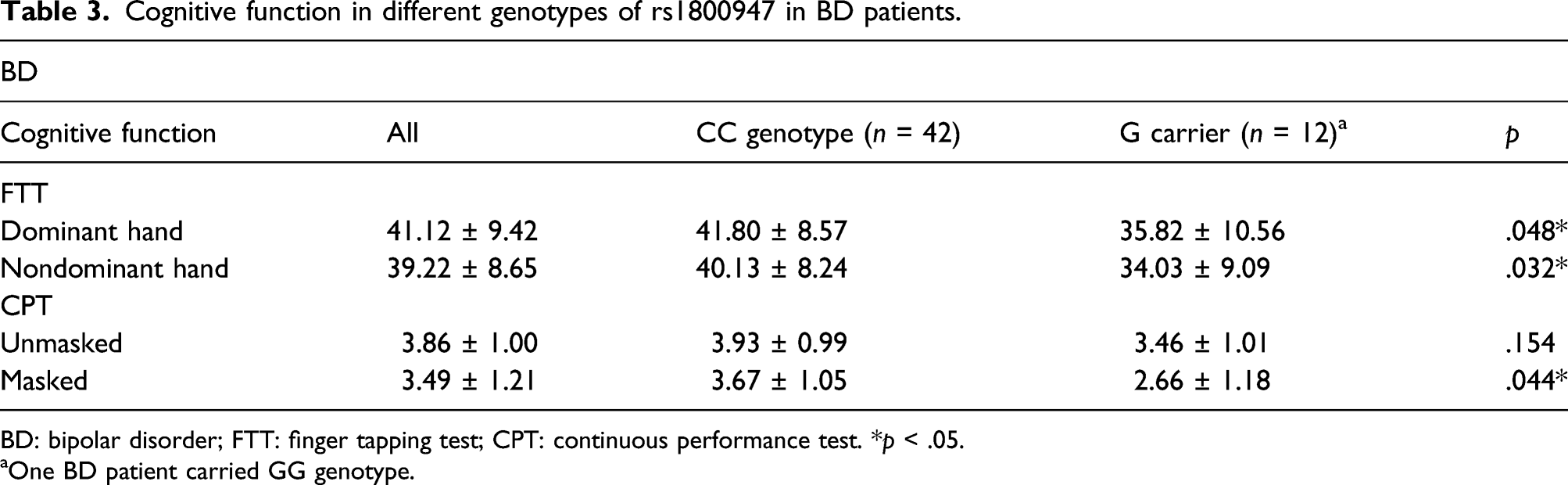
BD: bipolar disorder; FTT: finger tapping test; CPT: continuous performance test. *
aOne BD patient carried GG genotype.
Cognitive function in different genotypes of rs1130864 in BD patients.

BD: bipolar disorder; FTT: finger tapping test; CPT: continuous performance test.
aNo BD patients carried AA genotype.
Cognitive function in different genotypes of rs1205 and rs2794520 in BD patients.

The frequencies of
BD: bipolar disorder; FTT: finger tapping test; CPT: continuous performance test. *
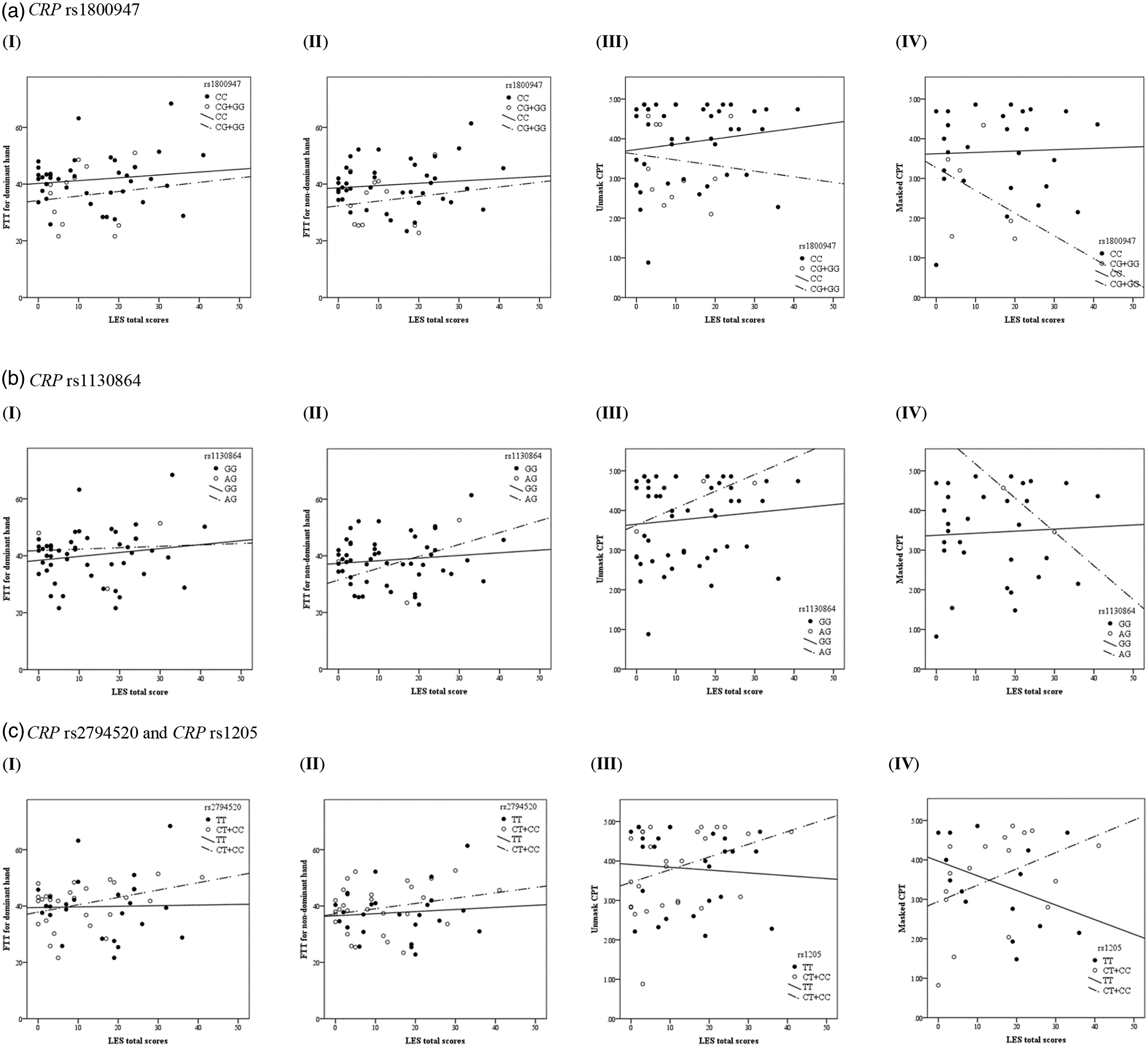
Interaction effect of
Discussion
Our aims in the current study were to conduct a genetic approach in drug-naïve depressed BD patients to clarify the association between CRP and the risk of BD and to further investigate the polymorphisms of
The causal effect of plasma CRP levels on BD risk and the frequencies of
According to the Mendelian randomization analysis, therefore, we suggested that BD might be caused by reverse causality (BD causes elevated plasma CRP levels). There were some possible contributors influencing the causal relationship. First, the reverse causality suggested that BD could cause elevated plasma CRP levels through the immune system via progressive neuropathological processes. In the CNS, alterations in the expression of tumor necrosis factor alpha (TNF-α) produced by astrocytes, microglia, and neurons have been suggested as important factors involved in synaptic transmission, synaptic plasticity, neurodevelopment, neurotoxicity, and the regulation of neuronal survival in cases of BD.46,47 The increased expression of TNF-α could shift this balance and play a role in cognitive impairment among chronic BD patients. 48 In addition, TNF-α induced the central recruitment of circulating monocytes, which produced more proinflammatory cytokines and sustained the inflammatory status, even in cases of peripheral inflammation. 49 Therefore, CRP, synthesized by hepatocytes in response to inflammatory processes, could be found at increased levels in mania/hypomania and depressive episodes and remained higher in euthymic patients than in control subjects.1,21 Second, confounding factors, such as environmental factors, life event changes, and comorbidities, could directly affect plasma CRP levels or BD. Elevated plasma CRP levels were associated with shared risk factors for BD, such as smoking, obesity, hyperlipidemia, and diabetes mellitus.1,50,51 In addition, elevated plasma CRP levels were also associated with medications for BD. Moreover, plasma CRP level could vary across mood states in BD patients,1,21,22 and CRP concentrations are higher during mania than during depression and euthymia. Third, recent studies have suggested that the hypothalamic pituitary adrenal (HPA) axis and CRP are involved in dysbiosis of the gut microbiome in psychiatric disease.52,53 The HPA axis is a central system in response to stress, regulating stress homeostasis and influencing CRP synthesis. Altering the neuroendocrine system results in a change in gut microbiota composition, disturbance of intestinal barrier function, and activation of the immune system.52,53 Proinflammatory status and low-grade inflammation in the dysfunctional intestinal mucosal barrier could also activate the HPA axis and further influence CNS function.53,54 Considering these confounding factors, we carefully recruited BD patients who were drug-naïve, in a depressed stage, and at first diagnosis of BD to exclude these possible biases. Taken together, the mutual interactions between plasma CRP and BD indicated that the inflammatory system is involved in one of the complex mechanisms of mood disorder and suggests a potential therapeutic role.
Growing evidence has shown that CRP appears to be a peripheral biomarker that reflects peripheral and central inflammation in MDD patients,55-57 although it is difficult to measure CRP levels in the CNS in clinical practice. Plasma CRP was significantly correlated with cerebrospinal fluid (CSF) CRP. 56 Plasma CRP was also correlated with plasma and CSF inflammatory cytokines, including TNF and interleukin (IL)-β. In addition, plasma CRP and CSF inflammatory markers were significantly associated with depressive symptom severity in MDD patients.56,57 Furthermore, a brain imaging study demonstrated that plasma CRP was not only highly predictive of depressive symptoms but also associated with reduced functional connectivity within a widely distributed network, such as the ventral striatum, parahippocampal gyrus/amygdala, and posterior cingulate cortex in patients with depression. 58 These alterations were centralized in the ventral medial prefrontal cortex, representing an important role for the effects of inflammation on network function in the whole brain. Therefore, peripheral CRP could be an appropriate marker with clinical utility for identifying patients with mood disorders.
Furthermore, since immune dysfunction, such as elevated plasma CRP levels, has been strongly implicated in the pathophysiology of BD, targeting the immune system is a strategy for the treatment of bipolar disorder.59,60 As such, numerous clinical trials have investigated the effects of anti-inflammatory agents in the treatment of BD.60-62 Adjunctive anti-inflammatory agents have demonstrated antidepressant effects with N-acetylcysteine (NAC), pioglitazone, minocycline, and nonsteroidal anti-inflammatory drugs in the acute phase of bipolar depression.63-68 In contrast, some studies reported that adjunctive NAC, aspirin+NAC, or infliximab did not show significant efficacy when compared with placebo in cases of acute BD depression unless subgroup analysis was performed.69-71 Adjunctive celecoxib and NAC showed potential antimanic effects,61,72 while there are limited studies concerning euthymic stage patients. 73 Overall, reports have shown that anti-inflammatory agents in the treatment of bipolar depression have moderate effects and good tolerance. 62 Moreover, regardless of illness phase, previous studies also suggested that anti-inflammatory agents are likely most beneficial in patients with mood disorder when subgrouped by immune dysregulation or by childhood maltreatment.59,71 Since low-dose memantine serves as an anti-inflammatory agent through the BDNF signaling pathway,74,75 BD patients with high initial plasma CRP levels receiving memantine adjunct treatment have a reduced risk of inflammation and metabolic imbalance. 59 Additionally, a report showed that the plasma CRP level could predict the differential responses to antidepressants, 76 and clinical trials of tumor necrosis factor antagonists and omega-3 fatty acids demonstrated the role of the initial plasma CRP level in predicting individual treatment responses in patients with major depressive disorder.77,78 Larger studies using samples stratified by inflammatory status are required in the future to examine the roles of inflammation and adjunctive immune modulating agents in the treatment outcome of BD. Taken together, plasma CRP has a role as a sensitive inflammatory marker, which could be linked not only to BD pathophysiology but also to the treatment outcome associated with BD.
Considering the stratification of the polymorphisms of the
Although the data were presented carefully, this study has certain limitations. The first limitation is the relatively small sample size of BD patients. Although the relatively small sample size may limit the interpretation of the results, the association between plasma CRP level and BD had a statistical power of 0.8. The second limitation is that the causality between plasma CRP levels and BD should be considered with caution given the potential biases and limitations of Mendelian randomization analyses, such as general health status, population stratification of genetic variations, and pleiotropy. In the current study, to decrease the bias and confounding factors, all BD patients were drug naïve and at their first diagnosis of BD. Additionally, the frequencies of the
Conclusions
In summary, our study suggested that drug-naïve, depressed BD patients who were at their first diagnosis had higher plasma CRP levels than control subjects, although the causal relationship between CRP and BD risk was not significant in the genetic analysis of Mendelian randomization. Furthermore, our study suggested that not only the genotypes of
Supplemental Material
sj-pdf-1-iji-10.1177_03946320221084835 – Supplemental Material for Roles of C-reactive protein polymorphisms and life event changes on cognitive function in bipolar patients receiving valproate
Supplemental Material, sj-pdf-1-iji-10.1177_03946320221084835 for Roles of C-reactive protein polymorphisms and life event changes on cognitive function in bipolar patients receiving valproate by Po See Chen, Li-Yi Tang and Hui Hua Chang in International Journal of Immunopathology and Pharmacology
Footnotes
Acknowledgments
The authors would like to thank Chih-Ying Lin for administrative support.
Author contributions
Conceptualization, H.H.C.; methodology, L.Y.T.; software, L.Y.T.; validation, P.S.C. and H.H.C.; formal analysis, L.Y.T.; investigation, P.S.C. and H.H.C.; resources, P.S.C. and H.H.C.; data curation, L.Y.T.; writing—original draft preparation, P.S.C. and H.H.C.; writing—review and editing, H.H.C.; supervision, H.H.C.; project administration, L.Y.T.; funding acquisition, H.H.C. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science and Technology of Taiwan [MOST 105-2320-B-006-014, MOST 106-2320-B-006-040, MOST 107-2320-B-006-071, MOST 108-2320-B-006-047-MY3] and the National Cheng Kung University Hospital [NCKUH-10301003, NCKUH-10509004, NCKUH-10802013, NCKUH-10902014].
Ethics approval
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Board for the Protection of Human Subjects at National Cheng Kung University Hospital (IRB No. A-ER-104–031).
Informed consent
The Institutional Review Board for the Protection of Human Subjects at National Cheng Kung University Hospital has approved the research protocol (IRB No. A-ER-104-031). All participants, recruited from outpatient settings at the National Cheng Kung University Hospital, provided written informed consent regarding their willingness to participate in the research.
Data availability statement
The data that support the findings of this study are available from National Cheng Kung University. Restrictions apply to the availability of these data, which were used under license for this study. The data presented in this study are available on request from the corresponding author with the permission of National Cheng Kung University.
Supplementary Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
