Abstract
Aims
Cytokines, soluble mediators of immunity, are key factors of the innate and adaptive immune system. They are secreted from and interact with various types of immune cells to manipulate host body’s immune cell physiology for a counter-attack on the foreign body. A study was designed to explore the mechanism of Toxoplasma gondii (T. gondii) resistance from host immune response.
Methods and results
The published data on aspect of host (murine and human) immune response against T. gondii was taken from Google scholar and PubMed. Most relevant literature was included in this study. The basic mechanism of immune response starts from the interactions of antigens with host immune cells to trigger the production of cytokines (pro-inflammatory and anti-inflammatory) which then act by forming a cytokinome (network of cytokine). Their secretory equilibrium is essential for endowing resistance to the host against infectious diseases, particularly toxoplasmosis. A narrow balance lying between Th1, Th2, and Th17 cytokines (as demonstrated until now) is essential for the development of resistance against T. gondii as well as for the survival of host. Excessive production of pro-inflammatory cytokines leads to tissue damage resulting in the production of anti-inflammatory cytokines which enhances the proliferation of Toxoplasma. Stress and other infectious diseases (human immunodeficiency virus (HIV)) that weaken the host immunity particularly the cellular component, make the host susceptible to toxoplasmosis especially in pregnant women.
Conclusion
The current review findings state that in vitro harvesting of IL12 from DCs, Np and MΦ upon exposure with T. gondii might be a source for therapeutic use in toxoplasmosis. Current review also suggests that therapeutic interventions leading to up-regulation/supplementation of SOCS-3, IL12, and IFNγ to the infected host could be a solution to sterile immunity against T. gondii infection. This would be of interest particularly in patients passing through immunosuppression owing to any reason like the ones receiving anti-cancer therapy, the ones undergoing immunosuppressive therapy for graft/transplantation, the ones suffering from immunodeficiency virus (HIV) or having AIDS. Another imortant suggestion is to launch the efforts for a vaccine based on GRA6Nt or other similar antigens of T. gondii as a probable tool to destroy tissue cysts.
Introduction
This table illustrates the role of specific cytokines either in resistance or susceptibility against T. gondii. The cytokines’ source/s, main function/s, synergistic, and antagonistic relations with other cytokines.

IFN (Interferon), IL (Interleukin), TNF (Tumor Necrosis Factor), TGF (Transforming Growth Factor).
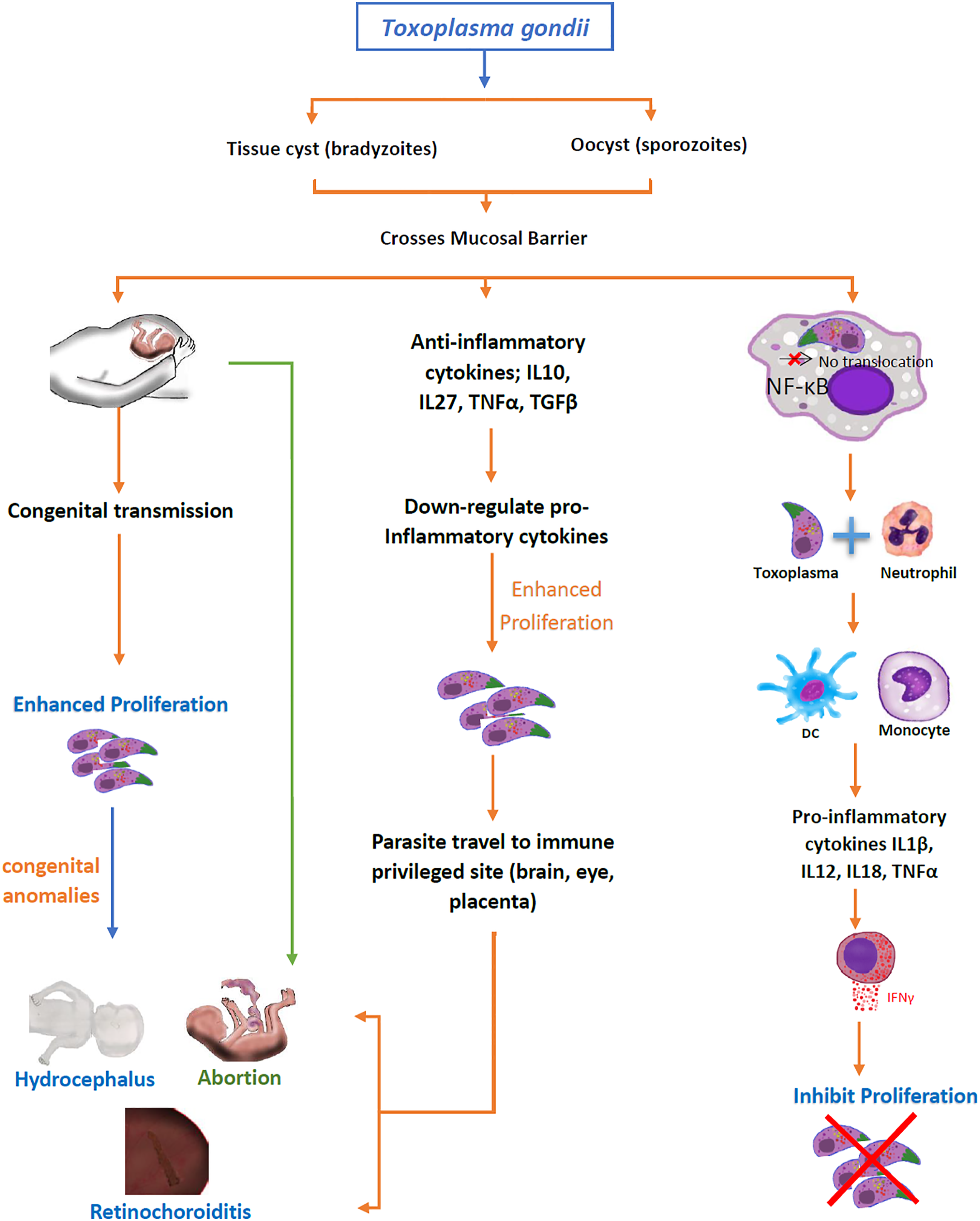
Outcome of toxoplasmosis in case of resistance and susceptibility.
Immunological studies on human infections have clearly concluded that cell-mediated immunity and IFNγ are paramount in the control of any infection particularly caused by the intracellular pathogen (T. gondii). 8 The inability of the humoral response alone (antibodies) to prevent Toxoplasma reactivation is evident by the fact that most of the HIV-infected people lack effective immunity hence exhibit symptoms of T. gondii infection and reactivation, even in the presence of high titers of specific IgG. 9
Pro-inflammatory cytokines are among the key factors to initiate and maintain innate as well as acquired immunity to restrict proliferation of Toxoplasma. A variety of cytokines are produced upon activation of APCs and cells of the adaptive immune system (B and T cells). The differences in cytokinome can be speculated at the different stages of infection, due to intra- or extra-cellular nature of pathogens as well as due to diversity of the host genetic makeup. 10 The indirect role of cytokines against T. gondii in leading to either resistance or susceptibility also depends upon the stage of parasite in the host and the induction and modulation of pro-inflammatory cytokines driven by the particular parasite strain as well as the robustness of the host’s immune profile. Different typical and atypical strains of T. gondii exist globally, and have been specifically studied in America and Europe. This parasite is identified having three distinct genotypic lineages in humans: type I strain (RH-88), type II strain (ME49, and DEG), and type III strains (CEP and VEG). 11 The genetic moieties in these strains result into a highly varied level of virulence7,12 which inflicts diverse pathological effects in host by a variety of cytokine pathways as well as owing to the wide range of interactions through a vast diversity of host and parasite molecules interacting each other, with some known and some unknown footprints. The known footprints include but not limited to inherent-oxidative stress, 13 a diversity of IRGs (Immunity Related GTPases) 12 of host with a locus on chromosome 11, 14 Z-DNA binding protein 1 (ZBP1), receptor-interacting serine/threonine-protein kinase 3 (RIPK3) of host. 15 Likewise, the wide range of parasite molecules includes a diversity of rhoptry proteins (e.g. ROP5, ROP18),12,16 dense granule proteins (e.g., GRA5, GRA12, GRA16, GRA24) 17 ; small GTPase immunity-associated proteins (GIMAPs 4, 5 & 6) 18 of T. gondii. This review describes the cytokinome (Table 1) in toxoplasmosis and their interactive role for development of host’s susceptibility and resistance toward T. gondii infection.
Database search
A search was conducted on Science Direct (https://www.sciencedirect.com/science/search) and PubMed (https://www.ncbi.nlm.nih.gov/pubmed) using “host immune response and resistance of toxoplasmosis” to achieve the relevant literature for this study. The searched period ranged from 1980 until 15 September 2021, yield 649 publications. The literature having mechanism of immune response and pathogen resistance were included in this study. Whereas for the last 5 years, the data were searched from PubMed using the same key words as mentioned above and majority of the articles with novel insights into the immune response mechanism as well as host parasite interactions were included in this review. Although maximum efforts have been done to review the literature on “host immune response and resistance of toxoplasmosis”, certainly there are limitations of this review. Hence, this should not be considered a review encompassing all the new literature on ‘Toxoplasma gondii” and “Toxoplasmosis”. This is because of the fact that 24,912 and 13,928 results are retrieved from Science Direct and PubMed, respectively, while using a key word “Toxoplasmosis” with the same time period as mentioned above. Likewise, 23,158 and 15,750 results are displayed from Science Direct and PubMed, respectively, with a key word “Toxoplasma gondii” for the same time period. Similarly, slightly modified key words, that is, “host immune response of toxoplasmosis” displays almost 7000 (6969) results in Science Direct. 1) Virulence of T. gondii strains
In humans, the regulatory cytokines profile depends on the T. gondii strain. Among three notable strains (type I, II and III) of T. gondii, the type I is more virulent as compared to type II and type III strains. In type I (RH strain) infected cells, the translocation of NF-κB does not take place resulting in the production of anti-inflammatory (IL10, IL27, and TGFβ1) cytokines which are higher as compared to uninfected cells.19–23 These anti-inflammatory cytokines enhance the proliferation of T. gondii. It was found that with a significantly high level of TGFβ1 in the blood as well as in the aqueous humor of the acutely intraocular Toxoplasma-infected host, it may adversely interfere with the effective cellular immune response leading to an increased mortality and extensive ocular tissue damage with tachyzoites observed in the pigment epithelium layers. 24 Consequently, Th2 and Treg responses are enhanced in comparison with a primary ocular infection. 25 ME49 is a type II strain that induces the translocation of NF-κB-light-chain-enhancer from the cytoplasm to the nucleus of activated B cells, splenocytes 26 and bone marrow–derived MΦ 27 which induce the production of pro-inflammatory cytokines (IL1β, IL12, IL18, TNFα, IFNγ, and IL12p40) by thioglycolated MΦ/cell lines19,28. Whereas, the production of anti-inflammatory cytokines are lower in ME49-infected cells than that of uninfected cells 21 speculating CD36-mediated engagement of low virulence strains, with macrophages. 29 Recent evidence also demonstrates the role of Toxoplasma’s parasitophorous vacuole-membrane–associate dense granule proteins in modulating parasitic virulence while interacting the host body’s resistance mechanisms like GRA12 of Toxoplasma was identified as a major virulence factor to counter the host’s IFNγ. 30
Host immune response
A) Innate immune response
In early stages of infection, dendritic cells (DC), macrophages (MΦ), natural killer (NK), and neutrophils (Np) interact in a coordinated way to provide the first line of defense in the form of innate immune response leading to develop adaptive immunity.31-33 The innate immune response is elicited against toxoplasmosis in the form of IL12 production upon interaction with antigen (Ag). The release of IL12 from MΦ, DCs, and Np is essential for the release of IFNγ from NK cells (innate immune response) and T lymphocytes (adaptive immune response) via antigen presentation.
34
IFNγ has been shown to induce guanylate binding proteins (GBPs) in a murine model of toxoplasmosis, thereby, these GBPs accumulate on the surface of intracellular parasites potentially causing parasitic destruction, thus displaying an active role of intracellular autonomous immunity.
35
The increased susceptibility toward T. gondii infection is due to the depletion of NK cells, MΦ, or DCs which have a significant involvement for innate immune response against the infection.
36
The mechanism of innate immune response initiate upon interaction of toll-like receptors (TLRs) with ligands expressed on T. gondii surface, thereby starting intracellular signaling pathways through engagement of the myeloid differentiation domain-88 (MyD88). These are the universal adaptor proteins involved in signaling of all TLRs except TLR-3. The study on MyD88 deficient mice model was impaired to induce primary protection in acute infection of T. gondii (RH strain).
37
Studies on mouse with targeted inactivation of MyD88 showed that DCs work as antigen-presenting cells (APC) and are responsible for the increased susceptibility to T. gondii infection. However, there was no effect on MΦ and Np. The MyD88 deficient mice masked the production of IL12 from DCs and IFNγ by NKs to initiate innate immune response. It explains a central role of DCs in the coordination of innate immune response against T. gondii infection and predicts the increased susceptibility towards infection if DCs are recognized defective in early encounter of T. gondii infection.
38
B) Cellular immune response
In T. gondii infection, the strong resistance to re-infection as well as the hindrance to reactivation of chronic infection is based on the host’s cell-mediated immunity.39,40 The synergistic role of CD4+ and CD8+ T cells for the development of acquired immunity was understood from targeted experiments on C57BL/6 mice vaccinated with temperature sensitive mutant strain of T. gondii (ME49).41,42 The development of complete immunity against a virulent strain (type I) is dependent on IFNγ synthesis from NK and T cells. The immunocompetent host activates either T cells or NK cells for encountering parasitic invasion. In a previous study, the MHC I (lack of CD8+ cell stimulation) impaired mice (beta 2m-deficient mice) surprisingly showed high resistance against T. gondii following vaccination. This enhanced immunological response in the absence of CD8+ cells showed the involvement of NK cells activated by IL12 upon parasitic invasion. 43
Generally, the CD8+ T cells are the major source of IFNγ production against most of the T. gondii strain.
44
The c-Rel expression regulated by NF-κB is widely dominant in hematopo
CD8+ T immune cells having a TCR Vβ8.1, 8.2+ phenotype produce protection against tissue cyst development in mouse model. Transfer of CD8+ cells induced by N-terminal of dense granule protein-6 (GRA6Nt) of parasite has been shown to clear T. gondii cysts from the brains of infected mice that were deficient in T cells (Sa et al. 49 2017), further highlighting the role of cytotoxic T cells in the induction of protective immunity against T. gondii. A genetically resistant strain of mice having a H-2d haplotype helped discover these specific type of cytotoxic T cells, whereas H-2Ld was found as a major antigen-presenting molecule to CD8+ T cells to achieve this objective of tissue cyst elimination.
The immune response of CD8+ cells is more dominant alike effector cells than CD4+ cells. Nevertheless, CD4+ helper T cells are direly required for an effective functioning of CD8+ cells.50,51 The correlation of CD4+ and CD8+ `cells proved to be the main scaffold of cytokine trafficking in mice host.
Regulatory T (T reg) cells are required for the maintenance of immunological self-tolerance and immune homeostasis by actively suppressing the pathological and physiological immune responses.52,53 An IL2 knocked-out mouse model orally infected with lethal dose of T. gondii, showed highly Th1 cell type-polarized mucosal immune response. Such effect contributed to the incapacity of T reg cells to perform effector responses and consequently led to immuno-pathogenesis.54,55 Besides this, T reg cells of infected mice expressed lower levels of Bcl-2 and increased levels of apoptotic markers than that of naive mice. It is suggested that de-regulation in the T reg cells is a consequence of these impaired cells turnover. 54 Besides this, there was found a gradual weight loss and significant delayed mortality in T reg-transferred toxoplasmosis-infected mice associated with lower level of IFNγ and TNFα. Additionally, higher cyst number and parasite load in brain of those mice were observed. 56 Furthermore, activity of T reg cells results in the death of proliferating T cells which favors the multiplication of pathogen in the murine model. 57 In a pregnant T. gondii-infected mouse model, numbers of splenic CD4+CD25+-T reg cells and placental Foxp3+ cells decreased synchronously. During infection, the reduction of splenic CD4+CD25+-T reg cells was associated with apoptosis (Bcl-2) induced by proliferating T cells. 58 Additionally, injection of pregnant mice with excretory–secretory antigens (ESA) of T. gondii also causes fetal death associated with apoptosis of CD4+CD25+ T reg cells by down-regulating their Bcl-2 expressions and Bcl-2/Bax ratio. It could be partly prevented by adoptive transfer of CD4+CD25+ T reg cells from normal to infected pregnant mice. 59
The high level of Th1 cytokines (IL2 and IFNγ) were reported to be produced by CD4+ cells upon interaction with tachyzoite. 50 It is found that MyD88 is effectively involved in the development of Th1 response. 60 Generally, the Th1 cell-mediated immunity builds the resistance against the T. gondii infection by IFNγ production via Th1 effector cells. 61 The role of Th1 cytokines (IFNγ, IL12, and TNFα) for susceptibility to toxoplasmosis has been witnessed with the absence of any of these pro-inflammatory mediators as previously studied.42,62 Moreover, other cytokines like IL2, IL6, IL7, IL15, IL18, and IL23 are also associated with the development of strong immunogenic response. 60 Recent evidence demonstrates TLR-11–independent activation of inflammasome for driving CD4+ T-cell–derived IFNγ-mediated host resistance to T. gondii. 7 GRA24-driven protective immunity mediated through p38 MAPK activation, IL12 production, and independent of MyD88 pathway has been evidenced, recently, through use of bicistronic IL12YFP reporter mice on MyD88+/+ and MyD88-/- genetic backgrounds, MyD88+/+ and MyD88-/- bone marrow–derived macrophages as well as exploiting parasites species named as uracil auxotrophic Type-I stain of T. gondii cps1-1 and cps1-1:Δgra24. 63
In contrast, various experiments have demonstrated that the cytokines involved in Th2 response also play a detrimental role for enhancing the susceptibility to T. gondii infection.
64
The modulation of Th2 response is mainly carried out by IL4 and IL10. Both cytokines increase the host susceptibility to T. gondii in early infection.
103
Nevertheless, the regulatory function of Th2 cytokines has been unveiled. The evidence from T. gondii infection (Type II stain) to the IL4-knockout mice resulted in less susceptibility to toxoplasmosis.
65
Shoot-up levels of inflammatory cytokines were detected in IL10 knockout mice causing early resistance to Toxoplasma.64,66 C) Cytokines and other inflammatory mediators playing a role against T. gondii
Moreover, the gp130 transducing component has common structural features to IL6 and IL27.
116
This IL27 is critically involved in curtailing the infection induced inflammatory pathology and functions antagonistically to IL6. The mice lacking gp130 signal transducer (gp130 Y757F mice) upon infection with T. gondii (ME49), showed high parasite burdens and increased mortality having low IL12 and IFNγ titer.
117
IL6 is a cytokine that more pronouncedly functions to resist the Toxoplasma-induced encephalitis in murine model.
118
Previously, the protective role of IL6 was questioned in various studies that illustrated the role of IL6 in increased intracellular multiplication of T. gondii.
110
Later studies defined the critical role of IL6 in progression of T. gondii infection as the IL6-deficient mice rapidly switched to severe states of toxoplasmosis such as Toxoplasma encephalitis
119
(Figure 2). Pathways for host susceptibility and resistance in human Toxoplasmosis, particularly as a result of IL6.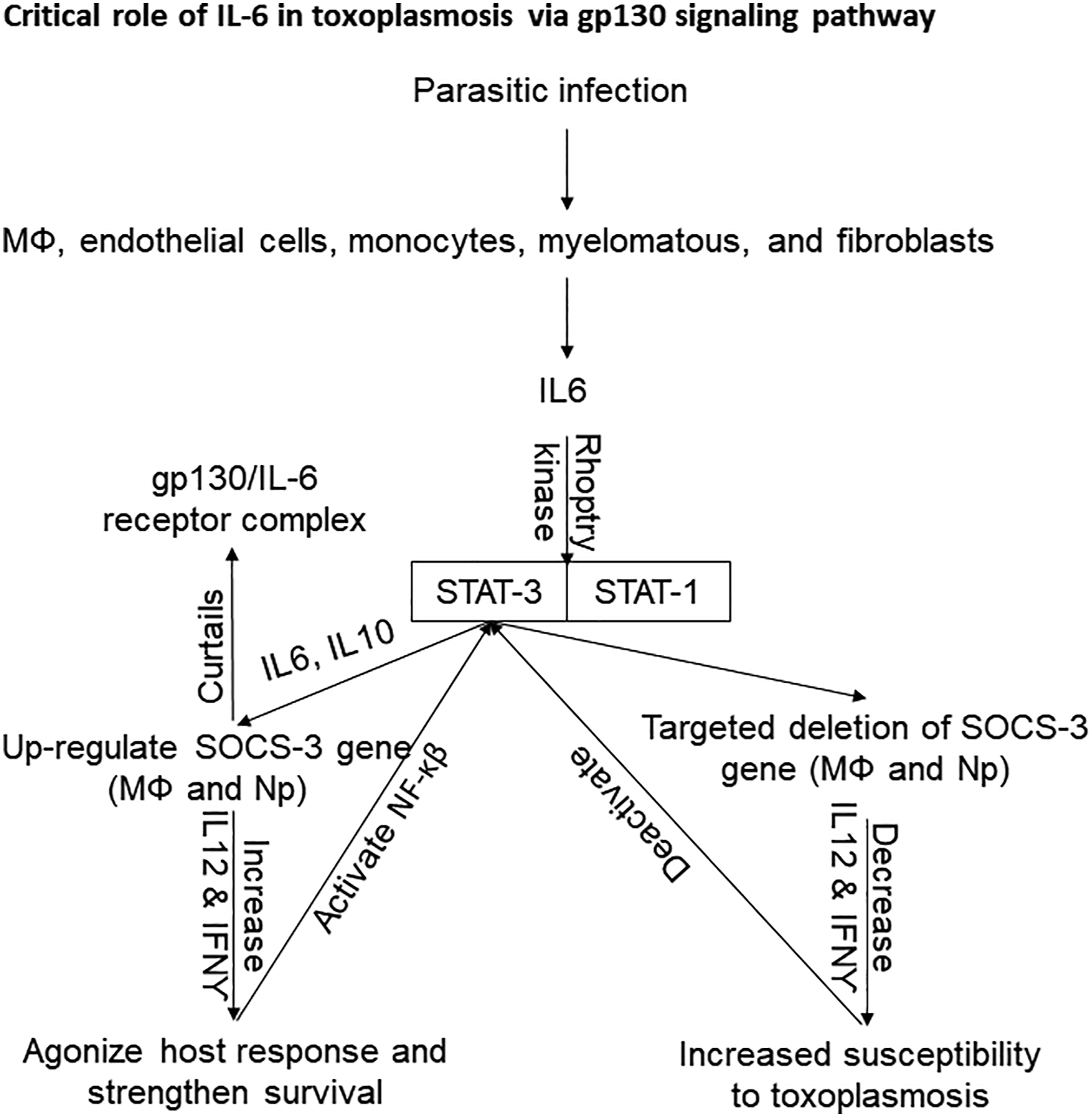
In spite of gastrointestinal sites, TGFβ also has a dominant role as an anti-inflammatory agent in brain and eyes. 188 TGFβ signals the spleen cells to secrete anti-inflammatory cytokine such as IL10 that is synergistically involved in checking the pro-inflammatory secretions from NK cells and CD4+ T cells in these immune-privileged sites (eyes, brain, and placenta). 189 The production of IL6 by innate immune response functions antagonistically to TGFβ and suspends its protective role for immune privileged sites (eyes and brain) susceptible to hyper-inflammation. 190
2) Toxoplasmosis and pregnancy
Toxoplasmosis is more important in pregnant women and immune compromised patients with respect to abortion, hydrocephalus, and retinochoroiditis. In pregnancy, Th2 immune response becomes activated which favors the proliferation of Toxoplasma. Briefly, in acute phase of T. gondii infection, certain cytokines (TGFβ1, TNFα, IL4, IL5 IL7, IL10, and IL17A) and chemokines (CXC, C, and CX3C families) play an important role as protective immune response.191,192 These pro-inflammatory cytokines down-regulate anti-inflammatory cytokines which travel to immune-privileged sites (brain, eyes, and placenta) to favor the existence of corpus luteum in the presence of low progesterone and 17β estradiol in pregnant women.
23
Apoptosis of placental cells may end up in fetal resorption, congenital anomalies (hydrocephalus and retinochoroiditis), or abortion
191
(Figure 1). Briefly, toxoplasmosis with lymphadenitis has been reported with higher levels of chemokines (CXCL8/IL8, CXCL9, and CXCL10) in pregnant women. Additionally, levels of VCAM1, CCL2, and CCL5 are lower in pregnant than in non-pregnant women.
193
The levels of ICAM1, CXCL9, CXCL10, MCSF, and TNFβ were up-regulated in acutely toxoplasmosis-infected Colombian pregnant women. Whereas, the levels of Eotaxin (Et), TGFβ, TNFα, IFNγ, IL2, IL4, IL15, CXCL1, and stem cell factor (SCF) were down-regulated in pregnant American acute cohorts.
194
In congenital toxoplasmosis, it was found that serum levels of IFNγ and IL5 were greatly increased during active stage of retinochoroiditis. In contrast, IL10 production was low during inflammatory stage and significantly higher in patients with inactive lesions.
195
The cytokine profile of acute toxoplasmosis-infected patients varies with geographical localities. 4) Perspectives for immunomodulation, therapy, vaccine, and other anti-parasitic challenges
The exploration of deep knowledge on the role of cytokines in toxoplasmosis should open new avenues for therapeutic measures based on immunomodulation. For instance, the use of IL17A antagonist inhibited the ocular toxoplasmosis in European patients. 196 Similarly, inhibition of parasite kinases in South American toxoplasmosis patients enhances the expression of IFNγ.163,197,198 This difference in inhibition sites might be strain dependent. Recent analysis of the cytokines profile in congenital toxoplasmosis199,200 indicates that modulation of cytokines through immuno-modulatory peptides could be assayed as immune adjuvants. 201 Such approaches need to be explored for the control of toxoplasmosis in humans. Etanercept (a soluble TNF-receptor fusion protein), widely used to treat autoimmune disease, activates the conversion of bradyzoites (chronic toxoplasmosis) to tachyzoites (acute toxoplasmosis) through down-regulation of pro-inflammatory cytokines (TNF, IL-1beta, and IL6). 202 It would be interesting to try to achieve a sterile immunity in an experimental model of chronic toxoplasmosis, at first transforming bradyzoites to tachyzoites through use of Etanercept but not too long after this, treating the tachyzoites to eliminate the parasite from the host body.
A recently identified drug target for T. gondii is an endonuclease named as cleavage and polyadenylation specificity factor subunit-3 (CPSF3) that has a role in mRNA processing in eukaryotes. This has been demonstrated by strong in vitro anti-parasitic activity by use of benzoxaborole (AN3661), a drug molecule that targets wild-type CPSF3. The parasites that were found resistant to this drug molecule displayed mutations in the TgCPSF3. Recapitulation of the similar resistant phenotype of the parasite through generation of mutations in the wild-type CPSF3 while exploiting CRISPR/Cas9, further strengthened the importance of this new therapeutic target against T. gondii. 203
One of the most exciting areas of research is to explore the means and effects of intervention strategies on how various strains of T. gondii can modulate host’s transcriptome 204 and non-coding RNAs including mircoRNA and long non-coding RNA. 205 Similarly, exploring how T. gondii exploits exosomes in modulating host immune response 206 as well as how therapeutic interventions designed for heme-deficient conditions affect infection outcome, 207 remains interesting areas of research.
Given the assumed fact that around one third population of world is harboring Toxoplasma in chronic form, that is, tissue cysts, why not to plan a vaccine to eliminate the tissue cysts from human population and other hosts seropositive to this infection, with a vaccine (based on GRA6Nt or other similar antigens) 49 that should be capable of eliminating tissue cysts.
Conclusions
Different factors are responsible for the pathogenesis of Toxoplasmosis and the survival of host. These factors include versatile genetic makeup of different strains of T. gondii, complicated immunological background of hosts, biochemical interaction among certain cytokines, invasion strategies of parasite as well as the immunogenicity of antigens encountered with host’s immune cells. The type of cytokines production depends on the strain of Toxoplasma. The IL10 and TGFβ1 production were higher in type I strain and lower in type II and III strain of toxoplasmosis. The production of IL12 was higher upon exposure of pathogen to DCs, MΦ, Np, NK cells, and T cells which is essential for the release of IFNγ. The production of IL12 switches the NK cells for release of IFNγ which develops resistance against T. gondii infection in host. Impairment in the production of IL12 may lead to demolish IFNγ resulting to develop host sensitivity for T. gondii infection. Moreover, IL6 also has critical role for gp130 signaling pathway for the up- and down-regulation of SOCS-gene which is responsible for the susceptibility and resistance of toxoplasmosis (Figure 2). The basic switching of pro- and anti-inflammatory cytokines in acute and chronic phases of toxoplasmosis is direly required for understanding the development of disease. Such cytokines are involved in the development of resistance and susceptibility of Toxoplasma in host. Agonist and antagonist effect of host cytokines network leads to the chronic condition of disease. In vivo up- and down-regulation of desired cytokines (IFNγ, IL6, IL12, and SOCS-3) could be helpful to boost up the immune response of host for the control of toxoplasmosis. Moreover, the synergistic and antagonistic relations among cytokines need to be comprehended on molecular and biochemical basis. The most compelling results are related with a Th2-deviated response associated to virulent strains in South American patients. Type II strain has the ability to translocate NF-κB in the nucleus of mouse splenocytes and bone marrow–derived MΦ. It is the reason that Toxoplasma type I strain survives from host immune response rather than type II and III but the complete defeat of host’s immune response is not in the favor of parasite’s survival in the ecosystem. The survival of the host after entry of T. gondii, is essential for ensuring existence of both the host and the parasite as if parasite defeats the host’s immune response, it not only marks the death of the host but also of the parasite as parasite needs a viable host to ensure its own survival as well as for its transmission to next generations of the same host as well as to other host species. The current review findings state that in vitro harvesting of IL12 from DCs, Np and MΦ upon exposure with T. gondii might be a source for therapeutic use in toxoplasmosis. Current review suggests that therapeutic interventions leading to up-regulation/supplementation of SOCS-3, IL12, and IFNγ to the infected host could be a solution to sterile immunity against T. gondii infection. This would be of interest particularly in patients passing through immunosuppression owing to any reason like the ones receiving anti-cancer therapy, the ones undergoing immunosuppressive therapy for graft/transplantation, the ones suffering from immunodeficiency virus (HIV) or having AIDS.
Footnotes
Acknowledgements
N. Cardona and M. Murillo, Centro de Investigaciones Biomedicas, Universidad del Quindio, Avenida Bolivar 12N, Armenia (Q), Colombia, South America helped to add some important studies in the manuscript. Mr. Adeel Mumtaz Abbasi (MPhil Student) is acknowledged for drawing of figures of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
