Abstract
Background
Diffuse large B-cell lymphoma (DLBCL) is the most common subtype of non-Hodgkin lymphoma. The aim of this study was to comprehensively analyze the clinical, cytomorphological, and flow cytometry characteristics of DLBCL patients, with a focus on bone marrow involvement (BMI), to identify novel prognostic factors.
Methods
The clinical, cytomorphological, and flow cytometry characteristics of 220 DLBCL patients were collected from January 2017 to April 2024. Univariate and multivariate Cox regression analyses were performed to identify prognostic factors for overall survival (OS). Kaplan–Meier survival curves were generated. Logistic regression analyses were used to explore associations between clinical, cytomorphologic, and immunophenotyping features.
Results
The median age of the patients was 63 years, with 48.6% showing positive BMI. Multivariate analysis confirmed that age, lactate dehydrogenase (LDH) level, hemoglobin (HGB) level, and platelet count (PLT) were independent prognostic factors for OS. Compared with patients without BMI, patients with BMI presented significant differences in laboratory parameters, such as lower lymphocyte counts and elevated inflammatory marker levels. Cytomorphological analysis of bone marrow smears revealed associations between specific cell characteristics (e.g., large cell size, medium cytoplasmic volume, and pseudopod protrusions) and immunophenotypic markers (e.g., CD23, CD79b, and cKappa).
Conclusion
Age, LDH, HGB, and PLT were identified as independent prognostic factors for OS. The integration of cytomorphological and flow cytometry data may provide additional insights into disease biology. Furthermore, this study highlighted that DLBCL patients with BMI have lower lymphocyte, PLT, and elevated levels of markers such as LDH, high-sensitivity C-reactive protein and β2-microglobulin.
Keywords
Introduction
Diffuse large B-cell lymphoma (DLBCL) is the most common subtype of non-Hodgkin lymphoma (NHL), accounting for approximately 30–40% of all cases. 1 Despite advances in treatment strategies, such as the introduction of rituximab-based immunochemotherapy, a significant proportion of patients still relapse or have refractory disease, highlighting the need for better prognostic biomarkers or therapeutic targets. 2 The International Prognostic Index (IPI) and its revised version (R-IPI) have been widely used to stratify patients into different risk groups. However, these indices do not fully capture the biological heterogeneity of DLBCL, which is crucial for tailoring individualized treatment plans.3,4
Bone marrow involvement (BMI) in DLBCL has been increasingly recognized as a critical factor associated with poorer outcomes, reflecting more aggressive disease biology. 5 Additionally, the cytomorphological and immunophenotypic characteristics of DLBCL cells in the bone marrow may provide further insights into the biology and prognosis of the disease.6,7 Recent studies have suggested that the integration of cytomorphological and immunophenotypic data from bone marrow samples could enhance our understanding of the biological complexity of DLBCL and improve risk stratification. 8 However, comprehensive analyses that combine clinical, cytomorphological, and immunophenotypic features in DLBCL patients according to BMI are still limited. Such analyses could identify novel prognostic markers and guide more effective treatment strategies for high-risk patients.
In this retrospective study, we aimed to comprehensively analyze the clinical, hematological, cytomorphological, and flow cytometry characteristics of DLBCL patients, with a particular focus on BMI. By integrating these diverse variables, we sought to identify novel prognostic factors associated with BMI in DLBCL patients. Our findings contribute to the growing body of evidence supporting the use of multimodal approaches for risk stratification in DLBCL patients.
Materials and methods
Patients
A retrospective single institution cohort design was used, and the clinical data of patients were collected through the hospital information system. From January 1, 2017 to April 30, 2024, patients who were diagnosed with DLBCL according to the 2008 World Health Organization classification criteria based on pathology, who had not previously received any antitumor therapy, had no history of other malignant tumors or hematological diseases, and had complete clinical information and follow-up data were included. The Ethics Committee of Zhejiang Cancer Hospital approved this study (IRB-2024-552), and written informed consent was obtained from all the study participants. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional committee and with the Declaration of Helsinki. A total of 220 patients were enrolled. The flow chart for the inclusion of patients is shown in Figure 1. The data were assessed starting from June 1, 2024. The latest follow-up visit was on September 1, 2024. Overall survival (OS) was calculated from the date of diagnosis to the date of death.
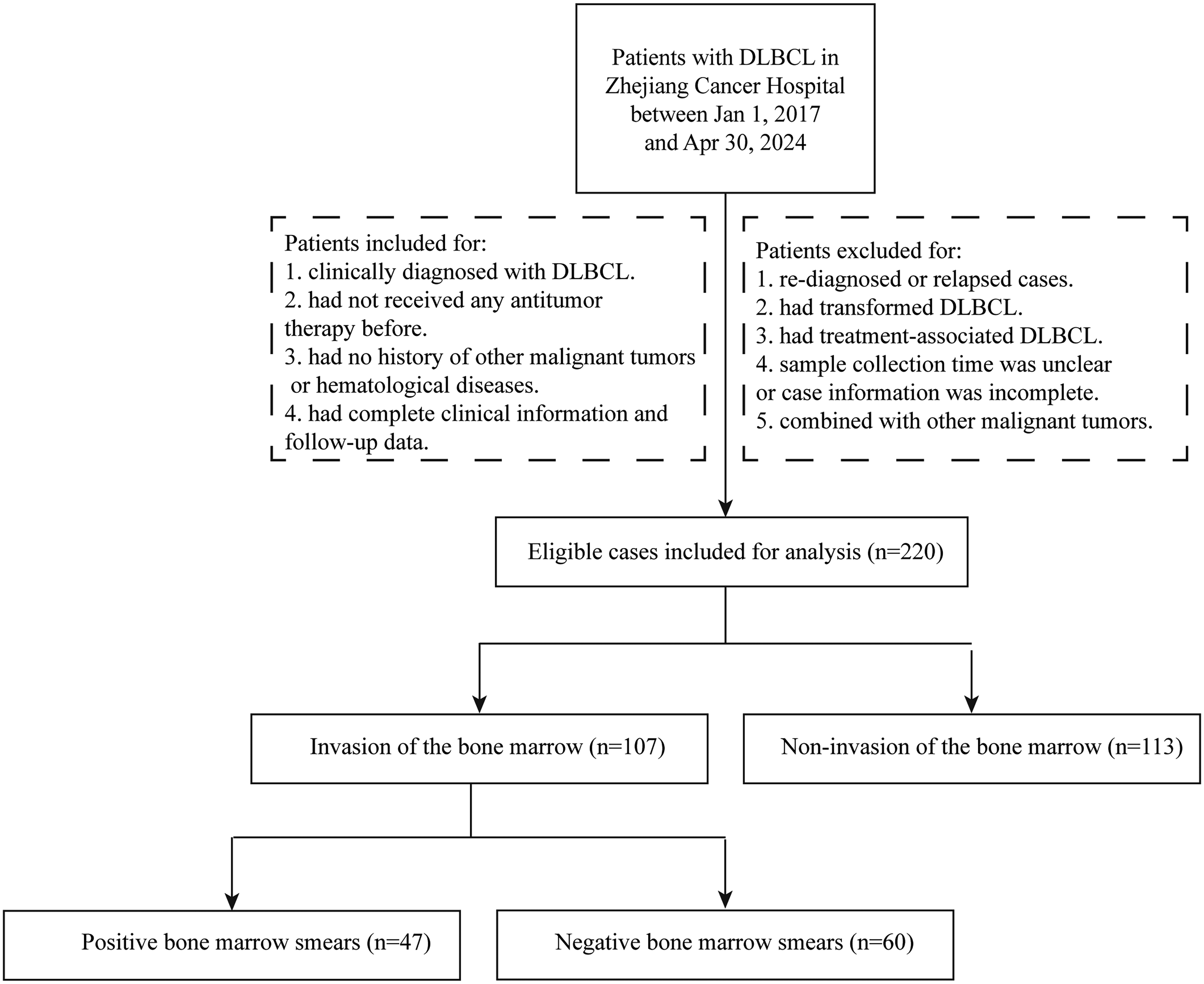
Flow chart for the inclusion of patients.
Data on the Ann Arbor stage, germinal center B cells (GCB), BMI, IPI, liver and spleen enlargement, number of extranodal sites, and routine laboratory tests were retrieved from patient charts. The cell-of-origin classification into GCB and non-GCB subtypes was determined using immunohistochemistry according to the Hans algorithm. The results of the bone marrow biopsies and the morphological characteristics of the DLBCL cells in the bone marrow smears were also collected. All the cytomorphological interpretations were independently completed by three experienced morphological experts. Cytomorphological characteristics included cell size, cell cytoplasmic volume, pseudopod protrusions, cytoplasmic vacuoles, chromatin, perinuclear faintly stained areas, nucleolar size, nucleolar number, nucleus number, tumor cell aggregation, phagocytosis, pathological divisional signs, etc. Cytomorphological characteristics of DLBCL were identified as normal, naïve, and mature lymphocytes. In addition, DLBCL cells in the bone marrow were analyzed by flow cytometry for the expression of CD5, CD10, CD19, CD20, CD22, CD23, CD38, CD79b, CD200, ki-67, FMC7, Kappa and Lambda.
Statistics
Statistical analysis was performed via SPSS 27.0 (SPSS Inc., Chicago, IL, USA) software. The laboratory parameter results were analyzed via receiver operating characteristic (ROC) curves to calculate cutoff values. The Cox proportional hazards regression model was applied to evaluate the hazard ratio (HR) for univariate and multivariate analyses. Multivariate analysis employed the stepwise forward (likelihood ratio) method. The Kaplan–Meier (KM) method was used to plot survival curves of the laboratory parameters. Comparisons of categorical or continuous variables between groups with and without BMI were performed via the chi-square test and Mann–Whitney U test. Correlations between the clinical characteristics, flow cytometry parameters, and cytomorphological characteristics of DLBCL cells were analyzed via binary or ordered multiclassified logistic regression.
Results
Baseline characteristics
The median age of the patients was 63 years (range 21–88 years). A total of 101 (45.9%) were male, and 119 (54.1%) were female. Among them, 133 (60.5%) patients were stage III/IV Ann Arbor, 63 (28.6%) were GCB type, 107 (48.6%) had positive BMI, 86 (39.1%) had an IPI ≥2, and 50 (22.7%) had systemic B symptoms (fever, night sweats, weight loss) at the time of diagnosis. Fifty-four patients (24.5%) had ≥2 extranodal sites. The clinical characteristics of the patients are shown in Table 1.
The clinical characteristics of 220 patients with diffuse large B-cell lymphoma (DLBCL).
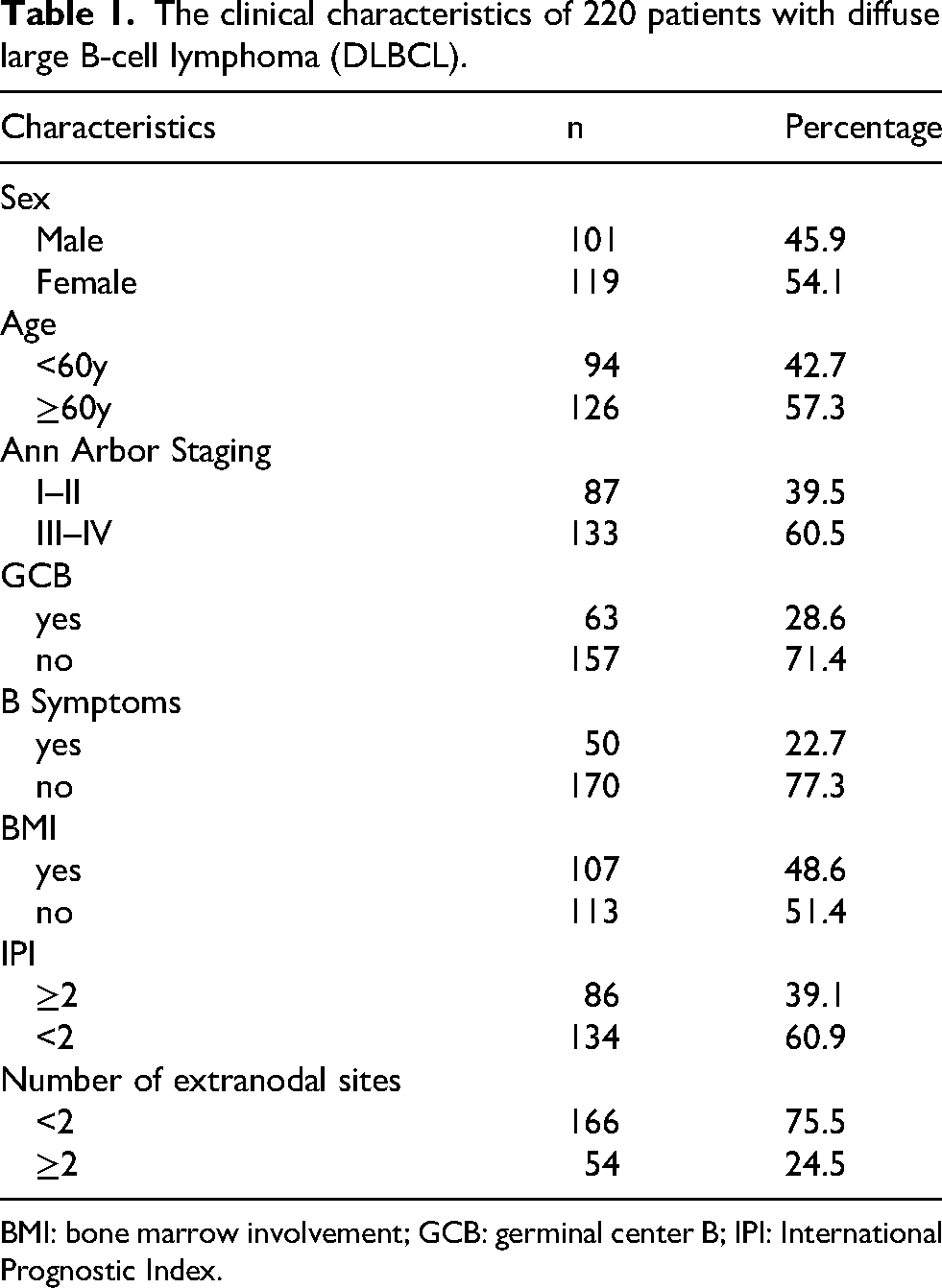
BMI: bone marrow involvement; GCB: germinal center B; IPI: International Prognostic Index.
Survival analysis
The optimal cutoff values for hematological parameters were determined according to the ROC curve (Supplementary Figure 1). The KM survival curves revealed that patients whose disease is associated with a poorer prognosis had the following laboratory parameter values: lactate dehydrogenase (LDH) ≥ 264.5 U/L, platelet count (PLT) < 126.5 × 109/L, hemoglobin (HGB) < 122.5 g/L, red blood cell count (RBC) < 3.905 × 1012/L, lymphocyte percentage (LYM%) < 23.05%, monocyte percentage (MONO%) ≧9.85%, monocyte count (MONO) ≥ 0.453 ×109/L, total protein (TP) < 67 g/L, albumin (ALB) < 39.65 g/L, albumin/globulin ratio (A/G) < 1.325, lactate dehydrogenase/albumin ratio (LAR) ≥ 6.755, creatinine (Cr) ≥ 65.15 μmol/L, uric acid (UA) ≥ 307.5 μmol/L, high-sensitivity C-reactive protein (hs-CRP) ≥ 12.685 mg/L, β2-microglobulin (β2-MG) ≥ 2898 μg/L, serum amyloid A (SAA) ≥ 22.5 mg/L, and D-dimer (DD) ≥ 550 ng/mL (Supplementary Figure 2). Univariable analysis and multivariate Cox regression analysis revealed that age (P = 0.001), LDH (P = 0.005), HGB (P = 0.017), and the PLT (P = 0.019) were independent factors for OS (Supplementary Table 1).
Comparison between groups with and without bone marrow invasion
There were statistically significant differences in age and stage (P < 0.05) but not sex or GCB status between the BMI and noninvolvement groups (data not shown). Compared with those in the nonBMI group, the LYM%, LYM, PLT, TP, ALB, and A/G levels were lower and the MONO%, LDH, hs-CRP, blood urea nitrogen (BUN), Cr, β2-MG, SAA, and DD levels were higher in the BMI group (P < 0.05) (Supplementary Table 2).
Cytomorphological features of DLBCL identified via bone marrow smears
The bone marrow smears of 47 DLBCL patients were available for analysis. The cytomorphologic features are shown in Figure 2. There were 25 patients (53.19%) who had medium-sized cells with a variety of cellular morphologies, which were rounded, round-like, or irregular; 21 (44.68%) had a medium cytoplasmic volume; 31 (65.95%) had pseudopod or protrusions; 22 (46.81%) had vacuoles; and 6 (12.76%) had perinuclear light-stained areas. The nuclei were diverse in morphology, including round shaped, round-like, or irregular, and 44 cases (93.62%) had a cell nucleus count of 1. The nuclei were mainly small and medium in size, and 20 patients (42.55%) had 1–2 nuclei; 32 (68.08%) had meticulous and sparse chromatin; 12 (25.53%) presented phagocytosis; 11 (23.40%) had tumor cell aggregation; and 17 (36.17%) had pathologic split images.
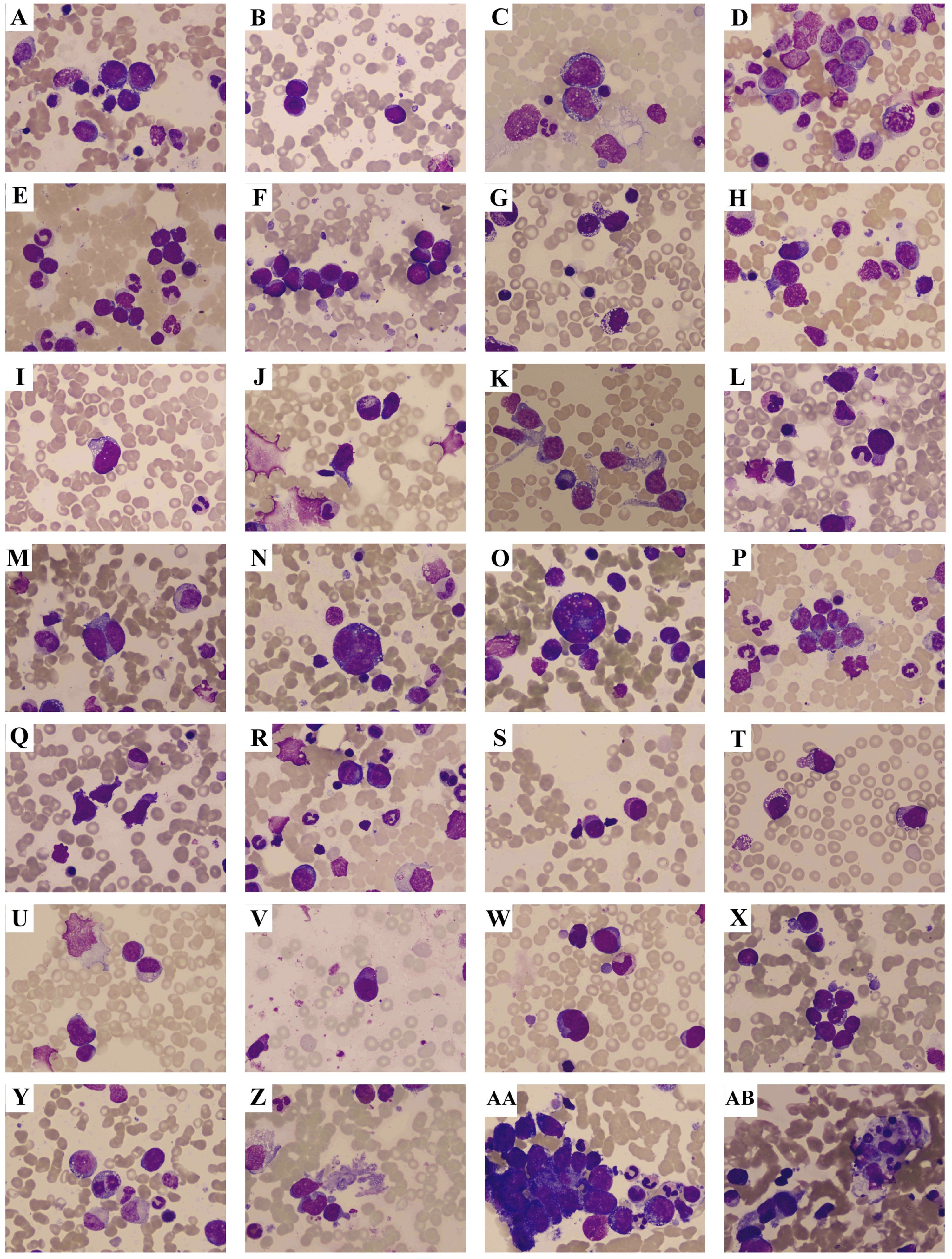
Bone marrow smears in diffuse large B-cell lymphoma (DLBCL) patients with bone marrow involvement (BMI). (a) Tumor cells of different sizes. (b) Medium-sized tumor cells. (c) Large tumor cells. (d) Abundant cytoplasmic volume. (e) Low cytoplasmic volume. (f) Medium cytoplasmic volume. (g) Large number of vacuoles. (h) Few vacuoles. (i) Medium volume vacuoles. (j) Only one cell with visible protrusion. (k) All cells appear as protrusions. (l) Some cells appear as protrusions. (m) 2 cell nucleus counts. (n) 3 cell nucleus counts. (o) 5 cell nucleus counts. (p) Irregularly shaped nuclei. (q) Pike shaped nuclei. (r) Rounded nuclei. (s) 1–2 nucleoli. (t) 3 or more nucleoli. (u) Without nucleoli. (v) Large nucleolus. (w) Small nucleolus. (x) Medium-sized nucleolus. (y) Pathologic split image. (z) Cytoplasm. (aa) Tumor cell aggregation. (ab) Hemophagocytosis.
Correlations between the cytomorphological features of DLBCL cells in bone marrow smears and the clinical characteristics or flow cytometry results
The dependent variables included DLBCL cytomorphologic features in bone marrow smears, and the independent variables included age, sex, stage, hepatosplenomegaly, number of extranodal organs, and the presence of DLBCL cells expressing CD5, CD10, CD19, CD20, CD22, CD23, CD38, CD79b, CD200, Ki-67, FMC7, Lambda, and kappa, which were analyzed via logistic regression (Table 2).
Correlations between diffuse large B-cell lymphoma (DLBCL) cytomorphologic features in bone marrow smears with clinical characteristics or flow cytometry results.
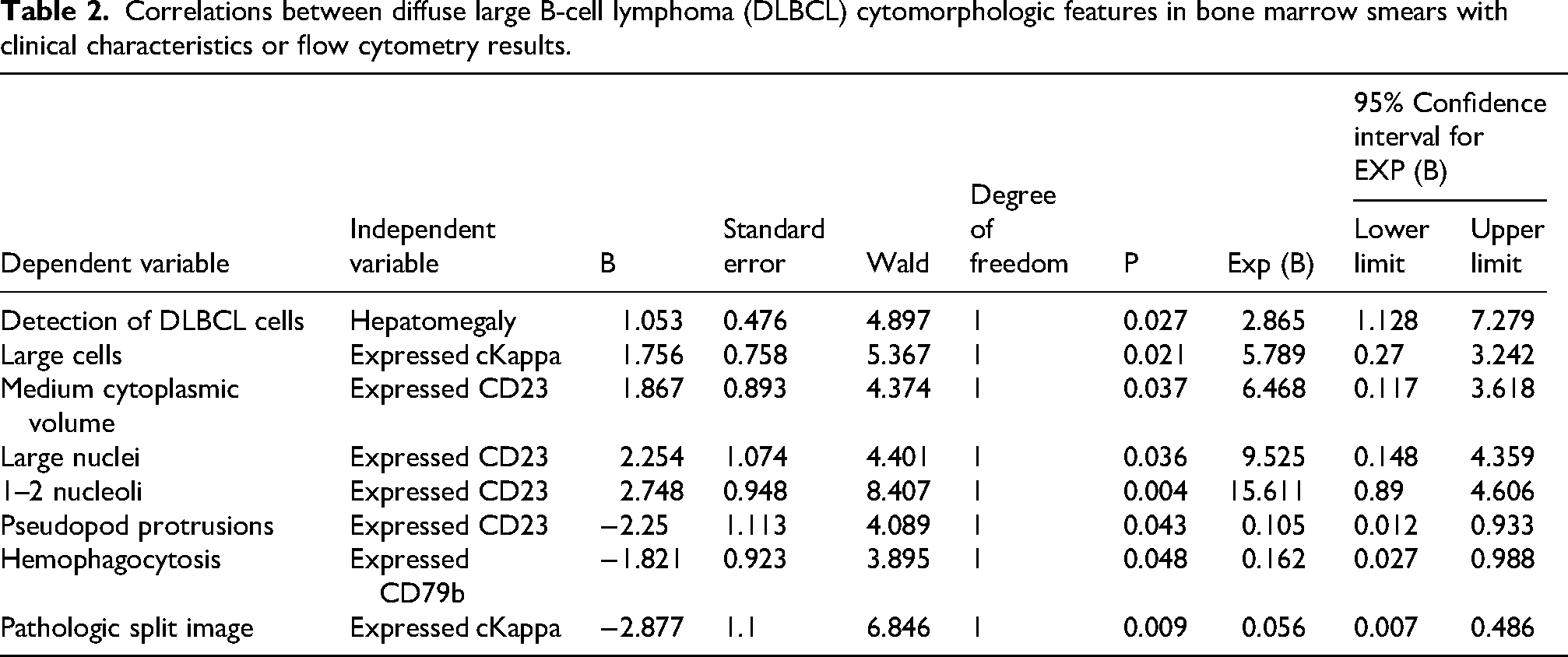
The dependent variables were set as follows: tumor cell size (1 = large, 2 = medium, 3 = small), cytoplasmic volume (1 = little, 2 = medium, 3 = large), cytoplasmic vacuoles (1 = yes, 2 = no), pseudopod or protrusions (1 = yes, 2 = no), nucleolus size (1 = none or small, 2 = medium, 3 = large), the number of nucleoli (1 = none, 2 = 1–2, 3 = 3), the presence of plasmodesmata (1 = yes, 2 = no), the occurrence of phagocytosis (1 = yes, 2 = no), and the occurrence of a pathologic split image (1 = yes, 2 = no).
The results revealed that the detection of DLBCL cells in bone marrow smears was associated with hepatomegaly. Large cells were associated with cKappa expression. Medium cytoplasmic volume, large nuclei, 1–2 nucleoli, and pseudopod protrusions were associated with CD23 expression. The occurrence of phagocytosis was associated with CD79b expression. Pathologic split images were associated with cKappa expression.
Discussion
Our study provides a comprehensive analysis of the clinical characteristics, biochemical parameters, and cytomorphology of BMI in patients with DLBCL. We found that age, blood LDH and HGB levels, and the PLT were independent prognostic factors for OS in DLBCL patients. These findings are consistent with previous studies that identified these factors as important predictors of survival in patients with DLBCL.9-12 LDH is an intracellular enzyme that can convert pyruvate into lactate in the tumor cell cytosol via the Warburg effect. 13 Both the IPI and the National Comprehensive Cancer Network (NCCN)-IPI have identified LDH levels above the upper limit of normal as key factors for predicting the outcomes of patients. Notably, in the assessment of the treatment efficacy of targeted agents such as tisagenlecleucel and glofitamab, elevated LDH is also the most robust predictor of inferior outcomes.14,15
BMI is associated with poorer outcomes and may reflect more aggressive disease.16,17 The combination of flow cytometry and bone marrow biopsy analysis is a relevant method for detecting DLBCL bone marrow infiltration. 18 Our analysis revealed significant differences in laboratory parameters between patients with and without BMI, including lower LYM% and elevated inflammatory markers such as hs-CRP and β2-MG. These findings suggest that BMI is associated with systemic inflammation and immune dysregulation, which may contribute to the poor prognosis observed in these patients. 19
The immunophenotypic profile of DLBCL cells further provides insights into the biological behavior of the disease.20,21 The expression of specific markers, such as CD5, CD10, CD19, CD20, and CD79b, has been linked to distinct molecular subtypes of DLBCL, such as the GCB and ABC subtypes, which have different clinical outcomes.22,23 However, the relationships between these immunophenotypic markers and cytomorphological characteristics in bone marrow smears have not been extensively studied. Here, we found that the cytomorphological characteristics of DLBCL cells in bone marrow smears, such as their cytoplasmic volume, hemophagocytosis, and pseudopod protrusions, were associated with specific immunophenotypic markers, such as CD23 and CD79b. These associations provide valuable information for understanding the biological behavior of DLBCL cells and may help explain the aggressive clinical course observed in patients with BMI. The integration of cytomorphological and flow cytometry data represents a novel approach to risk stratification in DLBCL, offering a more comprehensive understanding of the disease's biological heterogeneity. Our findings suggest that large cell size, medium cytoplasmic volume, and pseudopod protrusions may serve as morphological indicators of aggressive disease. Additionally, the associations between hemophagocytosis and CD79b expression, as well as between pathologic split images and cKappa expression, highlight the potential role of these features in disease progression and response to therapy, which warrants further investigation.
In addition, we included biochemical parameters such as BUN, fibrinogen, β2-MG, SAA, Cr, and UA in our analysis to identify potential prognostic factors for DLBCL. Although these markers are currently not the standard prognostic indices for DLBCL, previous studies have suggested their potential value in lymphoma prognosis assessment. For example, earlier research has demonstrated that elevated plasma fibrinogen levels are significantly associated with advanced disease stage, the presence of B symptoms, and elevated serum LDH and β2-MG levels at diagnosis in DLBCL patients. High fibrinogen levels also appear to be independent predictors of both OS and disease-free survival in DLBCL patients.24,25 β2-MG could improve the discriminatory performance of the NCCN-IPI in a retrospective study. 26 Additionally, SAA and misfolded transthyretin protein have been proposed as potential predictive biomarkers for relapsed/refractory DLBCL. 27 However, these variables were not independent factors for OS in our study. These findings warrant further validation in future prospective studies.
Conclusion
The limitation of our study is the lack of molecular investigations, such as FISH or NGS. However, we identified age, LDH levels, HGB levels, and the PLT as independent prognostic factors for OS. We also demonstrated that the morphological and immunophenotypic characteristics of DLBCL cells in the bone marrow may provide additional insights into disease biology and prognosis. These findings underscore the need for further research to better understand the biological mechanisms underlying BMI in DLBCL patients and to develop more effective treatment strategies for high-risk patients.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155251371061 - Supplemental material for Prognostic value of clinicopathological features of diffuse large B-cell lymphoma with bone marrow involvement
Supplemental material, sj-docx-1-jbm-10.1177_03936155251371061 for Prognostic value of clinicopathological features of diffuse large B-cell lymphoma with bone marrow involvement by Yuan Jin, Xiaofang Yao, Yahui Zhou, Linmei Zhong, Yi Yao, Jie Zhou, Zhiyu Wang, Yejiang Bao and Xu Qian in The International Journal of Biological Markers
Footnotes
Author contributions
X.Q. conceived and designed this study. J.Y., X.Y., Y.Z., L.Z., Y.Y., J.Z., Z.W. performed the experiments. X.Q. drafted the manuscript. X.Q. and Y.B. revised the manuscript. All authors approved the final manuscript. Y.J., X.Y., Y.Z., L.Z., and Y.Y. contrbuted equally to this article.
Funding
This study was partially supported by Hangzhou Institute of Medicine Chinese Academy of Sciences (2024ZZBS12, X.Q.).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
