Abstract
Background
Circular (circ)RNAs are essential regulators in cancer development and progression. CircPVT1, derived from exon 2 (410 nucleotides) of PVT1 gene located at 8q24, has been widely recognized as an oncogenic circRNA frequently upregulated in various human cancers. This study aimed to assess the diagnostic accuracy of circPVT1 for human cancers.
Methods
Articles published up to July 2024 were searched across four databases (PubMed, EMBASE, Web of Science, and Cochrane databases). A meta-analysis was conducted under a random effects model and the diagnostic performance was evaluated using receiver operator characteristic curve analysis. Subgroup analysis of circPVT1 in different cancer types and tissues was performed.
Results
Overall, 12 studies (1246 patients) were included for diagnostic outcome synthesis. The pooled sensitivity was 0.83 (95% CI, 0.77–0.88) and specificity of 0.80 (95% CI, 0.73–0.87), with an area under the receiver operator characteristic curve of 0.89 (95% CI, 0.86–0.91), highlighting the robust diagnostic value of circPVT1. Multiple studies have revealed that circPVT1 functions as a micro RNA sequester to modulate downstream gene expression, affecting various malignant behaviors in cancers.
Conclusion
This study enhances our understanding of the role and mechanism of circPVT1 in human cancers and supports its potential as a promising diagnostic biomarker for various cancer types.
Introduction
Cancer is considered the primary cause of death and is expected to become the leading cause of premature mortality in most countries during this century. 1 The lifetime risk of developing cancer by age 74 is 20.2%, and cancer imposes highest clinical, social, and economic burden in terms of cause-specific disability-adjusted life years compared to all other human diseases. 2 Early detection is crucial for enhancing quality of life, improving survival rates and reducing the financial burden associated with cancer treatments, which tend to be more expensive at advanced stages. 3 Cancer screening programs have shown the potential to reduce mortality rates from various common cancer types and lessen the overall cancer burden. 4 In recent years, multi-cancer early detection tests have become a promising screening tool, utilizing molecular analysis of tumor-related markers in body fluids to identify various cancers. 5 However, the biomarkers currently used in clinical practice are inefficient and lack accuracy, making it essential to develop more precise diagnostic biomarkers for cancer.
Recently, circular RNAs (circRNAs), a novel class of non-coding RNAs characterized by their closed-loop structure formed through back-splicing, have become an active area of research. 6 Compared with RNAs, circRNAs display greater stability in tissues and various bodily fluids (including serum, saliva, and urine), highlighting their strong potential as valuable biomarkers in human diseases. 7 Furthermore, evidence is emerging that circRNAs are implicated in various cellular processes through multiple functions, such as sponging microRNAs (miRNAs), interacting with RNA-binding proteins, regulating transcription, and potentially translating into proteins. 8 For example, circRNA CDR1as/ciRS-7, the first circRNA identified to function a miRNA sequester, has been reported as a reliable diagnostic and prognostic biomarker with high accuracy and efficiency for solid tumours.9,10 Additionally, a recent systematic review and meta-analysis revealed that circSHPRH, which acts as a miRNA sponger or encodes a protein to perform its functions, held promise as an important diagnostic and prognostic biomarker for solid tumours. 11
The circRNA hsa_circ_0001821, also referred to as circPVT1, is derived from exon 2 (410 nucleotides) of plasmacytoma variant translocation 1 (PVT1) gene located at 8q24, a well-known region linked to cancer susceptibility. 12 Recent evidence suggests that circPVT1 functions as an oncogenic non-coding RNA, playing an important role in the tumorigenesis and progression of various malignant conditions by acting as a competing endogenous RNA. 13 Moreover, elevated levels of circPVT1 are related to poor prognosis in cancer patients, supporting the idea that circPVT1 could serve as a potential biomarker in human cancers. 14 Thus, this study conducted a systematic review and meta-analysis of published research to assess the diagnostic value of circPVT1 in cancers. Several indexes were evaluated to determine whether circPVT1 could serve as a promising biomarker for diagnostic prediction in human cancers.
Methods
Protocol and registry
The protocol was registered at the International Prospective Register of Systematic Reviews (PROSPRO) at https://www.crd.york.ac.uk/prospero/ (ID: CRD42024618010).
Search strategies
A comprehensive search was conducted in PubMed, EMBASE, Web of Science, and Cochrane online databases up to July 13, 2024. The following search terms were used in the search strategy: “circular RNA PVT1” or “circPVT1” or “hsa_circ_PVT1” or “circ_PVT1” or “circ-PVT1” or “circ_0001821” or “hsa_circ_0001821”.
Study selection
The process of study selection is shown in Figure 1. The titles and abstracts of records from three electronic databases were screened independently by two authors. The full text of the articles was assessed subsequently. Eligible studies should meet the following criteria: (i) using the expression profile of circPVT1 for the diagnosis of cancers; and (ii) providing sufficient data to obtain the true positive (TP), false positive (FP), false negative (FN) and true negative (TN). Systematic reviews, meta-analyses, abstracts, and meeting reports were excluded. All disagreements during study selection were resolved through consensus, and when no consensus was reached it was resolved by a third author (J.C.).
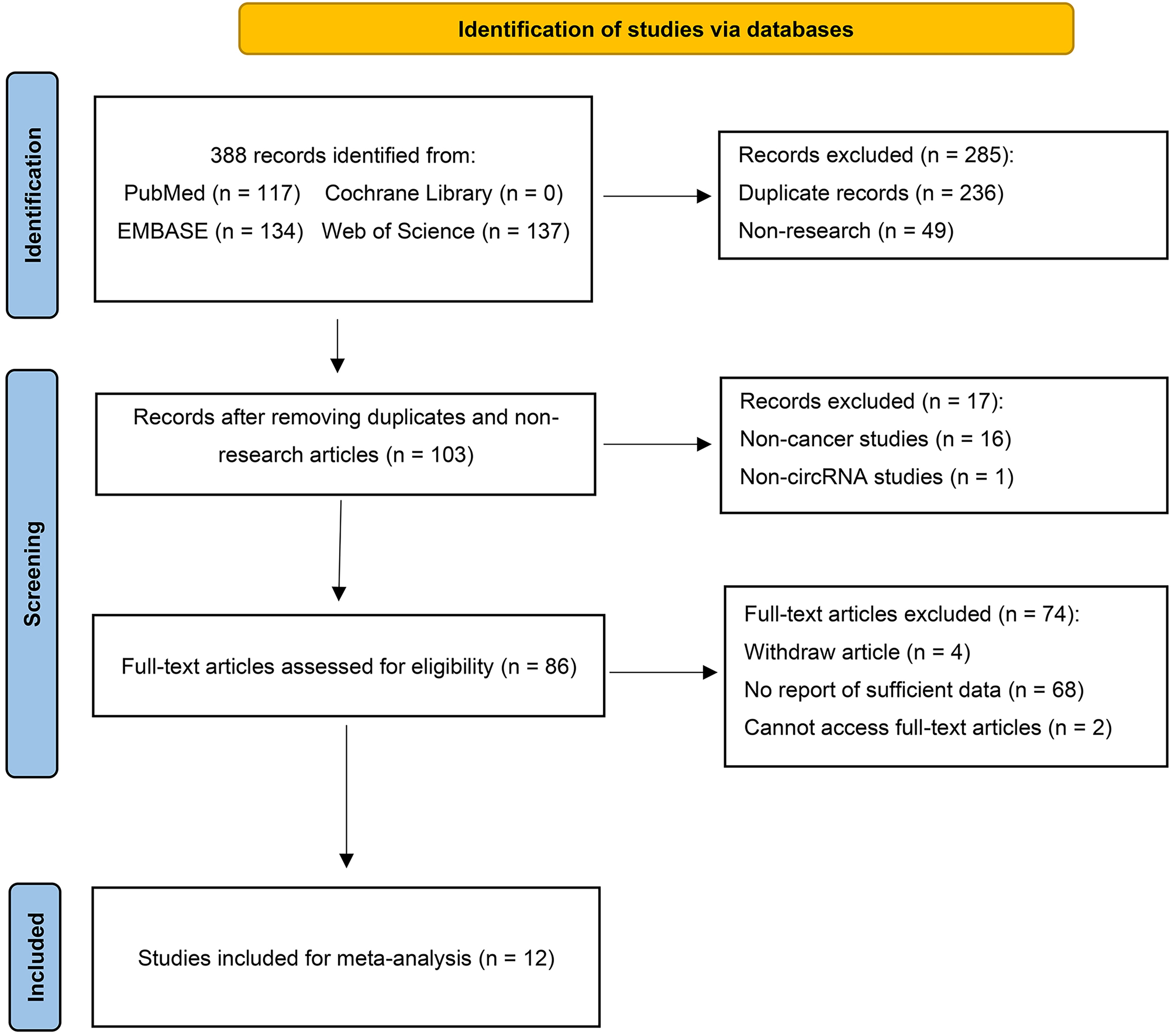
Flow diagram illustrating the selection progress, including the steps of identification, screening, and inclusion.
Data extraction and methodology quality assessment
One author (C.Z.) independently performed the data extraction and quality assessment, while another author (X.M.) checked the information. The following items were collected from the text, tables, or figures, of included articles: first author, publication year, cancer and tissue types, sample size, sensitivity, specificity, TP, FP, FN, TN, and area under the curve (AUC). For articles not reporting sensitivity and specificity, these two items were identified through the receiver operating characteristic (ROC) curve by using the Youden index to identify the optimal threshold. All inconsistencies were resolved by consults. The quality of the included articles was evaluated using the Quality Assessment of Diagnostic Accuracy Studies 2 (QUADAS-2). A study with at least three unclear risk or high risk of bias was considered as low quality.
Statistical analysis
Stata 17 software was utilized for data analysis. The random-effects model was used for data synthesis. Higgins’ I2 was used to evaluate the heterogeneity, with the value greater than 50 considered statistically significant. Subgroup analyses and meta-regression analyses were conducted based on cancer types and sample types, while the sensitivity analysis was performed to assess the robustness of results by excluding studies with small sample sizes of less than 100. Additionally, publication bias was evaluated by the Deek's funnel plot.
Results
Characteristic of included studies
The detailed process of study selection is presented in Figure 1. A total of 388 records were identified from four databases, and 12 articles (1246 patients) were included for the diagnostic meta-analysis. Among these studies, 10 different cancer types were implicated, including: breast cancer, acute myeloid leukemia, oral squamous cell carcinoma, gastric cancer, colorectal cancer, lung cancer, hepatocellular carcinoma, clear cell renal cell carcinoma, bladder cancer, and osteosarcoma. Additionally, the overall characteristics of the included studies are presented in Table 1. The quality of the included studies was assessed using QUADAS-2 scale (Supplemental Figure 1).
Characteristics of included studies of diagnostic test accuracy.
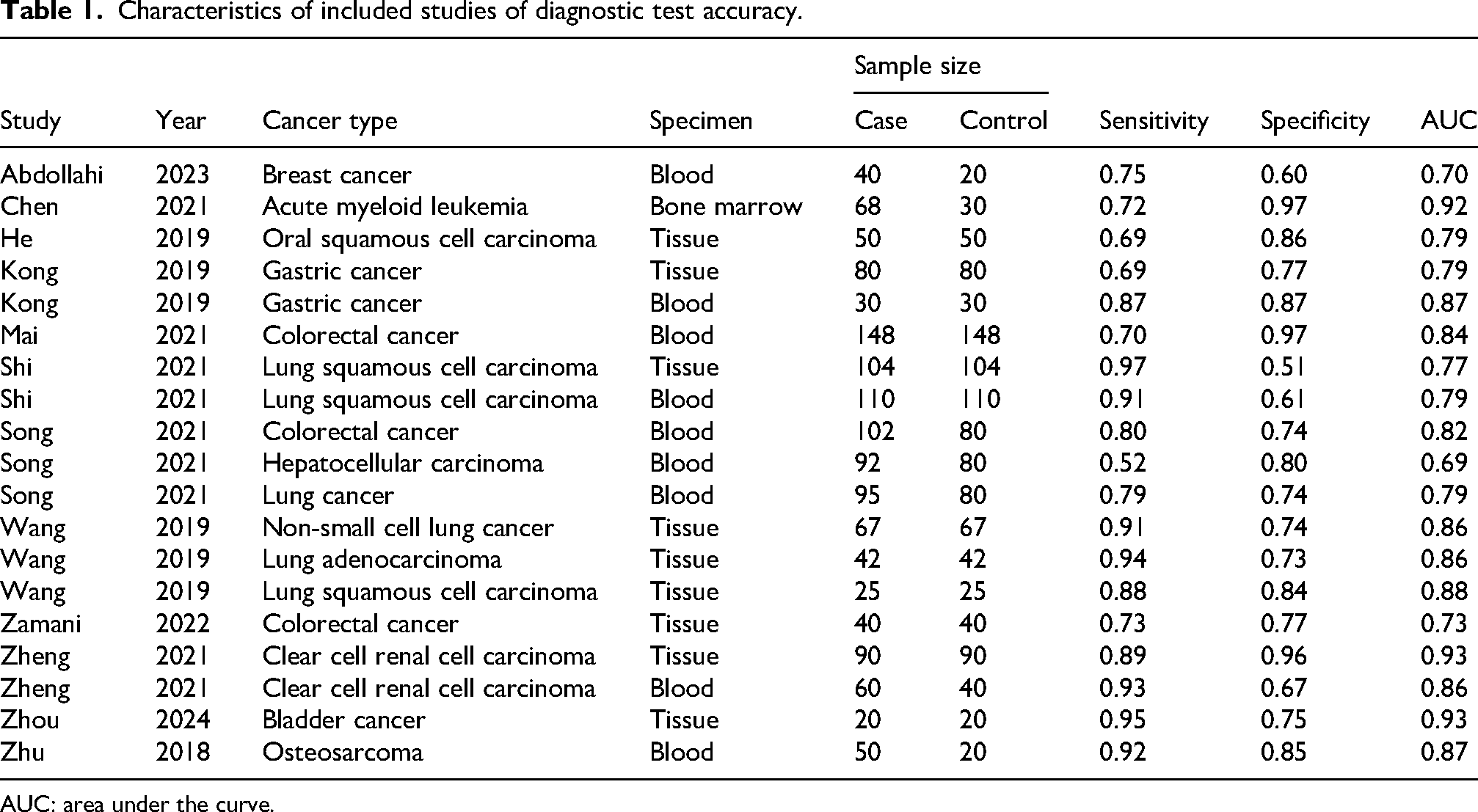
AUC: area under the curve.
Diagnostic accuracy analysis
To evaluate the diagnostic efficacy of circPVT1 in cancers, the pooled sensitivity, specificity, and AUC were calculated. The results revealed a combined sensitivity of 0.83 (95% confidence interval (CI), 0.77–0.88) and specificity of 0.80 (95% CI, 0.73–0.87) (Figure 2(a)). Additionally, an AUC of 0.89 (95% CI, 0.86–0.91) was shown by the summary receiver operating characteristic (SROC) curve (Figure 2(b)). The funnel plot analysis indicated no evidence of publication bias (P = 0.72, Figure 2(c)). Furthermore, Fagan's nomogram was used to estimate the post-test probabilities of circPVT1, showing that the post-test probability rose to 52% with a positive likelihood ratio (LR) of 4 and dropped to 5% with a negative LR of 0.21 (Figure 2(d)). These findings suggest that circPVT1 has a good diagnostic value for cancers.
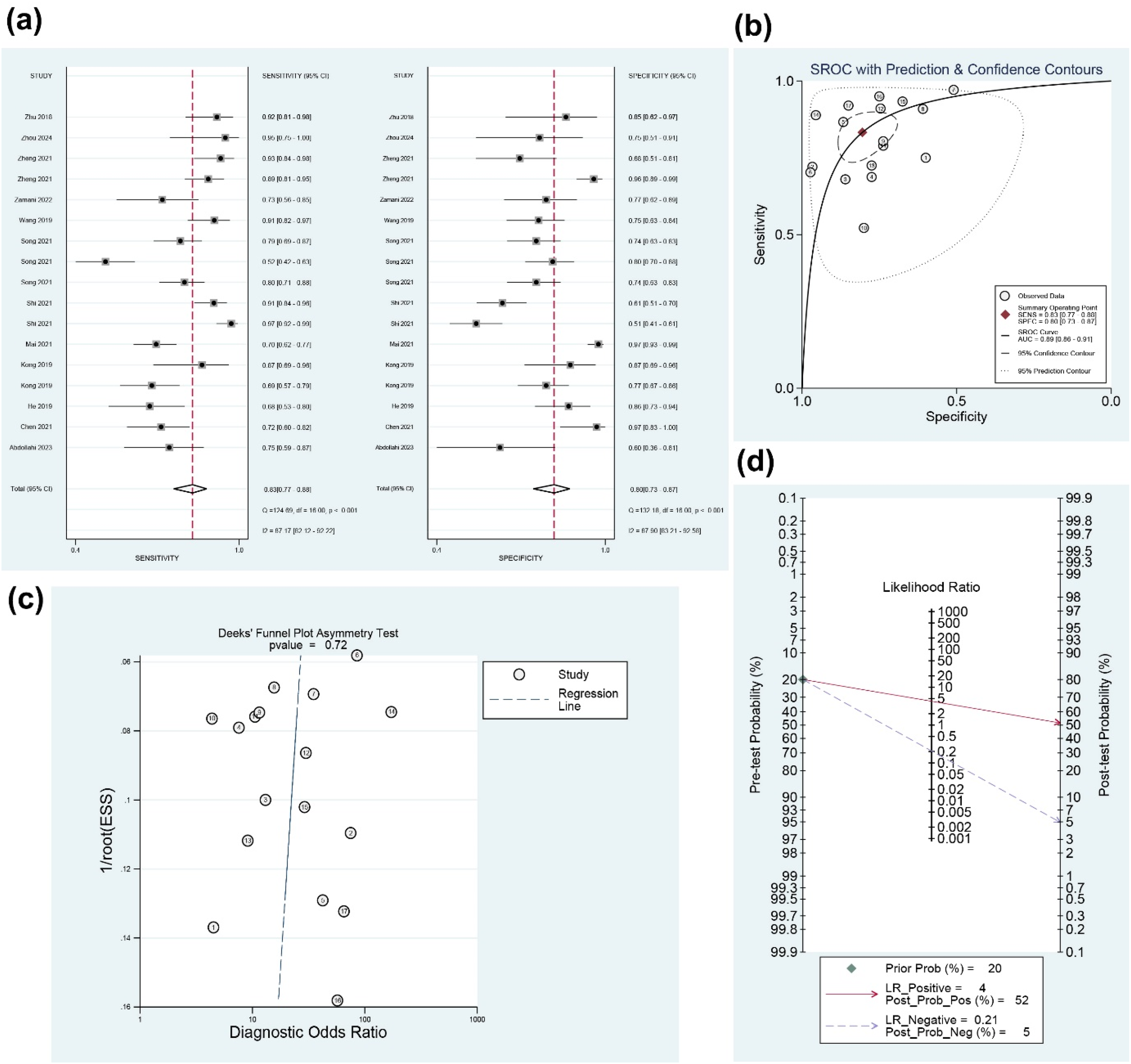
(a) The forest plot of sensitivity and specificity of circPVT1 in the diagnosis of human cancers. (b) The summary receiver operator characteristic curve of circPVT1 in diagnosis of human cancers. (c) The funnel plot for publication bias. (d) Fagan's nomogram was used to evaluate the post-test probabilities of circPVT1.
Subgroup analysis, meta-regression analysis, and sensitivity analysis
To assess the diagnostic value of circPVT1 for cancers, further subgroup analysis was performed according to cancer types (lung cancer and colorectal cancer) and sample types (tissue and blood). The results showed that the high diagnostic accuracy persisted across the subgroups (Table 2). Moreover, a meta-regression analysis was conducted, which showed that cancer types and sample types may result in statistical heterogeneity (Supplemental Figure 2). Given the considerable variation in sample size among the included studies, a sensitivity analysis was performed by excluding studies with sample sizes smaller than 100. The sensitivity analysis yielded results that were nearly identical to those of the overall analysis, suggesting the relative stability of the pooled outcomes (Table 2).
Summary of subgroup and sensitivity analysis for diagnostic accuracy of circPVT1 in cancers.
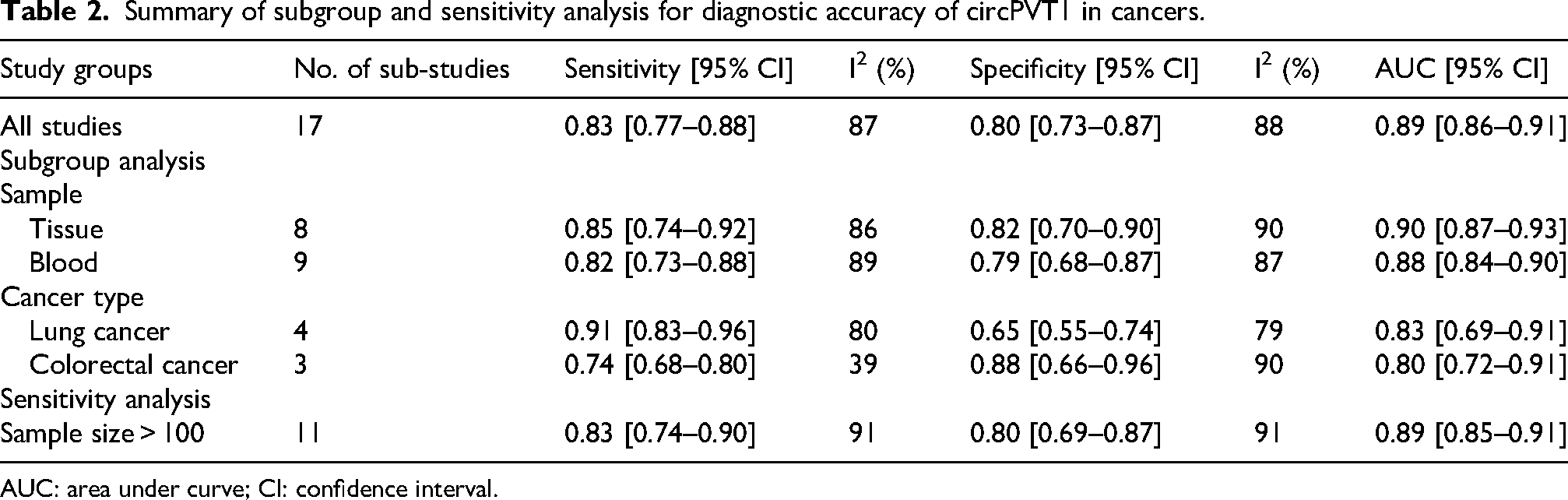
AUC: area under curve; CI: confidence interval.
Regulatory role of circPVT1 in cancers
Given the upregulation of circPVT1 in multiple human cancers, emerging evidence has demonstrated its regulatory role in cancer progression. CircPVT1 plays an important role in the progression of human cancers, including proliferation, apoptosis, migration, invasion, glycolysis, epithelial-mesenchymal transition, and drug resistance (Supplemental Table 1). The regulatory mechanisms of circPVT1 reported in various cancers were shown in Figure 3.
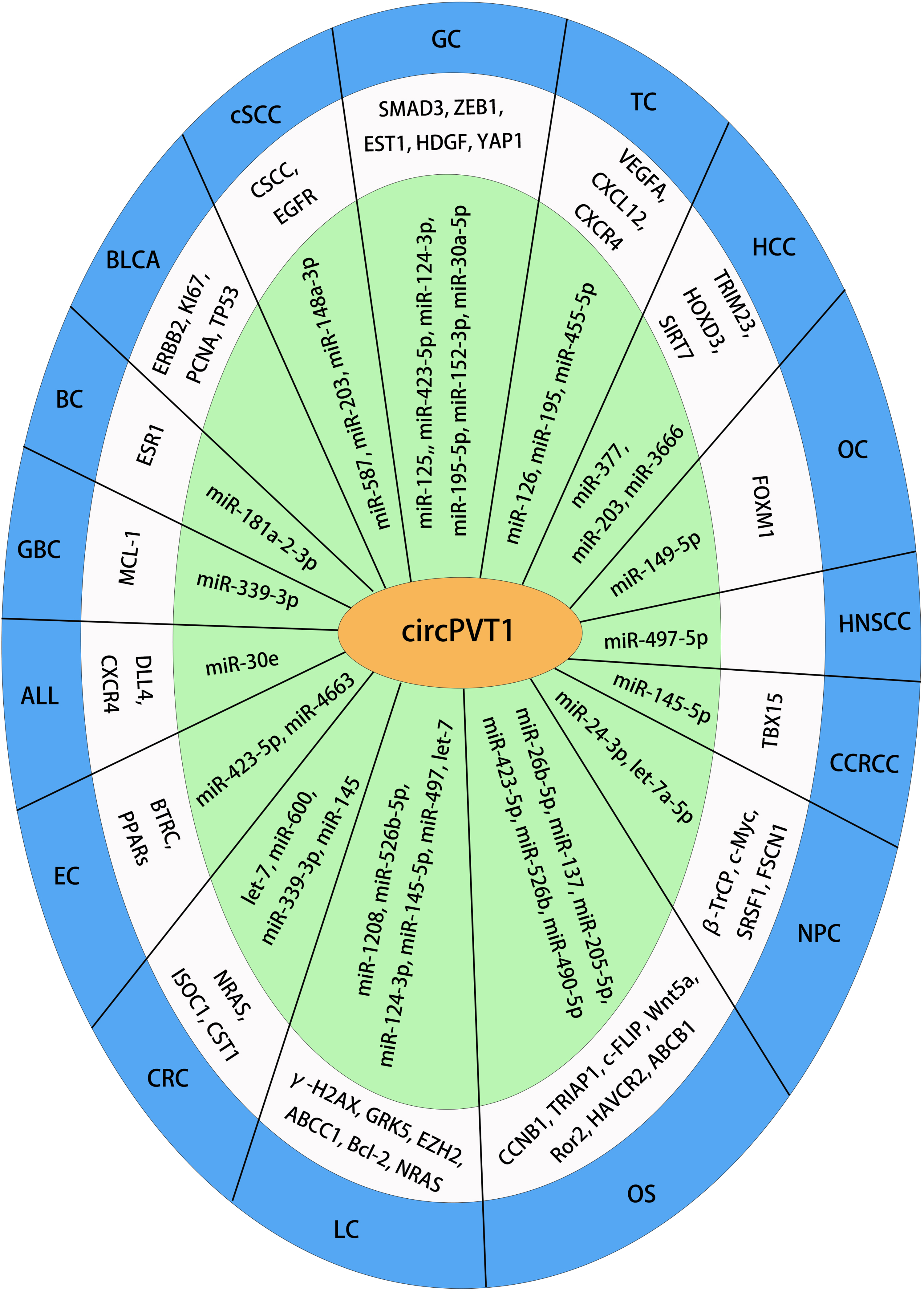
A schematic diagram illustrating the molecular mechanisms of circPVT1 across various human cancers. The green section highlights miRNAs sequestered by circPVT1; the white section depicts the target genes and proteins linked to circPVT1.
Discussion
This systematic review and meta-analysis aimed to evaluate circPVT1's diagnostic performance in human cancers, synthesizing data from 12 studies. The combined results suggested a high diagnostic accuracy, with a sensitivity of 0.83 and specificity of 0.80, as well as a 0.89 AUC of the SROC. These findings demonstrate that circPVT1 shows promise as a favorable diagnostic marker for cancers.
In clinical practice, multiple protein biomarkers, such as alpha-fetoprotein (AFP), carcinoembryonic antigen (CEA), prostate-specific antigen, cancer antigen 125 (CA125), carbohydrate antigen 19-9 (CA19-9) and cancer antigen 15-3 (CA15-3), are extensively utilized for diagnosing different types of cancer. 15 However, the sensitivity and specificity of individual biomarkers are limited for cancer detection, so it is suggested that using a panel of biomarkers provides a better diagnostic performance. 16 CircRNAs are characterized by their conservation, abundance, stability, and specificity, making them suitable as biomarkers in diseases. 17 A flurry of recent research has indicated that circRNAs have a good diagnostic performance and serve as candidate biomarkers for various diseases, including cardiovascular disease, lung cancer, and osteosarcoma.18–20 Furthermore, specific circRNAs, such as CDR1as/cirRS-7 and circ-SHPRH, have been identified as promising diagnostic biomarkers for various solid cancers.9,11 To our knowledge, this is the first study to assess the overall diagnostic value of circPVT1 in human cancers using meta-analysis. The combined results of circPVT1 showed a good diagnostic value with an AUC of 0.8 in human cancers. Furthermore, in compared to commonly used cancer biomarkers, such as CEA for colorectal cancer, AFP for hepatocellular carcinoma, neuron-specific enolase for lung cancer, alkaline phosphatase for osteosarcoma, as well as CEA, CA19-9 and CA125 for gastric cancer, circPVT1 showed superior diagnostic accuracy.21–23 Moreover, a panel of circPVT1 and other biomarkers cumulatively possess a higher diagnostic accuracy for cancer diagnosis.21,23 Thus, these findings suggest that circPVT1 could serve as a reliable marker for diagnosing human cancers, further supporting its correlation with cancer risk.
The application of circPVT1 in human cancer diagnosis might be associated with its overexpression in cancers. CircPVT1 is overexpressed in various types of human cancer, acting an oncogenic role in the occurrence and progression of cancers. 24 A previous meta-analysis showed that the upregulation of circPVT1 is closely linked to poor clinical outcomes, including advanced stage, metastasis, chemoresistance, and shorter overall survival. 14 Furthermore, recent research has shed light on the regulatory mechanisms underlying circPVT1 in the progression of multiple cancers. 25 For instance, circPVT1 acts as a competing endogenous RNA (ceRNA) to modulate various axes, contributing to non-small cell lung cancer progression.26–28 Additionally, several ceRNA axes involving circPVT1 (circPVT1/miR-423-5p/Wnt5a/Ror2, circPVT1/miR-526b/FOXC2, circPVT1/miR-205-5p/c-FLIP, and circPVT1/miR-490-5p/HAVCR2) play a role in regulating migration and invasion in osteosarcoma.29–32 Notably, circPVT1 has been reported that it can be packaged into exosomes and promotes malignant behavior. 33 In our study, the overexpression of circPVT1 in blood showed comparable diagnostic performance to that in tissue for diagnosing cancers. These findings suggest that circPVT1 is likely packaged into exosomes to exert its functions, indicating its potential as a biomarker associated with various aspects in cancer, including tumorigenesis, progression, prognosis, and treatment response. In this study, cicrPVT1 achieve the levels that the positive LR was 4 and negative LR was 0.21, recommending the sufficient robustness of circPVT1 for clinical application. However, the relationship of circPVT1 expression levels among cancer tissue and blood remains poorly understood, underscoring the challenge and the need for further investigation into circPVT1's potential as a blood biomarker in early cancer diagnosis.
This meta-analysis identified circPVT1 as a reliable biomarker with high diagnostic accuracy in human cancers. The sensitivity analysis, which excluded studies with small sample size, confirmed the stability of the pooled outcomes, with results remaining largely unchanged. However, there are several limitations in this study. A significant degree of heterogeneity was observed among included studies, likely due to variations in threshold effect, sample sizes, and patient populations. Our meta-regression also indicated that sample types and cancer types could be sources of significant heterogeneity. CircPVT1 may have different diagnostic performance across different tissues and cancers. The threshold effect is regarded as the most obvious source of heterogeneity in systematic reviews and meta-analyses of diagnostic test accuracy. 34 In this study, cut-off values of circPVT1 in the included studies have not been reported, which might be different among studies, leading to concerns regarding applicability. For example, circPVT1 exhibited higher sensitivity but lower specificity in lung cancer compared to colorectal cancer. Furthermore, variability in sample sizes across studies contributed to the high heterogeneity in the analysis of diagnostic variables. Moreover, most samples tested in Chinese groups led to population bias, posing a challenge for generalizing the findings to broader populations of cancer patients. As circPVT1 expression may vary across different ethnic diverse populations, further validation in more diverse cohorts is needed. Additionally, due to the absence of prospective and double-blind studies in diagnostic value investigation, some degree of bias was unavoidable. Thus, more high-quality, multicenter and prospective diagnostic studies with large sample sizes and cut-off values are needed for further validation.
Conclusions
This study summarized the diagnostic performance of circPVT1 in human cancers. Although the heterogeneity existed among included studies, circPVT1 showed a high diagnostic accuracy for cancers, indicating its potential as a valuable biomarker in cancer diagnosis. Further large-scale clinical studies across various cancer types are required to validate and broaden the applicability of circPVT1 in cancer diagnosis.
Abbreviations
alpha-fetoprotein
area under the curve
cancer antigen 125
cancer antigen 15-3
carbohydrate antigen 19-9
carcinoembryonic antigen
competing endogenous RNA
confidence interval
circular RNAs
false negative
false positive
likelihood ratio
microRNAs
plasmacytoma variant translocation 1
Quality Assessment of Diagnostic Accuracy Studies 2
summary receiver operating characteristic
true negative
true positive.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155251362862 - Supplemental material for Circular RNA PVT1 as a potential biomarker for human cancers: A systematic review and meta-analysis
Supplemental material, sj-docx-1-jbm-10.1177_03936155251362862 for Circular RNA PVT1 as a potential biomarker for human cancers: A systematic review and meta-analysis by Xiaoyan Ma, Junheng Chen and Chunbin Zhou in The International Journal of Biological Markers
Supplemental Material
sj-docx-2-jbm-10.1177_03936155251362862 - Supplemental material for Circular RNA PVT1 as a potential biomarker for human cancers: A systematic review and meta-analysis
Supplemental material, sj-docx-2-jbm-10.1177_03936155251362862 for Circular RNA PVT1 as a potential biomarker for human cancers: A systematic review and meta-analysis by Xiaoyan Ma, Junheng Chen and Chunbin Zhou in The International Journal of Biological Markers
Supplemental Material
sj-jpg-3-jbm-10.1177_03936155251362862 - Supplemental material for Circular RNA PVT1 as a potential biomarker for human cancers: A systematic review and meta-analysis
Supplemental material, sj-jpg-3-jbm-10.1177_03936155251362862 for Circular RNA PVT1 as a potential biomarker for human cancers: A systematic review and meta-analysis by Xiaoyan Ma, Junheng Chen and Chunbin Zhou in The International Journal of Biological Markers
Supplemental Material
sj-jpg-4-jbm-10.1177_03936155251362862 - Supplemental material for Circular RNA PVT1 as a potential biomarker for human cancers: A systematic review and meta-analysis
Supplemental material, sj-jpg-4-jbm-10.1177_03936155251362862 for Circular RNA PVT1 as a potential biomarker for human cancers: A systematic review and meta-analysis by Xiaoyan Ma, Junheng Chen and Chunbin Zhou in The International Journal of Biological Markers
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
