Abstract
Background
The relationship between serum uric acid and breast cancer remains uncertain. This meta-analysis aimed to elucidate the dose-response association between elevated serum uric acid levels and breast cancer risk.
Methods
We systematically searched PubMed, Embase, and Web of Science for observational studies evaluating serum uric acid and breast cancer risk in adult women. Risk ratios (RRs) with 95% confidence intervals (CIs) were calculated using a random-effects model.
Results
Ten studies were included. High serum uric acid was associated with an increased breast cancer risk (RR: 1.34; 95% CI: 1.03–1.73; P = 0.03; I2 = 78%). Meta-regression analysis revealed that the cutoff for high serum uric acid positively correlated with breast cancer risk (coefficient = 0.24; P < 0.001), explaining heterogeneity (residual I2 = 0%). Subgroup analysis demonstrated a high serum uric acid was significantly related to an increased breast cancer risk in studies with a cutoff value ≥ 5.4 mg/dL (RR: 1.95; P < 0.001), but not in those with a cutoff value < 5.4 mg/dL (RR: 0.97; P = 0.44). The dose-response meta-analysis revealed a U-shaped association between serum uric acid levels and the risk of breast cancer (P for nonlinearity = 0.013). The risk of breast cancer fell until around 4.5 mg/dL of serum uric acid, and increased afterward.
Conclusion
Serum uric acid may be nonlinearly associated with the risk of breast cancer (U-shaped). The risk of breast cancer increases with serum uric acid above 4.5 mg/dL, and a higher association between serum uric acid and the increased risk of breast cancer could be observed in studies with cutoff of serum uric acid > 5.4 mg/dL.
Introduction
Breast cancer (BC) is the most frequently diagnosed malignancy among women worldwide and represents a significant public health burden. 1 Globally, it accounts for approximately 24.5% of all cancer cases and 15.5% of cancer-related deaths in women.2,3 The prognosis of BC varies considerably based on the stage at diagnosis, tumor biology, and treatment options. Despite advances in early detection and therapy, BC remains a leading cause of cancer mortality. 4 Numerous risk factors for BC have been identified, including age, genetic predisposition, family history, reproductive history, hormonal factors, lifestyle factors such as obesity and alcohol consumption, and environmental exposures. 5 While these factors contribute to BC risk, there is a critical need to identify novel risk factors that could improve early detection and prevention strategies. 6
Uric acid (UA) is the end product of purine metabolism in humans. 7 It has dual roles in physiology, acting both as an antioxidant and a pro-oxidant depending on its concentration and the environment. 8 Physiologically, UA is mainly excreted by the kidneys, and its levels can be influenced by diet, genetic factors, renal function, and metabolic conditions. 9 Elevated serum UA levels, often seen in conditions like gout, hypertension, and metabolic syndrome, 10 have raised interest in its potential role in cancer development. 11 The biological characteristics of UA are complex. As an antioxidant, UA can scavenge reactive oxygen species (ROS) and protect cells from oxidative damage. 12 However, in high concentrations, UA may contribute to pro-oxidant effects, promoting inflammation, endothelial dysfunction, and oxidative stress, which are potential mechanisms linking UA to carcinogenesis. 12 Additionally, hyperuricemia may enhance insulin resistance and influence adipokine production, 13 further contributing to cancer risk.
Previous studies investigating the relationship between serum UA levels and BC risk have yielded inconsistent results. Some studies have suggested a positive association, indicating that high UA levels may increase BC risk,14–18 while others have found no significant relationship or even a protective effect.19–23 These conflicting findings highlight the need for a comprehensive evaluation of the available evidence. The objective of this meta-analysis is to systematically assess the association between serum UA levels and the risk of BC. By synthesizing data from observational studies, we aim to clarify whether elevated serum UA is a significant risk factor for BC and to explore potential sources of heterogeneity in the findings.
Methods
The study adhered to Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) 2020 24 and the Cochrane Handbook for Systematic Reviews and Meta-analyses 25 guidelines for conducting this meta-analysis, including for the study design, data collection, statistical analysis, and results interpretation. The protocol of the meta-analysis has been registered at PROSPERO with the identifier CRD42024579163.
Literature search
To identify studies pertinent to this meta-analysis, we searched the PubMed, Embase, and Web of Science databases using an extensive array of search terms, which included: (a) “uric acid” OR “hyperuricemia” OR “urate”; (b) “breast”; and (c) “cancer” OR “carcinoma” OR “tumor” OR “malignancy”. The search was limited to research involving human subjects and we only included studies published as full-text articles in English within peer-reviewed journals. The detailed search syntax for each database is shown in Supplemental File 1. Additionally, we manually reviewed the references of relevant original and review articles to identify further pertinent studies. The literature was assessed from the inception of the searched databases up to June 29, 2024.
Inclusion and exclusion criteria
The inclusion criteria for potential studies were defined according to the PICOS framework:
The exclusion criteria included reviews, editorials, meta-analyses, preclinical studies, studies without the evaluation of serum UA, studies with the serum UA analyzed as a continuous variable only, and studies without the risk of BC. If two or more studies with overlapping populations were found, the study with the largest sample size was enrolled for the meta-analysis.
Study quality evaluation and data extraction
The literature search, study identification, quality assessment, and data extraction were conducted independently by two authors, with any disagreements resolved through discussion with the corresponding author. Study quality was evaluated using the Newcastle–Ottawa Scale (NOS), 26 which assesses the selection, control of confounders, and outcome measurement and analysis, with scores ranging from 1 to 9, where 9 signifies the highest quality. The data collected for analysis included the study details (author, year, country, and design), participant characteristics (source, sample size, age, and menopausal status), cutoff values for defining a high serum UA, follow-up periods for longitudinal studies, the methods used to validate the diagnosis of BC, the number of patients with BC, and the variables adjusted when analyzing the association between the serum UA and BC.
Statistics
The association between the serum UA and the risk of BC was analyzed using risk ratios (RRs) and 95% confidence intervals (CIs), comparing the high versus low serum UA index groups. For studies that reported hazard ratios (HRs), these were used directly as RRs. The RR values and their standard errors were computed from 95% CIs or P-values and logarithmically transformed for variance stabilization. To assess heterogeneity, we used the Cochrane Q test and I² statistics, 27 with I2 > 50% indicating considerable heterogeneity. A random-effects model was applied to integrate the results, accounting for study variability. 25 A sensitivity analysis by excluding one study at a time was performed to evaluate the robustness of the finding. A univariate meta-regression analysis was performed to investigate the influences of study characteristics in continuous variables on the association between serum UA and BC, such as the sample size, the mean age of the women, and the cutoff values for defining a high serum UA. 25 Predefined subgroup analyses were performed to explore the effects of various factors based on the study-level data, such as geographic region (Asian, African, or western countries), study design (cohort vs. case-control studies), mean ages of the women, and cutoff values for defining a high serum UA. Medians of the continuous variables were used as the cutoff values for defining subgroups to keep the number of studies in each subgroup balanced. In addition, we conducted a dose–response meta-analysis to calculate pooled estimates from the natural logarithm of RRs and 95% CIs across categories of serum UA levels, using data of studies comparing the risk of BC among women of at least three categories of serum UA. For each category of serum UA, we used the midpoint of serum UA levels if the mean or median level per category was not reported. When extreme categories were open-ended, we used the width of the adjacent interval to calculate an upper or lower cut-off value. The method described by Greenland and Longnecker 28 was used for linear dose-response analysis. Nonlinear dose-response analysis was performed using restricted cubic splines with three knots at 10%, 50%, and 90% percentiles of the distribution. 29 Publication bias was evaluated using funnel plots and visual inspection for asymmetry, supplemented by Egger's regression test. 30 Analyses were performed using RevMan (Version 5.1; Cochrane Collaboration, Oxford, UK) and Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Results
Study inclusion
The study inclusion process is illustrated in Figure 1. Initially, 932 potentially relevant records were identified from the three searched databases, with 213 firstly excluded due to duplication. A subsequent screening of the titles and abstracts led to the further exclusion of 698 studies, primarily because they did not align with the objectives of the meta-analysis. The full texts of the remaining 21 records were reviewed by two independent authors, resulting in the exclusion of 11 more studies for various reasons, as detailed in Figure 1. Finally, 10 observational studies remained and were deemed appropriate for inclusion in the quantitative analysis.14–23
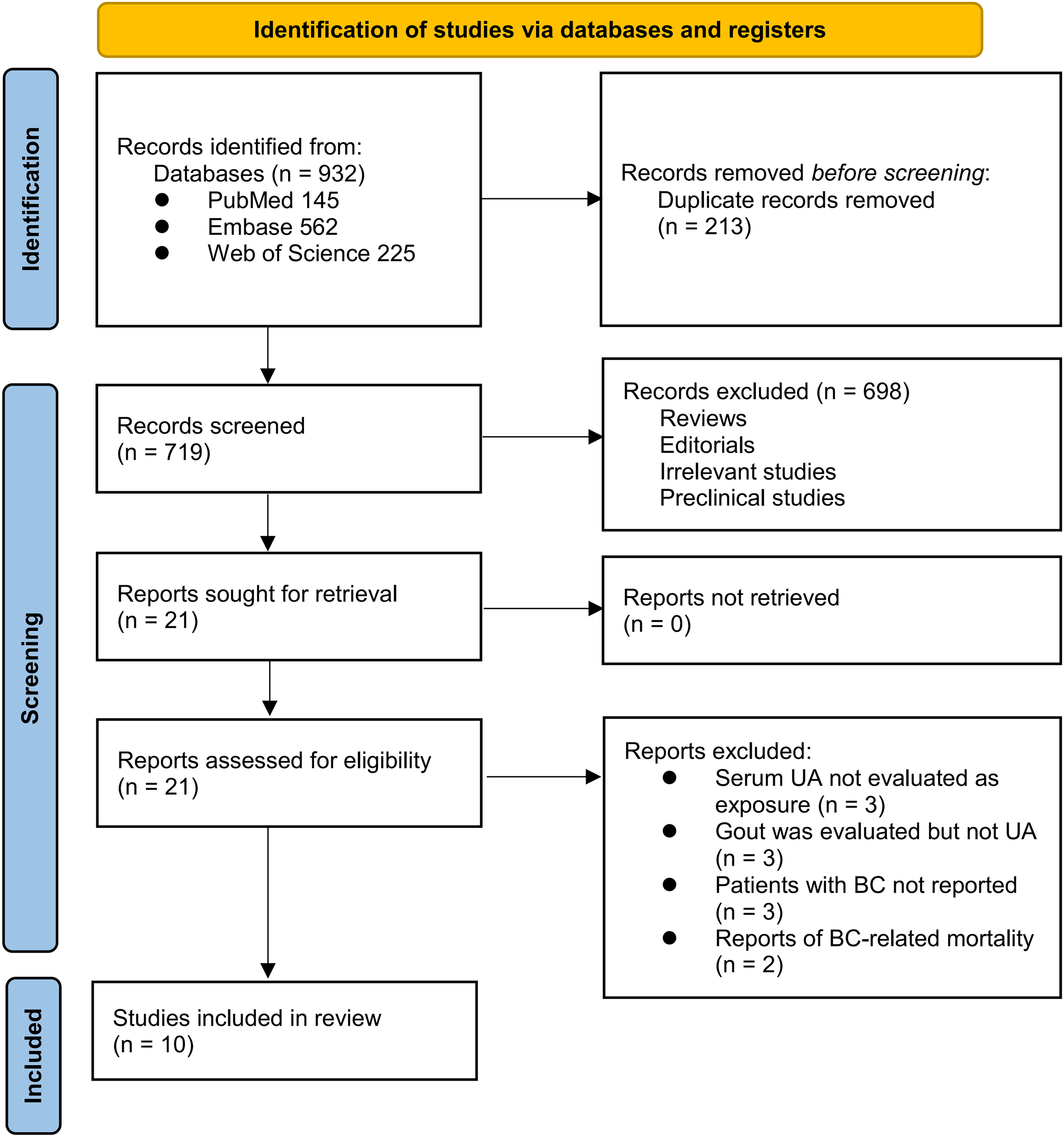
PRISMA flowchart of database search and study inclusion.
Overview of the study characteristics
Table 1 shows the summarized characteristics of the available studies included in the meta-analysis. Overall, four prospective cohort studies,15,19,21,23 one retrospective study, 22 and five case-control studies14,16–18,20 were included in the meta-analysis. These studies were published from 1988 to 2023, and were conducted in the United States, Australia, Italy, France, China, Sweden, and Ethiopia. Overall, 354,423 female participants were included, with the mean ages ranging from 44.1 to 61.4 years. Only three studies reported the menopausal status of the included women.14,15,18 The cutoff values for defining a high serum UA varied among the included studies from 2.0 to 7.4 mg/dL. The follow-up durations for the cohort studies were 4.5 to 19.5 years. The validation of the diagnosis of BC was via pathological diagnosis in three studies,17,18,22 via medical records in five studies,14,16,19–21 and via cancer registry in two studies.15,23 Overall, 12,368 participants were diagnosed as BC. Potential confounding factors, such as age, body mass index, smoking, alcohol drinking, and metabolic factors were adjusted to a varying degree when the association between serum UA and the risk of BC was reported. The NOS scores of the included studies were seven to nine, suggesting an overall good study quality (Supplemental Table 1).
Characteristics of the included studies.
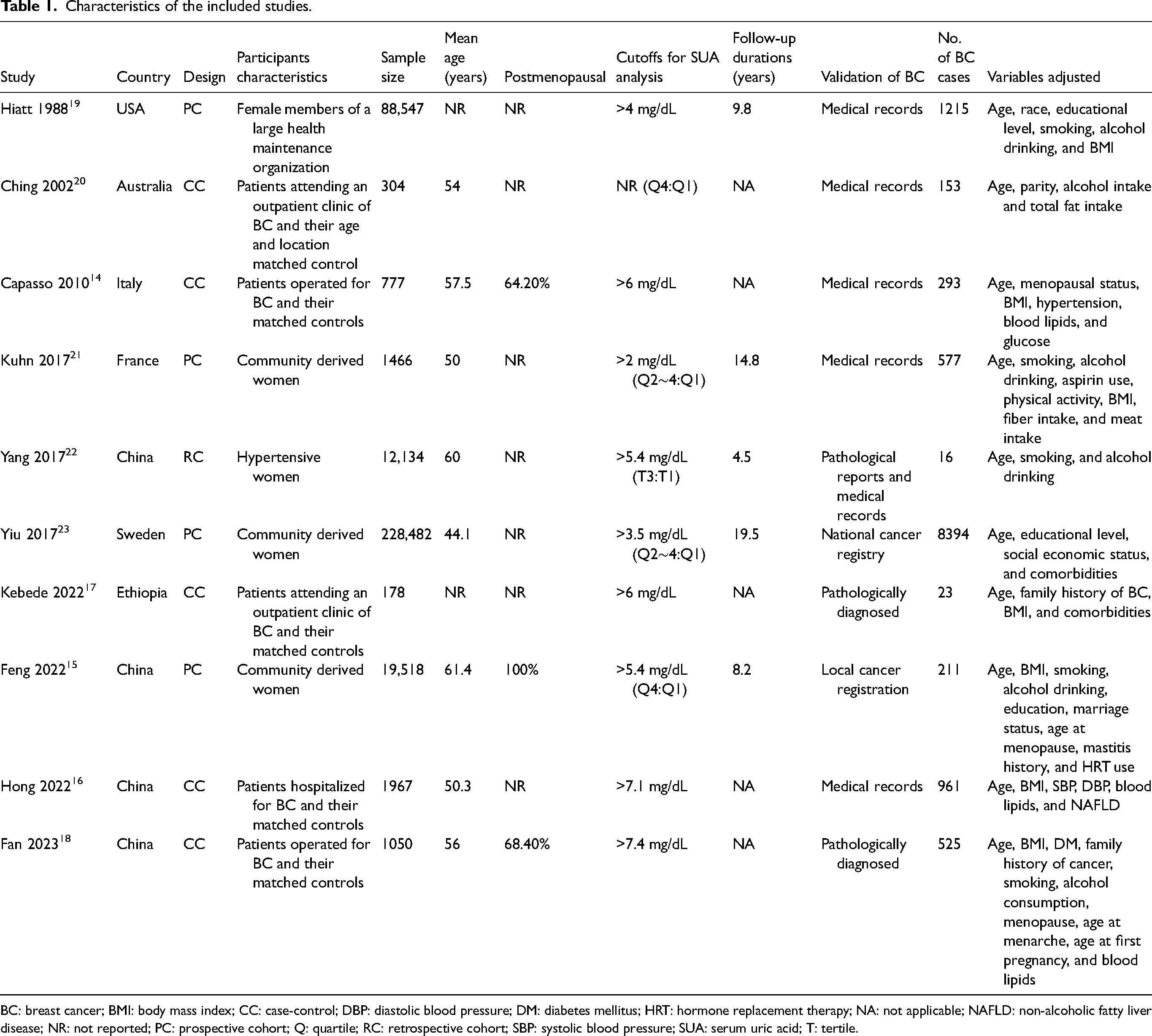
BC: breast cancer; BMI: body mass index; CC: case-control; DBP: diastolic blood pressure; DM: diabetes mellitus; HRT: hormone replacement therapy; NA: not applicable; NAFLD: non-alcoholic fatty liver disease; NR: not reported; PC: prospective cohort; Q: quartile; RC: retrospective cohort; SBP: systolic blood pressure; SUA: serum uric acid; T: tertile.
Association between serum UA and the risk of BC
Overall, the pooled results of the ten studies14–23 suggested that women with a higher level of serum UA may be associated with an increased risk of BC as compared to those with a lower level of UA (RR: 1.34; 95% CI: 1.03–1.73; P = 0.03; Figure 2(a)) with significant heterogeneity (I2 = 78%). Sensitivity analysis by excluding one study at a time did not significantly change the results (RR: 1.26–to 1.49; Pall < 0.05), indicating that the overall results were not significantly influenced by any single study. This suggests the robustness of our findings. Further univariate meta-regression analysis suggested that the cutoff for defining a high serum UA was positively correlated with the RR for the association between serum UA and BC (coefficient = 0.24; P < 0.001; Figure 2(b) and Supplemental Table 2), which adequately explained the source of heterogeneity (residual I2 = 0%). However, variables such as mean age of the included women or NOS scores of the included study did not seem to significantly modify the results (P = 0.30 and 0.32, respectively; Supplemental Table 2). Further subgroup analysis suggested a significant association between a high serum UA and the risk of BC in studies from China and Africa, but not for studies from western countries (P for subgroup difference = 0.007; Figure 3(a)). The subgroup analysis did not suggest different results between cohort and case-control studies (P for subgroup difference = 0.11; Figure 3(b)). The subgroup analysis suggested a significant association between a high serum UA and the risk of BC in studies with the mean ages of the women ≥ 55 years, but not for those < 55 years (P for subgroup difference = 0.02; Figure 4(a)). However, the I2 for the subgroups of western country, cohort studies, case-control studies, women < 55 years were ≥ 50%, suggesting the results should be interpreted with caution. Finally, a high serum UA was significantly related to an increased risk of BC in studies with cutoff value of serum UA ≥ 5.4 mg/dL (RR: 1.95; 95% CI: 1.55–2.46; P < 0.001; I2 = 1%), but not in studies with cutoff value of serum UA < 5.4 mg/dL (RR: 0.97; 95% CI: 0.90–1.05; P = 0.44; I2 = 0%), which fully explained the source of heterogeneity (P for subgroup difference < 0.001; Figure 4(b)). In addition, using data from the four studies which involve at least three categories of serum UA,15,18,19,23 the dose–response meta-analysis revealed a U-shaped association between serum UA levels and the risk of BC (P for nonlinearity = 0.013, Supplemental Figure 1). The risk of BC fell until around 4.5 mg/dL of serum UA levels and increased afterward.
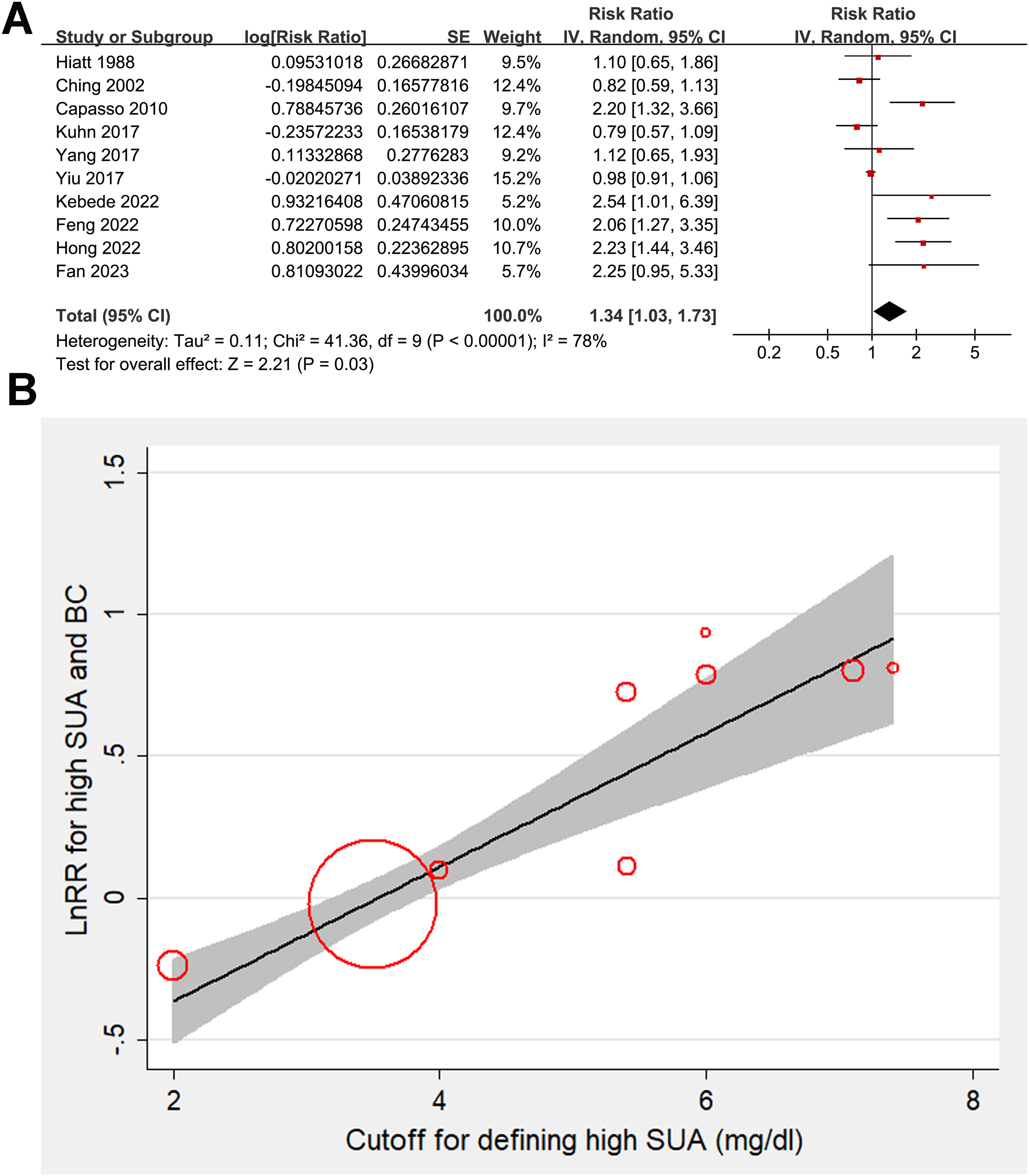
Forest plots and meta-regression analysis for the association between serum UA and the risk of BC in women. (a) Plots for the overall meta-analysis regarding the association between serum UA and the risk of BC in women. (b) Plots for meta-regression analysis evaluating the influence of cutoff values for a high serum UA on the results of the meta-analysis.
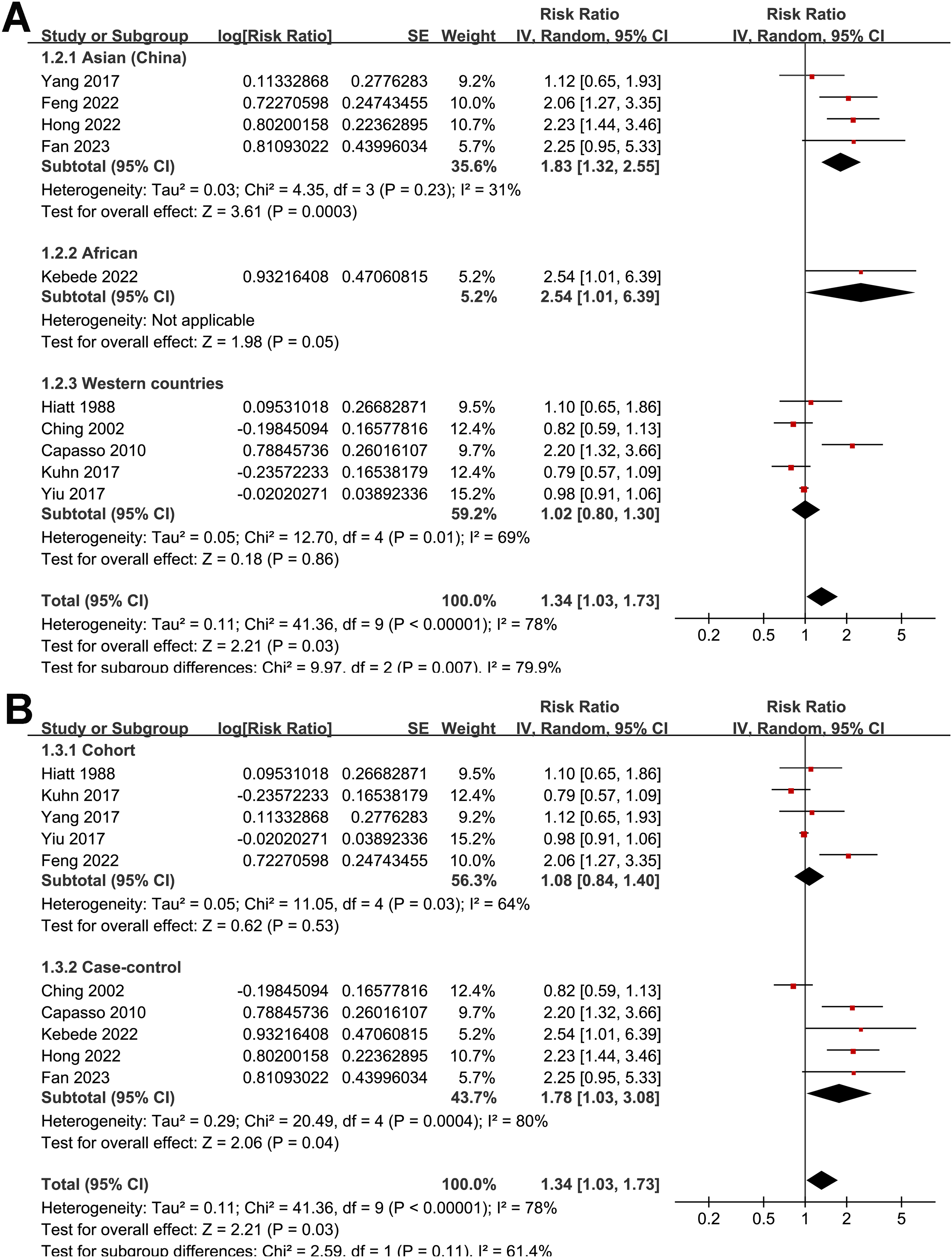
Forest plots for the subgroup analyses. (a) Subgroup analysis according to the study country. (b) Subgroup analysis according to the study design.
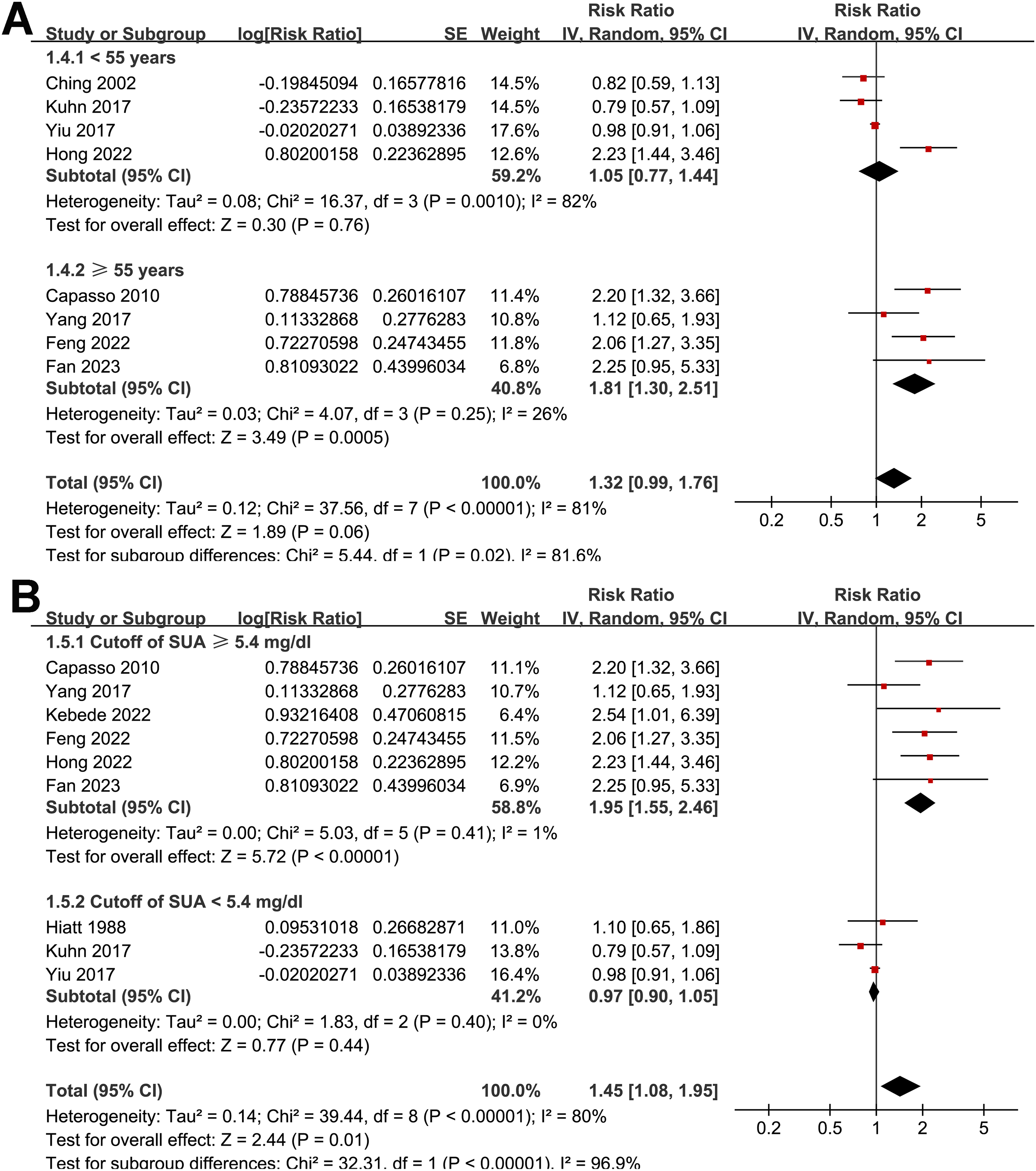
Forest plots for the subgroup analyses. (a) Subgroup analysis according to the mean ages of the included women. (b) Subgroup analysis according to the cutoff values for defining a high serum UA level in each study.
Publication bias
Upon visual inspection, the funnel plots for meta-analysis of the association between the serum UA and the risk of BC appeared symmetrical, indicating a low likelihood of publication bias (Supplemental Figure 2). Additionally, Egger's regression test results (P = 0.24) also supported this conclusion by suggesting a low risk of publication bias.
Discussion
This meta-analysis comprehensively evaluated the association between serum UA levels and the risk of BC across 10 observational studies involving 354,423 female participants. Our findings indicate that women with the highest category of serum UA levels have an increased risk of BC compared to those with the lowest levels, with a pooled RR of 1.34. The analysis revealed significant heterogeneity among the studies, which was explained by the cutoff values for defining high serum UA levels. Specifically, a significant association between high serum UA and BC risk was found in studies with a cutoff value of UA ≥ 5.4 mg/dL, but not in studies with a lower cutoff value. Moreover, the dose-response meta-analysis revealed a U-shaped association between serum UA levels and the risk of BC, and the risk of BC fell until around 4.5 mg/dL of serum UA and increased afterward. Taken together, these results suggested that serum UA may be nonlinearly associated with the risk of BC (U-shaped). The risk of BC increases with serum UA above 4.5 mg/dL, and a higher association between serum UA and the increased risk of BC could be observed in studies with cutoff of serum UA > 5.4 mg/dL.
The U-shaped association identified in the dose-response meta-analysis underscores the complex relationship between serum UA and risk of BC, highlighting a potential threshold effect. The findings suggest that UA levels above 4.5 mg/dL may be a significant risk factor for BC, particularly in studies with a cutoff value > 5.4 mg/dL. The potential mechanisms linking the nonlinear relationship between serum UA and risk of BC in women may be multifactorial. UA has dual roles as an antioxidant and a pro-oxidant.8,11,12,31 At physiological levels, UA can act as a scavenger of ROS, protecting cells from oxidative stress.11,12 This may explain a potential inverse relationship between serum UA and the risk of BC in dose-response meta-analysis when serum UA < 4.5 mg/dL. However, elevated UA levels may shift this balance, promoting oxidative stress, inflammation, and endothelial dysfunction—all of which are implicated in carcinogenesis.11,12 High UA levels can also induce insulin resistance 32 and alter adipokine profiles, 33 contributing to a pro-tumorigenic environment. Moreover, hyperuricemia may lead to the activation of pro-inflammatory pathways, such as the NF-κB signaling pathway, 34 further facilitating cancer development. 35 These mechanisms may explain an increased risk of BC when serum UA is above 4.5 mg/dL.
Our meta-regression analysis demonstrated that the cutoff for defining high serum UA significantly influenced the association with BC risk. This finding underscores the importance of considering the threshold values used in individual studies when interpreting the results. The significant association observed in studies with a UA cutoff ≥ 5.4 mg/dL suggests that higher UA levels may have a more pronounced effect on BC risk. However, the heterogeneity within subgroups defined by study country and age of women indicates that other factors, such as genetic and environmental differences, may also play a role. The subgroup analysis revealed a significant association between high serum UA and BC risk in studies from China and Africa, but not in studies from western countries. Similarly, a significant association was found in studies with the mean ages of the women ≥ 55 years, but not in studies with younger women. These findings should be interpreted with caution due to the substantial heterogeneity within these subgroups, suggesting that additional factors may influence the relationship between serum UA and BC risk.
The strengths of this meta-analysis include its adherence to PRISMA guidelines, comprehensive literature search, rigorous data extraction, and quality assessment using the NOS scores. The large sample size enhances the statistical power of our findings and allows for more reliable conclusions. Furthermore, the use of meta-regression and subgroup analyses provided insights into the sources of heterogeneity and the potential dose-dependent effect of serum UA on BC risk. However, several limitations should be acknowledged. First, the observational nature of the included studies precludes establishing causality. This meta-analysis included a mix of prospective cohort, retrospective, and case-control studies to comprehensively evaluate the relationship between serum UA levels and BC risk. While this approach broadens the scope of evidence and enhances statistical power, it may also introduce heterogeneity due to differences in study design. Subgroup analyses did not reveal statistically significant differences between study designs (P for subgroup difference = 0.11), but the potential for design-related bias cannot be excluded. Therefore, these findings should be interpreted with caution, and future meta-analyses focusing on a single study design, such as prospective cohort studies, may provide more methodologically consistent insights. Second, this meta-analysis was conducted using study-level data, as individual patient data were not available. Subgroup analyses, such as those stratified by age and cutoff of serum UA, were performed based on the mean ages and cutoffs used in each study, with the medians of continuous variables used as cutoff values to balance the number of studies in each subgroup. While this approach provides insights into potential age and cutoff-related differences, it does not allow for overlapping cohorts across subgroups, which is a limitation of study-level meta-analyses. Future research incorporating individual patient data would allow for a more precise assessment of age and UA-cutoff-specific associations. The variability in the cutoff values for defining high serum UA among studies may introduce bias and affect the comparability of results. In addition, subgroup analyses in this meta-analysis were conducted by stratifying studies to achieve a balanced number of studies per subgroup rather than based on sample size. While this approach ensures comparability, it limits the ability to assess whether sample size is a factor contributing to nonsignificant findings in certain subgroups. This limitation underscores the need for future research with individual patient data to provide more precise subgroup analyses. Moreover, the lack of data on menopausal status in most studies limits our ability to assess its impact on the association between UA and BC risk. Additionally, residual confounding by unmeasured or inadequately measured factors, such as dietary habits and genetic predispositions, cannot be ruled out. Finally, publication bias, although assessed and found to be low, remains a potential concern in any meta-analysis.
From a clinical perspective, our findings suggest that for women with serum UA > 4.5 mg/dL, a further elevated serum UA level could be a modifiable risk factor for BC, particularly in older women and those with higher UA levels. Monitoring and managing serum UA levels might be considered as part of a broader strategy for BC risk reduction, especially in populations with high prevalence of hyperuricemia. However, these findings should be interpreted with caution, and further research is needed to confirm the observed associations and to elucidate the underlying mechanisms. Prospective cohort studies with standardized definitions of high serum UA, detailed assessments of confounding factors, and data on menopausal status are warranted to validate our findings. Additionally, mechanistic studies exploring the role of UA in BC pathogenesis could provide valuable insights into potential therapeutic targets.
Conclusions
In conclusion, this meta-analysis suggests that serum UA may be nonlinearly associated with the risk of BC (U-shaped). The risk of BC increases with serum UA above 4.5 mg/dL, and a higher association between serum UA and the increased risk of BC could be observed in studies with cutoff of serum UA > 5.4 mg/dL.
The findings highlight the potential role of UA as a modifiable risk factor for BC and underscore the need for further research to confirm these associations and to explore the underlying mechanisms. Clinicians should be aware of the possible link between hyperuricemia and BC risk, and consider monitoring UA levels as part of a comprehensive approach to BC prevention. Future studies should aim to address the limitations of the current evidence and provide more definitive answers regarding the role of serum UA in BC development.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155241313469 - Supplemental material for Association between serum uric acid and the risk of breast cancer: A meta-analysis of observational studies
Supplemental material, sj-docx-1-jbm-10.1177_03936155241313469 for Association between serum uric acid and the risk of breast cancer: A meta-analysis of observational studies by Liping Yang, Ziyun Yu, Jin Zhu, Longbo He, Lieliang Wang and Qingfeng Luo in The International Journal of Biological Markers
Supplemental Material
sj-docx-2-jbm-10.1177_03936155241313469 - Supplemental material for Association between serum uric acid and the risk of breast cancer: A meta-analysis of observational studies
Supplemental material, sj-docx-2-jbm-10.1177_03936155241313469 for Association between serum uric acid and the risk of breast cancer: A meta-analysis of observational studies by Liping Yang, Ziyun Yu, Jin Zhu, Longbo He, Lieliang Wang and Qingfeng Luo in The International Journal of Biological Markers
Supplemental Material
sj-docx-3-jbm-10.1177_03936155241313469 - Supplemental material for Association between serum uric acid and the risk of breast cancer: A meta-analysis of observational studies
Supplemental material, sj-docx-3-jbm-10.1177_03936155241313469 for Association between serum uric acid and the risk of breast cancer: A meta-analysis of observational studies by Liping Yang, Ziyun Yu, Jin Zhu, Longbo He, Lieliang Wang and Qingfeng Luo in The International Journal of Biological Markers
Supplemental Material
sj-docx-4-jbm-10.1177_03936155241313469 - Supplemental material for Association between serum uric acid and the risk of breast cancer: A meta-analysis of observational studies
Supplemental material, sj-docx-4-jbm-10.1177_03936155241313469 for Association between serum uric acid and the risk of breast cancer: A meta-analysis of observational studies by Liping Yang, Ziyun Yu, Jin Zhu, Longbo He, Lieliang Wang and Qingfeng Luo in The International Journal of Biological Markers
Supplemental Material
sj-docx-5-jbm-10.1177_03936155241313469 - Supplemental material for Association between serum uric acid and the risk of breast cancer: A meta-analysis of observational studies
Supplemental material, sj-docx-5-jbm-10.1177_03936155241313469 for Association between serum uric acid and the risk of breast cancer: A meta-analysis of observational studies by Liping Yang, Ziyun Yu, Jin Zhu, Longbo He, Lieliang Wang and Qingfeng Luo in The International Journal of Biological Markers
Footnotes
Author contributions
Liping Yang and Qingfeng Luo designed the study. Liping Yang and Ziyun Yu performed database search, literature review, study identification, and quality evaluation. Liping Yang, Jin Zhu, Longbo He, and Lieliang Wang performed statistical analyses. Liping Yang and Qingfeng Luo interpreted the results. Liping Yang drafted the manuscript. All authors revised the manuscript and approved the submission.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article. The template data collection forms and data used for all analyses including the analytic code can be obtained upon reasonable request from the corresponding author.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical consideration
The data is collected from published studies and does not violate any ethical guidelines.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
