Abstract
Objective
The poor prognosis of cervical cancer patients leads to an annual increase in mortality, while microRNAs are involved in various cancers, including cervical cancer. This study aimed to investigate the clinical value and possible effect of miR-29b-2-5p on the progression of cervical cancer.
Methods
The expression level of miR-29b-2-5p in cervical cancer tissues and cells was analyzed by polymerase chain reaction. The Kaplan–Meier curve was used to evaluate the role of miR-29b-2-5p in cervical cancer prognosis. The independent prognostic factors of cervical cancer were explored by the multivariate Cox regression analysis. The effect of miR-29b-2-5p on the proliferation, migration, and invasion of cervical cancer cells was determined by in vitro cell experiments.
Results
A significantly downregulated miR-29b-2-5p expression was observed in cervical cancer tumor tissues and cervical cancer cells compared with the adjacent tumor tissues (tissues of the negative surgical margin) and H8 cells, respectively. Higher miR-29b-2-5p expression correlated with a better 5-year progression-free survival of cervical cancer. MiR-29b-2-5p was also associated with the indicators (tumor size, tumor differentiation, FIGO (International Federation of Gynecology and Obstetrics) stage, and invasion depth) of the progression of cervical cancer tumors. And miR-29b-2-5p, along with tumor size, tumor differentiation, FIGO stage, histology type, and invasion depth, were independent prognostic factors for poor cervical cancer prognosis. MiR-29b-2-5p showed a suppressive effect on the proliferation, migration, and invasion of cervical cancer cells.
Conclusions
MiR-29b-2-5p was downregulated in cervical cancer tumor tissues and could serve as an independent prognostic factor for cervical cancer. The overexpressed miR-29b-2-5p could be considered a tumor suppressor to inhibit the progression of cervical cancer.
Introduction
Cervical cancer (CC) is the fourth most prevalent cancer in women globally, and it is responsible for approximately 8% of all female cancer-related mortalities annually. 1 Notably, the incidence and mortality rates of CC are considerably higher in developing countries. 2 In addition, the age of onset is trending younger in the incidence of CC. 3 The most prevalent form of CC is squamous cell carcinoma, predominantly attributable to persistent exposure to the human papillomavirus (HPV). 4 Another common type of CC is adenocarcinoma and the prognosis is worse than squamous cell carcinoma. 5 CC may be asymptomatic in its early stages and as the tumor progresses, it will cause irreparable damage to CC patients. With the rapid progress of modern medicine, the mortality rate of CC has been falling gradually. 6 The primary intervention for early-stage CC is radical trachelectomy. However, postoperative infection, significant bleeding, and bladder and bowel dysfunction are common adverse effects of surgical techniques. 7 Treatment options for CC patients with refractory and metastatic advanced tumors are restricted, and the prognosis remains poor. 7 Consequently, an investigation into the molecular mechanisms underlying the progression of CC is beneficial in enhancing the comprehension of CC and in identifying novel therapeutic targets for the clinical management of CC. The identification of prognostic biomarkers of CC also represents a critical step towards predicting the likelihood of a poor prognosis and enabling the implementation of timely interventions for CC patients to avoid the progression.
MicroRNAs (miRNAs) constitute a category of endogenous small non-coding RNAs, with a length of approximately 20–24 nucleotides. A single miRNA may regulate multiple genes, and conversely, a single gene may be regulated by several miRNAs. MiRNAs are implicated in a range of essential biological processes, including early development, lipid metabolism, cell proliferation, migration, invasion, and differentiation. For instance, miR-214-3p could suppress the cell migration and invasion of CC by regulating the expression of THBS2 8 and overexpressed miR-760 could suppress the migration of Hela cells. 9 Furthermore, the dysregulation of miR-587, miR-19-3p, and miR-362 has also been demonstrated to exert significant regulatory effects on the cellular functions associated with CC progression.10–12 Given the regulatory function of miRNAs in fundamental cellular processes, an expanding cohort of researchers is engaged in the investigation of miRNAs as a potential novel therapeutic target of diseases and biomarkers for disease diagnosis and cancer prognosis.
MiR-29b-2-5p was observed to be dysregulated in various diseases such as dysfunctional digestive system, breast cancer, and colorectal cancer, and could serve as a diagnostic biomarker for various kinds of diseases.13–15 A study investigating miRNA involvement in lymph node metastasis (LNM) in patients with early CC revealed a strong correlation between miR-29b-2-5p and LNM, and the results indicated that CC patients with decreased miR-29b-2-5p expression were more likely to develop LNM. 16 A meta-analysis of the competing endogenous RNA (ceRNA) regulatory network associated with CC was conducted using a network of public databases and the results indicated that a lower miR-29b-2-5p expression level was associated with a worse prognosis in CC. 17 However, the association between CC prognosis and miR-29b-2-5p expression lacks the support of clinical trials. In light of the aforementioned information, we postulate that miR-29b-2-5p is involved in the progression of CC. This study was conducted to validate the correlation between miR-29b-2-5p expression and CC progression, the expression level of miR-29b-2-5p in CC patients was analyzed and the effect of miR-29b-2-5p on CC cells was also evaluated.
Subjects, materials, and methods
This research was a prospective study and included two parts: clinical research and an examination of the cell experiments. The clinical study was conducted to explore the clinical significance of miR-29b-2-5p in CC and the association between miR-29b-2-5p expression and CC prognosis. To further substantiate the role of dysregulated miR-29b-2-5p expression in CC progression, cell experiments were conducted to evaluate the effect of miR-29b-2-5p in CC, which may provide potential therapeutic target for CC treatment.
Subjects and materials
Clinical subjects
A total of 147 patients diagnosed with CC were enrolled in this study. All subjects underwent a Thinprep Cytologic Test (TCT) and HPV analysis at The Third Hospital of Shijiazhuang between January 2019 and December 2021. Subjects with squamous/glandular cell lesions and a positive HPV result underwent further examination by colposcopy and pathological biopsy. This study was permitted by the Ethics Review Committee of The Third Hospital of Shijiazhuang, and all subjects signed the informed consent after understanding the purpose of this research.
Inclusion criteria: (a) The ages of the patients were more than 18 years old; (b) patients were first diagnosed with CC by pathological diagnosis; (c) the pathological types were squamous cell carcinoma (hereinafter referred to as squamous) and adenocarcinoma; (d) No anti-cancer treatment before surgery was received by the patients; and (v) the clinical data of patients was complete.
Exclusion criteria: (a) Subjects with IIB stage and more advanced stage that require radiotherapy and chemotherapy; (b) patients combined with severe cardiovascular and cerebrovascular diseases and abnormal liver and kidney function; (c) patients under pregnancy or lactating; (d) patients with any infectious diseases; (e) patients with any immunodeficiency diseases and other malignant tumors; and (f) metastatic CC originating from the vagina, vulva, or endometrium.
Tissues
Surgical resection was performed to remove the tumor tissue of the CC subjects. Negative surgical margins of all the resected tumors were confirmed by pathological examination. The tumor tissues and the adjacent normal tissues (tissues of the negative surgical margin) of CC subjects were collected and kept at −80°C for later research. Pathological confirmation of the tumors was conducted by at least two clinical pathologists.
Cells
The H8 cells (immortalized human normal cervical epithelial cells) and four cell lines (SiHa, Hela, CasiKi, and C-33A) 18 were sourced from Sangon Biotech (Shanghai, China).
Methods
Clinical general data collection
Key indicators, including age, menopausal status, and tumor status including tumor size, tumor differentiation, International Federation of Gynecology and Obstetrics (FIGO) stage, histological type, and invasion depth of the tumor, were recorded and evaluated. A 24- to 60-month follow-up visit was conducted for all subjects, and a study of the 5-year progression-free survival was performed. Pertinent indicators such as survival time and the progression of the tumors used for progression-free survival data analysis were also collected in this study.
Analysis of mir-29b-2-5p expression levels in tumor tissues and CC cells
The collected tissues obtained from the subjects were smashed, and a Trizol reagent (Sigma, St Louis, MO, USA) was added to the tissues and cells for total RNA extraction. NanoDrop-2000 (Thermo-Fisher, Waltham, MA, USA) was utilized to evaluate the purity and concentration of the extracted RNA. Then the Invitrogen SuperScript reverse transcription kit (Thermo-Fisher) was used to reverse RNA to cDNA. The quantification of miR-29b-2-5p was analyzed on the 7500 quantitative reverse transcription-polymerase chain reaction (qRT-PCR) system (Applied Biosystems, Foster City, CA, USA) using the SYBR kit (Invitrogen, Waltham, MA, USA). Finally, equation 2−ΔΔCt was used to calculate the expression level of miR-29b-2-5p. U6 was selected as an internal reference.
Cell culture and transfection
The RPMI 1640 medium (Thermo Fisher) with 10% fetal bovine serum (FBS, Gibco, Big Cabin, OK, USA) was used for culturing cells, and the culture conditions were 37°C with 5% CO2.
Before transfection, the cells were counted and seeded on 96-well plates at a density of 1 × 103 cells per well and cultured in RPMI 1640 medium at 37°C with 5% CO2. The negative control (mimic NC) and miR-29b-2-5p mimic were sourced from Ribobio (Guangzhou, China). Overexpression of miR-29b-2-5p was performed by transfection of miR-29b-2-5p mimic. Transfection of mimic NC and miR-29b-2-5p mimic in Hela and SiHa cells was performed with the help of Lipofectamine 2000 (Invitrogen). Post-transfection, the cells were cultured in an incubator (Thermo) for 24–72 h.
The proliferation analysis of CC cells
The CKK-8 assay was utilized for the analysis of proliferation in both Hela and SiHa cells. After transfection with miR-29b-2-5p or mimic NC for different durations (0, 24, 48, and 72 h), each well of the plates was added with 10 μL of CKK-8 solution (Dojindo Molecular Technologies, Tabaru, Japan). Subsequently, the plates were incubated for another two hours. The absorbance at OD 450 of each well was analyzed within 15 min after incubation.
The migration and invasion analysis of CC cells
The evaluation of cell migration and invasion was conducted through the utilization of a transwell assay. Following a 24-h starvation period, the Hela and SiHa cells transfected with mimic and mimic NC were counted and then seeded in the upper chamber of the transwell plates (Corning, NY, USA). In the migration assay, the upper chamber was supplemented with FBS-free RPMI 1640 medium, while the bottom chamber was provided with RPMI 1640 complete medium, which served as the chemoattractant. The transwell plates were incubated for 24 h at the condition of 37°C with 5% CO2. Subsequently, the bottom chamber was removed, and the cells on the subface of the upper chamber were immobilized using methanol (Merck, Darmstadt, Germany) and stained with 0.1% crystal violet (Solarbio, Beijing, China). Following a 30-min interval, the subface of the upper chamber was rinsed with phosphate-buffered saline (PBS), and the cells on the subface of the upper chamber were observed and counted using a microscope (Nikon, Tokyo, Japan). In the invasion assay, Matrigel (BD Biosciences, San Jose, CA, USA) was pre-coated on the membrane in the upper chamber before seeding the cells, with the remaining procedures conducted in accordance with the migration assay.
Statistical analysis
The data analysis and diagram creation were conducted using SPSS and GraphPad Prism, with all data presented as the mean value ± standard deviation (SD). The clinical data exhibited a characteristic of normal distribution which was verified by the Probability-Probability (P-P) Plot. The statistical significance of the observed differences was assessed by the chi-square test and the t-test. The Kaplan–Meier curve was employed to evaluate the 5-year survival rate, and a multivariate Cox regression analysis was utilized to investigate independent prognostic factors for CC. Results were considered significant if the P value was less than 0.05.
Results
The mir-29b-2-5p expression level in CC tumor tissues and cells
The comparison of miR-29b-2-5p between the CC tumor tissues and normal para-cancer tissues was revealed in Figure 1(a). MiR-29b-2-5p expression levels were visibly downregulated in CC tumor tissues compared to normal tissues and the downregulation of miR-29b-2-5p expression was also observed in CC cells (CasiKi, Hela, SiHa, and C-33A) when compared to H8 cells (Figure 1(b)). The Hela and SiHa cell lines were selected for further research according to the expression level of miR-29b-2-5p. In comparison to the control group, a significant elevation in miR-29b-2-5p expression was observed in the miR-29b-2-5p mimic group in both Hela and SiHa cells. This result demonstrates the efficacy of the transfection process, indicating successful overexpression of miR-29b-2-5p in CC cells (Figure 1(c)).
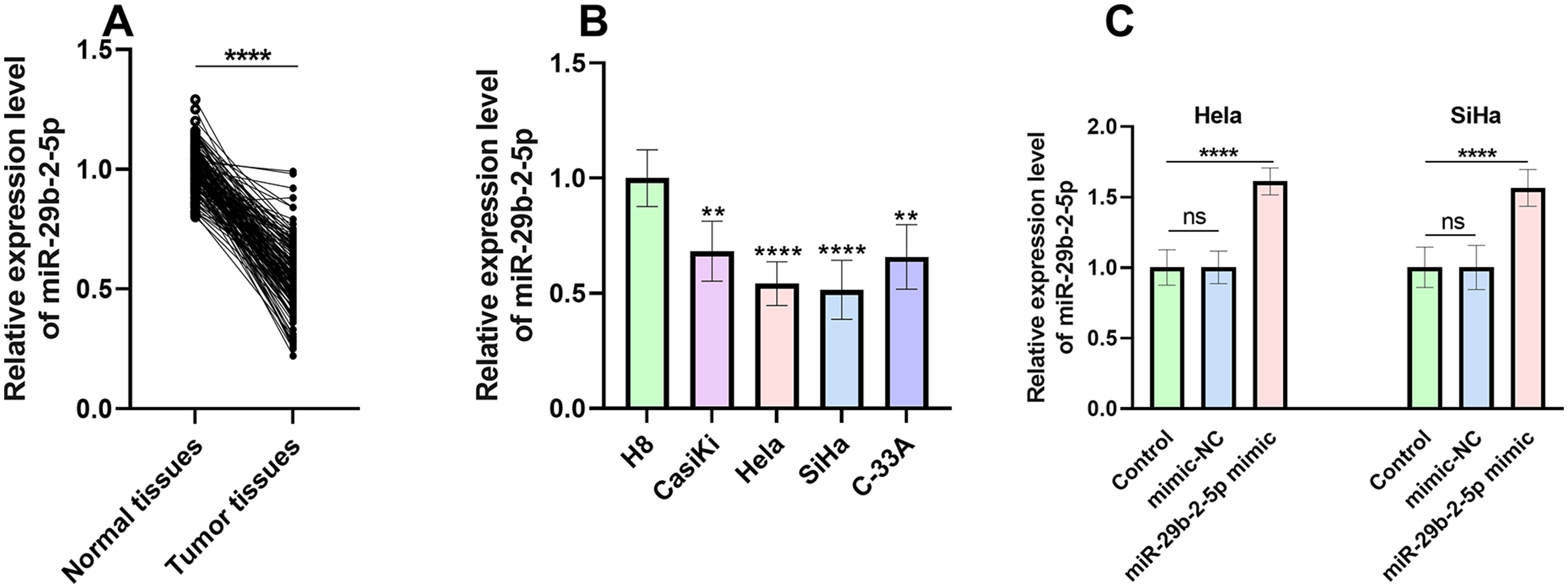
MiR-29b-2-5p expression in tissues and CC cells. (a) The comparison between the expression level of miR-29b-2-5p in normal adjacent tissues and tumor tissues. (b) The miR-29b-2-5p expression in CC cells. (c) The miR-29b-2-5p expression was significantly upregulated in CC cells after the transfection of miR-29b-2-5p mimic compared with the control group. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
The association between mir-29b-2-5p expression level and clinical indicators
A high miR-29b-2-5p expression group (n = 72) and a low miR-29b-2-5p expression group (n = 75) were categorized according to the average miR-29b-2-5p expression level in the CC tumor tissues. The correlation between clinical indicators (age, menopausal status, tumor sizes, tumor differentiation, FIGO stage, LNM, histology, and invasion depth) and miR-29b-2-5p expression levels was shown in Table 1. The expression level of miR-29b-2-5p showed no significant association with indicators such as age, menopausal status, and the histology types of the tumors, while the miR-29b-2-5p expression level was significantly correlated to tumor size (P = 0.018), differentiation (P = 0.026), FIGO stage (P = 0.017), and invasion depth (P = 0.010). Compared with the CC subjects with a high miR-29b-2-5p expression, more CC subjects with a low miR-29b-2-5p expression developed bigger tumors, poorer tumor differentiation, an advanced FIGO stage, and a deeper invasion depth, suggesting a close association between the low miR-29b-2-5p expression level and the positive progression of CC tumors. To further substantiate the correlation between tumor indicators and miR-29b-2-5p expression levels, a comparative analysis of miR-29b-2-5p levels in subjects exhibiting the aforementioned significant tumor indicators (tumor size, differentiation, FIGO stage, and invasion depth) was conducted and the result was presented in Supplementary Figure (SF) 1. There was a notable downregulation of miR-29b-2-5p expression in subjects presenting with a tumor size exceeding 2 cm, poor differentiation, FIGO stage IIA, and deep invasion compared with subjects with smaller tumor size, high or moderate differentiation, FIGO stage IA and IB, and superficial invasion. The close association between miR-29b-2-5p expression and the tumor indicators (tumor size, differentiation, FIGO stage, and invasion depth) was confirmed.
The correlation between the expression level of miR-29b-2-5p and the CC indicators.

CC: cervical cancer; FIGO: International Federation of Gynecology and Obstetrics.
The role of mir-29b-2-5p expression level on the prognosis of CC
The Kaplan–Meier survival curve was utilized to evaluate the 5-year progression-free survival (Figure 2). A better progression-free survival was observed in the high miR-29b-2-5p expression group compared with the low miR-29b-2-5p expression group, substantiating the association between low miR-29b-2-5p expression and the poor prognosis of CC. The independent prognostic factors for CC prognosis were assessed by a multivariate Cox regression analysis (Table 2). And miR-29b-2-5p was confirmed as an independent prognostic factor for predicting the poor prognosis of CC, with an hazard ratio (HR) value of 0.444 (95% confidence interval (CI) = 0.230–0.860, P = 0.016). Another four indicators, including tumor size (HR = 2.024, 95% CI = 1.073–3.818, P = 0.030), tumor differentiation (HR = 0.407, 95% CI = 0.185–0.896, P = 0.026), FIGO stage (HR = 2.700, 95% CI = 1.105–6.597, P = 0.029), histology (HR = 2.056, 95% CI = 1.049–4.032, P = 0.036), and invasion depth (HR = 3.054, 95% CI = 1.150–8.110, P = 0.025), were also identified as the independent prognosis factors for CC poor prognosis. According to the HR values, the miR-29b-2-5p expression level was positively correlated with a better prognosis of CC, while the bigger tumor size, advanced FIGO stage, positive LNM, histology, and deeper invasion depth were correlated with a poor prognosis of CC.
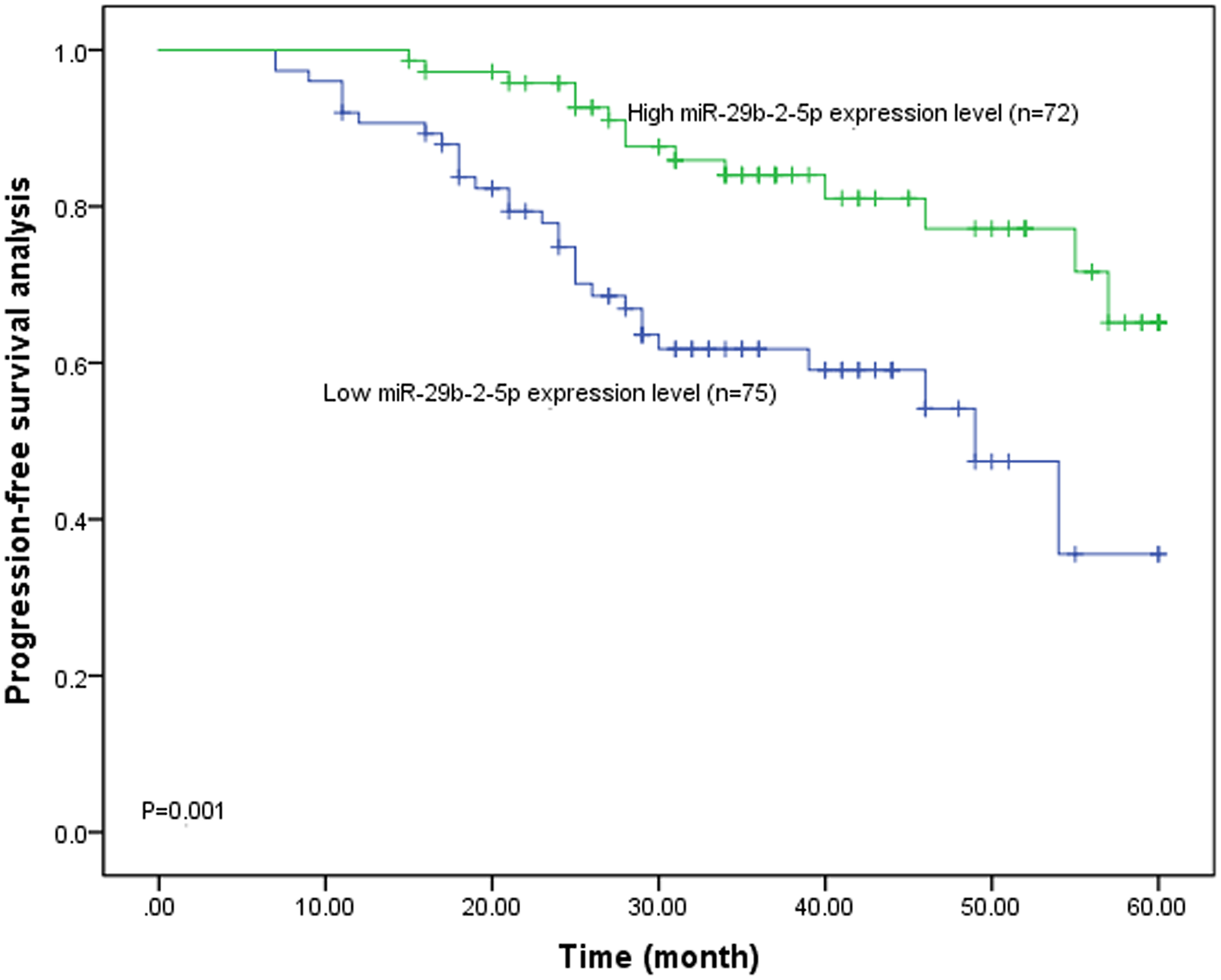
The Kaplan–Meier curve of CC subjects with different miR-29b-2-5p expression.
The evaluation of independent prognostic factors of CC poor prognosis by multivariate Cox regression analysis.
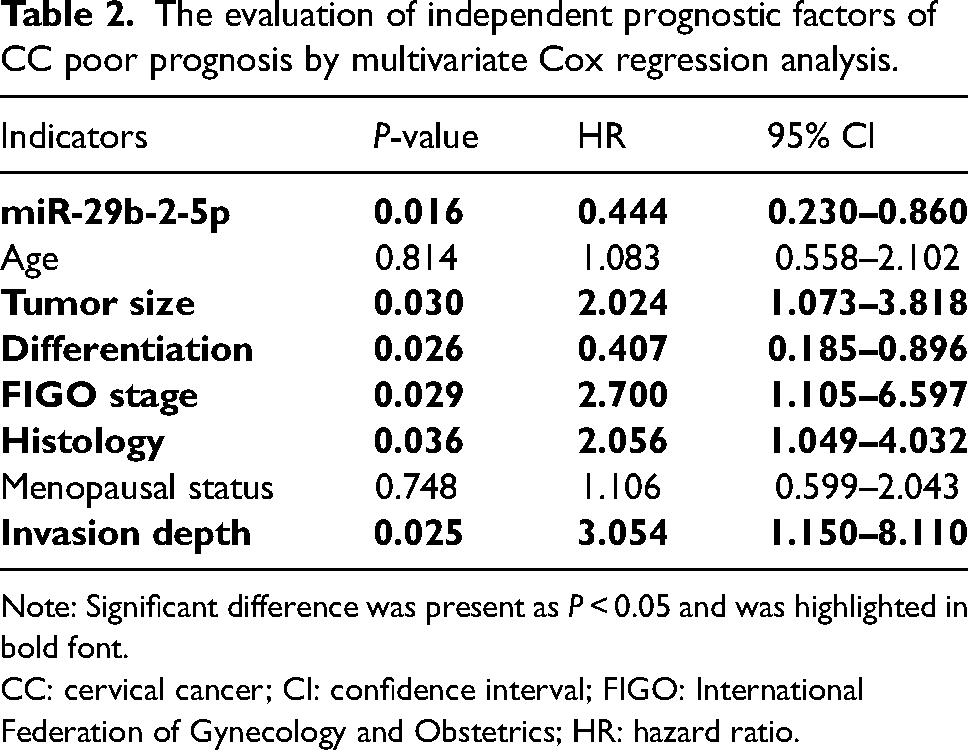
Note: Significant difference was present as P < 0.05 and was highlighted in bold font.
CC: cervical cancer; CI: confidence interval; FIGO: International Federation of Gynecology and Obstetrics; HR: hazard ratio.
The effect of mir-29b-2-5p expression level on the proliferation, migration, and invasion of Hela and SiHa cells
After 72 h of transfection of miR-29b-2-5p mimic in the Hela (Figure 3(a)) and SiHa (Figure 3(b)) cells, a notable reduction in the OD 450 value was observed in both the Hela and SiHa cells of the miR-29b-2-5p mimic groups in comparison to the control group, suggesting an inhibitory effect of miR-29b-2-5p on the proliferation of CC cells.
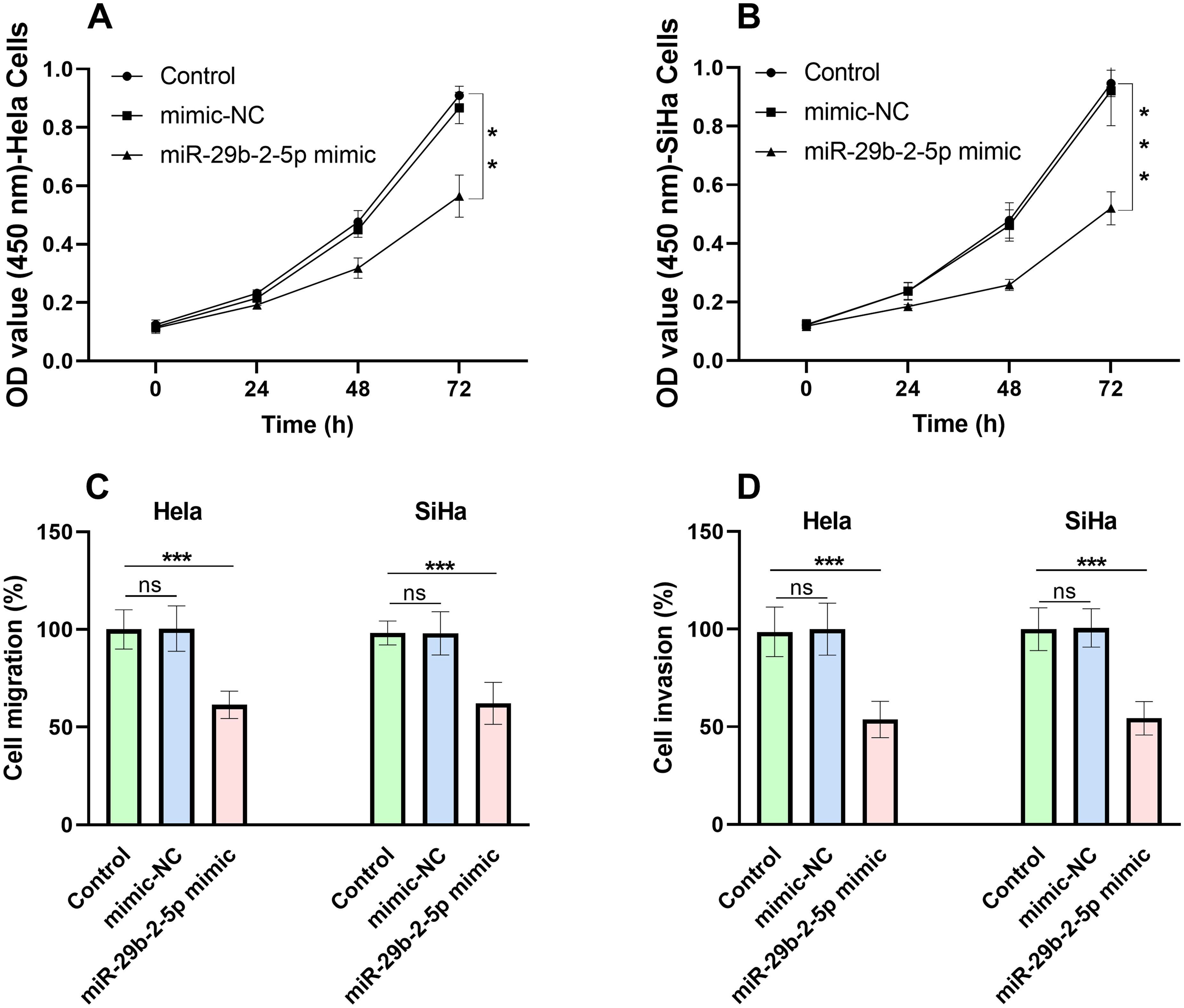
The proliferation, migration, and invasion of CC cells after the transfection of miR-29b-2-5p mimic. The proliferation of Hela (a) and SiHa (b) cells was obviously suppressed by the upregulated miR-29b-2-5p expression. The migration (c) and invasion (d) of Hela and SiHa cells were also significantly inhibited by the overexpressed miR-29b-2-5p. **P < 0.01, ***P < 0.001.
In the migration and invasion assay, the migration (Figure 3(c)) and invasion (Figure 3(d)) rates were visibly decreased in both Hela and SiHa cells of the miR-29b-2-5p mimic groups compared with the control groups, illustrating that the overexpressed miR-29b-2-5p could suppress the migration and invasion of CC cells.
Discussion
The existing treatment options for CC inevitably result in adverse effects on patients. 7 Despite CC patients accepting standard therapy, a significant proportion (29–38%) still experience a relapse, and the 5-year survival rate of advanced CC after recurrence is less than 5%.19,20 It is therefore imperative to discover a novel treatment for CC and identify an effective biomarker for CC prognosis to prevent the occurrence of CC progression. miRNAs attracted considerable attention due to their diverse biological functions, and for instance, dysregulated miR-29b-2-5p expression was observed in various diseases such as Alzheimer's disease, glioma, and pancreatic ductal adenocarcinoma.21–23 In our study, PCR was employed to analyze the miR-29b-2-5p expression due to its high sensitivity, high specificity, rapidity, simplicity, and low cost. MiR-29b-2-5p was found to be downregulated in CC and was identified as an independent prognostic factor for the prognosis of CC. Furthermore, miR-29b-2-5p could be considered the tumor suppressor of CC, which may provide a novel clinical therapeutic target for CC treatment.
Downregulation of miR-29b-2-5p was observed in both CC tumor tissues and four CC cell lines, demonstrating the protective value of a high expression level of miR-29b-2-5p on CC development. Tumor size, tumor differentiation, and depth of invasion are commonly employed indicators for the assessment of the severity of CC. These factors are explicitly delineated in the FIGO staging system, which serves as a guiding framework for the therapeutic management of CC. The FIGO stage was a proven risk factor for the overall survival (OS) rate of CC and significantly influenced the prognosis.24,25 Tumor size is correlated with the FIGO stage and larger tumors increase the risk of local invasion, metastasis of CC tumors, and treatment failure.26,27 Also, poor tumor differentiation and deeper invasion depth indicate greater tumor malignancy and worse prognosis.28,29 The invasive tumor cells could pass through the wall of the lymphatic canal, fall off with lymph fluid to the confluent lymph node, and grow the same tumors. According to the association between the expression level of miR-29b-2-5p and CC-related indicators, the close relationship between the expression of miR-29b-2-5p and CC severity was confirmed, and CC subjects with a low expression of miR-29b-2-5p were more likely to develop larger tumor size, poor tumor differentiation, advanced FIGO stage, and deeper invasion depth, which may demonstrate a poor prognosis. In patients with CC who exhibit lower expression of miR-29b-2-5p, a combination of surgical and radiotherapeutic or chemotherapeutic intervention may prove more efficacious in achieving therapeutic efficacy and reducing the risk of CC progression. In the 5-year progression study, a more favorable progression-free survival was observed in CC subjects with a high expression level of miR-29b-2-5p compared to CC subjects with a low expression level of miR-29b-2-5p, which provides further evidence of the close association between higher miR-29b-2-5p expression and a more favorable prognosis of CC. The multivariate COX regression results identified miR-29b-2-5p as an independent prognostic factor for CC, along with tumor size, tumor differentiation, FIGO stage, histology type, and invasion. Of the identified indicators, miR-29b-2-5p demonstrated the greatest significance in predicting poor prognosis for CC and the evaluation of miR-29b-2-5p expression was more effective, accurate, and convenient. It is recommended that greater attention be paid to CC patients with relatively low miR-29b-2-5p expression levels, with an increased frequency of re-examination to monitor the patient's condition and prevent the progression of CC. Proliferation, migration, and invasion represent fundamental cellular functions. In contrast to normal cells, which are capable of proliferation for a limited number of cycles, cancer cells can proliferate indefinitely. The growth of a tumor is dependent upon the proliferation of tumor cells, whereas the migration and invasion of cells are integral to the progression of tumor infiltration and metastasis. 30 In this study, the effect of the miR-29b-2-5p expression on cell proliferation, migration, and invasion was evaluated to explore the potential regulatory mechanism of miR-29b-2-5p on CC and the inhibitory effect of miR-29b-2-5p on tumor progression was identified, which indicated that miR-29b-2-5p may serve as a novel therapeutic target for CC treatment in the clinic.
Some limitations still exist in this research, such as the comparatively small sample size and the insufficient investigation of the regulatory mechanism of miR-29b-2-5p in CC. Only 147 patients with CC participated in this research, and this relatively small sample size may not reflect all the general characteristics of patients with CC worldwide. POU2F2 was reported to be dysregulated in CC in a previous study 31 and showed the highest target score with miR-29b-2-5p. Thus, miR-29b-2-5p may interact with POU2F2 to influence the development of CC, which should be investigated in further studies.
In summary, miR-29b-2-5p was downregulated in CC tumor tissues and could serve as an independent prognostic factor for CC prognosis. The overexpression of miR-29b-2-5p has been identified as a tumor suppressor, capable of inhibiting the progression of CC. This study may provide a novel therapeutic target for CC treatment.
Footnotes
Authors' contributions
Study concept and design: Wenhui Zhang and Qinghan Liu; analysis and interpretation of data: Qinghan Liu, Xi Chen, Wei Shang, Jinwei Cao, and Huijuan Zhao; drafting of the manuscript: Wenhui Zhang and Feng Jian; critical revision of the manuscript for important intellectual content: Qinghan Liu, Xi Chen, Wenhui Zhang, and Feng Jian; statistical analysis: Wei Shang, Qinghan Liu, and Xi Chen. Qinghan Liu and Xi Chen contributed equally to this study.
Availability of data and materials
All data generated or analyzed during this study are included in this article. Further enquiries can be directed to the corresponding author.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Consent for publication was obtained for every individual person's data included in the study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Approval was obtained from the ethics committee of The Third Hospital of Shijiazhuang. The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Hebei Provincial Medical Science Research Project (grant number 20231640).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
