Abstract
Biobanks are valuable tools for developing and applying scientific research and international cooperation through the collection of biological materials and their associated data. Systematic research following the Preferred Reporting Items for Systematic Review and Meta-Analysis guidelines was conducted in late 2022 in PubMed and Scopus, and generated 17 articles to be reviewed in depth and critically assessed using the Critical Appraisal Skills Programme Checklist due to the limited available data; 12 relevant health organizations and government websites outside of peer-reviewed journals were also included. Our research identified 44 biobanks in Latin America. In general, there is a lack of regulation and legislation guaranteeing the stored materials’ quality and institutional collaboration. We believe a consensus needs to be reached regarding the terminology and definitions used for biobanks. The design for informed consent should also be agreed upon to ensure the privacy of the data shared among institutions. In conclusion, in Latin America, there is a clear need for government support in creating specific procedures for biobanks and providing further support for existing biobanks.
Introduction
Globalization and recent discoveries in the “omics” field have increased the need to process large amounts of biological samples and link findings with epidemiological characteristics. This represents a significant challenge, not only for obtaining samples but also for ensuring their quality for analysis in the short or long term. 1 Biobanks—a collection of biological samples amassed for research purposes—are an optimal solution, permitting cooperative research and global collaborations to expedite the development and application of scientific knowledge through biological materials and their associated data.
Biobanks are also critical platforms where various disciplines, including molecular and genetic epidemiology, molecular pathology, pharmacogenomics, and pharmaco-proteomics, converge. 2 The samples stored in biobanks have widespread applications in diverse areas; they can be used to investigate how genetic changes can affect cancer risk, how material from cancer patients can be used to assess efficacy of available therapies, and be valuable for exploring innovative treatment approaches for cardiovascular and neurological diseases.3–5 Moreover, biobanks enable the investigation of patient–drug sensitivities and are particularly useful for monitoring natural disease history through sequential sampling. 6 Finally, they are key instruments in the fields of metabolomics and lipidomics by providing crucial insights into disease progression and prognosis. 7
Worldwide leading biobanks have millions of stored samples that have been used for research and innovation in healthcare systems. Derived from government, academic, and industry collaboration, telehealth tools, such as Electronic Health Records, can be integrated with biobanks, thus linking genomic research and health systems. 8 Despite this, studies show that up to 80% of the samples stored in these biobanks are of European ancestry, with an underrepresentation of other ethnic groups. 9
Latin America has a rich ethnic diversity; however, biological specimens from this region are not adequately represented in both American and European biobanks. Therefore, there is a need for biobanking development in these countries. For regional biobanks to be established they must meet international standards and regulations.
Such standards not only ensure safe and ethical management of samples but also include guidelines for applying informed consent, correct sample collection and storage, data management, and controlling access to samples and data. In Latin America, meeting these criteria can be challenging. Difficulties include the recruitment of trained personnel and adequate sample management and accrual; as a result, the collections of biological specimens can be of diverse quality. There is even variation in the interpretation of the term biobank and consequently the legislation around it. 10 The aim of this article is to review the establishment of biobanks and repositories throughout Latin America and identify its strengths and limitations to achieve its optimal biobank function.
Search strategy and selection criteria
A bibliographic search was conducted following the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) Statement in PubMed and Scopus databases using the following search strategy: (Biobank* OR Repository* OR Biorepository*) AND ((“Latin America” OR Mexico OR Mexican OR Brazil OR “Puerto Rico” OR Chile OR Colombia) AND (law OR Ethic* OR Regulation*)). The PRISMA checklist is available in Supplementary Table 1. The search was performed during September and October 2022. The inclusion criteria were peer-reviewed articles written in English and Spanish published from 2012 to 2022 on the implementation of biobanks, the quality and management of biospecimens, and the ethical aspects and legislation of biobanking in Latin America. To provide a comprehensive analysis of biobanking in Latin America, and due to the limited availability of literature on the subject, there was no exclusion criterion regarding study type; further research was carried out in relevant health organizations and government websites outside of peer-reviewed journals on the existing legal and ethical framework for biobanks in Latin America. Two researchers carried out data collection independently and assessed titles and abstracts to identify eligible articles. Each researcher individually assessed the selected articles using the Critical Appraisal Skills Programme Checklist (Supplementary Table 2). To decide whether to include an article, the researchers resolved all the discrepancies by consensus. The process of gathering information from health organizations and government websites was also carried out by the same two researchers; discrepancies of whether or not to include a certain document were resolved by consensus with a third researcher. Only articles published between 2019 and 2022 were included.
Characteristics of studies.

BB: biobank; BCH: Barretos Cancer Hospital; FDA: US Food and Drug Administration; HIPAA: Health Insurance Portability and Accountability Act; n.d.: no data; PCR: polymerase chain reaction; qPCR: quantitative PCR.
Current biobanks in Latin America.

Results
The search in Scopus returned 106 articles and PubMed returned 121: in total 227 articles were retrieved, 28 duplicates were removed, 153 articles were excluded by title and abstract screening, 46 articles were reviewed, and 29 were excluded because they did not address biobanking in Latin America as the focus of the article or only marginally addressed biobanks. The remaining 17 articles were reviewed in-depth and retained for the review, other articles were added by cross-referencing, and 12 additional reports retrieved from websites and organizations were included. Figure 1 shows the corresponding PRISMA flow diagram for this search and for the identification of studies using other methods. The search of other websites retrieved a total of 1227 documents. There were multiple reasons for excluding webpages: not having data published between 2019 and 2022, not written in English or Spanish, considered duplicate information (discussing the same biobank or regulation), and for not belonging to the official page of the institution, government, or health organization. Of the initial 1227 sites, a further 1167 were excluded by title, leaving 60 sources considered eligible. Of these a further 31 webpages were removed for not providing sufficient information about the respective biobank or regulation, and 17 were excluded due to concerns about the reliability of the published information, leaving only 12 webpages with relevant information.
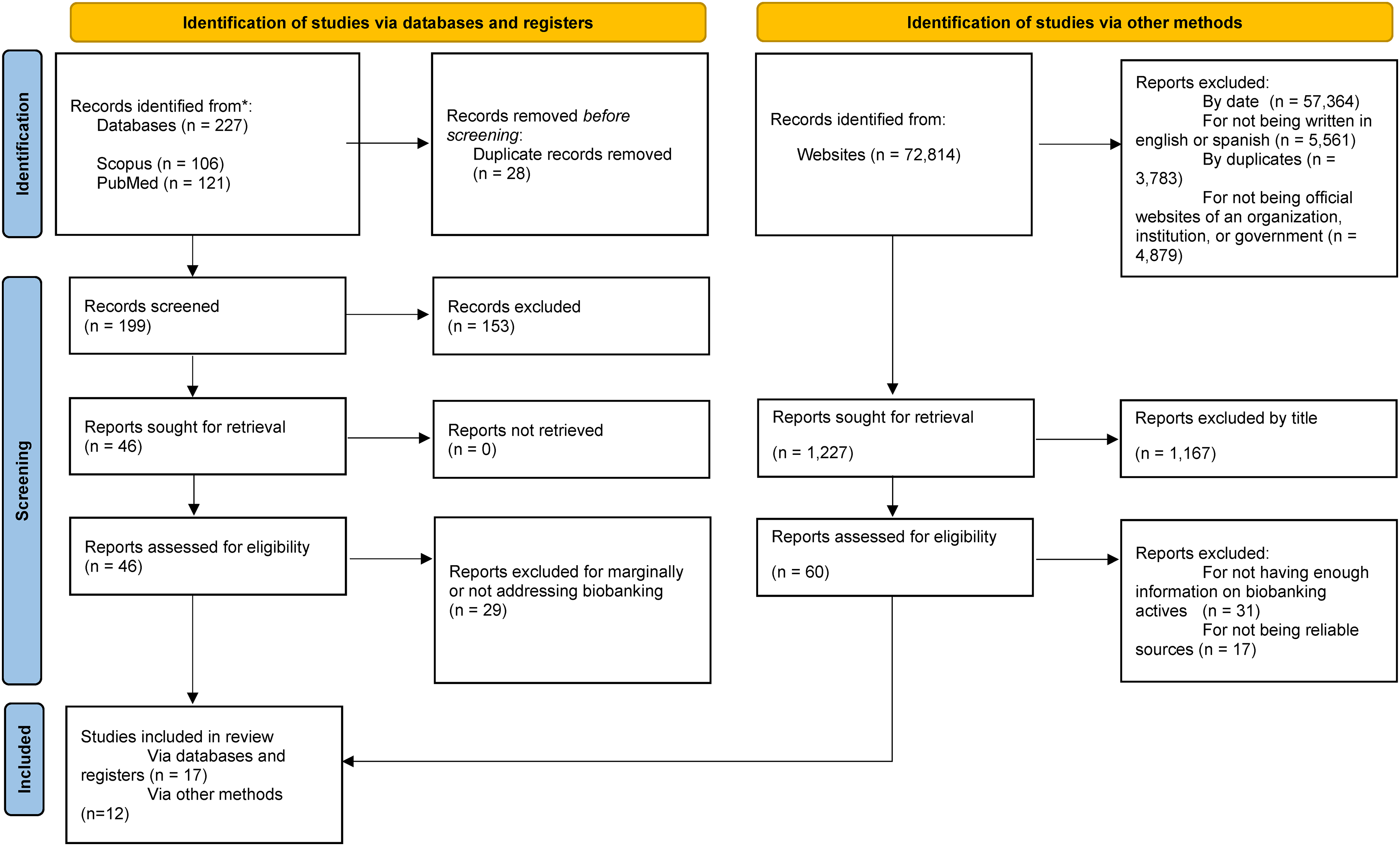
PRISMA flowchart.
Finally, our research identified 44 biobanks in Latin America; 89% were disease-oriented and 11% were population-based biobanks. Most of the biobanks were cancer-oriented (85%), although some were also dedicated to infectious diseases (8%) or neurological conditions (5%), and only 1 was oriented to metabolic disease. Chile had the highest number of biobanks with 11, followed by Mexico (n = 10) then Argentina (n = 6). There was a lack of legal and regulatory frameworks in most countries, though Brazil had the most robust regulations. Further details on these biobanks are shown in Table 2.
Characteristics of biobanks compared to biorepositories
Two terms are commonly used to refer to the collection and storage of biological samples: biobank and biorepository. However, despite sources using them interchangeably, they are not the same. According to the International Organization for Standardization (ISO) in Biotechnology—Biobanking—General requirements for biobanking 20387, one of the few norms addressing biobanks, a “Biobank” is a “legal entity or part of a legal entity that performs biobanking” and defines “biobanking” as the “collection, preparation, preservation, testing, analyzing, and distributing defined biological material as well as related information and data.” 25 In contrast, the International Society for Biological and Environmental Repositories (ISBER) defines “repositories” as “a formally managed physical or virtual entity that may receive, process, store, and/or distribute specimens and/or samples and their associated data as appropriate in support of current or future use […] it is a biorepository if the specimens are biological specimens.” 26 This means researchers can search the catalogs of a biorepository for a specific specimen to cover their particular needs. However, the key component that distinguishes biobanks from biorepositories is the governance mechanisms that enable biobanks to fully consolidate themselves as robust research institutions.
Biobanks can also form alliances with other national and international institutions to exchange biological material that is not meant to be static; for this reason, the biological samples must be closely associated with their epidemiological data, and the organization of collections must ensure an easy and accessible way to find material.
Due to these core differences, this distinction between biobank and biorepository is essential because while some activities are shared or similar, each has a purpose that might influence regulations and procedures.
Importance of governance in biobanking
The term governance encompasses several characteristics inherent to institutions, initially perceived as the capacity to exert control over the institution, its activities, and its members. However, within the context of biobanking, governance extends beyond mere control; it comprises formally constituted regulatory bodies, legal instruments, advisory and internal regulation committees, and internal policies specific to each biobank—and even encompasses the culture and values of the organization. 27 Kaye et al. 27 define “governance” as a complex compound of institutions, systems, structures, processes, procedures, practices, relationships, and leadership behavior. This multifaceted definition encapsulates the exercise of social, political, economic, and managerial/administrative authority in public or private affairs management. In essence, governance is composed of three fundamental elements: individuals, including the workforce, decision-makers, and institutions; internal procedures encompassing all biobanking activities; and a legal framework that encompasses not only governmental regulations but also the internal policies of each institution.
The significance of governance in biobanks is underscored by its relevant role in providing clarity and operational efficiency. 28 It establishes a transparent framework for rules and procedures, ensuring a standardized approach. Through the implementation of a robust governance system, ethical and legal research practices are upheld, providing a solid foundation for integrating biobanking efforts.
Types of biobanks
There is no consensus on one unique biobank classification due to their inherent heterogeneity and diversity. These organizations exhibit diverse approaches, encompassing differences in dedicated diseases, types of collected samples, inclusion criteria for patients in studies, varied sample processing methods, study designs, and overall size. The diverse nature of these organizations poses a challenge in creating a standardized classification system. Recognizing this complexity, Coppola et al. 29 propose a more comprehensive classification, categorizing biobanks into two primary groups: population-based and disease-oriented. 29
Population-based biobanks are characterized by extensive patient cohorts and relatively fewer inclusion criteria. 30 The primary goal of these biobanks revolves around exploring the interplay of genetic susceptibility and exposure to external factors in the development of diverse diseases. These biobanks predominantly collect DNA aliquots from all individuals, with a lesser emphasis on procuring tissues or other sample types. Among the largest population-based biobanks are the Danish National Biobank and the UK Biobank with over 5 million and 500,000 individuals, respectively.50,54
Disease-oriented biobanks are more prevalent, typically manifesting as smaller-scale repositories designed with a specific disease focus. Their primary objective consists of following disease pathogenesis and identifying potential therapeutic targets. These types of biobanks are valuable tools for case-control studies, basic research, and certain clinical trials. They store a variety of tissues and may range from specific organ samples to several organs throughout the body for the study of diseases such as cancer.29,30 Disease-oriented biobanks are often part of collaborative efforts among institutions; this collaborative effort enhances the capacity to undertake extensive research initiatives with significant scientific and medical implications. An example of a disease-oriented biobank is the AIDS Specimen Bank (ASB) of the University of California, San Francisco (UCSF) created in 1982 as a response to the HIV and AIDS pandemic. 32
Because of the limited information found on biobanks in Latin America, little data were retrieved regarding their functions and capacities. Identifying which institutions were biobanks or biorepositories was not possible, hence Table 2 uses the term “biobank” indistinctly since this was the denomination used by the organizations.
Latin America biobank legislation overview
Regarding laws and legislation at the international level, biobanks have their ethical framework in The Belmont Report of 1979 proposed by the National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research, which establishes the ethical principles for research in humans, taking as basic principles autonomy, justice, beneficence, and non-maleficence. The International Society for Biological and Environmental Repositories (ISBER) monitors, promotes, and influences the regulations and policies concerning biobanks and biorepositories worldwide. Another international institution is the ISO, which is dedicated to establishing international standards and guidelines. These quality standards aim to establish the minimum requirements for trust, reliability, and safety for the proper functioning of biobanks and their operation in all countries, and allow biobanks to obtain a certification that guarantees compliance with the quality standards proposed by the organization. It is evident that the purpose of these guidelines is to standardize and homogenize biobanking by addressing some key aspects, such as: the most appropriate type of informed consent to safeguard the integrity and privacy of the study subjects; the adequate method for the samples’ obtention; the ownership of the sample, since there is still no consensus whether the biospecimen belongs to the institution or the subject; the storage and protection of biological samples and data; the right of the subject to receive genetic, medical advice, or information about the genetic data obtained; and the right to partake in the benefits of the research.
Brazil. Brazil is the only country in Latin America with national standards and guidelines for biobanks. Brazil was the pioneer country to create specific national guidelines addressing the principles and standards for biobank operations and their bioethical, technical, and legal aspects. 23 Biobanking activities are based on the National Guidelines for Biorepositories and Biobanks published by the National Ministry of Health in 2011 and the Resolution of the National Health Council (Conselho Nacional de Saúde; CNS) 441/11. 36 These guidelines define biobanks as collections of human bio-specimens associated with their sociodemographic and medical data for research and non-profit purposes. They state that the collection of every sample must include informed consent for each study or a single consent permitting authorization for future studies. Guidance is provided on the legal ownership of the stored biological material of the research subjects. The guidelines also mention that the biobank has the responsibility for the indefinite preservation of biological material while the sample is still viable. The rights of study subjects are also outlined, and these include the right to know all the information derived from the sample; this includes potential genetic risk for hereditary diseases if the subject decides to be told.
Puerto Rico. Since Puerto Rico is an unincorporated territory of the United States of America, many of the federal laws also apply to this country. The main regulations that govern biobanking are the 45 CFR 46 or Common Rule of the Office, Human Research Protections, which are regulations for the protection of human subjects in research, and the US Food and Drug Administration regulations that deal with all biomedical interventions that may lead to the development of a new treatment, drug, or diagnostic/prognostic biomarker. The Health Insurance Portability and Accountability Act (HIPAA) Privacy Rule establishes the requirements to protect the privacy of health information and limits the use of the data obtained in the sample without the subjects’ consent. The Occupational Safety and Health Administration (OSHA) has guidelines on biosafety and sample handling. 13
Mexico. The Mexican legal framework governing the National Health System is divided into different regulations; however, the central axis is the General Health Law, which provides research development. The regulations and laws applicable to genomic research and tissue and sample handling are described below, highlighting the most relevant aspects concerning biobanks.
Mexican General Health Law. In its Fifth Title of Health Research, the Mexican General Health Law establishes the foundations for research on human health, within which the requirements for protecting the integrity of the research subject and the minimum requirements of the institution conducting the research are included. Title Fourteenth Article 317 and 317 Bis addresses the requirements so that organs, blood and plasma, cells, plus other tissues and their components from which genetic material can be obtained can leave the country. In general, there is no authorization for this scenario; however, Article 317 Bis mentions that they may be transferred outside the national territory for population genomic research, for which the corresponding permits must be obtained from the National Institute of Genomics and the Ministry of Health. Article 317 Bis 1 mentions that such material may not be used for purposes other than those established in the permit. 48
Regulation of the General Health Law on Research for Health (Mexico). The Regulation of the General Health Law on “Research for Health, in its Second Title Chapter I” discusses the ethical aspects of research in human beings in which dignity, confidentiality, the protection of individuals and rights and welfare of the research subject are defined. The Second Title Chapter VI of the “Research on Organs, Tissues and their Derivatives, Products, and Corpses of Human Beings” states the importance of the processes related to obtaining and processing said samples for research purposes. The Third Title Chapter III discusses new research areas, and confirms that any research that has not previously been considered in these regulations must be authorized by the Ministry of Health with the corresponding scientific foundations and such research must be aligned to the provisions of the Law. 20 In this regulation, even though some ethical dilemmas are alluded to regarding the dignity, confidentiality, and protection of research subjects and the handling of samples and tissues obtained from them, nothing concrete is mentioned about the existence of biobanking.
Mexican Official Normative. The Official Mexican Standard (Mexican Official Normative; NOM) NOM-004-SSA3-2012 for the Medical Record discusses informed consent, and emphasizes the patients’ autonomy, which means that they have the right to decide whether to receive care or participate in clinical research. 48 Subsection 10.1.2 of this NOM mentions the procedures that require informed consent, which include organ and tissue donation and clinical research in humans. In subsection 10.1.3, there is an opportunity for health professionals to make a specific type of informed consent if considered pertinent for other activities different from the ones mentioned in the NOM; this feature can be used for the creation of a variety of informed consents that have been used internationally.
In addition to this, the NOM-012-SSA3-2012 establishes the minimum criteria for the execution of research projects for health in humans. 55 These sets of regulations emphasize the importance of informed consent, the role of the researchers, the rights and protection of the research subjects, and the content and authorization of the research protocols by the different committees and the institutions where the research is carried out.
These guidelines and legislation cover some aspects of biobanking; however, nowhere in the Mexican Health Law are biobanks explicitly mentioned, making Mexican biobanking regulations scattered, fragmented, and mostly unclear, resulting in challenges for genomic research and the development of biobanks. The implementation of specific legislation based on international guidelines is necessary to standardize and improve the function of Latin American biobanks and biorepositories.
Discussion
Biobanks are helpful tools within the research area that can accelerate significant breakthroughs and discoveries. In other geographical regions as the UK and US, biobanks have enhanced research and potentiated its scope. In Latin America, there is a lack of standardization regarding definition, procedures, and legal framework, diminishing their impact in this region. The difference between the terms “biobank” and “biorepository” should be initially well established in guidelines and legislation to lessen confusion. Similarly, the development of more intuitive databases facilitates the exchange of samples and allows global collaboration, ensuring data protection and donors’ privacy. A consensus should be reached regarding general terminology, search terminology, and informed consent design to ensure the legality of the data shared on all continents. Adequate informed consent and protection of a participants’ privacy and confidentiality are paramount to maintain biobanking initiatives. Establishing clear and robust ethical guidelines in alignment with international standards will not only safeguard the rights of participants but also foster collaborations and data-sharing initiatives among countries in the region. This goal could be fulfilled with the experience and expertise of organizations such as ISBER and ISO; their guidelines and good practice recommendations would be useful for building a legal framework that could be adopted by countries interested in global collaboration.
Biobanks are key in advancing personalized medicine and translational research, serving as valuable resources for studying complex diseases and exploring their specific treatments. In the systematic search, many studies emphasize access to high-quality biological samples and associated data, and consider that biobanks can bridge the gap between basic research and clinical applications, serving an essential need for the region. In personalized medicine, where individual genetic variances and the influence of external factors significantly impact treatment responses, biobanks enable the identification of biomarkers, validate therapeutic targets, and foster the development of more effective treatments for these complex diseases. This review underscores a critical need for higher standards in quality assurance and quality control, plus requisite certification and accreditation.
In Latin America, among all the biobanks listed in Table 2, 89% are disease-oriented, while only 11% are population-based. These findings align with existing literature29,30,53; with population-based projects being less frequent and disease-oriented biobanks more prevalent, the majority are dedicated to cancer (85% in Latin America). When comparing these biobanks with their counterparts in Europe and the US, such as the UK Biobank or the All of Us Research Program, respectively, similarities and differences emerge. One prominent distinction lies in their scale: while the UK Biobank boasts around 500,000 participants and the All of Us program aims for at least 1 million participants, the largest biobanks in Latin America encompass no more than 100,000 participants.47,49,50 This discrepancy could largely be attributed to funding mechanisms. In Latin America, most biobanking projects rely on small grants or institutional funding, contrasting with the UK Biobank, which operates as a charitable entity receiving contributions from various organizations and government sources. 1 However, long-term financial sustainability remains a concern across all biobanks, with no guaranteed funding continuity. 50 Moreover, disparities exist in knowledge and data-sharing practices among regions. In Latin America, biobanks predominantly share data with external researchers collaboratively, while the UK Biobank and All of Us prioritize open access, ensuring equitable data accessibility for bona fide researchers.
The Latin American biobanks (Figure 2) have the capacity to recruit a large amount of population through both public and private sectors, as well as existing alliances among these countries. This can be boosted by creating national campaigns promoting and raising awareness of biobanks and their benefits. However, some weaknesses and areas of opportunity need to be addressed to ensure biobanks’ success in Latin America. An important barrier to the creation of biobanks and biorepositories is funding. Most biobanks are non-profit facilities; thus, the operating costs need to be considered to guarantee continued function with the highest quality standards. Coupled with this, the high equipment costs, maintenance, training, or even the consideration of emergencies, such as natural disasters, may be a low priority in Latin American countries.
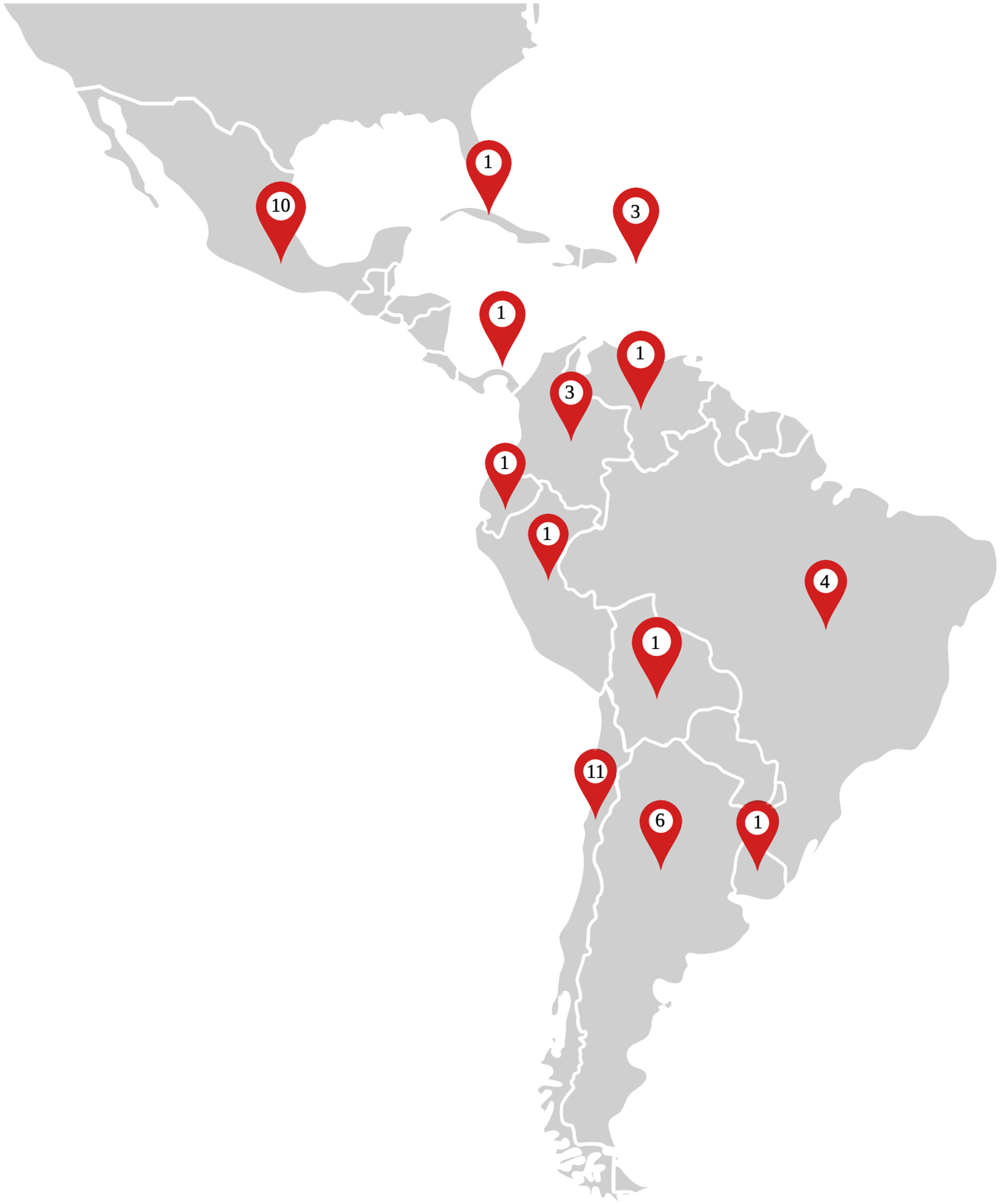
Distribution and quantity of biobanks across Latin America.
Governance is an essential aspect of biobanking for the biobanks to be established as robust and recognized institutions. In the case of the UK Biobank, effective governance has proven indispensable in securing regulatory approvals for its operations and fostering robust relationships with stakeholders.50–52 Brazil possesses the region's most robust legal and regulatory framework, enabling their biobanks to be effective organizations. Interestingly, Brazil does not have the highest number of biobanks; perhaps the governance of biobanks in this country has facilitated the pooling and convergence of biobanking efforts in the creation of large multicentric biobanks. Countries with a more significant number of biobanks, such as Mexico and Argentina, lack specific regulations and legal frameworks. In addition, the presence of many small biobanks may contribute to the lack of uniformity in procedures and the absence of governance, hindering the consolidation of efforts to establish themselves as recognized institutions.
Conclusion
The advancement of biobanking in Latin America is contingent upon governmental initiatives aimed at reviewing, developing, legislating, and regulating biobanks, thereby facilitating the establishment of governance and their evolution into robust research institutions. This supportive infrastructure will notably benefit already established biobanks, fostering a cost-effective investment for the Latin American region. As governments take proactive measures to provide a regulatory framework, biobanks can significantly contribute to medical and scientific progress, offering valuable resources for research and personalized medicine initiatives. The implementation of effective governance mechanisms ensures ethical conduct, legal compliance, and transparent operations, thereby enhancing public trust and facilitating collaboration with various stakeholders in the pursuit of advancements in healthcare and biomedical research.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155241239672 - Supplemental material for A systematic review of biobanks in Latin America: Strengths and limitations for biomedical research
Supplemental material, sj-docx-1-jbm-10.1177_03936155241239672 for A systematic review of biobanks in Latin America: Strengths and limitations for biomedical research by J. Adrián Rivera-Alcántara, Natalia Esparza-Hurtado, Gabriela A. Galán-Ramírez, Ivette Cruz-Bautista, Roopa Mehta, Carlos A. Aguilar-Salinas and Alexandro J. Martagon in The International Journal of Biological Markers
Supplemental Material
sj-xlsx-2-jbm-10.1177_03936155241239672 - Supplemental material for A systematic review of biobanks in Latin America: Strengths and limitations for biomedical research
Supplemental material, sj-xlsx-2-jbm-10.1177_03936155241239672 for A systematic review of biobanks in Latin America: Strengths and limitations for biomedical research by J. Adrián Rivera-Alcántara, Natalia Esparza-Hurtado, Gabriela A. Galán-Ramírez, Ivette Cruz-Bautista, Roopa Mehta, Carlos A. Aguilar-Salinas and Alexandro J. Martagon in The International Journal of Biological Markers
Footnotes
Author contributions
JARA, NEH: Conceptualization, investigation, methodology, writing – original draft & editing. AJM: Conceptualization, supervision, writing – original draft, review & editing. CAAS, GAG-R, ICB, & RM: Supervision, writing – review & editing. JARA and NEH contributed equally to this work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
No ethical approval was required for this manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Guarantor
AJM.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
