Abstract
The expression of programmed cell death-ligand 1 (PD-L1) on circulating tumor cells offers a noninvasive method for the detection of PD-L1 expression in lung cancer, and could serve as a potential surrogate for cancer tissue. However, discrepant results make it difficult to apply PD-L1 on circulating tumor cells to clinical practice. Therefore, we conducted a meta-analysis to investigate the diagnostic value of PD-L1 on circulating tumor cells in lung cancer. To identify the relationship between the expression of PD-L1 on circulating tumor cells and lung cancer, the PubMed, Web of Science, Embase, China National Knowledge Infrastructure, and Wanfang databases were searched from inception to March 2023. The pooled sensitivity, specificity, positive likelihood ratio, negative likelihood ratio, diagnostic odds ratio, and the corresponding 95% confidence intervals were calculated to assess the diagnostic performance of PD-L1. We also conducted subgroup and sensitivity analyses. A total of 11 studies including 472 lung cancer patients were included in our study. The overall performance in terms of pooled sensitivity and specificity was 0.72 (0.52–0.86) and 0.54 (0.25–0.81), respectively. The positive likelihood ratio, negative likelihood ratio, and area under the curve were 1.57 (0.87–2.84), 0.52 (0.30–0.90), and 0.70 (0.66–0.74), respectively. Deeks’ funnel plot test indicated no publication bias. Our analysis demonstrated that positive PD-L1 expression on circulating tumor cells (CTCs) exhibited a moderate diagnostic value in lung cancer, and CTCs may serve as a feasible alternative tissue analysis for the detection of PD-L1 in lung cancer.
Introduction
Lung cancer is the second most commonly diagnosed cancer worldwide and the first common cause of cancer-related mortality according to 2020 global cancer statistics. 1 In the past few decades, encouraging progress in molecular targeted treatment for lung cancer therapy has been achieved. 2 Immune checkpoint inhibitors (ICIs) were developed to target inhibitory checkpoint molecules. Among them, blockade of programmed cell death ligand 1 (PD-L1) interaction with its receptor programmed cell death 1 (PD-1) is a major breakthrough in the individualized treatment of lung cancer based on the individual genetic background, 3 and is currently used as first- and second-line therapies for non-small-cell lung cancer (NSCLC).
Researchers have demonstrated that the elevated level of PD-L1 in tumor tissues enables it to escape immune surveillance by inhibiting the activation of immune cells, and targeted PD-L1 could be beneficial for lung cancer patients with a high expression of PD-L1.4,5 While some objections have been raised, recent studies have indicated that a large number of patients with high PD-L1 expression in tumors do not benefit from the first-line pembrolizumab (anti-PD-1) antibody, and this activity has also been observed in patients with PD-L1-negative tumors.6–8 The reason for these findings may be the spatio-temporal heterogeneity of PD-L1 expression in the tumor and the different detection methods.
It is well known that cancer evolves in the course of disease, especially under the influence of treatment. Considering the heterogeneity of tissues, the development of new metastatic sites and dynamic changes of PD-L1 expression may impact the status of PD-L1 in the process of disease evolution and treatment. 9 Hence, utilization of tumor tissue for real-time assessment of the tumor profile may not accurately reflect the current PD-L1 protein expression status. Currently, liquid biopsy has attracted wide attention due to its low invasion, convenient collection, and dynamic real-time characteristics. 10 Circulating tumor cells (CTCs) may enter the blood circulation after being separated from the tumor tissue of the primary tumor or metastatic tumor; thus, CTCs hold promise to better reflect the heterogeneity of tumors compared to tissue biopsies. Studies have found that the expression of PD-L1 in CTC is twice as high as in tumor tissue, and the expression of PD-L1 in CTC can predict the prognostic value of cancer.11,12 Also, research has found that CTC could be used as a substitute for tumor tissue and as a material source for detecting gene changes and expression of the therapeutic target. However, controversial diagnostic value of PD-L1 expression on CTCs in lung cancers has been raised. Some researchers13,14 suggested that there is no correlation between PD-L1 expression on CTCs and on tumor tissue. Other studies showed that there is 93% concordance between tissue and CTCs. 15
The inconsistent results may be due to the different therapeutic regimens, CTC enrichment methods, PD-L1 detection methods, or tumor stages. Therefore, we performed a meta-analysis to systematically assess the diagnostic value of PD-L1 expression on CTCs in lung cancers.
Materials and methods
We systematically searched the PubMed, Web of Science, Embase, China National Knowledge Infrastructure (CNKI) and Wanfang databases from inception to March 2023. The search key words were as follows: (lung cancer and/or neoplasm or carcinoma) and (CTCs or circulating tumor cells) and (programmed death ligand 1 or PD-L1 or CD274).
To retrieve the relevant articles, two independent reviewers first screened the titles and abstracts, then the full texts of articles were further accessed. The differences between reviewers were resolved through discussion or consultation with the third researcher until a consensus was reached. Articles were included if they met the following inclusion criteria: (a) lung cancer must be histologically or cytologically confirmed; (b) PD-L1 must be detected both by circulating tumor cells and in tumor tissue; and (c) sufficient data were available to construct the 2 × 2 contingency table. In addition, the exclusion criteria were: (a) reviews, letters, or case reports; (b) animal or cell-line articles; (c) tumor tissue and blood samples were unpaired; and (d) inefficient in providing data for a 2 × 2 contingency table.
Data retrieval and quality assessment
Two researchers extracted information from eligible studies. The information was: first author, publication year, country, number of samples, CTC enrichment platform, PD-L1 detection method, and necessary data, including specificity (SPE), sensitivity (SEN), and area under the curve AUC). In addition, in order to evaluate the quality of the included articles, a Diagnostic Accuracy Study Quality Assessment 2 (QUADAS-2) was conducted. The QUADAS-2 tool mainly consists of four parts: (a) patient selection, (b) index test, (c) reference standard, and (d) flow and timing. Each domain is used for bias assessment, and the first three domains are used to evaluate the clinical applicability of these studies.
Statistical analysis
Stata 12.0 was adopted for the meta-analysis, and P < 0.05 was considered statistically significant. Based on the eligible articles, pooled SEN and SPE, positive likelihood ratios (PLR), negative likelihood ratios (NLR), diagnostic odds ratios (DOR), and corresponding 95% confidence intervals (CIs) were calculated using a random effects model. Moreover, the summary receiver operator characteristic (SROC) curve and the area under the SROC curve (AUC) were calculated to estimate the diagnostic performance of PD-L1 on CTCs for lung cancer.
Heterogeneity was assessed by Cochran's Q and I-squared (I2) tests, and P < 0.05 or I2 > 50% indicated significant heterogeneity. A subgroup analysis was conducted by study region, tumor stage, detection method, and treatment. Moreover, a Fagan nomogram was adopted to evaluate the clinical utility. Deeks’ funnel plot was used to examine publication bias.
Results
Literature retrieval and selection
Initially, we searched 258 articles in total from PubMed (196), Web of Science (34), EMBASE (15), CNKI (10), and Wanfang (3). Among them, 25 articles were excluded because of duplicates. After screening for the title and abstract, 215 articles were removed for the following reasons: reviews, case reports, meeting, not describing PD-L1 on CTCs with lung cancer, and animal or cell-line articles. Then, the full texts of the remaining articles were reviewed. Due to insufficient data, CTCs and tissues did not match, and 7 articles were eliminated. Finally, 11 eligible studies were included in our meta-analysis. The flow chart of the whole selection process is shown in Figure 1.
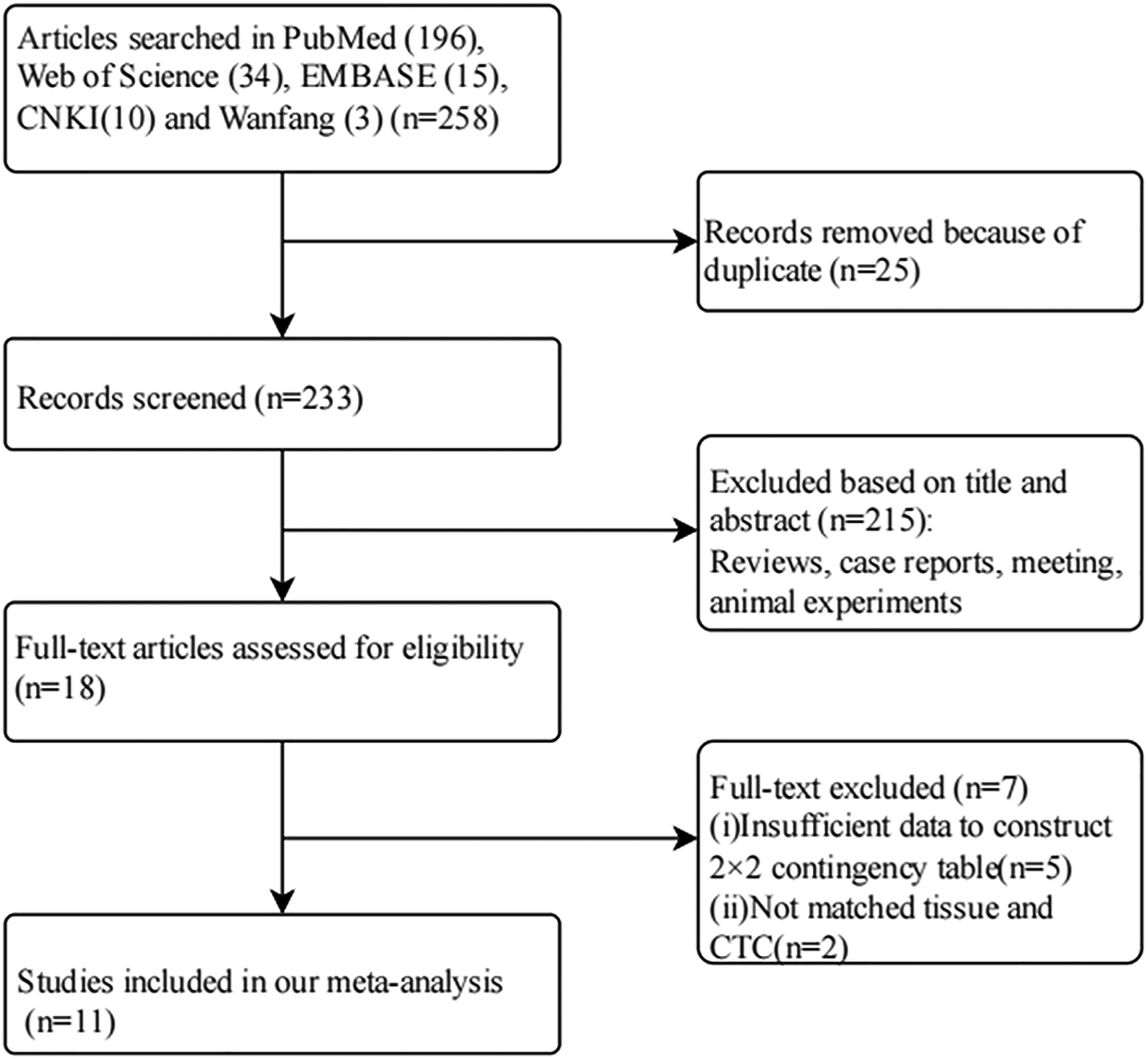
Flow chart for the literature search and study selection.
Study characteristics and quality assessment
A total of 11 studies with 472 patients were included in our meta-analysis. Five studies16–20 were mainly from Asia, and 621–25 contained a large European population. Five studies16–19,24 included patients with all stages of lung cancer, and 620–23,25 included only patients with advanced lung cancer. Patients in 7 of the studies were new patients,16–21,24 while patients in 4 of the studies were previously treated.15,22,23 The isolation by size of epithelial tumor cells (ISET)15,16,22 were used in 3 articles to separate CTCs; four articles adopted Cell Search17,18,23,25 to separate CTCs, while the label-independent Parsortix system, 21 MCA system, 19 microfilter platform, 24 and negative enrichment method 20 were used in the other 4 articles. In 4 studies15–17,22 PD-L1+ CTCs cut off ≥1%; 5 studies18,20,21,23,25 PD-L1+ CTCs cut off ≥1; 1 study 24 PD-L1+ CTCs cut off ≥3; and 1 study 19 PD-L1+ CTCs cut off ≥1. The main characteristics of the studies are summarized in Table 1. According to the assessment of the QUADAS2 checklist, the quality of research articles was not “low risk,” making them fit for further analysis. (Supplementary Figure 1, available online at www.biological-markers.com—Assessment of the risk of bias).
Main characteristics of included studies.
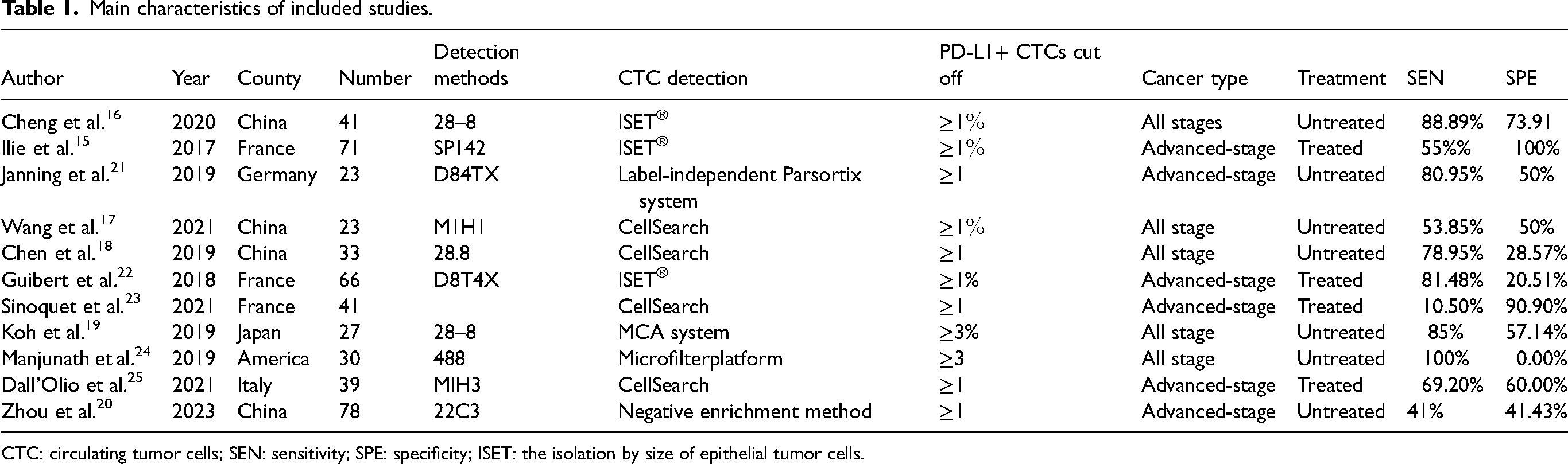
CTC: circulating tumor cells; SEN: sensitivity; SPE: specificity; ISET: the isolation by size of epithelial tumor cells.
Diagnostic value of PD-L1 on CTCs
We calculated the sensitivity, specificity, positive likelihood ratio, negative likelihood ratio, DOR, and the corresponding 95% CIs. The overall SEN and SPE were 0.72 (0.52−0.86) and 0.54 (0.25–0.81), respectively, (Figure 2). The pooled PLR and NLR were 1.57 (0.87–2.84) and 0.52 (0.30–0.90), respectively. (Supplementary Figure 2, available online at www.biological-markers.com—Forest plot of positive likelihood ratio and negative likelihood ratio.) The DOR was 3 (1.06–8.52). (Supplementary Figure 3, available online at www.biological-markers.com—Forest plot of diagnostic odds ratio.) In addition, the AUC of the SROC curve was 0.70 (0.66–0.74) (Figure 3); thus, the diagnostic value of PD-L1 on CTCs was moderate.
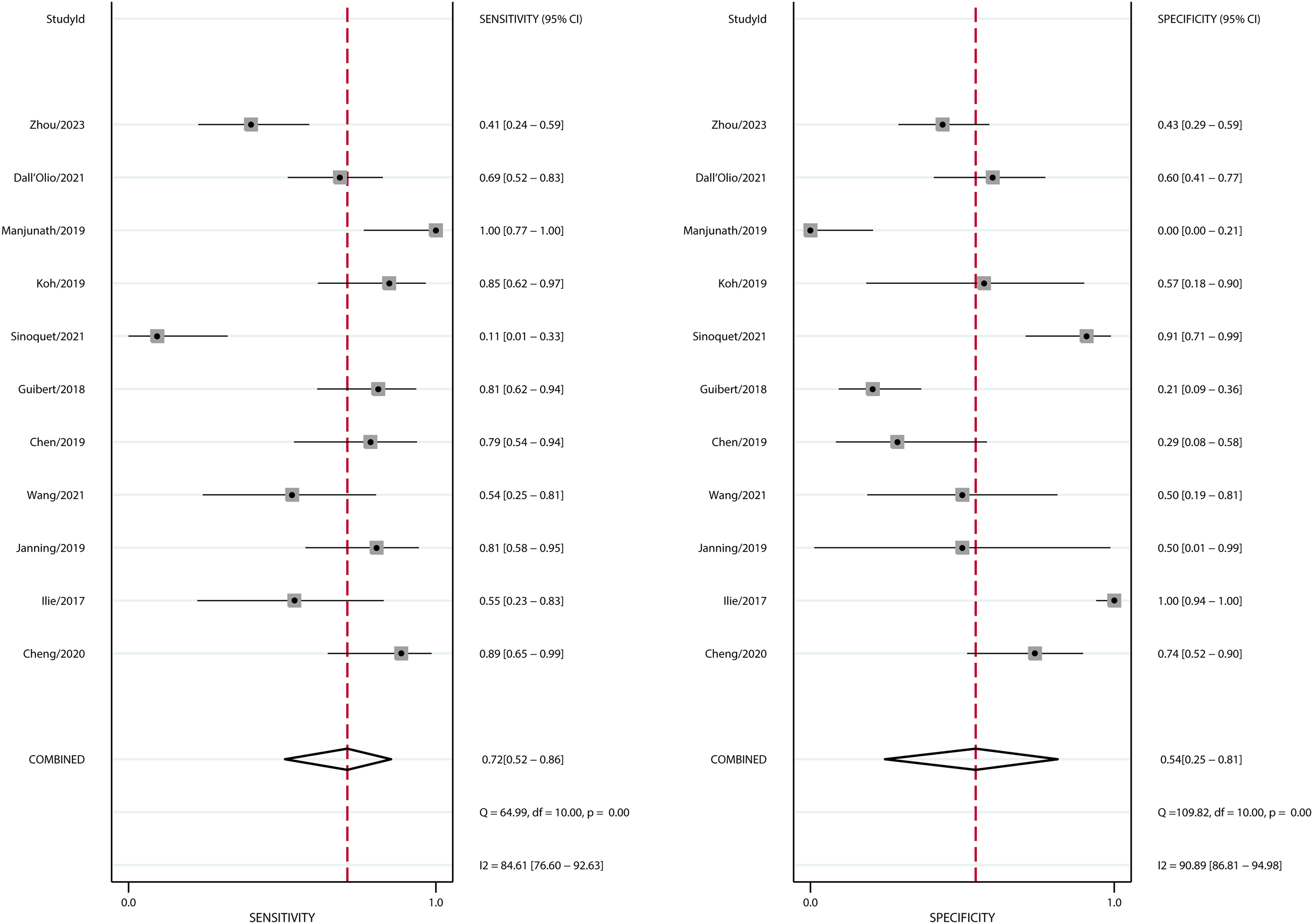
Forest plot of the pooled sensitivity and specificity. PD-L1 on CTCs has good sensitivity and specificity to lung cancer.
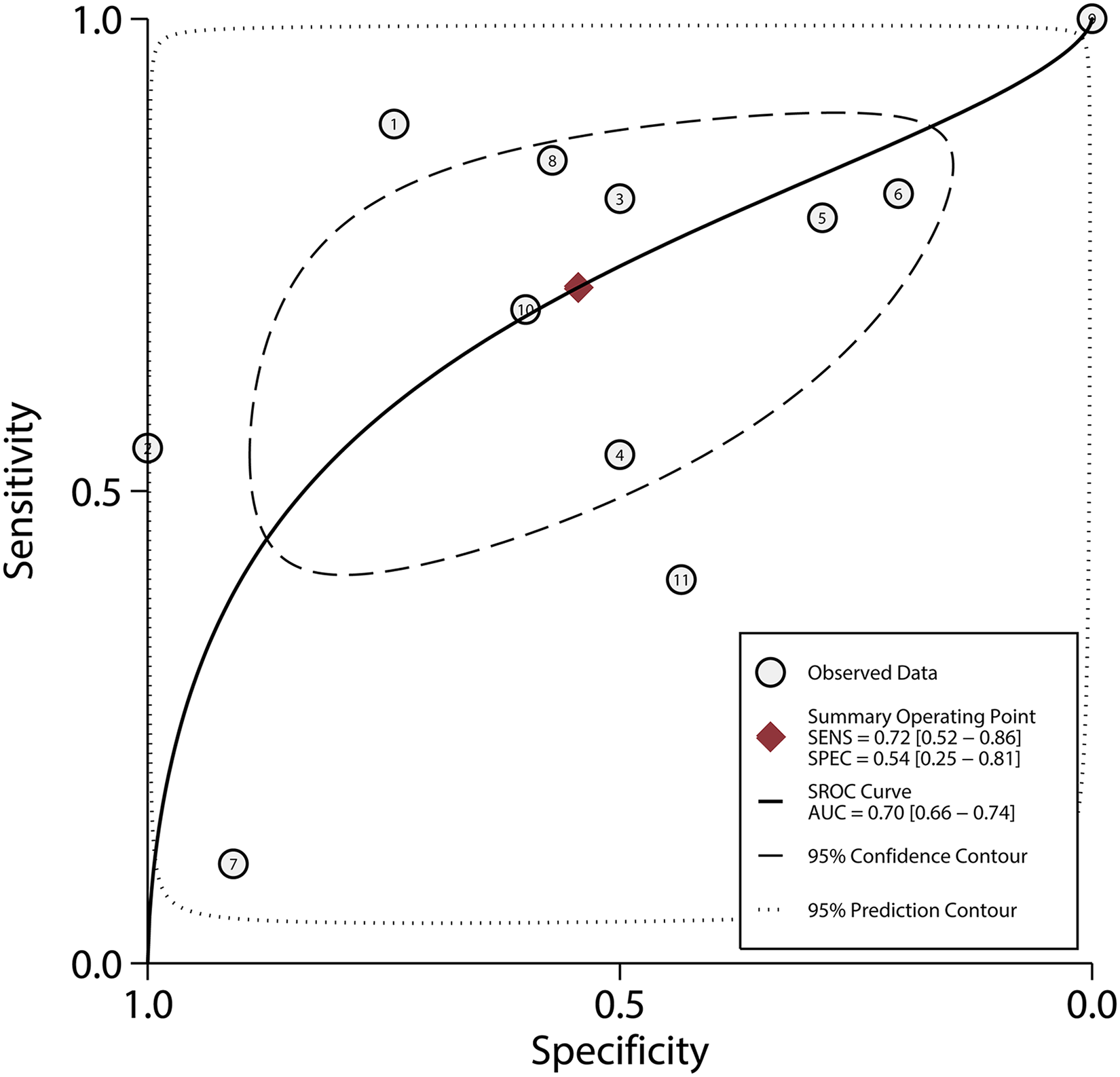
Summary receiver operating characteristic curve. The diagnostic value of PD-L1 on CTCs is moderate.
Heterogeneity and subgroup analysis
Significant heterogeneity was observed, with I2 values of 84.61 and 90.89 for sensitivity and specificity, respectively. This indicated that there is significant heterogeneity in the study, so further analysis followed the random effects model. To find the potential source of this heterogeneity, we carried out a SEN analysis. However, we did not find any evidence of a threshold effect. Therefore, a subgroup analysis based on study region, CTC enrichment method, tumor stage, and tumor treatment was carried out. The SEN, SPE, SROC, and DOR of the ISET® enrichment method were higher than those of the CellSearch enrichment method (0.77; 95% CI 0.56, 0.89 vs. 0.54; 95% CI 0.24, 0.82), (0.89; 95% CI 0.09, 1.00 vs. 0.63; 95% CI 0.35, 0.85), (0.82; 95% CI 0.78, 0.85 vs. 0.62; 95% CI 0.58, 0.66), and (27; 95% CI 1, 1402 vs. 2; 95% CI 1, 4), respectively. The SEN, SPE, SROC, and DOR of PD-L1 CTC cut off ≥1% were higher than those cut off ≥1 (0.72; 95% CI 0.55, 0.85 vs. 0.56; 95% CI 0.27, 0.81), (0.79; 95% CI 0.19, .98 vs. 0.54; 95% CI 0.29, 0.77), (0.77; 95% CI 0.73, 0.80 vs. 0.56; 95% CI 0.52, 0.61), and (10; 95% CI 1, 122 vs. 1; 95% CI 1, 3), respectively. Patients with all stages were slightly more sensitive and higher diagnostic performances than advanced patients (0.85; 95% CI 0.65, 0.95 vs. 0.56; 95% CI 0.33, 0.77, respectively) and (0.76; 95% CI 0.72, 0.79 vs. 0.66; 95% CI 0.62, 0.70, respectively), but had lower SPE (0.35; 95% CI 0.10, 0.73, vs.0.72; 95% CI 0.30, 0.94, respectively).Untreated patients were slightly more sensitive than patients with treatment (0.80; 95% CI 0.59, 0.91, vs. 0.53; 95% CI 0.24, 0.81, respectively), but had lower SPE (0.38; 95% CI 0.18, 0.64, vs. 0.84; 95% CI 0.29, 0.98, respectively) (Table 2).
Subgroup analysis of included studies.
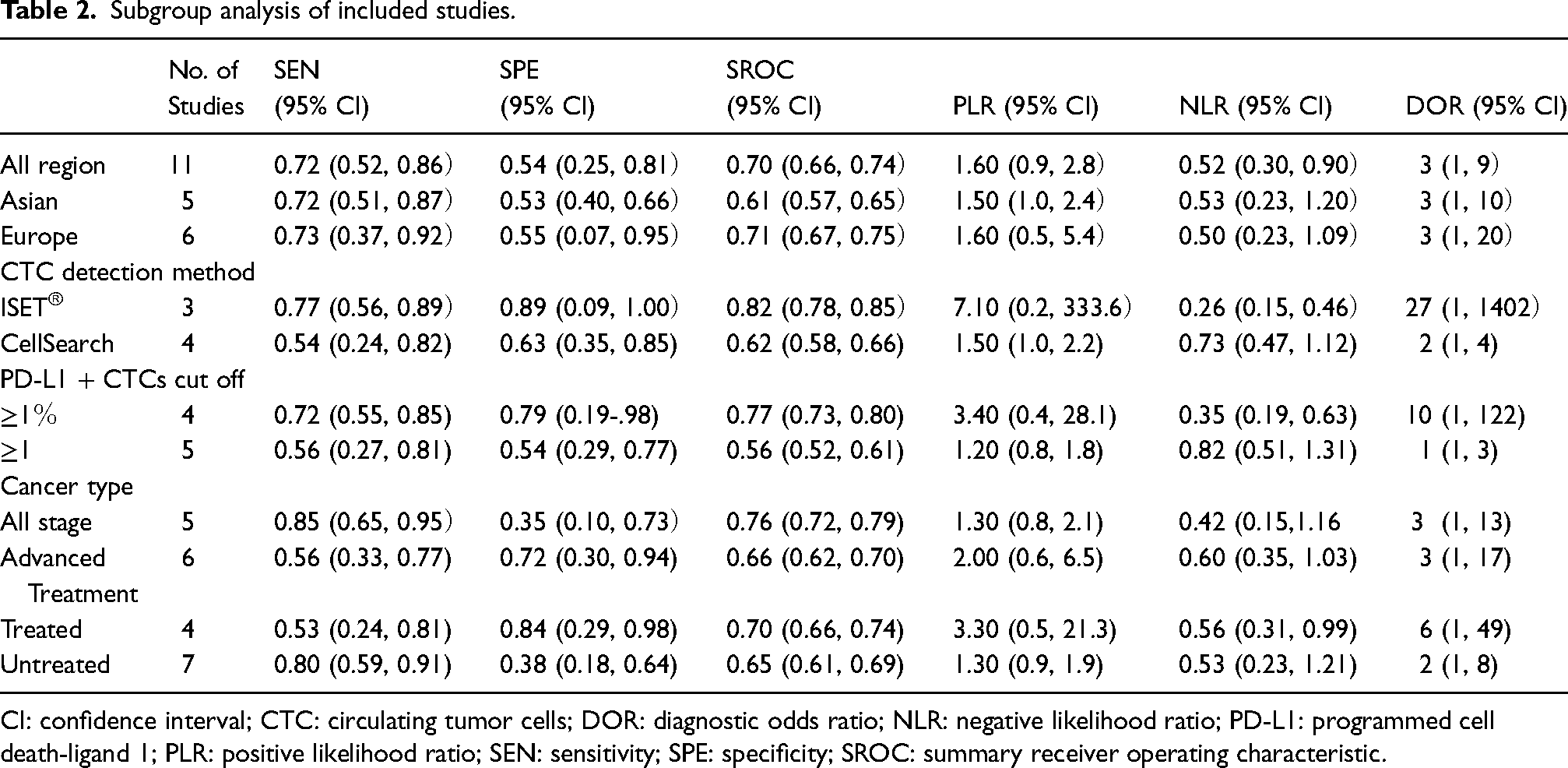
CI: confidence interval; CTC: circulating tumor cells; DOR: diagnostic odds ratio; NLR: negative likelihood ratio; PD-L1: programmed cell death-ligand 1; PLR: positive likelihood ratio; SEN: sensitivity; SPE: specificity; SROC: summary receiver operating characteristic.
Publication bias
Publication bias is consider as the main factor that affecting diagnostic performance, so we conducted Deek's funnel plot asymmetry test to assess the presence of publication bias. The shape of the funnel shows no sign of asymmetry, and no significant publication bias was observed in Deeks’ funnel plot (P = 0.52). (As shown in Supplementary Figure 4, available online at www.biological-markers.com—Deek's funnel plot of potential publication bias.)
Fagan's nomogram
The Fagan plot illustrates the clinical utility of PD-L1 on CTC in the diagnosis of lung cancer. The results indicated that the positive post-test probability of diagnosing lung cancer would increase to 61%, while the negative post-test probability of diagnosing lung cancer would decrease to 34%, with a pre-test probability of 50%. (As shown in Supplementary Figure 5, available online at www.biological-markers.com—Fagan's nomogram of PD-L1 expression CTCs for the diagnosis of lung cancer.)
Discussion
Recent studies have demonstrated that liquid biopsy could be a better substitute in cancer diagnosis and prognosis. Researchers have shown that positive PD-L1 expression on CTCs predicts a better survival prognosis for ICI treatment,26,27 and monitoring PD-L1 expression on CTCs could be an additional biomarker for precision medicine that may help in determining the response to immunotherapies. 28 Some meta-analyses focus on the predictive or prognostic value of PD-L1 expression on CTCs in lung cancer patients. However, so far, there is no meta-analysis available that majorly focuses primarily on the value of PD-L1 expression on CTCs for diagnosing in lung cancer. The diagnostic results of PD-L1 on circulating tumor cells for lung cancer are inconsistent, and no consensus has been reached. Ilie et al. 15 revealed that among 71 evaluable samples with matched-tissue and CTCs, there is 93% concordance between tissue and CTCs. Cheng et al. 16 showed that the consistency of PD-L1 expression in CTC and tumor tissue (TT) was 80% in 59 non-small cell lung cancers. While Guibert et al. 22 observed that there is no correlation between tissue and CTC PD-L1 expression (r = 0.04, P = 0.77). Also, Wang et al. 17 found the same result, and the correlation coefficient of PD-L1 in tissue and CTC was 0.20. Due to the discrepant results among these studies, it is difficult to apply PD-L1 to clinical applicability. Therefore, our work emphasizes the potential of PD-L1 expression on CTCs in lung cancer diagnosis. We reviewed 11 studies of PD-L1 expression on CTCs, and performed a systematic meta-analysis to clarify whether CTCs may serve as a feasible alternative tissue analysis for the detection of PD-L1 in lung cancer.
Our study indicates moderate diagnostic performance for blood-based PD-L1 expression on CTCs for lung cancer. However, significant heterogeneity was observed in our study. To find the source of heterogeneity, we analyzed the factors that may affect heterogeneity. To assess patients from different countries, we stratified the meta-analysis by the CTCs enrichment platform and found that the ISET® enrichment method had higher SEN, SPE, SROC, and DOR than the the CellSearch enrichment method. Although CellSearch is the only CTC enrichment platform approved by the US Food and Drug Administration in metastasized breast, prostate, and colorectal cancer, our results show that it remains challenging in lung cancer. Due to the low detection rate of CTC in lung cancer, in order to improve the detection rate of CTC, more and more detection methods (such as ISE, the label-independent Parsortix system, the MCA system, a microfilter platform, the negative enrichment method, etc.) are used to enrich CTC. Currently there is no well-standardized CTCs enrichment platform; however, it is hoped that there will be a CTC enrichment platform with higher detection capability in the future. Our results are consistent with other findings; that is, compared with the CellSearch method, the ISET® method has a higher detection rate of CTC.29,30 Given the small number of studies, further studies are needed to confirm our results.
The included studies used different thresholds to detect PD-L1 expression on circulating tumor cells, with some setting the threshold of PD-L1 + CTC to ≥1 and some setting the threshold of PD-L1 + CTC to ≥1%. Our meta-analysis results indicated that the threshold of PD-L1 + CTC to ≥1% had higher SEN, SPE, SROC, and DOR than the threshold of PD-L1 + CTC to ≥1. Due to the limited number of publications used for each method, further research is necessary.
The results of the subgroup analysis demonstrated that patients with all stages of lung cancer had slightly higher SEN than advanced patients. It may be that the expression of PD-L1 changes dynamically with the progression of the disease. 31
Additionally, untreated patients had slightly higher SEN than patients with treatment. The reason may be that patients receiving chemotherapy, radiotherapy, or ICI treatment will also have changes in PD-L1 expression. In addition, some researchers found that the number or proportion of PD-L11 + CTCs decreased when treating patients with response, but the expression of PD-L11 + CTCs increased or remained unchanged in patients without response.
According to the above analysis, PD-L1 expression on CTCs exhibited a moderate diagnostic value in lung cancer, and CTCs may serve as a feasible alternative tissue analysis for the detection of PD-L1 in lung cancer. However, there are some limitations to our study. First, although we tried to collect all the articles, some data were lost because our study was restricted to articles published only in English or Chinese. Second, there was obvious heterogeneity in our study, which may be caused by the study region, cancer stage, treatments, CTC enrichment, and PD-L1 detection methods. Third, most of the included studies had very small sample sizes. More large-scale studies with PD-L1expression on CTCs are needed to validate the findings of our meta-analysis
Conclusion
Our analysis demonstrated that CTCs serve as a liquid biopsy that is effective for the detection of PD-L1 in lung cancer.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155231192674 - Supplemental material for Diagnostic value of programmed cell death-ligand 1 expression on circulating tumor cells in lung cancer: A systematic review and meta-analysis
Supplemental material, sj-docx-1-jbm-10.1177_03936155231192674 for Diagnostic value of programmed cell death-ligand 1 expression on circulating tumor cells in lung cancer: A systematic review and meta-analysis by Meng Cui, Zhiyong Wan, Jia Yang, Dan Liao, Yang Yang and Yin Xiang in The International Journal of Biological Markers
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
