Abstract
BACKGROUND:
The PD-L1 antibody atezolizumab has shown promising efficacy in patients with advanced non-small cell lung cancer. But the predictive marker of clinical benefit has not been identified.
OBJECTIVE:
This study aimed to search for potential predictive factors in circulating blood of patients receiving atezolizumab.
METHODS:
Ten patients diagnosed with advanced non-small cell lung cancer were enrolled in this open-label observing study. Circulating immune cells and plasma tumor markers were examined in peripheral blood from these patients before and after atezolizumab treatment respectively. Relation between changes in circulating factors and anti-tumor efficacy were analyzed.
RESULTS:
Blood routine test showed that atezolizumab therapy induced slightly elevation of white blood cells count generally. The lymphocyte ratio was increased slightly in disease controlled patients but decreased prominently in disease progressed patients in response to atezolizumab therapy. Flow cytometric analysis revealed changes in percentage of various immune cell types, including CD4
CONCLUSIONS:
The circulating immune cell ratios and plasma tumor marker levels were related with clinical efficacy of atezolizumab therapy. These factors could be potential predictive marker for anti-PD-L1 therapy in advanced non-small cell lung cancer.
Introduction
Outcome for metastatic non-small cell lung cancer (NSCLC) is still poor both in the world and China [1, 2]. One of the main reasons for therapy failure is escape of tumor cell from the immune surveillance. It was found that the PD-1/PD-L1 axis plays important role in tumor immune modulation [3, 4]. PD-1 is an immunoinhibitory receptor expressed mainly on the surface of activated T cells [5, 6]. Binding with its ligand PD-L1 which is expressed on tumor cells, PD-1 will be activated and exert a negative effect on immune responses by dephosphorylating key downstream proteins of the antigen receptor and therefore provides a possible mechanism of escape immune surveillance [7, 8]. The anti-PD-1/PD-L1 checkpoint inhibitors have shown prominent efficacy for treatment of advanced NSCLC [9, 10].
Atezolizumab, a fully human monoclonal antibody against PD-L1, has shown anti-tumor efficacy in various metastatic cancers [11, 12, 13, 14, 15]. In patients with advanced, previously treated NSCLC, atezolizumab yielded a significant improvement in overall survival compared with docetaxel [16, 17]. However, despite the success of atezolizumab in NSCLC, a major remaining challenge is identifying which patients will benefit from anti-PD-1/PD-L1 therapy [18]. Although it was found that PD-L1 expression on both tumor cells and tumor-infiltrating immune cells is independently predictive of survival improvement with atezolizumab [19]. Predictive biomarkers in peripheral blood for the PD-1/PD-L1 blockade therapy remain controversial and are still under investigation. The aim of this study is to observe the influence of atezolizumab on peripheral blood parameters and to explore potential predictive factors from blood of advanced NSCLC patients receiving atezolizumab therapy.
Material and methods
Patients and blood samples
Patients with stage IIIB/IV NSCLC after failure of platinum based chemotherapy were prospectively enrolled between September 2016 and January 2017, receiving intravenous 1200 mg of atezolizumab every 3 weeks until disease progression or unacceptable toxicities. Relevant clinical information and blood samples were collected before and after 2 cycles of injection of atezolizumab. Besides, complete blood cell counts with differential count and serum chemistry were carried out at baseline and before each drug infusion as routine exam. All patients gave their informed consent for use of clinical data for scientific purposes, approved by the Institutional Review Board of Peking University Cancer Hospital and Institute.
Response evaluation
Clinical examination, CT scans of chest and abdomen, cerebral MRI/CT and ultrasound scan of lymph nodes was performed at baseline and then every 6 weeks (2 cycles of atezolizumab). The overall response was assessed according to the Response Evaluation Criteria in Solid Tumors (RECIST 1.1 version), categorizing as complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD). Adverse events (AEs) were continuously monitored and assessed in all treated patients and were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events, version 3.0. AEs were managed using protocol-specific guidelines.
Demographic and clinical characteristics of patients
Demographic and clinical characteristics of patients
Abbreviations: ECOG
For phenotype analysis of immune cell subsets, patient peripheral blood was subjected to Ficoll Hypaque density centrifugation, and the buffy layer containing peripheral blood mononuclear cells (PBMCs) were stained with specific antibodies for flow cytometric analysis. Thereafter, for membrane staining, single cell suspensions were incubated for 30 minutes at 4
Analysis of circulating biomarkers
Blood routine examination and plasma tumor markers comprising CEA, CA125 and CA199, were evaluated in peripheral blood samples. Routine blood tests were performed by electrical impedance with a Beckman coulter LH750 instrument (Beckman Coulter, Inc., Brea, CA, United States). The CEA, CA125 and CA199 were assayed by the micro-particle chemiluminescence analysis using the Abbott ARCHITECT i2000SR immuno analyser (USA). All test procedures were performed according to the equipment operation procedure, calibration solution and reagents were original kits from manufacturer. The reference interval of these tumor markers were that: CEA 0–5 ng/ml, CA125 0–35 U/ml, CA199 0–37 U/ml.
Statistical analyses
Statistical analyses were conducted using GraphPad Prism and SPSS 19.0 software. Categorical variables were summarized as frequency counts and percentages, measured data as medians and ranges. The Wilcoxon rank-sum and Jonckheere-Terpstra tests were used for the comparison of different cell subsets between patient groups. Differences among two groups were statistically analyzed using a two-tailed Student’s t test. Values of
Results
Clinical characters, safety and response
In this study, 10 patients with advanced NSCLC received atezolizumab regardless their PD-L1 status, including 5 cases adenocarcinoma, 4 cases squamous carcinoma and 1 case adenosquamous carcinoma (Table 1). The median age was 64 years (range 45–66 years). Previous chemotherapy agents include: gemcitabine, pemetrexed, vinorelbine, combined with cisplatin or carboplatin. One patient received crizotinib. No EGFR TKIs was delivered. All patients received no more than 2 lines chemotherapy previously.
PD-L1 blockade influences the frequencies of CD4
Patients received atezolizumab for a median of 4 cycles (range 1 to 10 cycles), which corresponded to a median treatment duration of 3 months (range 1 to 7 months). Eight patients experienced a treatment-related AEs; grade 1 AEs were reported in 5 cases, including 3 cases fatigue, 1 case rash, 1 case numbness; grade 2 AE was reported in 1 patient as immune-mediated interstitial pneumonitis, which resolved promptly with systemic corticosteroid treatment. Two cases of grade 3 immune-mediated AEs were reported, including one case treatment-related liver hemotoma, and one case heart failure which led to treatment discontinuation. No treatment-related grade 4 to 5 AEs was reported.
In the first evaluation after 2 cycles of therapy, 4 patients were evaluated as SD, 1 patient was PR, and 5 patients were PD. Three patients quit the study, the others continue treatment. In following cycles, 4 patients quit study because of secondary disease progression or adverse effects; three patients were still under treatment now. The median duration of survival follow-up was 4.83 months (95% CI: 3.73–6.43 months). Overall, the median progression-free survival (PFS) was 3.73 months (95% CI: 1.5 months to not reached), and the median overall survival (OS) was not reached.
According to the best therapy response, patients were divided into arm A (SD and PR) and arm B (PD). The disease control rate is 2/5, 3/4, and 0/1 for adenocarcinoma, squamous and adenosquamous type respectively; in smoking patients the disease control rate is 5/7 while in non-smoking patients it is 0/3. All 10 patients were in ECOG 0–2 at base line.
Blood routine examination was performed in all 10 patients. The white blood cells count, circulating lymphocyte ratio and neutrophil-to-lymphocyte ratio (NLR) were analyzed (Table 2). After 2 cycles of therapy, the average white blood cell (WBC) count increased slightly comparing with baseline in both arm A and B. No significant differences were seen in either baseline average WBC or WBC variation between two arms.
PD-L1 blockade decreases the frequencies of Treg and MDSC cells. PBMC from patients with NSCLC were used. A. Representative flow cytometric analysis of Treg cells and pooled data (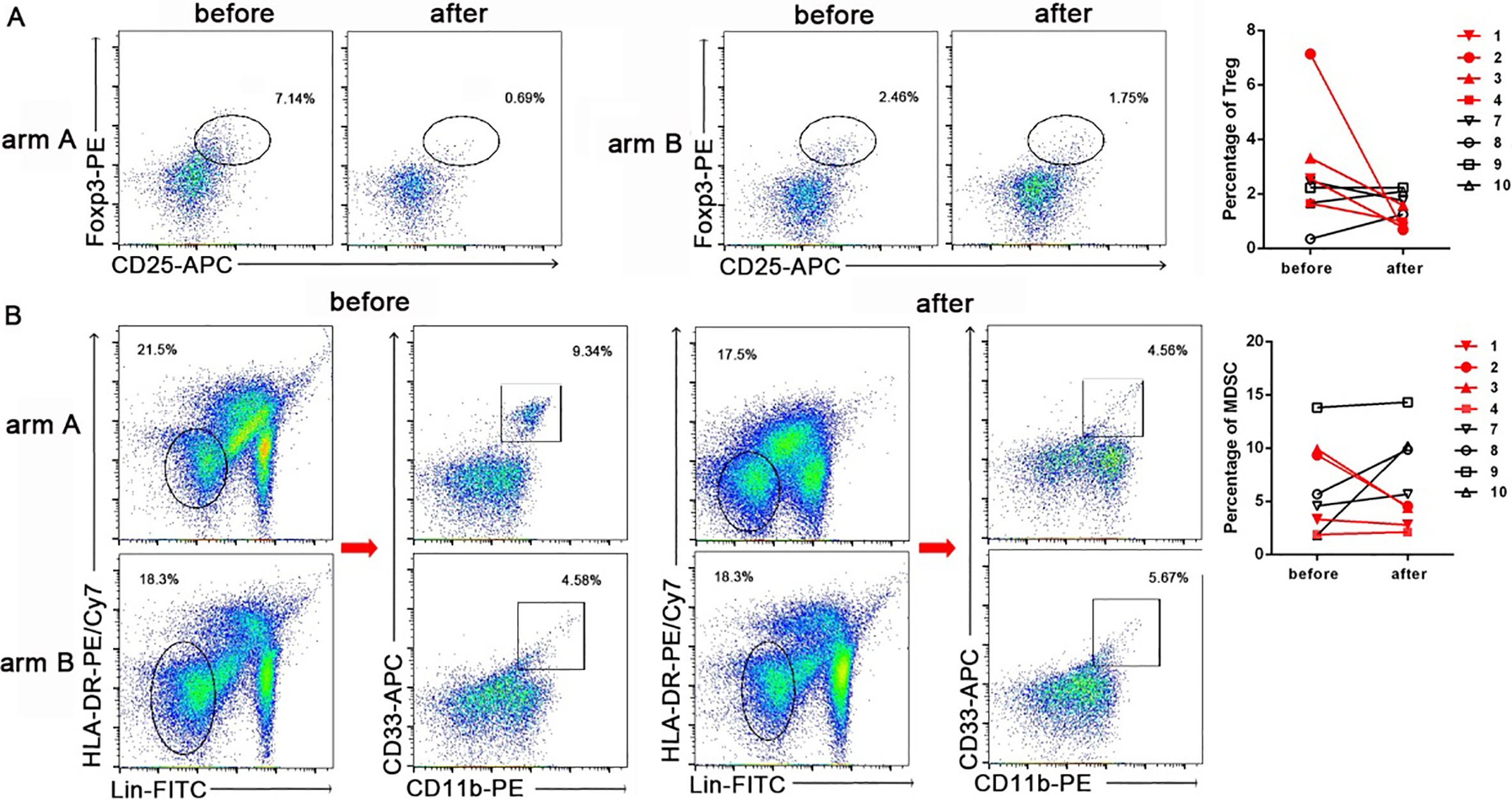
In comparison with baseline, the lymphocyte ratio was changed in response to atezolizumab therapy. The lymphocyte ratio decreasing was seen in 1 patient in arm A and 4 patients in arm B. The average lymphocyte ratio increased slightly in arm A but decreased sharply in arm B, suggesting lymphocyte ratio increasing was associated with disease control while decreasing was associated with disease progression. Moreover, the post-treatment NLR decreased apparently in arm A, but increased slightly in arm B, although there was no obvious difference between pre-treatment NLR of arm A and B on account of the small sample size. These might predict that the reduction in NLR after atezolizumab treatment was associated with a higher objective response rate.
WBC count, lymphocyte ratio and NLR in peripheral blood of NSCLC patients before and after atezolizumab treatment
WBC count, lymphocyte ratio and NLR in peripheral blood of NSCLC patients before and after atezolizumab treatment
We assessed PBMCs from 8 NSCLC patients before and after 2 cycles of atezolizumab therapy. Flow cytometric analysis was performed in PBMC of 8 patients, including 4 patients in arm A (NO. 1–4) and 4 patients in arm B (NO. 7–10). As shown in Fig. 1A, the percentage of CD4
Given the constant interaction of PD-1 with PD-L1 in the tumor microenvironment, we next sought to determine whether PD-1 expression in T-cell was affected by anti-PD-L1 therapy. In comparison with base line, PD-1 expressing CD8
Along with checkpoint blockade of T-cell function, multiple cell types are involved in tumor-mediated immunosuppression, including regulatory T cells (Treg) and MDSC. We next investigate whether atezolizumab treatment affects Treg and MDSC subsets in peripheral blood of patients. Compared with baseline, both Treg and MDSC frequency were downregulated after atezolizumab treatment in arm A. On the contrary, we did not observe the percentage of Treg and MDSC in arm B were descending, indeed they were increasing slightly (Fig. 2). Collectively, our findings showed that downregualtion of Treg and MDSC subsets was associated disease control in atezolizumab therapy.
The plasma levels of CEA, CA199 and CA125 changed prominently after 2 cycles of atezolizumab therapy (Fig. 3). In arm A, more than half patients showed marker decreasing. The CEA decreased in 2 cases, CA199 decreased in 4 cases, CA125 decreased in 2 cases; in arm B, only 1 case showed CEA decreasing, neither CA199 nor CA125 decreasing was seen. Interestingly, all 3 markers decreased in patient NO. 4 who achieved PR, while for patient NO. 8 and NO. 10 who experienced fast disease progression, the CA125 level increased over 300% comparing with baseline. Our data showed that, if a patient had either all 3 markers increasing or at least 2 markers level increase over 50% above baseline, the patient would be classified into disease progression, otherwise, he/she will show disease control.
Alteration of plasma tumor marker levels in response of atezolizumab therapy. The vertical value indicates percent of marker level alteration in comparison with baseline after 2 cycles atezolizumab therapy; horizontal number indicates patients NO. 1–10.
To date, reliable predictive marker for PD-L1 antibody activity in lung cancer was still lacking. Although pre-existing T-cell infiltration [20, 21, 22] and the presence of PD-L1 in tumors may be used as indicators of clinical response [9, 10], blood-based profiling to predict the efficacy of PD-1/PD-L1 blockade has not been widely explored.
In current study, we investigated different circulating markers in blood of patients receiving atezolizumab therapy. We found that atezolizumab affects circulating lymphocyte ratio in NSCLC. The ratio increased slightly in disease controlled patients but decreased prominently in disease progressed patients. Our finding was in coincidence with previous studies. It has been reported that both disease control and survival were significantly associated with increasing absolute lymphocyte count and decreasing FoxP3/regulatory T cells between baseline and the end of dosing in patients treated with ipilimumab [19, 23, 24]. Similar results were also reported with pembrolizumab, which is an anti-PD-1 antibody [25]. These data suggests that large scale lymphocytes could be motivated into circulation by checkpoint inhibitors and migrate into lesions to kill tumor cells, indicating lymphocyte ratio monitoring could potentially predict immunotherapy response.
Tumor infiltrating lymphocytes have been found to have predictive value with respect to the natural history of primary cancers [26, 27, 28]. There were reports that the baseline density and location of T cells in metastatic melanomas have predictive value in the treatment outcome of anti PD-1/PD-L1 therapies [29]. But it is challenging due to the difficulties in obtaining tissue samples during therapy, we analyzed the peripheral blood samples and found that decrease of CD4
A previous study reported that high PD-1 expression on peripheral CD4
As we know, this is the first report that decrease of circulating Treg, MDSC and PD-1 expressing CD8
Plasma tumor markers level reflect tumor burden. In traditional chemotherapy and target therapy, tumor markers have shown value in evaluating therapy response. In this study, we found that levels of tumor markers changed after atezolizumab therapy. In disease controlled patients tumor markers decrease or stay stable generally, while in progressed patients either 3 kinds of markers increase or 2 markers increase over 50% from baseline. In prior studies, Le had reported that the CEA level decline after one dose of pembrolizumab was predictive of both progression-free survival and overall survival. The CEA response occurred well in advance of radiographic confirmation of disease control. In contrast, patients who had disease progression had rapid biomarker elevation within 30 days after the initiation of therapy [42]. In another study, Maria reported significant differences were observed in serum level of CEA and CYFRA21-1 between responders and non-responders among NSCLC patients receiving nivolumab [43]. We suggest that an optimized panel of tumor markers is required to help predict immunotherapy response in future.
Neutrophils are the first responders to acute tissue damage and infection, but in certain circumstances presence of neutrophils often correlates with cancer progression [44, 45]. Increasing NLR has been demonstrated to be a harbinger of a more dismal prognosis in various cancers, which is related to outcomes in immune checkpoint blockade, intratumoral levels of MDSCs and peripheral Tregs [44]. Recent study has shown that a high pretreatment NLR is associated with worse PFS and OS in nivolumab-treated NSCLC patients [46]. While Suh et al. have found that low NLR or decrease in post-treatment NLR at week 6 after anti-PD-1 antibody were associated with better PFS and OS, supporting that post-treatment NLR is associated with response rate and survival in patients with NSCLC treated with anti-PD-1 antibody [47]. Our study also showed that reduction in NLR after atezolizumab treatment was associated with better response rate, suggesting that the NLR can be used as a predictor for response to immunotherapy. Moreover, in lung cancer patients, the tumor markers, including the CEA, CA125 and CA199, are mainly released from cancer cells, elevation of these serum markers are usually associated with advanced stage or disease progression. The increased NLR could promote tumor progress and increase the inflammatory reaction locally, increase the permeability of vascular wall, leading to elevation of tumor markers. Prior studies had shown the combination of NLR with tumor marker could reflect the dynamic interaction between tumor cells and the immune microenvironment, and has improved prognostic value for cancer patients [48, 49]. In this study we did not analysis this because of the sample size limitation, but it is worth to explore the predictive value of the combination of NLR with tumor markers in future large scale studies about immunotherapy.
This study has some limitations. The small sample size made it difficult to take satisfying statistic analysis and our data did not reach statistic significance, future large cohort studies are warranted to explore the predict role of circulating biomarkers. Due to the complexity of immune system, we did not analyze more immune factors. It is necessary to combine flow cytometry and mass cytometry to probe the phenotypes and functions of multiple immune cell subsets simultaneously. A whole-view profiling of immune characteristics will eventually yield correlates of responsiveness to therapy.
Conclusions
In conclusion, we found atezolizumab induce alterations in levels of the circulating blood cells and tumor makers in advanced NSCLC patients. Increased lymph ratio, decrease in Treg, MDSC and tumor marker are associated with good response. Liquid biopsy could be a non-invasive and effective method to investigate prognostic and predictive circulating biomarkers for NSCLC patients receiving anti-PD-L1 therapy.
Footnotes
Acknowledgments
This work was supported by Beijing Natural Science Foundation (Commission No. 7162038); Open Project funded by Key laboratory of Carcinogenesis and Translational Research, Ministry of Education/ Beijing (2017 Open Project – 4); and the Beijing Municipal Administration of Hospitals’ Youth Programme (Commission No. QML20161101).
Conflict of interest
None.
