Abstract
Purpose
Aspartate beta-hydroxylase (ASPH) is a transmembrane protein involved in cancer progression, which has been shown to imply a worse prognosis in several solid tumors. The aim of the present study was to further investigate the prognostic value of ASPH in early breast cancer.
Methods
ASPH expression was investigated through immunohistochemistry in a cohort of 153 breast cancer patients with long-term follow-up, and correlated with clinical–pathological features plus all-cause and breast-cancer-specific mortality. Appropriate statistics were utilized.
Results
ASPH negatively correlated with all-cause and breast-cancer-specific mortality.
Conclusions
The results of this cohort study support the prognostic value of ASPH in early breast cancer.
Introduction
Breast cancer represents the leading cause of cancer death among women, mainly in western countries. 1 Disease prognosis and treatment choices are still tailored on clinical–pathologic features and more specifically on the definition of subtypes on the basis of immunohistochemistry (IHC) assessment of estrogen receptor (ER), progesterone receptor (PgR), Ki-67, and human epidermal growth factor receptor2 (HER/2). 2 More recently, multigene platforms have been implemented and tested in order to improve actual ability to prognosticate both for patient clinical outcome and for response to currently available treatments 3 ; however, not all these platforms have been prospectively validated and, most importantly, they are not applicable to all patient subsets. Moreover, they are costly and not all of them are available in all countries and/or reimbursed by local health systems. This provides the rationale to continue searching for individual genes or protein candidates that are easily assessable at a low cost, in the hope of identifying novel prognostic (predictive) markers and, likewise, further refining the information provided by clinical–pathologic features.
Aspartate beta-hydroxylase (ASPH) is a transmembrane protein that hydroxylates aspartyl and asparagine residues in EGF-like domains of transformation-associated proteins, including NOTCH receptors and ligands. The subsequent interaction NOTCH/ligands, activates the transcription of downstream target genes like HES1, HEY1, and Cyclin D1, which in turn promote cell migration, invasion, and metastasis.4,5 ASPH overexpression has been observed in multiple malignancies and is associated with poor prognosis and shortened survival. 4 To our knowledge, only a couple of studies have investigated the role of ASPH in breast cancer progression.6,7 Zhang et al. 6 reported on the relationship between ASPH expression and the pathologic characteristics of breast cancer. These investigators found a correlation of ASPH expression with cancer type, node involvement, and tumor node metastasis (TNM) stage as well as with poor survival. However, no correlation was found with recurrence-free survival. Shimoda et al. 7 found that ASPH expression negatively correlated with recurrence-free survival of luminal B breast cancer patients who had received adjuvant endocrine therapy. However, no data were reported relative to survival. Both studies suggest a putative prognostic role of ASPH expression in selected groups of breast cancer patients. The results of these studies are not easily comparable to each other. Therefore, additional studies are required to better define the role of ASPH in breast cancer.
Taking advantage of the experience previously achieved by our research group in studying the expression of ASPH in colon and prostate cancer,5,8 and considering the availability of a set of tissue micro-arrays (TMA) obtained from tumor samples with markedly long follow-up (which have been used in previous studies),2,9 we were interested in re-evaluating ASPH expression in early breast cancer.
Materials and methods
Patient selection and ethical considerations
We identified a cohort of 153 patients who had a histologically confirmed diagnosis of breast cancer between January 1985 and November 1990. The cohort was selected from a larger cohort of women submitted to breast surgery at our Institution and in particular from a cohort of 200 breast cancer patients for whom TMAs were available. Supplementary Figure 1 presents the consort diagram. TMA material, which has been utilized in previous studies,2,9 had undergone IHC typing to define breast cancer subtypes according to the criteria adopted by the 2013 St. Gallen Consensus Conference. 10 ER status was defined positive for nuclear staining of ≥1% of cells according to the American Society of Clinical Oncology (ASCO) recommendations. 11 The cut-off point discriminating between “low” and “high” PgR was <20% and ≥20%, respectively. Definition of HER/2 status was based on the 2013 ASCO/College of American Pathologists recommendations: positive if IHC 3 + ; negative if IHC 1 + or 0, IHC 2 + requiring confirmation of gene amplification by FISH. 11 A threshold of 20% was used to discriminate between “low” (<20%) and “high” (≥20%) Ki67 tumors.
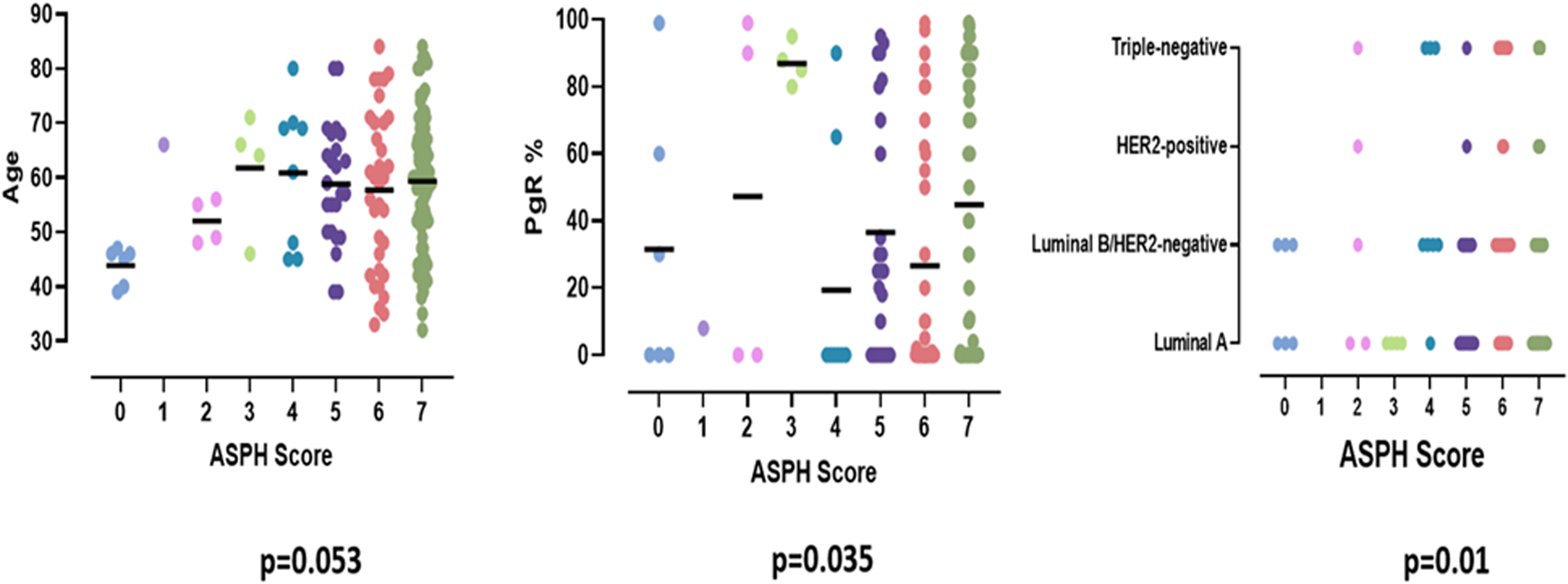
Correlation of ASPH score with age, PgR status and tumor subtype (ANOVA).
Study protocol was approved by the Ethics Board of Regione Liguria, Italy (N.367/2021-DB id 11592). The Ethics Board members were aware of the fact that (a) the great majority of the patients included in the cohort had died; (b) a long time had elapsed since surgery (median follow-up time exceeding 30 years); and (c) since the patients who were still alive at the last check had been lost to follow-up, it would not be possible to trace the few remaining patients to obtain their written informed consent. Data were managed according to the Italian Data Protection Authority Prescriptions, which, in similar cases, allow the use and publication of data for scientific purposes (http://www.garanteprivacy.it).
ASPH determination
All TMAs had been constructed from formalin-fixed and paraffin-embedded neoplastic tissue, as detailed previously. 9 Two 3-micron thick sections were newly microtome cut and mounted on super frost slides (Thermo-Scientific, Braunschweig, Germany). IHC was performed on the automated immunostainer Benchmark Ultra (Roche Diagnostics, F. Hoffmann-La Roche Ltd, Basel, Switzerland). The selected antibody, following our previous experiences5,8 and that of the aforementioned previous studies on breast cancer,6,7 was anti-ASPH (clone A-10) (Santa Cruz Biotechnology, Inc. Dallas, USA), which is a mouse monoclonal antibody raised against amino acids 382–681 mapping within an internal region of human ASPH. Positive and negative controls from ASPH expressing or non-expressing colorectal cancers 8 were used to optimize the IHC procedure. Briefly, sections were deparaffinized and antigen retrieval was performed using EDTA borate buffer (pH8) then heated up to 98°C for 36 min.
An initial explorative assessment was carried out on full slide sections of anonymized breast cancer cases with the specific aim of ensuring that ASPH staining was homogeneously expressed and that TMA analysis could be fruitful. ASPH was indeed homogeneously expressed throughout the neoplastic tissue sections without differences between the invasive edge and the central portion of the tumor. Staining was therefore performed on all available TMA samples.
ASPH is a type II membrane protein involved in calcium homeostasis and is located in the endoplasmic reticulum giving a membranous and cytoplasmic positivity. 8 The intensity of immunostaining and the proportion of immuno-colored cells were both considered to grade ASPH expression. Namely, the numbers representing staining intensity and the percentage of cells stained, as detailed below, were added together to generate a numerical total score.
The intensity of immunostaining in each core was scored as 0 = negative, 1 = weak, 2 = moderate, or 3 = strong. Supplementary Figure 2 shows representative immunostaining images of ASPH in breast cancer tissue sections relative to staining intensity.
The proportion of cell staining positively were scored as 0 = no cells, 1 = 1–25% of cells, 2 = 26–50% of cells, 3 = 51–75% of cells, and 4 = 76–100% of cells.
The median ASPH total score value in our cohort was 6 (range 0–7). This value was arbitrarily utilized for statistical analysis of mortality because it performed best in respect to the other percentile values tested (data not shown).
Outcomes of interest
Cohort study outcomes of interest were the correlation of ASPH expression with clinical–pathologic characteristics and with all-cause and breast-cancer-related mortality. Deaths occurring after any well-documented breast cancer relapse were arbitrarily defined as breast-cancer-related; all other deaths were defined as breast-cancer-unrelated. All-cause mortality included both breast-cancer-related and breast-cancer-unrelated deaths. Data were retrieved from patients’ medical records. Mortality data missing in the medical records were obtained by consulting the local mortality registry or the registry offices of the patients’ place of residence. All events occurring by the dead line of September 30, 2020 were recorded. Pandemic COVID-19 events did not allow us to further update data.
Independent variable
The cohort study independent variable was ASPH expression categorized according to the total score value obtained as previously detailed.
Control variables
The following variables, categorized as per Supplementary Table 1, were abstracted via chart review: patient age at diagnosis, menopausal status, tumor size, nodal status, tumor stage (TNM version 2009), ER, PgR, Ki67, and HER/2 status, tumor subtype (i.e., luminal A, luminal B, HER/2-like, triple negative according to the St. Gallen criteria, as previously detailed), and receipt of adjuvant systemic treatment.
Statistical analysis
The one-way ANOVA followed by Bonferroni's post hoc test were applied to compare the ASPH total score with the control variables. The curves of mortality were constructed using the Kaplan–Meir method, and treatment effect comparisons were obtained through the log-rank test. 12 Cox proportional hazards regression models 13 were used to check for prognostic features, to estimate hazard ratios and 95% confidence intervals for ASPH score comparisons, and to test for interaction between variables. Age (≤59 years vs. >59 years), menopausal status (premenopausal vs. postmenopausal) tumor size (≤2cm vs. >2cm), nodal status (negative vs. positive), tumor stage (I vs. II vs. III), and treatment (no adjuvant therapy vs. adjuvant therapy) were included in the models. Cumulative incidence estimates for breast-cancer-related and breast-cancer-unrelated deaths were performed to check for competing risks. The difference between cumulative incidence curves in the presence of competing risks was tested using Fine and Gray's test. 14 All probability values were obtained from two-sided tests and a P-value <0.05 was considered statistically significant. All statistical tests were carried out using IBM-SPS package (version 27.0), and STATASE11.
Results
Correlation of ASPH expression with clinical-pathologic characteristics
Statistically significant correlations were found between ASPH expression and PgR level and tumor subtype, (Figure 1), which shows that lower ASPH scores were more frequent in PgR rich and in Luminal A and B subtypes while higher ASPH scores were more frequent in HER/2-like and triple-negative subtypes. The correlation with patient age was borderline significant. No significant correlation between ASPH score and the other co-variables including ER level were found (data not shown).
Correlation of ASPH expression with mortality
At the time of the present analysis 117 deaths had occurred. Of these, 72 were judged to be cancer-related, as previously defined, and 45 to be cancer unrelated. Table 1 shows the prognostic value of the ASPH score and of the other co-variates after univariate and multivariate analysis. In the univariate model patient age, menopausal status, tumor size, nodal status, tumor stage, adjuvant treatment, and ASPH score all significantly predicted the all-cause mortality risk. The prognostic value of PgR status, Ki67 value, and tumor subtype did not reach the statistical significance and for this reason these variables were not included in the multivariable analysis. This latter analysis confirmed the independent prognostic value of age, menopausal status, tumor size, and ASPH total score. Cumulative mortality curves relative to all-cause mortality risk according to ASPH score are shown in Figure 2. Of the patients with ASPH total scores above the median value, 87% were found to have died by the 30-year follow-up compared to 73% of those with ASPH scores under the median; these differences maintained statistical significance even after adjusting for the other co-variates. Interestingly, the competitive risks analysis showed that there was no obvious difference in the risks of breast-cancer-unrelated mortality according to ASPH score value, while there was a substantial impact of ASPH expression on breast-cancer-related mortality; however, the differences were borderline significant due to the small size of the population of breast-cancer-related deaths (n = 72; Figure 3). The impact of ASPH total score value on all-cause mortality was quite homogeneous in all the subgroups, being more evident in (a) younger patients; (b) patients with a T size >2 cm; (c) patients with pN1 tumors; (d) patients with stage II disease; and (e) patients who had received adjuvant therapy (Figure 4(a)). Subgroup analysis of breast-cancer-related mortality showed specular findings (Figure 4(b)).
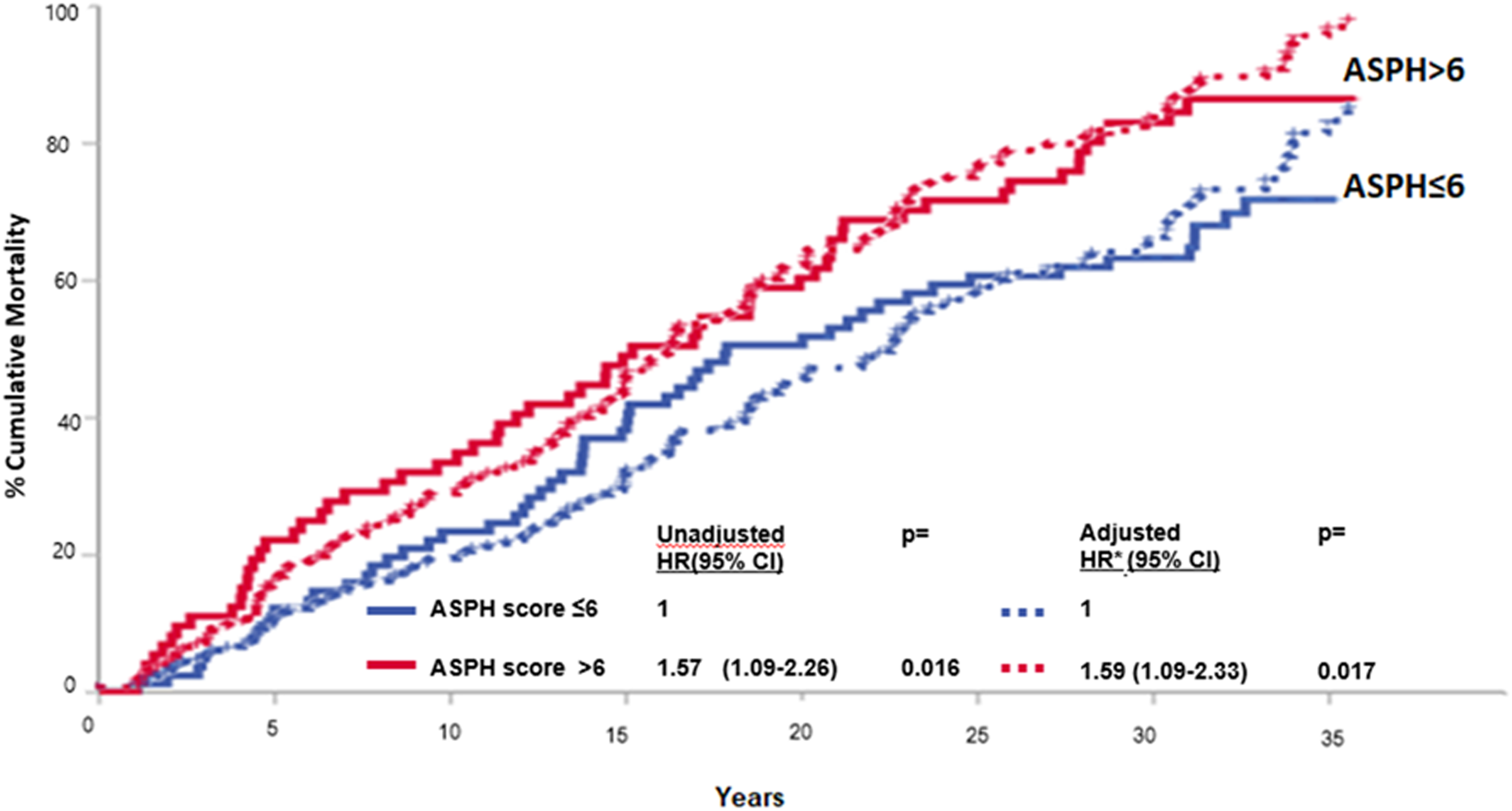
All-cause mortality by ASPH total score.
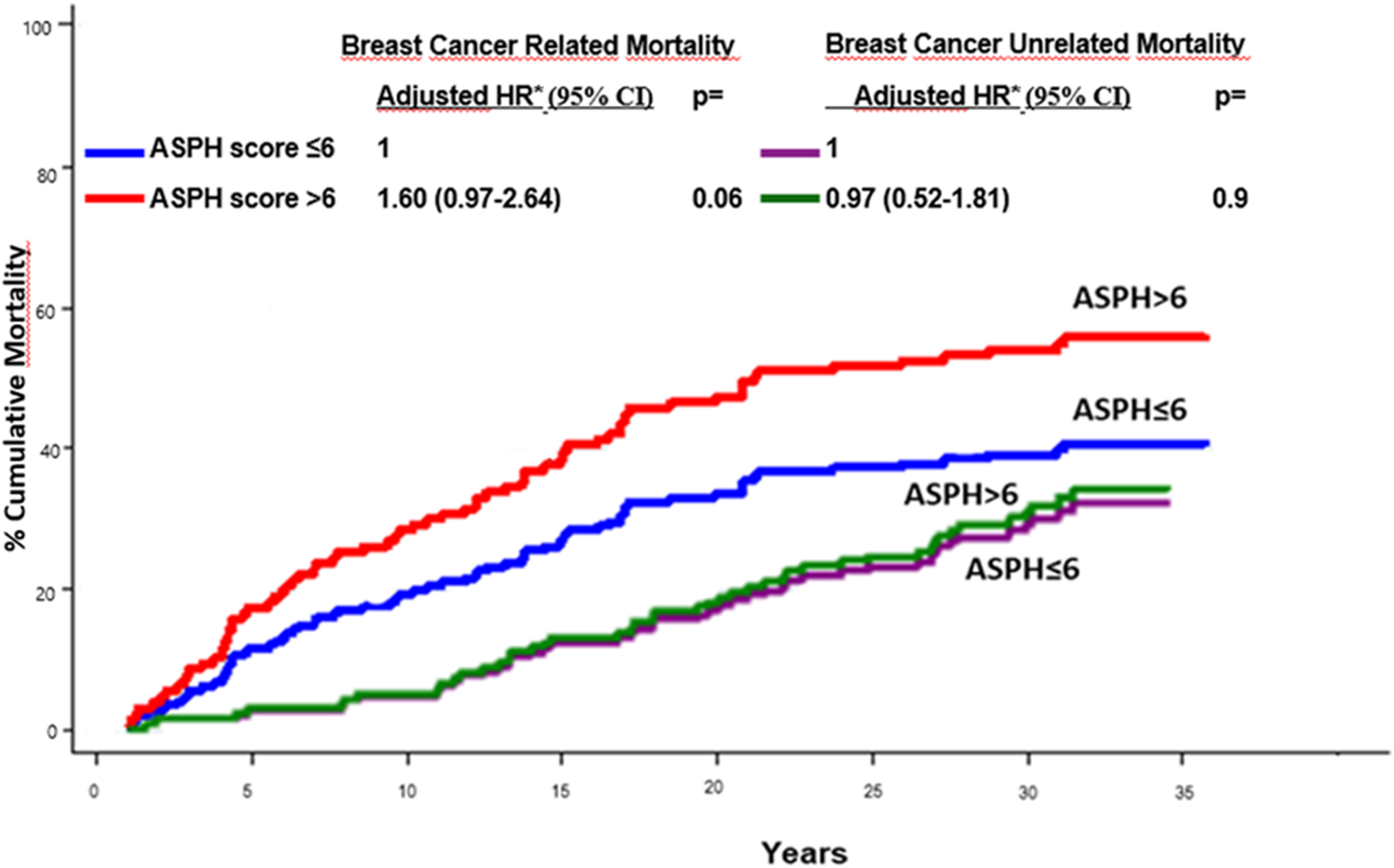
Mortality by ASPH total score in breast cancer related (n = 72) and breast cancer unrelated (n = 45) mortality groups.
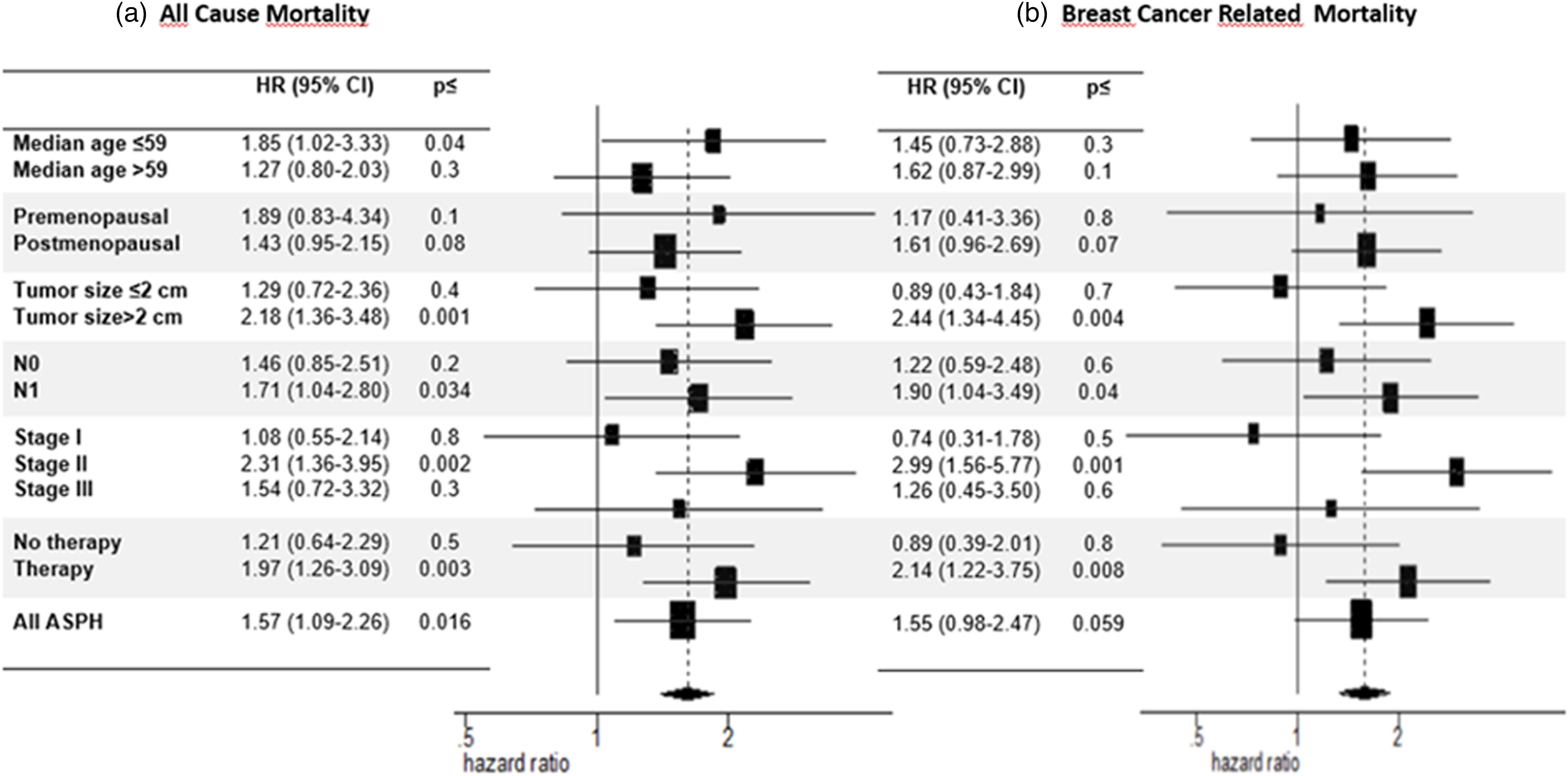
Forest plot of all-cause (a) and breast-cancer-related (b) mortality risk according to ASPH total score (>6 vs. ≤6).
Univariate and multivariate analysis of all-cause mortality risk.
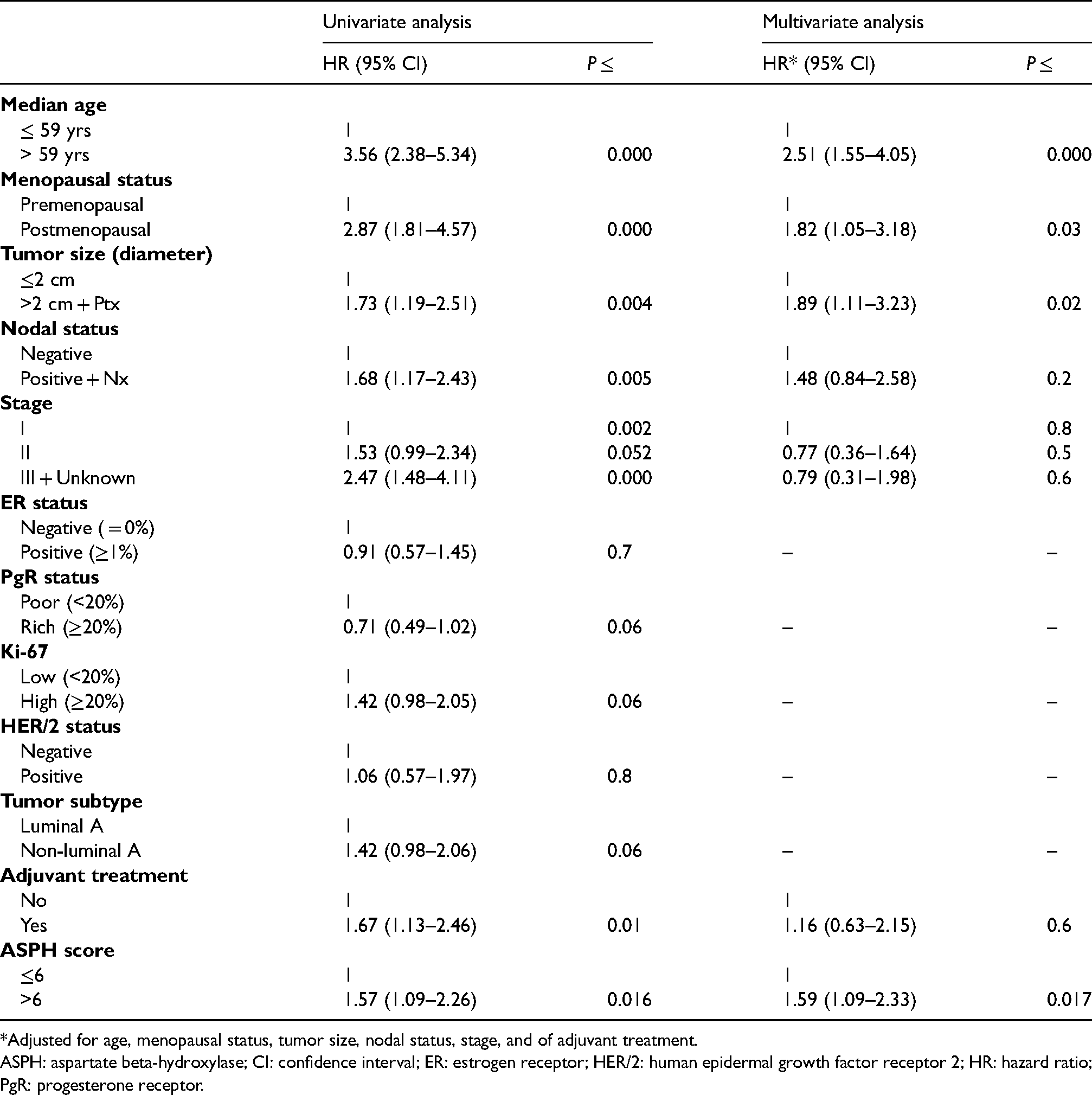
*Adjusted for age, menopausal status, tumor size, nodal status, stage, and of adjuvant treatment.
ASPH: aspartate beta-hydroxylase; CI: confidence interval; ER: estrogen receptor; HER/2: human epidermal growth factor receptor 2; HR: hazard ratio; PgR: progesterone receptor.
Discussion
Correlation of ASPH expression with clinical-pathologic characteristics
As already mentioned, lower ASPH scores were found in patients with higher PgR levels and in tumor subtypes luminal A and luminal B, which are known to bear a better prognosis and a higher response to endocrine therapy. 2 In contrast, higher ASPH levels were found in HER/2-like and triple-negative cancers, which are less responsive or unresponsive to endocrine therapy and bear a worse prognosis. 2 Our findings partially fit in with those previously reported by Zhang et al. 6 who observed a close relationship between ASPH expression and TNM stage, though no correlation with molecular subtypes was observed here. However, differences in the methods used to evaluate the ASPH expression score and those concerning the cohorts of patients included in these two studies should be taken into adequate account. Nevertheless, both studies demonstrate that ASPH expression appears to correlate with poor prognostic features, though both analyses should be considered merely explorative, due to the small size of the patient cohorts included in both studies.
Correlation of ASPH expression with mortality
It is worth noting that the total ASPH score significantly predicted for all-cause mortality in the univariate model and maintained an independent predictive value in the multivariable model. This latter model showed that age and menopausal status were also independent predictors of all-cause mortality, but this should be expected as the risk of death is obviously associated with aging. However, the confounding effect of treatments that differed in the two menopausal settings might have contributed to the independent prognostic role of menopausal status. Adjuvant chemotherapy was in fact preferentially given to premenopausal women, independent of hormone receptor status. Moreover, postmenopausal women with node-negative tumors did not usually receive adjuvant therapy. How ASPH score might add to the prognostic information associated with T size has not been specifically investigated here, but it can be assumed by the forest plots of the subgroup analysis, given that the prognostic effect of ASPH score was more evident in patients with T > 2 cm tumors (Figure 4(a) and (b)). The results of competitive risk analysis (Figure 3) are of particular interest as they clearly show that ASPH expression is specifically correlated with breast-cancer-related mortality and that the prognostic effect related to ASPH expression is maintained constant over time, even after such a prolonged observation time. Again, lack of statistical significance is presumably due to the small number of patients included in each sub-cohort identified on the basis of the cause of death. Our data suggest that higher ASPH expression in the primary tumor represents a strong prognostic marker, which, negatively and continuously over time, influences the clinical outcome of early breast cancer patients especially those with larger tumors, nodal involvement, a more advanced TNM stage, and those who received adjuvant therapy. This last observation might be explained by the fact that, at the time cohort patients were managed, adjuvant treatment (either chemotherapy, or tamoxifen for 2–5 years or both) was mainly reserved to node-positive patients. Unfortunately, the numbers were too small to allow further analysis according to the type of adjuvant treatment received.
Our results concerning mortality fit in with those previously reported by Zhang et al., 6 even though our paper compared to theirs differs on many aspects. The survival data reported by those investigators were in fact retrieved from two informatic platforms (Oncomine and GEPIA datasets), which included only BRCA-positive women. Moreover, ASPH mRNA expression was utilized for clinical correlations. The Chinese study also provides information relative to the hub genes related to ASPH expression, which might have functional roles in the occurrence and progression of breast cancer. No mortality data were provided by Shimoda et al., 7 though their study clearly shows a negative correlation between ASPH expression and recurrence-free survival. However, again, the study was limited to a selected group of patients with ER-positive, node-negative tumors all of whom received adjuvant endocrine therapy but not chemotherapy. Even in this case, the expression of ASPH mRNA was evaluated and data were retrieved by informatic platforms. The data achieved by these investigators, both in their in vitro experiments and through bio-informatic investigations, support the putative prognostic role of ASPH expression in the analyzed population and the putative predictive value of the protein relative to the response to adjuvant endocrine therapy.
We acknowledge several limitations of our cohort study. It is retrospective and included a relatively small number of patients, who were randomly selected on the basis of the availability of their TMA: this did not allow us to perform subgroup analyses that might be appropriate (see prognostic significance of ASPH expression vs. type of adjuvant treatment) and it may slightly flaw the statistical significance of data. Finally, there is no doubt that staging migration has occurred over years for breast cancer as well as for the majority of tumors that benefit from screening. Therefore, it is more than likely that the population selected by us for this retrospective study does not fit in perfectly with the average population of patients who are presently diagnosed with breast cancer in everyday practice. Standards of care have also changed over time with respect to those utilized at the time our cohort patients were managed. However, our study is strong in providing solid data with such a long follow-up time (median follow-up time: 33 years, range1.1–35.7 years). This was crucial to assess the prognostic effect of ASPH expression over time, considering that not all the markers available in breast cancer have been proven so far to maintain their predictive value over time. Also, it provides mortality data stratified according to the cause of death, which allow us to show the specificity of ASPH expression with respect to breast-cancer-related mortality.
Conclusions
ASPH IHC determination is easy, affordable, and cheap, and may add important additional information to the other clinical–pathological variables. Standardization of the IHC technique and prospective validation of scoring methods and of cut-off is also required before being able to include an ASPH total score among the variables putatively valuable for decision making in early breast cancer. The findings of our cohort study need to be confirmed on a prospective basis in larger cohorts. However, it seems to us that our study and previous studies by Zhang et al. 6 and Shimoda et al. 7 point in the same direction and support the important prognostic role of ASPH expression in early breast cancer. Moreover, present evidence encourages interest in ASPH also as a potential predictor of the response to specific treatments such as endocrine therapy. This has also been suggested by Shimoda et al. 7 Lastly, ASPH may prove to be a novel therapeutic target 15 that might be particularly useful in those patients (i.e., triple-negative patients) who show high expression levels of the protein and a poor response to endocrine therapy and chemotherapy. 2
Supplemental Material
sj-docx-1-jbm-10.1177_03936155221108412 - Supplemental material for The prognostic value of aspartate beta-hydroxylase in early breast cancer
Supplemental material, sj-docx-1-jbm-10.1177_03936155221108412 for The prognostic value of aspartate beta-hydroxylase in early breast cancer by Paola Barboro, Alessandra Rubagotti, Silvia Poddine, Federica Grillo, Luca Mastracci and Francesco Boccardo in The International Journal of Biological Markers
Footnotes
Acknowledgments
The authors are grateful to Dr. Claudia Casella (Epidemiology Unit-Tumor Registry, IRCCS Policlinico San Martino, Genoa, Italy) for helping with the mortality data collection.
Author contributions
Boccardo F, Barboro P, and Rubagotti A conceived and designed the study; Rubagotti A performed the statistical analysis; Poddine S collected the breast tissues materials and cooperated to data collection; Grillo F and Mastracci L were involved in IHC analysis; Boccardo F, Barboro P, Rubagotti A, and Grillo F all contributed to writing the manuscript. All authors read and approved the final manuscript. Barboro P and Rubagotti A contributed equally and are co-first authors.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Conflicts of interest
The authors declare that they do not have any conflict of interest.
Ethics approval
Study protocol was approved by the Ethics Board of Regione Liguria, Italy (N.367/2021-DB id 11592).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Italian Ministry of Health funds, (grant number 5X1000 and “Ricerca Corrente” 2020–2021).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
