Abstract
Introduction
Head and neck squamous cell carcinomas (HNSCCs) are cancers with generally poor prognosis. Outcomes have not improved in decades, with more than half of the patients presenting with lymph node metastases at the time of diagnosis. A unique subtype of HNSCC, cancer of unknown primary of the head and neck (HNCUP) is associated with a poor outcome. Increased expression of the D2-40 gene (podoplanin) has been described for several human malignancies and has been associated with increased metastatic potential of cancer cells.
Methods
In order to examine the role of podoplanin in lymph node metastasis of HNSCC generally and HNCUP specifically, we evaluated the prognostic impact of podoplanin expression in HNSCC- (n = 68) and HNCUP-associated lymph node metastases (n = 30). The expression of podoplanin was analyzed by immunohistochemical staining of lymph node tissue samples and correlated with clinical and histopathological data.
Results
We found a non-significant tendency towards a higher podoplanin expression in HNCUP compared to HNSCC lymph node metastases and a significant correlation between a high podoplanin expression and advanced node-stage classification. Podoplanin expression had no significant impact on overall survival for both groups and did not correlate with human papillomavirus tumor status.
Conclusion
Taken together, our results suggest that upregulation of podoplanin may be associated with a stimulation of lymphatic metastasis in head and neck cancer.
Keywords
Introduction
Head and neck squamous cell carcinomas (HNSCCs) are the sixth most common cancer in the world, accounting for more than 5% of all human malignancies with 890,000 new cases and 450,000 deaths in 2018.1,2 The prognosis of patients with HNSCC is influenced by various factors, including comorbidities, and tumor primary site location and stage.3–6 Of particular importance, lymph node metastases are frequently found at the time of initial diagnosis, and the presence of node-positive disease markedly worsens a patient’s prognosis. As such, node-positive disease is treated with intensified therapy; for example, the addition of adjuvant treatment after surgery. 7 Due to the frequent delays of clinical diagnoses and a lack of new surgical and medical treatment methods, 5-year survival with HNSCC has not improved over the past 20 years and remains at 50–60%,8,9 which emphasizes an urgent need for new diagnostic tools for early cancer detection and innovative treatment approaches.
In 3–5% of newly diagnosed cases of malignant epithelial tumors, including HNSCC, no primary tumor can be found, leading to the diagnosis of “cancer of unknown primary” (CUP).10,11 There are 1.5–3% of all head and neck cancers that initially present as cervical lymph node metastases without an evident primary tumor, known as head and neck CUP (HNCUP)12,13 and are associated with a poorer prognosis compared to other HNSCCs. The mechanism by which certain primary tumors remain clinically occult remains largely unknown with several existing hypotheses on tumor evolution and development. 14
Podoplanin, also known as D2-40, is a 38 kDa mucin-type transmembrane glycoprotein expressed on endothelial cells of lymphatic vessels, but not blood vessels15,16 and is therefore suggested to be a specific marker of lymphatic tissue. It can also be found in kidney podocytes, type I pneumocytes, cardiomyocytes, placenta, or mesothelial cells.16–18 On a functional level, a role in lymphangiogenesis, platelet production, and immune response was recently described in the literature.19–22 Apart from its physiological role, podoplanin expression was also detected in various tumor cell types, including squamous cell carcinomas (SCCs) of the lung, skin, and HNSCC.23–25 In patients with oral leukoplakia, studies showed that high podoplanin expression can predict the risk of transformation into SCC.26,27 Additionally, the role of podoplanin as a prognostic biomarker was shown in oral cancer, with a worse prognosis for patients with high expression levels.26,28 Another study indicated an association of high podoplanin expression with sentinel lymph node metastases in carcinomas of the oral cavity and oropharynx. 29 A potential role of podoplanin in HNCUP has not been investigated to our knowledge.
Here, we analyzed and compared podoplanin expression in cervical lymph node metastases of HNSCC and HNCUP patients and correlated expression levels with clinical and histopathological features in order to further clarify the role of podoplanin in the metastatic behavior of head and neck cancers, and its validity as a prognostic biomarker.
Material and methods
Patients and tissue samples
In total, 68 HNSCC and 30 HNCUP patients all with histologically proven SCCs who were treated at the Department of Otorhinolaryngology, Head and Neck Surgery at the Saarland University Medical Center (Homburg, Germany) between 2009 and 2014, were included in the study. All patients gave their informed consent for the use of tissue samples and clinical data for our study. All experiments were carried out in accordance with the Declaration of Helsinki and the relevant national ethical standards. For immunohistochemical analyses, formalin-fixed paraffin-embedded (FFPE) tissue samples of cervical lymph node metastases were obtained for all patients. Details on clinical and histopathological data of the patients are shown in Table 1. The study was approved by the Saarland Medical Association ethics review board (index number 280/10).
Clinical data of head and neck squamous cell carcinomas (HNSCC) and cancer of unknown primary of the head and neck (HNCUP) patients.
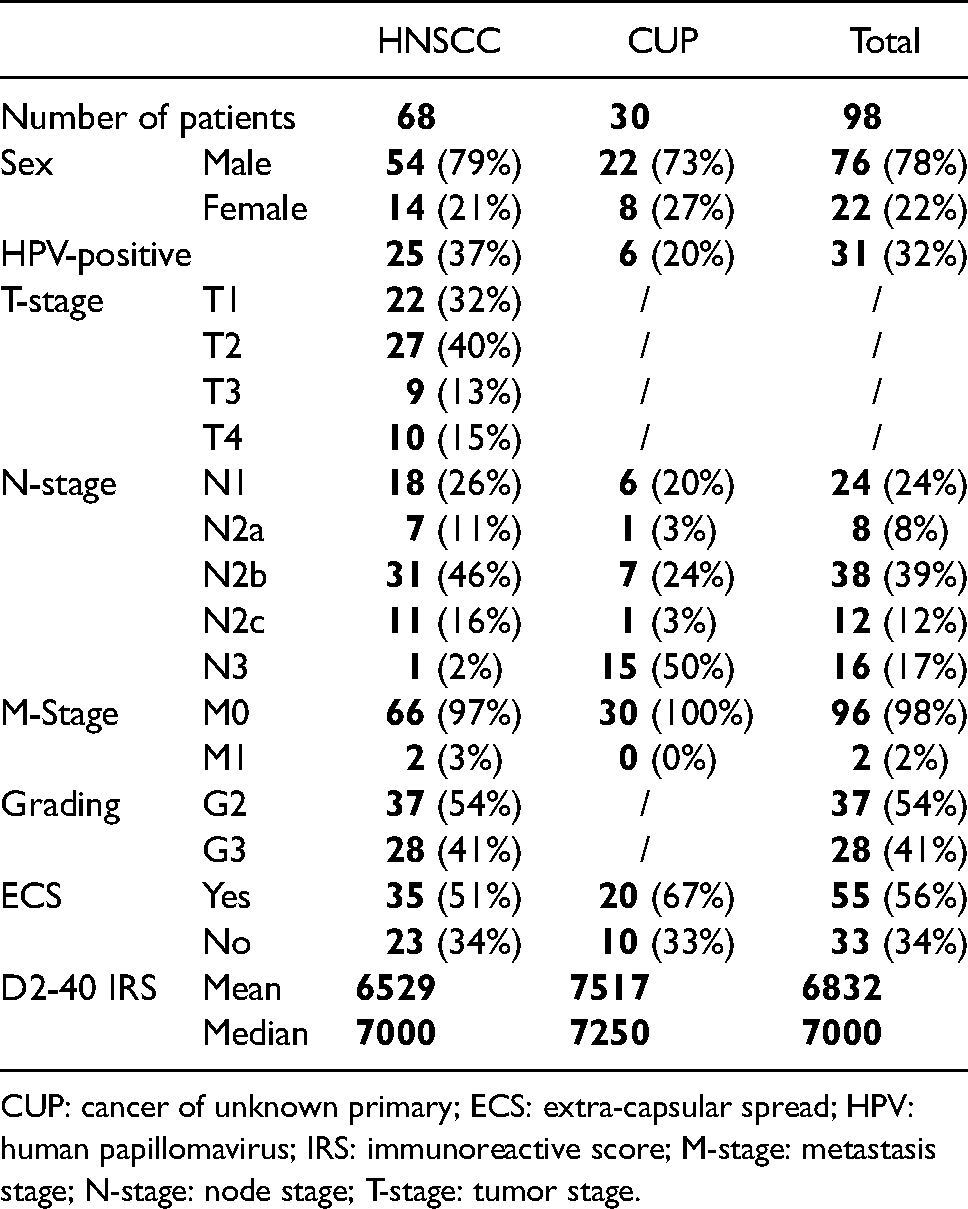
CUP: cancer of unknown primary; ECS: extra-capsular spread; HPV: human papillomavirus; IRS: immunoreactive score; M-stage: metastasis stage; N-stage: node stage; T-stage: tumor stage.
Immunohistochemistry
FFPE tissue sections were obtained and used for immunohistochemical staining, targeting podoplanin. Therefore, 4-μm sections were obtained using a Leica RM 2235 rotary microtome (Leica Microsystems, Wetzlar, Germany), transferred onto Superfrost Ultra Plus microscope slides (Menzel-Gläser, Braunschweig, Germany) and dried in an incubator at 37°C overnight. After deparaffinization, heat-induced epitope-unmasking was performed in a 10 mM citrate buffer (pH 6.0). After cooling down to room temperature, unspecific protein binding sites were blocked by incubation in 200 mL phosphate-buffered saline (PBS); Sigma Aldrich, St. Louis, MO, USA) and 6 g bovine serum albumin (BSA; Sigma Aldrich) under pH control (7.2) for 30 min at room temperature. Afterwards, the slides were incubated with the primary antibody (1:60 dilution in 1% BSA/PBS; anti-podoplanin, clone D2-40; ab77854, Abcam, Cambridge, UK) for 1 hour at room temperature. For visualization the REAL detection system Alkaline Phosphatase (Dako Agilent Technologies, Glostrup, Denmark) was used according to the manufacturer's instructions. Finally, the slides were counterstained with hematoxylin (Sigma Aldrich). Each analysis included negative controls by omission of the primary antibody and positive controls by staining healthy tonsil tissue from tonsillectomy patients that were treated at our department. Podoplanin-immunoreactivity was evaluated using an immunoreactive score (IRS) according to Remmele and Stegner. 30 In this scoring system, IRS values ranging from 0 to 12 are calculated based on the percentage of positively stained cells and the staining intensity of those cells (0–1: no expression, 2–3: mild expression, 4–8: moderate expression, 9–12: strong expression). Two single scores for the percentage of stained cells (0: no positive cells, 1: <10% of positive cells, 2: 10–50% positive cells, 3: 51–80% positive cells, 4: >80% positive cells) and for staining intensity (0: no reaction, 1: mild reaction, 2: moderate reaction, 3: intense reaction) are multiplied and result in the final IRS. All immunohistochemical staining was evaluated by three experienced examiners including one board-certified histopathologist
Human papillomavirus status
To determine whether the HNSCC and HNCUP tumors were human papillomavirus (HPV)-associated, a combination of HPV-DNA-polymerase chain reaction (PCR) and a simultaneous immunohistochemical detection of p16 and Ki67 was used as recently described. 31 DNA was extracted from the FFPE samples using the QIAamp DNA Blood Mini Kit according to the manufacturer’s instructions (Qiagen, Hilden, Germany). The following HPV-PCR was performed with the LightCycler 2.0 instrument (Roche, Mannheim, Germany) using the GP5 + /6 + primers as described by De Roda Husman et al. 32 SYBR green as well as gel electrophoresis were used for detection. After initial denaturation at 95°C for 15 min, 45 PCR cycles followed with denaturation at 95°C for 10 s, annealing at 45°C for 5 s and elongation at 72°C for 18 s. After amplification, a melting curve was performed at temperatures between 45°C and 95°C, with temperature increasing at a rate of 0.2°K s − 1. Melting Temperature™ for the HPV16-positive control was 79°C and 82°C for the HPV18-positive control. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) PCR was performed in parallel for each sample as a control as described in Ruprecht et al. 33 For the p16-Ki67 dual immunohistochemical staining the CINtec PLUS kit (Roche mtm Laboratories, Heidelberg, Germany) was applied according to the manufacturer's instructions. Each analysis included positive and negative controls.
Only tumors with a positive HPV-DNA-PCR result as well as a dual expression of p16 and Ki67 in lesional cells were rated as HPV-positive.
The Cancer Genome Atlas data analysis
In The Cancer Genome Atlas (TCGA) program, more than 10,000 samples from more than 20 different tumor entities were analyzed in a multicenter project using high-throughput genome sequencing techniques and bioinformatics. DNA as well as RNA sequencing data from those patients are publicly available and can be used for analyzing gene expression signatures of interest. To evaluate the influence of podoplanin expression in the TCGA-HNSC cohort (n = 518) on the patients’ overall survival (OS), RNAseq-based expression data and clinical data were analyzed using the publicly available software algorithm “Gene Expression Profiling Interactive Analysis” (GEPIA) as described by Tang et al. 34
Statistical analysis
GraphPad Prism 7.0 (GraphPad Software, La Jolla, CA, USA) and SPSS version 16 (IBM, Ehningen, Germany) were used for statistical analysis presuming a significance level of 5% (α = 0.05) and a statistical power of 80% (ß = 0.8). The existence of normal distribution of podoplanin expression was controlled by the Kolmogorov–Smirnov test, the Anderson–Darling test, the D’Agostino–Pearson test, and the Shapiro–Wilk test. Homogenous variance was checked by the Levine test. If parameters showed no normal distribution in at least one of the aforementioned tests, the non-parametric Mann–Whitney U test was used. In case of normal distribution in all of the aforementioned tests, a two-sided t-test was used. For multi-variable comparisons, a one-way ANOVA test was used. Significance levels and
Results
Podoplanin expression in lymph node metastases of HNSCC and HNCUP patients
Immunohistochemical analysis of podoplanin expression in lymph node metastases of HNSCC and HNCUP patients showed numerically higher expression levels in HNCUP compared with HNSCC patients, although this difference was not statistically significant (

Differential cytoplasmic expression of podoplanin in cervical lymph node metastases. (a) Weak expression: immunoreactive score = 1, (b) High expression: immunoreactive score = 12. Scale bar 200 µm.
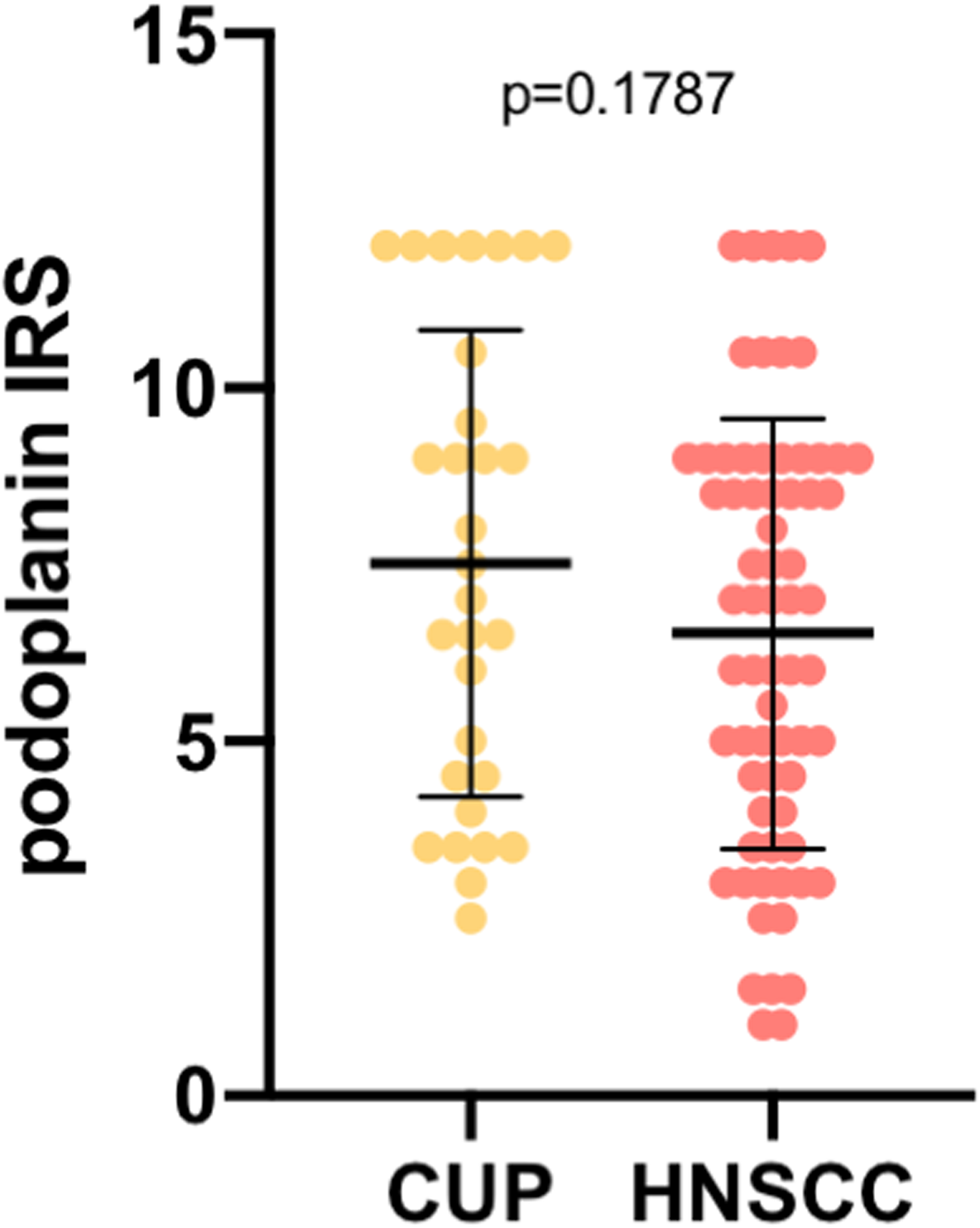
Podoplanin expression in lymph node metastases in patients with cancer of unknown primary of the head and neck (HNCUP) and head and neck squamous cell carcinomas (HNSCC). In the scatter plots, the mean is indicated by a black horizontal line with standard deviation illustrated by an error bar. Every dot represents one patient's data (Mann–Whitney U test).
Correlation of podoplanin expression with tumor node metastasis stages, tumor differentiation, and extracapsular spread
Correlation of podoplanin expression with (tumor node metastasis) stages revealed no significant association of expression levels with T category (Figure 3(a)). When correlating podoplanin expression with lymphatic metastasis indicated by N category, we found significantly higher levels of podoplanin in lymph node metastases of N3 stage compared to N2 (

(a) Podoplanin expression in lymph node metastases compared to the T stage of the primary tumor. In the scatter plots, the mean is indicated by a horizontal line with standard deviation illustrated by error bars. Every dot represents one patient's data (Mann–Whitney U test). (b) Podoplanin expression in lymph node metastases compared to the N stage. Patients with cancer of unknown primary of the head and neck (HNCUP) and head and neck squamous cell carcinoma (HNSCC) are grouped together and dealt with as a single cohort. In the scatter plots, the mean is indicated by a horizontal line with standard deviation illustrated by error bars. Every dot represents one patient's data (one-way-ANOVA-test). (c) Podoplanin expression in lymph node metastases with and without extracapsular spread (ECS) for HNCUP and HNSCC as a single cohort. In the scatter plots, the mean is indicated by a horizontal line with the standard deviation illustrated by error bars. Every dot represents one patient's data (Mann–Whitney U test).
When comparing podoplanin expression between lymph node metastases with and without extracapsular spread (ECS), we found significantly higher IRS values in ECS cases compared to non-ECS cases when HNCUP and HNSCC patients were grouped together (
Correlation of podoplanin expression with HPV tumor status
For every patient, HPV status was determined by a combination of HPV-DNA-PCR and p16 immunohistochemistry using the lymph node metastases FFPE material. Analysis of podoplanin expression in HPV-positive versus HPV-negative cases showed no significant association between HPV status and podoplanin IRS (
Prognostic significance of podoplanin in HNCUP and HNSCC patients
OS was analyzed for all HNCUP and HNSCC patients with a median follow up of 52 months (HNCUP: 39 months; HNSCC: 57 months) ranging from 1 month to 186 months. When comparing the OS between the HNSCC and HNCUP patients, we found a significantly shorter OS for the HNCUP group compared with the HNSCC group (

(a) Kaplan–Meier analysis of overall survival of cancer of unknown primary of the head and neck (HNCUP; red) and head and neck squamous cell carcinoma (HNSCC) patients (green). (b) Overall survival in relation to D2-40 expression. Kaplan–Meier analysis of overall survival of patients with low D2-40 expression (red; IRS 0–6) and with high D2-40 expression (green; IRS 7–12). HNCUP and HNSCC groups are combined and dealt as a single cohort. Censored data in (a) and (b) are indicated by black or grey dots, respectively (Log-rank test). (c) Kaplan–Meier analysis of overall survival of TCGA-HNSC patients with high (red; n = 259) versus low (blue; n = 259) podoplanin expression based on RNAseq data analysis by Tang et al. 34
Additionally, we analyzed the impact of podoplanin expression on the OS in the TCGA-HNSC cohort (n = 518) based on RNAseq data using the Gene Expression Profiling Interactive Analysis (GEPIA) as described by Tang et al.
34
In this cohort, high podoplanin expression in the primary tumor was associated with a significantly shorter OS (
Discussion
In this study, we analyzed podoplanin expression by immunohistochemical staining of lymph node metastases from 68 HNSCC and 30 HNCUP patients and correlated expression levels with clinical and histopathological data. We found significantly higher podoplanin expression in tumors with nodal metastases of higher N category and a non-significant tendency towards higher podoplanin levels in HNCUP cases compared to HNSCC cases. Survival analyses revealed no significant role of podoplanin as a prognostic biomarker in our cohort. Taken together, our results indicate a potential role of podoplanin in the molecular process of lymphatic metastasis and oral carcinogenesis.
Apart from our study, only a few other studies analyzed podoplanin expression in premalignant and malignant lesions of the oral/pharyngeal mucosa and addressed its potential role as a prognostic biomarker and molecular driver of oral carcinogenesis so far. Preuss et al. 35 analyzed podoplanin expression in 107 oropharyngeal carcinoma patients and found no significant correlation of podoplanin expression with the T, N, and M stages and tumor grading. In accordance with our results, no association of podoplanin levels with HPV tumor status, defined by p16 immunohistochemistry and HPV-DNA-PCR, was found. A study by Huber et al. 29 showed a significant association between podoplanin expression in the primary tumor and positive sentinel lymph node biopsies pointing towards a relevance of podoplanin in lymphatic metastasis of HNSCCs; these findings are consistent with our data.
With respect to a potential role of podoplanin as a prognostic biomarker in head and neck cancer, study results published in the literature in the past have been heterogeneous. While Yuan et al. 28 found a significantly shorter disease-free survival in a limited number of n = 60 oral tongue cancer patients with high-level podoplanin expression, Preuss et al. 35 did not identify any association between podoplanin expression and OS, which is in line with our results. Kreppel et al. 27 investigated podoplanin expression in tissue samples from 80 oropharyngeal cancer patients. In this cohort, the 5-year OS rate for patients with high levels of podoplanin expression was significantly lower than for patients with low and moderate expression. According to these results, high podoplanin expression was associated with a significantly shorter OS in the TCGA-HNSCC cohort as shown in Figure 4(c). This is in contrast to our results showing no significant association of podoplanin expression with survival. One possible explanation could be the much lower number of analyzed patients in our cohort (n = 98) compared to the TCGA-HNSC cohort (n = 518). Additionally, we analyzed podoplanin expression in lymph node metastases while TCGA data are based on primary tumor tissue sequencing. Further studies will be needed to clarify the influence of podoplanin on the prognosis of HNSCC patients.
From a functional point of view, our findings of a significant association between podoplanin expression and N-stages and ECS indicate a role of podoplanin in tumor cell metastasis, invasiveness, and aggressiveness. This theory is supported by Hamada et al., 36 who showed that high expression of podoplanin in tumor budding cells can predict neck lymph node metastasis in patients with low grade SCC of the tongue. Additionally, Martín-Villar et al. 17 showed that podoplanin can induced a reorganization of the actin cytoskeleton by recruitment of ezrin and the induction of cell-surface protrusions thus inducing tumor cell invasion and migration. Almeida et al. 37 showed that high podoplanin expression can predict perineural invasion in oral cancer. Mermod et al. 38 found a correlation of high levels of podoplanin expression with the presence of extracapsular spread by multivariate analysis. These data are supported by functional analyses, including cell culture and animal experiments in head and neck cancer as well as other human malignancies. Sun et al. 39 showed a downregulation of podoplanin expression by miR-363 in different head and neck cancer cell lines, which attenuated cancer cell metastasis and invasion. Scholl et al. 40 showed, in a mouse keratinocyte model, that ectopic expression of podoplanin in MCA3D cells induces cell surface extensions and increased cell motility, pushing these cells into a phenotypic conversion with the acquisition of malignant characteristics. Furthermore, lymph node metastasis was stimulated by podoplanin expression in MCA3D cells upon injection into mice. In a human breast carcinoma xenograft model, Cueni et al. 41 found that podoplanin expression enhanced lymphangiogenesis and metastasis to regional lymph nodes without affecting primary tumor growth, which perfectly fits with our observation of podoplanin expression correlating with N but not T stages.
A caveat to these findings is that lower number of patients in each cohort (n = 68 for HNSCC; n = 30 for HNCUP) may have limited statistical power, although our study represents one of the largest cohorts in the existing literature on podoplanin and HNSCC, and is the first study analyzing podoplanin expression in HNCUP patient tissue. Additionally, data on extracapsular spread and tumor differentiation were only available for a subset of all included patients, which limits the validity of statistical analyses.
Taken together, we report a significant correlation of high podoplanin expression with advanced N stages and ECS, indicating a potential role of podoplanin in migration, invasiveness, and lymphatic metastasis of head and neck cancer, without affecting patient prognosis. Additional clinical studies, including a higher number of cases and functional studies using cell culture and animal models, are needed to further elucidate the clinical and therapeutic relevance of podoplanin in head and neck cancer and its role in the molecular mechanisms of oral carcinogenesis.
Supplemental Material
sj-tif-1-jbm-10.1177_03936155221105524 - Supplemental material for Podoplanin expression in lymph node metastases of head and neck cancer and cancer of unknown primary patients
Supplemental material, sj-tif-1-jbm-10.1177_03936155221105524 for Podoplanin expression in lymph node metastases of head and neck cancer and cancer of unknown primary patients by Jan Philipp Kühn, Florian Bochen, Sandrina Körner, Bernhard Schick, Mathias Wagner, Sigrun Smola, Barbara Berkó-Göttel, Luc G.T. Morris, Jingming Wang, Alessandro Bozzato and Maximilian Linxweiler in The International Journal of Biological Markers
Supplemental Material
sj-tif-2-jbm-10.1177_03936155221105524 - Supplemental material for Podoplanin expression in lymph node metastases of head and neck cancer and cancer of unknown primary patients
Supplemental material, sj-tif-2-jbm-10.1177_03936155221105524 for Podoplanin expression in lymph node metastases of head and neck cancer and cancer of unknown primary patients by Jan Philipp Kühn, Florian Bochen, Sandrina Körner, Bernhard Schick, Mathias Wagner, Sigrun Smola, Barbara Berkó-Göttel, Luc G.T. Morris, Jingming Wang, Alessandro Bozzato and Maximilian Linxweiler in The International Journal of Biological Markers
Supplemental Material
sj-tif-3-jbm-10.1177_03936155221105524 - Supplemental material for Podoplanin expression in lymph node metastases of head and neck cancer and cancer of unknown primary patients
Supplemental material, sj-tif-3-jbm-10.1177_03936155221105524 for Podoplanin expression in lymph node metastases of head and neck cancer and cancer of unknown primary patients by Jan Philipp Kühn, Florian Bochen, Sandrina Körner, Bernhard Schick, Mathias Wagner, Sigrun Smola, Barbara Berkó-Göttel, Luc G.T. Morris, Jingming Wang, Alessandro Bozzato and Maximilian Linxweiler in The International Journal of Biological Markers
Supplemental Material
sj-docx-4-jbm-10.1177_03936155221105524 - Supplemental material for Podoplanin expression in lymph node metastases of head and neck cancer and cancer of unknown primary patients
Supplemental material, sj-docx-4-jbm-10.1177_03936155221105524 for Podoplanin expression in lymph node metastases of head and neck cancer and cancer of unknown primary patients by Jan Philipp Kühn, Florian Bochen, Sandrina Körner, Bernhard Schick, Mathias Wagner, Sigrun Smola, Barbara Berkó-Göttel, Luc G.T. Morris, Jingming Wang, Alessandro Bozzato and Maximilian Linxweiler in The International Journal of Biological Markers
Footnotes
Acknowledgments
The excellent technical support and experimental assistance of Monika Hoffmann, Ulrike Bechtel, and Barbara Linxweiler is gratefully acknowledged.
Author contributions
JPK and ML wrote the paper, conceived and designed the analysis, collected the data, contributed data and analysis tools and performed the laboratory work and the analysis. AB and BS contributed to the study design and concept. FB SK helped with the laboratory work. MW performed the histological examination of the lymph node metastasis, SS and BBG performed and evaluated the HPV status. LM and JW critically examined the manuscript and evaluated and corrected it.
All authors read and approved the final manuscript.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All authors assert that all procedures used met the approval by local ethics review committee.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a HOMFOR (Homburger Forschungsförderungsprogramm) grant and Else Kröner-Fresenius Stiftung grant (2017_A83) to ML.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
