Abstract
Background
The NLRP3 inflammasome is significantly associated with tumor development and metastasis in various malignancies. However, the significance of the NLRP3 inflammasome in pancreatic adenocarcinoma has not been fully determined. Therefore, we try to evaluate the expression of the NLRP3 inflammasome in pancreatic adenocarcinoma and analyzed its prognostic significance.
Methods
This cohort study enrolled 98 patients with primary pancreatic adenocarcinoma who received curative surgery. The NLRP3 inflammasome expression levels in cancer tissue were determined by immunohistochemistry, and compared with that of adjacent normal tissues. The association between NLRP3 inflammasome expression levels and baseline clinicopathological characteristics were also analyzed. Moreover, the correlation between NLRP3 inflammasome expression levels and survival was analyzed by log-rank test, and the survival curve was made by the Kaplan–Meier survival analysis.
Results
Expression of each NLRP3 inflammasome component in cancer tissue was higher than that in the adjacent normal tissues (all P < 0.05), including NLRP3, IL-1β, ASC, and Caspase-1. All four components of the NLRP3 inflammasome were closely associated with clinical stage and lymph node status (all P < 0.05). The Kaplan–Meier log rank test showed that the high expression level of the NLRP3 inflammasome was significantly related to poor overall survival in pancreatic adenocarcinoma patients.
Conclusions
NLRP3 inflammasome expression was upregulated in cancer tissue and closely associated with the prognosis of operable pancreatic adenocarcinoma.
Introduction
Pancreatic adenocarcinoma remains one of the most lethal digestive cancers, serving as a major constitution of cancer-related mortality and mortality worldwide, and presenting as a challenge for surgeons due to the high recurrence rate and robust chemoresistance, resulting in an overall 5-year survival rate of no more than 7%.1-3 Surgical resection is potentially considered as an only curative treatment approach for patients with pancreatic adenocarcinoma.4,5 However, when diagnosed with pancreatic adenocarcinoma, only 15%–20% of patients had a chance of undergoing curative surgery, and those who received curative resection had a postoperative 5-year survival rate of less than 20%.6,7 Recently, a large number of clinical and experimental studies focused on the prognosis analysis of pancreatic cancer and introduced various clinical and pathological prognostic indictors.8,9 However, no prognostic indictors concerning pathogenesis and development of pancreatic adenocarcinoma, and cannot be serves as a therapeutic target, and has no significance for promoting prognosis.
The occurrence and development of pancreatic cancer are closely related to chronic inflammation.8,9 The persistent inflammation in the tumor microenvironment is conducive to the survival and proliferation of tumor cells and intratumoral microvessels formation, thus promoting metastasis. 10 A variety of inflammatory cytokines participate in the development of tumor-related inflammation, including interleukin (IL)-1β and IL-18. 11 It has been confirmed that the secretion and releasing of IL-1β and IL-18 mainly resulted from activation of the NLRP3 inflammasome. 12 The NLRP3 inflammasome is a multi-protein complex, which is mainly distributed in cytoplasm, including the NLRP3 protein, ASC adapter protein, and pro-Caspase-1. It is activated to form an inflammasome complex under the stimulation of an infectious agent or tumor to produce Caspase-1-mediated inflammation, including the secretion of IL-1β and IL-18.13,14 In an experiment we reported that NLRP3 inflammasomes played curial roles in the proliferation and invasion of pancreatic cancer cells. 15 Therefore, in this present cohort study, we analyzed the expression characteristics of the NLRP3 inflammasome in patients with pancreatic cancer and their prognostic significance.
Materials and methods
Study design
This cohort study is a retrospective analysis of clinical and pathological data extracted from the institutional database. This study was approved by the Ethics Committee of the Chinese PLA General Hospital. Each patient provided written informed consent at the time of admittance.
Patients
All consecutive patients with primary pancreatic adenocarcinoma that received curative operations in the Chinese PLA General Hospital were enrolled. There were sufficient blank histological tissue slides for pathological staining studies in each enrolled patient. Inclusion criteria were: (a) resectable pancreatic adenocarcinoma according to preoperative enhanced magnetic resonance imaging; (b) histological examination of resected tissue samples; (c) age no younger than 18 years old; and (d) survival expectancy no less than 6 months. Exclusion criteria were: (a) patients that received any preoperative adjuvant therapies or palliative surgery; and (b) patients with acute and severe complications and comorbidities associated with prognosis. Demographic data of all enrolled patients were obtained from the clinical database, including sex, age, tumor location and size, T stage, lymph node status, and pathological differentiation. The T stage was assessed according to the UICC tumor node metastasis (TNM) classification. All patients were regularly followed up after being discharged, until to either the time of death or October 2015.
Immunohistochemistry
A tissue microarray with formalin-fixed paraffin-embedded pancreatic adenocarcinoma tissue specimens and corresponding paracancerous tissue from each enrolled patient was prepared. Immunohistochemistry (IHC) was performed according to an indirect immunoperoxidase method as previously reported, using The OptiView DAB IHC Detection Kit and OptiView Amplification Kit (Ventana Medical Systems, Tucson, AZ, USA). Briefly, sections of 4-µm thickness were incubated with specific primary antibodies following deparaffinization and rehydration, including rabbit anti-NLRP3 (1:250, Santa Cruz), rabbit anti-ASC (1:500, Santa Cruz), rabbit anti-caspase-1 (1:250, Santa Cruz), and rabbit anti- IL-1β (1:200, Santa Cruz) at 4°C for 24 h. After rinsing in phosphate-buffered saline, sections were incubated with horseradish-peroxidase-conjugated secondary antibodies (1:2000, DAKO) for 50 min at normal room temperature, then visualized by using the diaminobenzidine visualization method. The images were obtained with Nikon ECLIPSE LV 150N microscope camera and evaluated by Image Pro Plus software 6.0.
All results determination of all histological assessment were independently performed by two pathologists blind to any clinical data of patients. Both staining intensity and the percentage of positive tumor cells were evaluated as follows: (a) staining intensity: 0 = absence of staining; 1 = weak staining; 2 = moderate staining; and 3 = strong staining; and (b) percentage of positive tumor cells: 0 = 0–5% of staining; 1 = 5–25% staining; 2 = 25–50% staining; 3 = ≥50% staining. The final score was obtained by multiplying the staining intensity score and percentage score. The final score ≥4 was defined as high expression, <4 as lower expression.
Outcome evaluation and statistical analyses
Statistical analyses were performed by using SPSS statistical software version 20.0 (IBM, USA). All statistical tests were conducted via two-sided. P < 0.05 was considered as statistically significant.
Overall survival (OS) was considered as the primary outcome, which was defined as OS calculated as the duration between the date of surgery and the date of death from any cause or the endpoint. The survival analysis was performed by Kaplan–Meier analysis and comparisons between survival curves were conducted using the Log-rank test.
The expression characteristics of the NLRP3 inflammasome was considered as a secondary outcome. For comparisons of NLRP3 expression between pancreatic adenocarcinoma tissue and corresponding paracancerous tissue was analyzed by two-sided non-parametric Kruskal–Wallis and Mann–Whitney U-tests. The association of clinical variables and NLRP3 expression was evaluated with the Chi-square test
Results
Baseline characteristics
A total of 98 patients were enrolled in this study (58 males, 40 females, with a mean age of 58.4 ± 1.6 years). In terms of tumor location, 32 patients had a tumor on the pancreatic head and 66 patients had pancreatic body and tail cancer. For cancer clinical stage, 71 patients had TNM stage II tumor, 27 cases had a lesion with TNM stage I. Moreover, 53 cases had well/moderately differentiated adenocarcinoma, but 45 patients had a poor differentiated tumor. The details of the characteristics of all 98 patients are shown in Table 1.
Clinicopathological characteristics of all 98 patients with pancreatic adenocarcinoma.
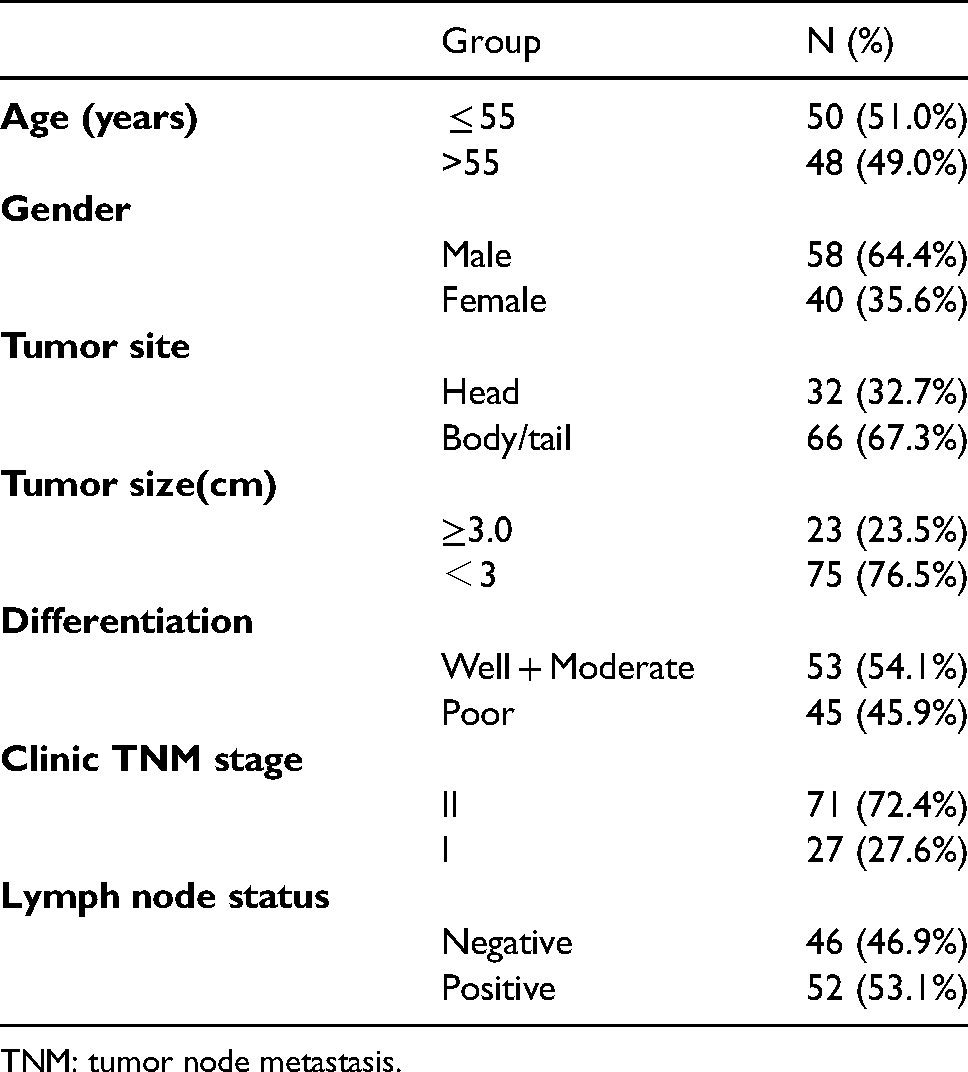
TNM: tumor node metastasis.
The expression status of NLPR3 inflammasome in pancreatic adenocarcinoma
NLPR3 inflammasome is mainly expressed in the cytoplasm. Expressions of NLRP3, ASC, Caspase-1, and IL-1β inflammasome components were upregulated in all pancreatic cancer tissue samples, whereas low expressions in adjacent normal pancreatic tissue were observed, as shown in Figure 1. According to the IHC scoring, among the 98 cases of pancreatic cancer: (a) 56 (57.1%) had a high NLRP3 expression and 42 had a low expression (42.9%); (b) 32 had a high ASC expression (32.7%) and 66 had a low expression (67.3%); (c) 47 (48.0%) had a high Caspase-1 expression and 51 (52.0%) had a low expression; and (d) 62 (63.3%) had a high IL-1β expression and 36 (36.7%) had a low expression (Table 2).
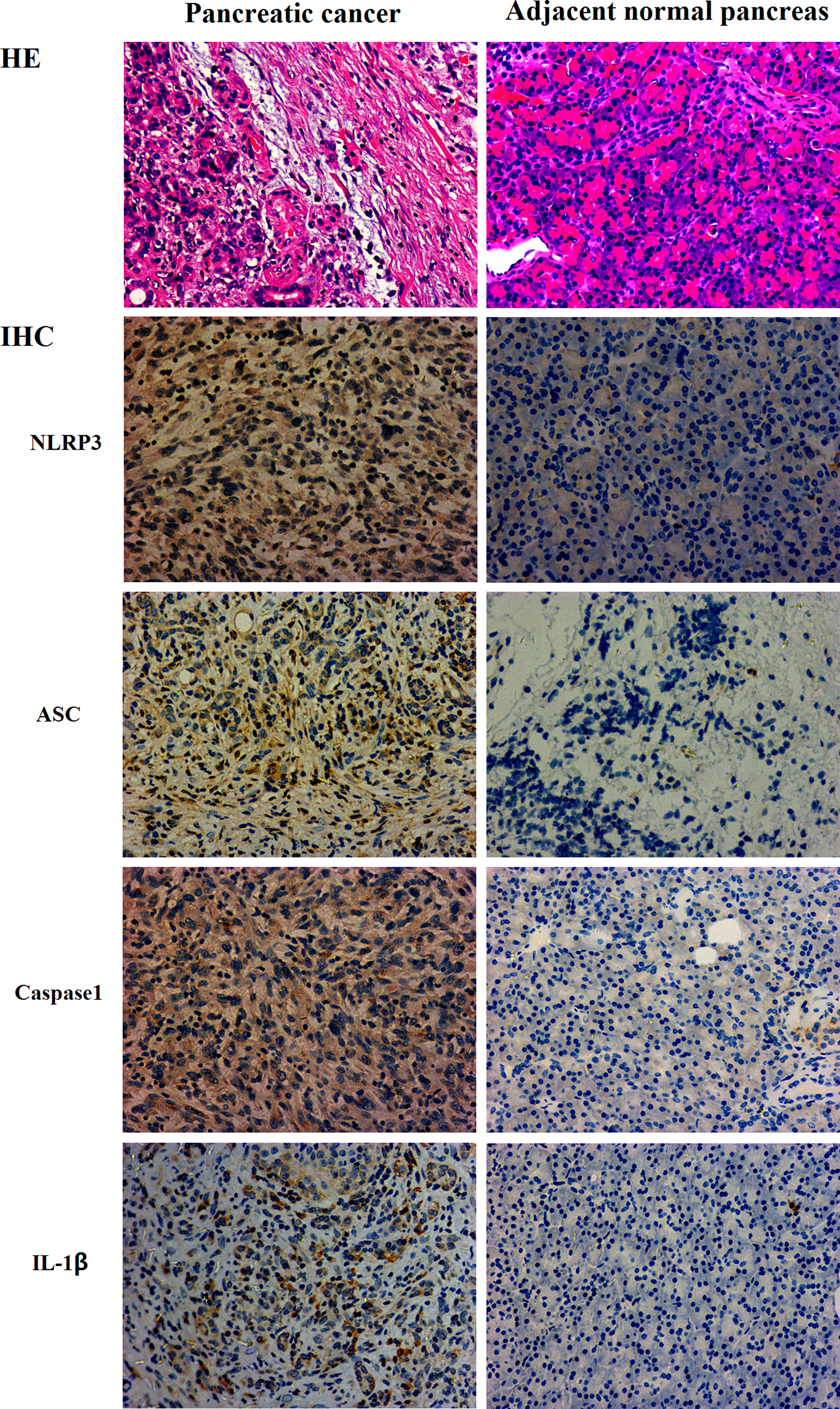
IHC staining and corresponding hematoxylin and eosin (HE) staining of NLRP3 inflammasome expression in pancreatic adenocarcinoma tissues and adjacent normal tissues (200x).
Expression of the NLRP3 inflammasome in pancreatic cancer tissues and adjacent tissues.

IL: interleukin.
Correlation between NLPR3 inflammasome expression and clinicopathological variables
According to the expression intensity of the NLPR3 inflammasome, all 98 cases were divided into high versus low groups for comparison. The high expressions of NLRP3 were significantly associated with a higher rate of positive lymph nodes (P <0.01) and a higher rate of TNM stage II tumor (P = 0.01). Patients with a high expression of ASC seemed to also have a higher rate of positive lymph nodes (P = 0.01) and worse clinical stage (P < 0.01). The same associations between high expression of Caspase-1 and positive lymph nodes and worse clinical stage were observed (both P < 0.05), respectively, as well as IL-1β (both P < 0.05), as shown in Table 3.
Correlations between the NLRP3 inflammasome expression and the clinicopathologic characteristics of patients with pancreatic adenocarcinoma.
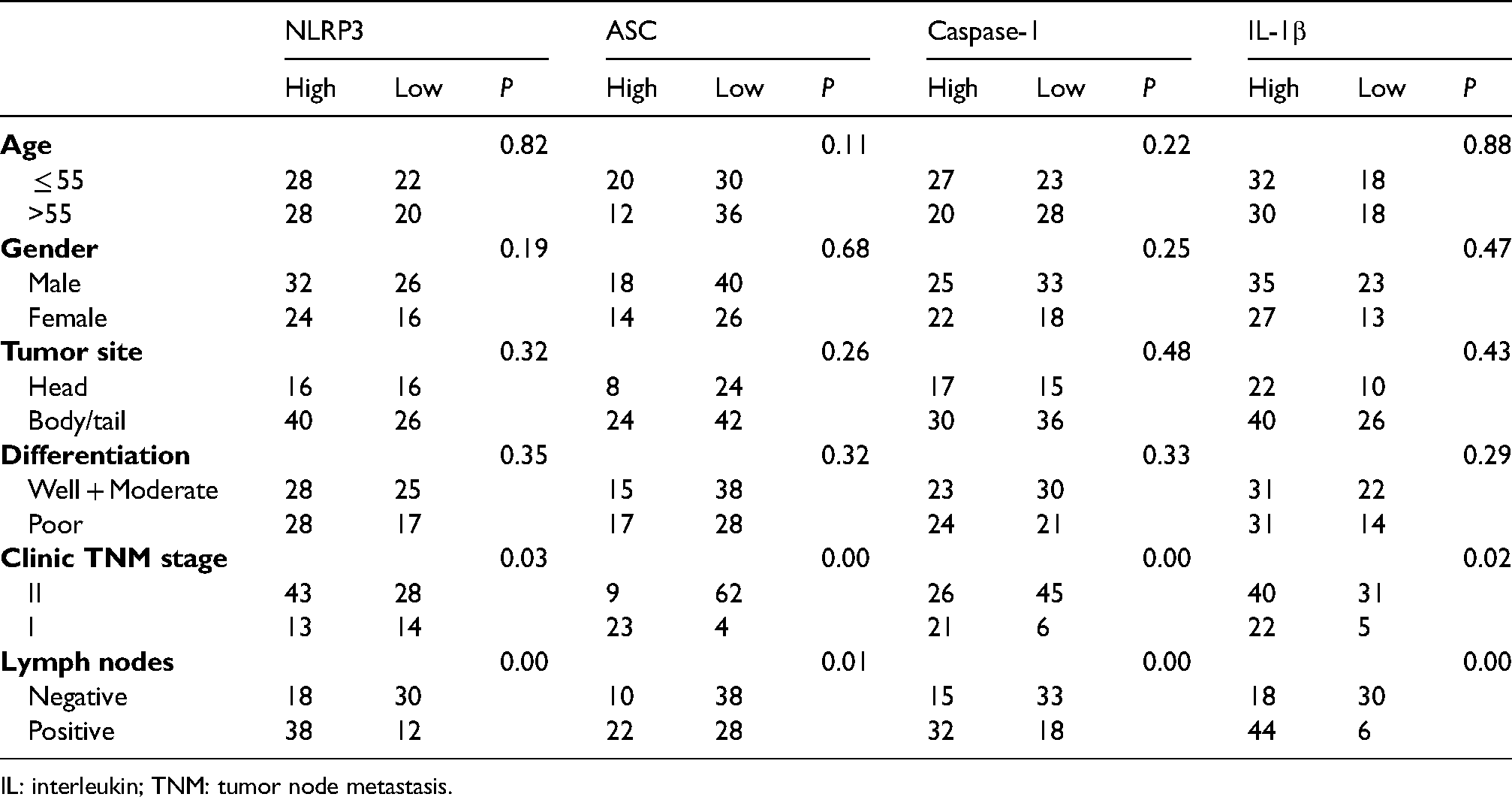
IL: interleukin; TNM: tumor node metastasis.
Prognostic value of NLPR3 inflammasome expression for pancreatic adenocarcinoma
Univariate survival analysis showed that the expressions of NLRP3, ASC, Caspase-1, and IL-1β were correlated with the prognosis of pancreatic cancer. Among them, the median survival time of patients with a high NLRP3 expression was significantly lower than in those with a low expression (12 months vs. 21 months, P < 0.01, Figure 2(a)). The median survival time of patients with a high expression of ASC was also significantly lower than in those with a low expression (13 months vs. 18 months, P < 0.01, Figure 2(b)). There was a significant association between the median survival time of patients and expression of Caspase-1 (13 months vs. 17 months, P < 0.01, Figure 2(c)). The prognosis of patients with a low IL-1β expression was better than those with a higher IL-1β expression (18 months vs. 13 months, P < 0.01, Figure 2(d)).
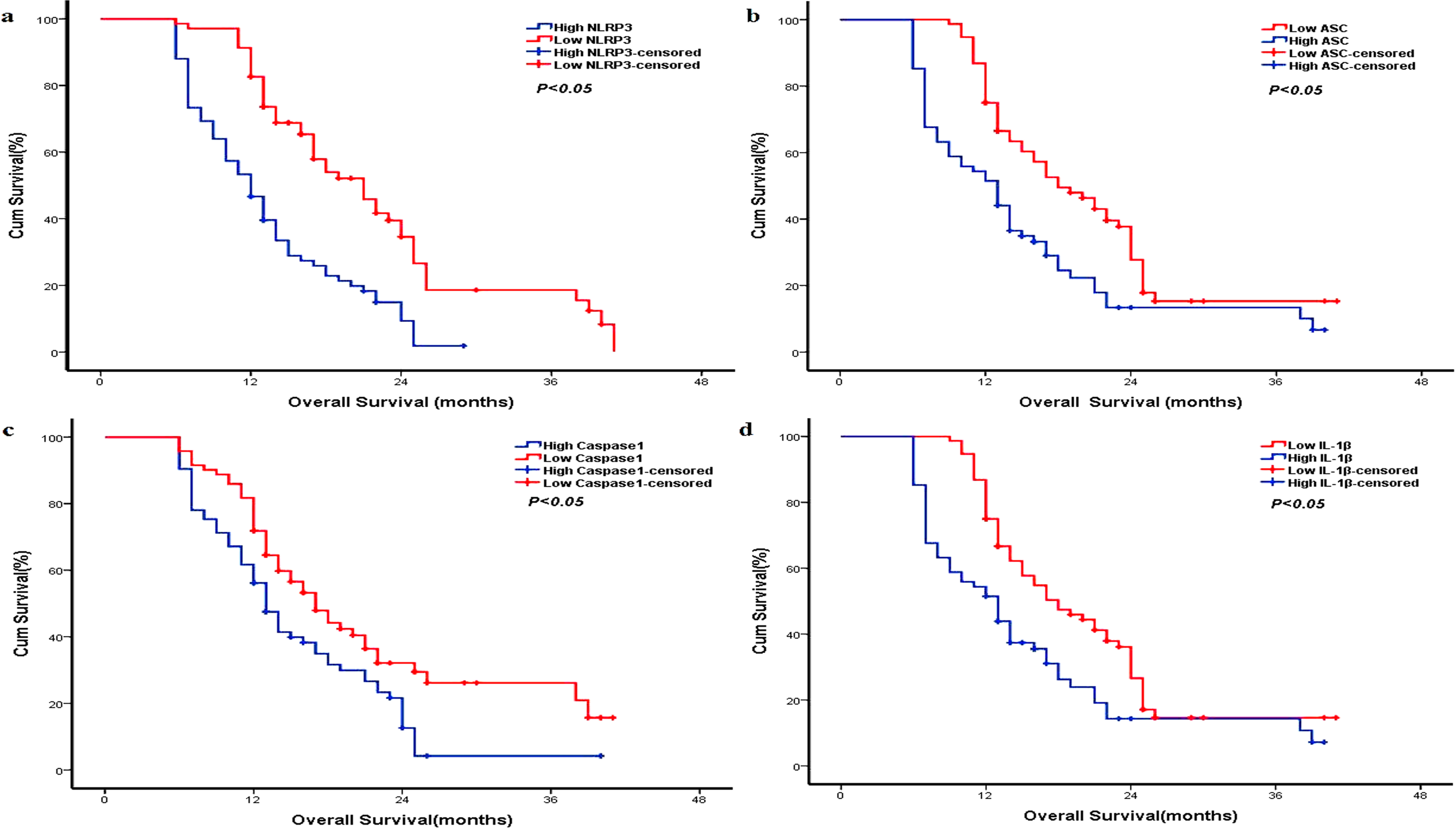
Prognostic significance of NLRP3 inflammasome for pancreatic adenocarcinoma. Median overall survival: (a) NLPR3 (12 months vs. 21 months, P < 0.01); (b) ASC (13 months vs. 18 months, P < 0.01); (c) Caspase-1 (13 months vs. 17 months, P < 0.01); and (d) IL-1β (18 months vs. 13 months, P < 0.01).
Discussion
The occurrence and development of pancreatic adenocarcinoma are potentially associated with inflammation. 15 Epidemiological studies have shown that the risk of pancreatic cancer in patients with chronic pancreatitis increased by 10–20 times. 16 Following the inflammatory response initiation, various types of inflammatory cells (including macrophages) release cytokines, which combine with macrophages to form endogenous signals to produce a microenvironment that is conducive to tumor occurrence and development, and inhibit anti-tumor immunity.17,18 Among these cytokines, IL-18 and IL-1β have been shown to be closely related to pancreatic adenocarcinoma. In detail, IL-18 can enhance Th1-type immune response, thereby producing large amounts of INF-γ and TNF-α, plus promoting chronic inflammation and forming the basis of tumorigenesis.19,20 Carbone et al. 21 conducted a study that enrolled 58 patients with pancreatic cancer, and found that expression of IL-18 in both tumor tissues and in serum was upregulated, and elevated serum IL-18 was associated with poor prognosis. IL-1β can promote the invasion ability of pancreatic cancer cells. 22 The secretion of IL-18 and IL-1β mainly depends on the activation of the NLRP3 inflammasome. Therefore, the NLRP3 inflammasome is more likely to participate in the development of pancreatic cancer.
In this study, we showed that the expression level of NLRP3 inflammasome components in pancreatic cancer tissues was higher than those in adjacent normal pancreatic tissue by IHC analysis, suggesting that the activation products of inflammasomes may be involved in the inflammatory background associated with pancreatic cancer. In addition, we found that the expression level of NLRP3 inflammasome components were related to the clinical stage of pancreatic cancer. In detail, patients with TNM stage II cancer had a higher expression level of NLRP3 inflammasome components than those with TNM stage I cancer. The clinical staging of tumors is a well-known factor associated with the malignancy of tumors and tumor burden. Moreover, patients with advanced tumors often have obvious inflammatory reactions. 23 Tumor burden stimulates the activation of the NLRP3 inflammasome and produces a large number of cytokines, which further promotes tumor development. This study also found that the expression of the NLRP3 inflammasome is related to lymph node invasion. For patients with positive lymph nodes, the anti-tumor immunity from lymph nodes was inhibited. 24 A high tumor burden stimulates the activation of the NLRP3 inflammasome, which in turn stimulates the inflammatory response. 25
The NLRP3 inflammasome is a multi-protein complex mainly expressed in the cytoplasm, which also functions as the activation platform for Caspase-1 to mediate the inflammatory response, and is an important factor for activating the secretion of IL-18 and IL-1β. 26 The inflammasome recruits Caspase-1 precursors and activates Caspase-1 precursors to Caspase-1 through autologous cleavage. The activated Caspase-1 further cleaves IL-18 and IL-1β precursors, thereby promoting secretion of biologically active IL-18 and IL-1β. The activation of inflammasomes is related to many diseases.14,27 At present, there are few relevant studies concerning the expression of inflammasomes in tumor tissues, and the conclusions are controversial. Kong et al. 28 showed that a highly expressed NLRP3 inflammasome was observed in human lung cancer cell lines and tissues. Wei et al. 29 found that the expression of the NLRP3 inflammasome in hepatocellular carcinoma tissues was down-regulated, and was related to clinical staging and pathological differentiation. In this study, we firstly showed that the expression level of NLRP3 inflammasome components in pancreatic cancer tissues was higher than that in adjacent normal pancreatic tissue, which was also associated with lymph node invasion and clinical stage.
Furthermore, in this study, survival analysis showed that a high expression of the NLRP3 inflammasome was associated with a poor prognosis of pancreatic cancer, which further suggested that the dysregulation of the NLRP3 inflammasome expression may be a risk factor for the progression of pancreatic cancer. Xue et al. 30 also showed a statistically significant correlation between the 5-year OS and disease-free survival after surgery and NLRP3 inflammasome expression levels in laryngeal squamous cell carcinoma patients. Feng et al. 31 showed that a high NLRP3 expression was related to poor clinical outcome in oral squamous cell carcinoma patients that received 5-FU treatment. However, the clear underlying mechanism for the association between cancer prognosis and NLRP3 inflammasome expression is still not fully confirmed. As one of the seven hallmarks of cancer, activation of inflammasomes may result in the inhibition of the natural killer and T-cell-induced anti-tumor immunity. 32 Moreover, the overexpression of IL-18 that resulted from activated NLRP3 inflammasome expression was also associated with the upregulation of myeloid-derived suppressor cells. 33 The NLRP3 inflammasome promotes lymph angiogenesis and metastasis by S1PR1 signaling. 34 However, the definite association between cancer prognosis and NLRP3 inflammasome expression still requires abundant prospective clinical and experimental data.
There are several limitations that may have influenced the generalization of the results of this study. We enrolled a limited number of clinical cases and ended up with a relatively small sample size. We did not obtain the recurrent status of all patients, and cannot analyze the recurrence-free survival, but only OS. Furthermore, this study was conducted in a third-grade class-A hospital, therefore, the results generally cannot be suitable in other lower-level hospitals.
In summary, in this study, we showed that expression of the NLRP3 inflammasome is upregulated in pancreatic cancer and is related to clinical staging and lymph node invasion, and high expression of the NLRP3 inflammasome is associated with poor prognosis, which further confirms that the NLRP3 inflammasome promotes tumorigenesis, and can serve as a risk factor for pancreatic cancer. Also, it could be considered as a therapy target.
Availability of data and materials
All data analyzed and generated during the current study are available from the corresponding author upon reasonable request
Footnotes
Authors’ contributions
(a) Conception and design: Hailiang Liu. (b) Administrative support: Hailiang Liu. (c) Provision of study materials or patients: Hailiang Liu. (d) Collection and assembly of data: Lina Zheng. (e) Data analysis and interpretation: Hailiang Liu and Lina Zheng. (f) Manuscript writing: All authors. (g) Final approval of manuscript: All authors.
Competing interests
The authors declare no conflict of interest with respect to this article.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of the Chinese PLA General Hospital. Written informed consent was obtained from individual patients.
