Abstract
Mechanical forces related to blood pressure and flow patterns play a crucial role in vascular homeostasis. Perturbations in vascular stresses and strain resulting from changes in hemodynamic may occur in pathological conditions, leading to vascular dysfunction as well as in vascular prosthesis, arteriovenous shunt for hemodialysis and in mechanical circulation support. Turbulent-like blood flows can induce high-frequency vibrations of the vessel wall, and this stimulus has recently gained attention as potential contributors to vascular pathologies, such as development of intimal hyperplasia in arteriovenous fistula for hemodialysis. However, the biological response of vascular cells to this stimulus remains incompletely understood. This review provides an analysis of the existing literature concerning the impact of high-frequency stimuli on vascular cell morphology, function, and gene expression. Morphological and functional investigations reveal that vascular cells stimulated at frequencies higher than the normal heart rate exhibit alterations in cell shape, alignment, and proliferation, potentially leading to vessel remodeling. Furthermore, vibrations modulate endothelial and smooth muscle cells gene expression, affecting pathways related to inflammation, oxidative stress, and muscle hypertrophy. Understanding the effects of high-frequency vibrations on vascular cells is essential for unraveling the mechanisms underlying vascular diseases and identifying potential therapeutic targets. Nevertheless, there are still gaps in our understanding of the molecular pathways governing these cellular responses. Further research is necessary to elucidate these mechanisms and their therapeutic implications for vascular diseases.
Introduction
Non-physiological stress and strain on vascular wall
Blood vessels are constantly exposed to two hemodynamic stimuli due to the pulsatile nature of blood pressure and flow: the wall shear stress (WSS), which is the frictional force per unit area exerted by the blood flow parallel to the vessel wall, and the intraluminal pressure, which stretches the vessel wall perpendicularly, promoting circumferential strain.1,2 Vascular cells sense and respond to these mechanical stimuli to maintain the integrity of the vasculature itself and to enable appropriate adaptations. 3 Despite vascular endothelial cells (ECs) and vascular smooth muscle cells (SMCs) are exposed to both types of mechanical cues, the shear stress resulting from blood flow is acting primarily on ECs of the intima, whereas SMCs of the media are primarily subjected and sense changes in intramural stress.1,3
In this context, several molecular pathways are known to be involved in the onset of cellular responses to the above-mentioned mechanical forces, 1 which result in the regulation of intracellular signaling, gene expression, and protein expression that maintain the physiological function of blood vessels. In physiological conditions, the biological response varies with changes in mechanical and chemical stimuli, maintaining a dynamic balance which allows to preserve vascular homeostasis. 4 However, when this balance is disturbed by non-physiological mechanical stimuli, cell signaling can lead to a responsive adaptation or damaging of the vessel wall, possibly contributing to diseases of the vasculature. In this regards, previous research has demonstrated that ECs exposed to unidirectional and elevated WSS exhibit a quiescent phenotype5,6 while disturbed flow characterized by low and oscillatory WSS, typically found in bifurcation or branching regions, has been suggested to trigger a proliferative, pro-inflammatory, pro-oxidant ECs state and an impaired vascular tone regulation. 5 In literature, it is also reported that SMCs manifest a diverse array of phenotypes, based on the type of artery and the presence of disease. Additionally, different loads can promote various cytoplasmic signals and gene expression patterns that modulate both SMCs’ structure and phenotype. 3 Typically, cyclic strains or stresses at levels that replicate physiological values result in maintaining physiological SMCs phenotypes. On the contrary, exposure to cyclic strains or stresses exceeding normal thresholds often triggers inflammatory pathways in SMCs.7,8
Perturbations in vascular stresses and strains caused by changes in blood pressure and hemodynamics may occur either under physiological conditions, such as during physical exercise, or pathological conditions related to vascular disease. 9 Changes in physiological vascular stresses develop also in arteriovenous shunt for vascular access in hemodialysis patients, as well as in artificial vascular graft and in some conditions also with mechanical circulatory support. Thus, over the past decades it has become evident that understanding how the local hemodynamic conditions affect the mechanotransduction of vascular cells is crucial for elucidating the underlying mechanisms of vascular homeostasis and disease.
Turbulent-like flow-induced vascular wall vibrations revealed through computational studies
Disturbed blood flow and “turbulence” in the vascular system is normally linked to pathological conditions. These flows feature rapid and seemingly random velocity and pressure fluctuations in time and space, with continuous energy transfer across scales. Such flows may share certain features with turbulence, but do not necessarily exhibit all of the well-known characteristics outlined by Tennekes and Lumley in 1972 or follow mathematical theories of high Reynolds number homogeneous and isotropic turbulence itself. 10
Furthermore, the presence of flow instabilities leading to vascular pathologies and adverse vascular remodeling is well-documented in various vascular districts. For instance, adopting medical image-based computational fluid dynamics (CFD) from magnetic resonance imaging (MRI) or computed tomography (CT) scans, with boundary conditions obtained from literature or patient-specific Doppler ultrasound, turbulent-like flow phenotypes have been documented in cerebral aneurysms, 11 as well as in carotid syphons 12 where atherosclerotic plaques commonly occur. Additionally, transitional flow has been found in the venous segment of hemodialysis arteriovenous fistula (AVF),13,14 a vascular region usually associated to neointimal hyperplasia, 15 which is a fibrotic-muscular thickening of the vessel wall caused by the migration of smooth muscle cells into the intimal layer of the vessel.16,17
Such turbulent-like flow can induce high-frequency vibrations in the vascular wall. Recent studies from our group 18 showed, through a fluid-structure interaction (FSI) approach, that transitional flow-induced high frequency pressure fluctuations caused the walls in the AVF vein to vibrate at frequencies up to hundreds of Hz. Moreover, these vibration amplitudes were found to be predominant at the inner curvature of the cephalic vein, the region where stenotic lesions typically develop. Furthermore, recent FSI studies have uncovered the presence of vascular wall vibrations also within cerebral aneurysms,10,19,20 unraveling broad-band, random vibrations which show similar characteristics with clinically observed bruits.21,22
All these evidence from “in silico” studies suggest that high-frequency vibrations may play a role in vascular mechanobiology across different vascular regions, highlighting the need for further exploration into this phenomenon.
Vascular wall vibrations in the onset of cardiovascular disease
Investigating the impact of high-frequency vibrations on the cardiovascular system has roots dating back to the latter half of the 20th century. During this period, experimental setups and animal clinical models were developed to explore thrills, bruits, and high-frequency vibrations, suggesting a potential link to the onset of vascular diseases. Already in 1963, Roach. 23 observed that the presence of a recordable bruits was a common feature in various diseases characterized by vascular stenosis. Wang et al. 24 found that patients with occlusive coronary arteries, where turbulent flow fluctuations were present, exhibited a typical increase in high frequencies that matched with acoustic signals recorded by a chest microphone. In the same period, other experiments25 –27 documented vascular wall vibrations that might contribute to the pathogenesis of structural fatigue affecting the vasculature subjected to non-physiological stimulus. Then, the relationship between bruits and pressure fluctuations in the post-stenotic region was numerically investigated in idealized vessels.28,29 Also, recent studies on carotid artery bifurcation 30 and cerebral aneurysm 31 reported turbulent flow-induced high-frequency vibrations. Therefore, based on the reported rationale, understanding how the frequency of blood flow- and pressure-induced vessel wall displacement impacts on vascular function is essential for elucidating the underlying mechanisms of vascular diseases. Thus, the aim of this review is to explore the existing literature regarding the effect of a high-frequency vibration on the behavior of vascular cells in terms of morphological, functional, and mechanobiological changes. We define as high-frequency vibrations, frequencies exceeding that induced by the normal heart rate, corresponding approximately to 1–2 Hz in humans and to 6 and 9 Hz in rats and mice, respectively (Figure 1). 32
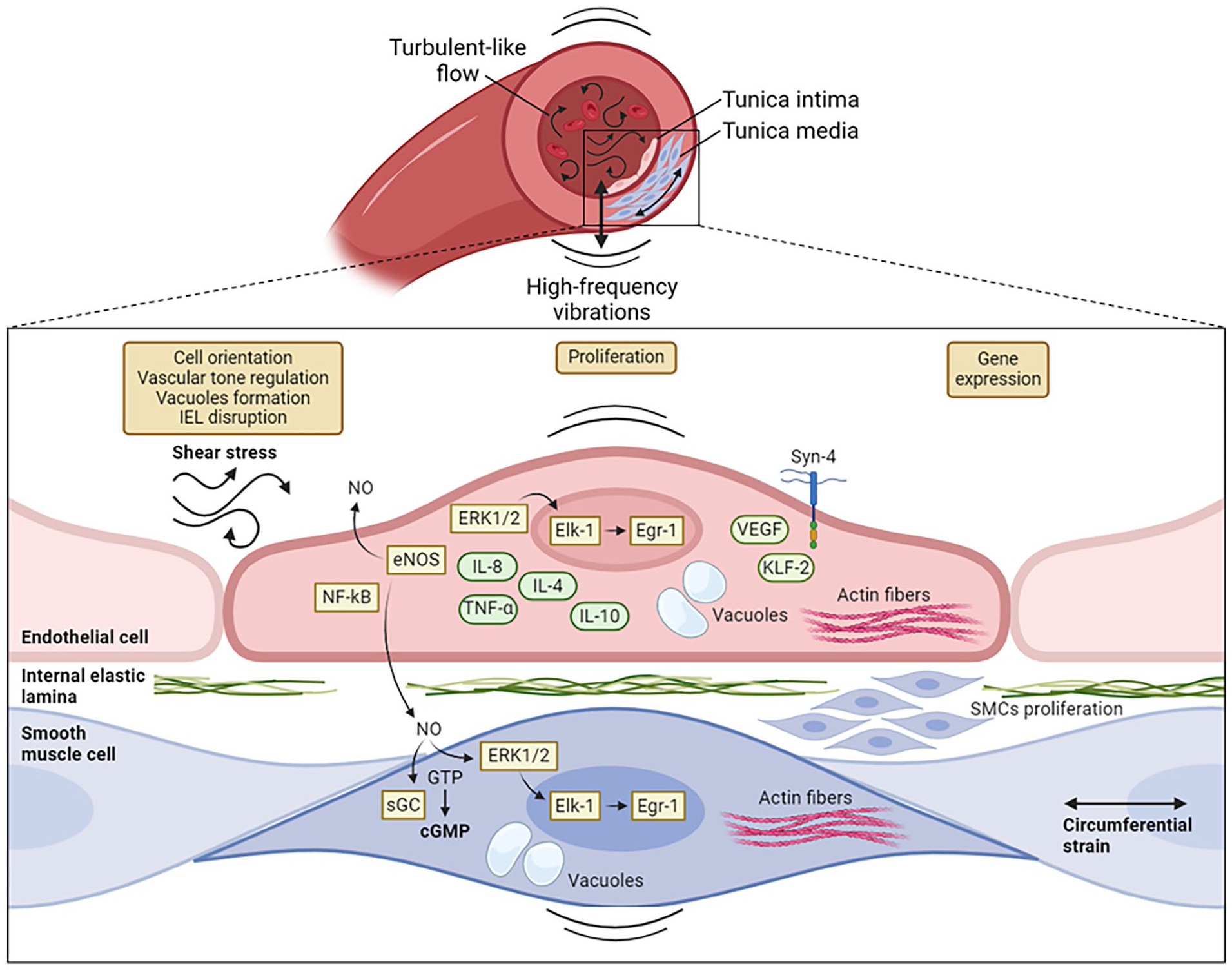
Biological response of vascular cells to flow-induced high-frequency vibrations. The schematic diagram reports some of the morphological, functional, and gene expression changes observed in endothelial (ECs) and smooth muscle cells (SMCs) exposed to stimuli at high-frequencies. Morphological changes include actin fibers alignment and cell orientation, vascular tone regulation, formation of double-membrane limited vacuoles, and disruption of the internal elastic lamina (IEL). Vibrations induce the proliferation of smooth muscle cells leading to a thickening of the intimal layer and promote in ECs and SMCs the expression of factors and the activation of pathways related to inflammation, oxidative stress and vascular dysfunction, including extracellular signal-regulated kinase 1 and 2 (ERK1/2), endothelial nitric-oxide synthase (eNOS), nuclear factor-kB (NF-kB), soluble guanylate cyclase (sGC), Krüppel-like factor 2 (KLF-2), vascular endothelial growth factor (VEGF), tumour necrosis factor alpha (TNF-α), interleukin-8 (IL-8), interleukin-4 (IL-4), and interleukin-10 (IL-10). Early growth response 1 (Egr-1). This figure was created using Biorender.com
Impact of high-frequency stimulation on vascular cells morphology, functions, and mechanobiology
Cellular mechanotransduction is an important biological process in living organisms, deeply investigated to assess the impact of biological mechanical cues such as the circumferential stretching force, the fluid shear stress and the extracellular matrix (ECM) stiffness on multiple pathophysiological processes. Signals are sensed by the ion channels and receptors on the cell membrane and affect cell-matrix communications and signal transduction, triggering changes in cytoskeleton structure and downstream signaling cascades which regulate several processes including cell differentiation, adhesion, migration, proliferation, secretion of factors, and ECM generation. 33
Many signaling pathways have been identified as involved in cell responding to mechanical stimuli. A large body of evidence is reported regarding the role of vascular endothelial growth factor (VEGF), Notch, platelet-derived growth factor (PDGF), Krüppel-like factor 2 (Klf2), endothelial nitric-oxide synthase (eNOS), endothelin, Rho family signaling molecules, mitogen-activated protein kinase (MAPK) signaling pathway, nuclear factor-kB (NF-κB) signaling pathway, and GTPase signaling pathway.34,35 Great attention was given to mechanosensors including integrins, the glycocalyx, primary cilia, G protein-coupled receptors, and ion channels36 –43 such as Piezo1. 44 In response that is, to fluid shear stress, several mechanosensory complexes are also activated, including vascular endothelial cell cadherin (VE-cadherin), VEGF receptor 2 (VEGFR2), and platelet endothelial cell adhesion molecule (PECAM-1). 41 Furthermore, activation of extracellular signal-regulated kinase 1 and 2 (ERK1/2) cascades is reported. 45
With regard to cellular responses to cues, several investigations have explored the connection between mechanical signal transduction and diseases. That being said, the study of vibrations of the blood vessel wall promoted by pressure fluctuations recently acquired interest, since a correlation with some vascular pathologies has been observed.19,20,23,31 Based on these studies, high-frequency vibrations appear to impact vascular cell functions, suggesting the role of a mechanobiological process that links high-frequency stresses within the vessel wall to adverse vascular remodeling. In the next paragraphs, we report the available evidence in the literature concerning the effect of high-frequency stimuli on the biology of vascular cells.
Morphological changes in vascular cells stimulated by high-frequency stimuli
Over the years, the potential of high-frequency stimuli in inducing cell morphological changes has been studied by exposing cultured cells or vessel segments to mechanical loadings such as vibration or cyclic stretching. It is reasonable that cells sense a cyclic circumferential strain when they are subjected to a vertical vibration, since their body is stretched perpendicularly to the axis of their displacement. That being said, this stress might play a role in the effect of vibrations on vascular cells.
Several in vivo studies investigated the impact of this stimulus on vascular histology. Krajnak et al. 46 explored the physiological effects of vibrations on the ventral tail artery of rats exposed to a 125 Hz-vibration. Their study revealed no apparent signs of trauma to the vascular smooth muscle or endothelial cell layer in arteries collected from both stimulated and unstimulated animals. However, arteries from vibrated rat tails exhibited larger involutions in the internal elastic lamina (IEL), indicative of vessel constriction, and compression of endothelial cells.
Furthermore, in 2002, Curry investigated the mechanism of vibration injury by exposing rat tails to a 60 Hz vibration. 47 Electron microscopy revealed numerous arterial regions with loss and thinning of endothelial cells, along with activated platelets coating the exposed subendothelial tissue. Both ECs and SMCs contained double membrane-limited, swollen vacuoles similar to those formed under massive vasoconstriction induced by direct norepinephrine application. 48 This similarity suggests the vasoconstrictive potential of vibrations. Additionally, the study identified an increase in chondroitin sulfate proteoglycan, which normally precedes smooth muscle migration, 49 in the extracellular matrix between SMCs. Since the denudation of the endothelial barrier triggers platelet adherence and their release of factors leading to smooth muscle proliferation and stenosis, together with factors that degrade the internal elastic lamina, Curry hypothesized that a similar scenario may occur when vibration disrupts the endothelium of the tail artery, resulting in degradation of the IEL. If this hypothesis is correct, prolonged vibrations should result in smooth muscle overgrowth and eventually to vascular occlusion.
Much evidence shows that mechanical stresses can also modulate cell orientation and actin fibers alignment. For instance, cyclic strain at low frequencies has been observed to induce vascular cells to align most frequently approximately 90° away from the direction of stretch.50 –53
In this regard, the orientation and structural network of the vascular SMCs layers that build up the arterial wall are very important for maintaining its mechanical strength and function and also for providing the mechanical compliance required for pulsatile blood flow.54 –56 The stress loading induces vascular smooth muscle cells to align in a frequency-dependent way, 50 and their response to cyclic mechanical loading appears to accelerate with higher frequencies. 51 However, this phenomenon was assessed only for frequencies lower than 2 Hz, indicating a need for further data on the outcomes of higher-frequency stimulation of these cells. Furthermore, ECs exhibit a greater degree of alignment in response to 1 Hz stretching compared to 0.1 Hz, resulting in a more attenuated effect, and to 0.01 Hz, which causes a complete lack of stress fiber orientation. Again, despite interesting studies on the response of these cells to low frequencies, information about the impact of high-frequencies is not present in the literature. 52 Human fibroblasts subjected to cyclic stress elongate along the direction perpendicular to the mechanical loading, and the timing of the cytoskeleton orientation is influenced by cell density and stimulus frequency. For subconfluent cells, the time needed for the alignment decreases as the frequency of stretch loading raise from 0.1 Hz to 1 Hz. However, no further acceleration in alignment is observed when the stimulus is increased up to 20 Hz. Conversely, confluent cells exhibit a shorter reorientation time, which tends to decrease with increasing frequency of the applied stimulus. These findings suggest that cell-cell contacts mediated by cadherins may play a significant role in the cellular response to mechanical strain. 53
Additionally, in a recent study by Mu et al., the impact of a stimulation with vibrations at a frequency of 31.5 Hz on the morphology of cultured ECs was assessed. They observed that in the absence of vibrations cells exhibited a flat, polygonal morphology, tightly packed in a paving stone pattern. However, this morphology was notably altered after mechanical stimulation, with cells acquiring a rounded morphology and a decreased distribution density. 57
Overall, these studies highlight those different frequencies of mechanical stimuli may induce diverse cellular responses in different cell types. Vascular cells adjust their shape and orientation to endure the cues they experience, leading to the remodeling of the vessel wall which aims at the maintenance of the structure, function, and mechanical integrity of the vessel. However, despite the large number of studies, primarily investigating histological modifications of the vessel wall, the molecular mechanism by which vibration leads to these changes remains unknown. Moreover, consistent observation of the effect of vibrations within the range of 50–150 Hz is not yet present in the literature.
Functional changes in vascular cells stimulated by high-frequency stimuli
Mechanical signals exert significant influence over numerous essential cellular functions, impacting processes such as cell proliferation, differentiation, and migration, and consequently governing critical phenomena including bone and cartilage growth, wound healing, and angiogenesis. 58 However, many aspects of the biochemical transduction of some mechanical stresses like vibrations are not known in detail, yet a profound understanding of the way cells respond to them is important for basic biological science, in the vascular field in particular.
It has been reported that a normal response of biological tissues to repeated stress is an increase in proliferation. 59 Therefore, it is reasonable to hypothesize that exposure of vascular cells to high frequency stimulation may induce changes in cell growth and vessel remodeling. However, there is limited research available on this topic.
In vivo, prolonged exposure to vibrations triggers vascular impairment. Histological changes observed in the peripheral arteries of workers subjected to the frequent use of vibrating tools, who manifested Raynaud’s phenomenon, 60 can be considered among the first evidence of the damaging potential of this mechanical stimulus. Raynaud’s phenomenon is one of the main symptoms of the hand-arm vibration syndrome (HAVS), which is characterized by dysfunction of the peripheral vascular and sensorineural systems, and cause an exaggerated vasoconstriction, especially in response to cold temperature-exposure.46,61
Several studies investigated the role of wall vibrations in inducing vascular remodeling. Okada et al. 62 demonstrated experimentally the occurrence of intimal thickening in peripheral arteries of rats after 90 days of exposure to a local vibration at 60 Hz. The same group, some years later showed that the small arteries of hind legs of rats exposed for 30 days to vibrations at 30 and 480 Hz manifested a thicker intimal layer. The disruption of internal elastic lamina and focal cell proliferation and the formation of collagen and elastic fibres was also observed. 60 Further research 63 revealed an important reduction in the lumen diameter of ventral tail arteries in rats exposed to vibrations at 250 Hz, accompanied by a significant increase in vascular smooth muscle thickness. Conversely, stimulation at frequencies of 62.5 and 125 Hz induced morphological changes that were not statistically significant.
Additionally, Bittle 59 proposed a hypothesis trying to correlate the hyperproliferation of the intimal layer with vascular vibrations. She reported that the contractile myofilaments of vascular smooth muscle cells and their attachment to the plasma membrane as forming a lattice network. This network allows internal cellular structures, such as the nucleus and Golgi complex, to move differently from the surrounding cytoskeletal structure, resulting in abnormal stresses on cell structures. Such abnormal stress, along with localized stretching of the membrane due to relative motion of the myofilaments, may activate cellular signaling pathways leading to increased cell growth or replication. She also assessed the impact of vibrations on cell proliferation by culturing vascular SMCs in growth environments simulating disturbed flow, with high-frequency vibrations and small amplitude motions. Among different combination of the two parameters, she found that only cells exposed to a displacement of 12 µm at 45 Hz exhibited significantly higher growth rates compared to cells cultured in static conditions. This suggests that both the frequency and amplitude of the stimulus may impact on cellular functions. At variance to this investigation, a study conducted in 2012 64 demonstrated that two murine myoblast cell lines, C2C12 and L6C11, exhibited reduced proliferation rates compared to control cells after being exposed to 30 Hz-frequency vibrations for one week. The cell index, a measure of cell quantity in a culture plate well, was significantly lower in treated wells. They also concluded that the decreased growth rate was not due to apoptosis activation, since no differences in apoptotic cell percentages were observed. Therefore, they suggested that mechanical vibration might induce micro cytoskeletal alterations, leading to a reduction in cycling capabilities of cells. This discrepancy among the studies available in the literature need further investigation to understand more in detail the effect of high-frequency vibrations in this cell phenotype.
High-frequency mechanical stimuli modulate gene expression in vascular cells
Exposure to high-frequency stimuli may modulate the expression level of some genes in vascular cells. It is known that altered blood flow hemodynamics triggers an adaptive response of the vessel wall involving migration and proliferation of vascular SMCs in the subintimal space 65 through the activation of intracellular signaling molecules such as MAPKs.66,67 The MAPK cascade, comprising ERK1 and ERK2, is well known for its role in mediating signal transduction induced by hemodynamic forces and growth factors. 68 This cascade activates the nuclear transcription factor Elk-1, crucial for the transcriptional activation of early growth response 1 (Egr-1), 69 which coordinates the expression of various endothelial and vascular smooth muscle cell proteins involved in the molecular signaling activated by hemodynamic cues, such as PDGF-A, PDGF-B, and transforming growth factor-β (TGF-b).68,70 Based on this rationale, Loth et al. 68 examined the distribution and activity level of ERK1/2 in a porcine arteriovenous graft model. This study showed that venous anastomosis exhibits areas of high-frequency vein-wall vibration (~300 Hz), which correlate with elevated activity levels of ERK1/2 and intimal thickening. In detail, a densitometric analysis revealed that ERK1/2 primarily localized in regions with the highest intensity of vibration within the intimal and medial SMCs. Molecular investigations using MAPK assay and Western Blot assay confirmed their enhanced activity along with increased activation of the downstream effector Elk-1. These findings provide initial evidence of a potential association between vein-wall vibration and the ERK1/2 mechanotransduction pathway. Furthermore, 4 weeks post-anastomosis, there was a modest increase in T lymphocyte and macrophage infiltration across the anastomotic regions, albeit not co-localized with activated ERK1/2 within the intima. 68 This suggests the possible existence of an ERK1/2-independent mechanism responsible for recruiting inflammatory cells to the damaged vessel site, although this aspect was not investigated in this study. Overall, these findings suggest that mechanical variables, such as turbulence-induced vein-wall vibrations, may significantly influence MAPK activation in intimal endothelial cells. However, given the proximity of the oscillation region to the flow branching area, it is challenging to distinguish their individual effects. Therefore, further research is necessary to elucidate the precise distribution of ERK1/2 and its role in mechanotransduction.
Another signaling molecule that has a pivotal role in regulating blood flow and oxygenation of tissues is nitric oxide (NO). Nitric oxide is produced and released into the blood by the endothelial cells that line the blood vessels and lymphatic vessels. 71 The effect of high-frequencies stimuli on NO release by ECs was already investigated in the early 1990s by Hutchenson, 72 who found that the molecule is produced as a function of pulsatility with an optimal frequency of pulsation of 2–8 Hz (120–480 cpm). The baseline pulsations in the human circulation are in the frequency range of 1–2 Hz and additional pulsations beyond these increase NO bioavailability via eNOS.72 –74 Vibration stimulates ECs to produce and release NO and crucial for its generation is the endothelial nitric oxide synthase. Despite the precise mechanisms by which vibrations influence endothelial mechanosensors to modulate eNOS activity are not fully understood, it seems that the endothelial cells mechanosensor-proteins Syndecan-4 (Syn4), VEGF, and KLF2 translate the physical force from the vibration into biochemical signals. Studies have demonstrated that vibroacoustic stimulation at 100 Hz induces Syn4 and VEGF expression.75,76 NO, in response, regulates blood flow and vascular tone by affecting vascular smooth muscle cells, activating guanylate cyclase (sGC), 77 and regulates the phosphorylation of ERK1/2. 78 Significant NO-release increment has been observed with different types of vibrations, including whole-body periodic acceleration at 6 Hz, 79 arm-applied vibration at 50 Hz, 80 chest-applied sonic vibration at 100 Hz, 81 and low amplitude-vibrations at various body surfaces ranging from 150 to 250 Hz. 82 Furthermore, nuclei of endothelial, smooth muscle, and adventitial cells from rat ventral tail arteries exposed for 4 h to a 60 Hz-vibration, showed an increased immunostaining of the Ca+2 activated nuclear factor of activated T-cells cytoplasmic 3 (NFATc3), induced by the upregulation and nuclear translocation of the factor. 47 Since NFAT is known to regulate cardiac hypertrophy, 83 it is plausible to consider its involvement in vibration-induced endothelial cell injury. Although the mechanism of action and protein expression influenced by NFAT were not characterized, evidence concerning this factor could be relevant for future research aimed at investigating the cellular and molecular mechanisms involved in the early stages of vibration injury.
Recent research suggests that high-frequency mechanical vibration can promote inflammation and oxidative stress. Indeed, the stimulation of cultured cells increases the expression of the inflammatory factor NF-κB, 57 and the inflammatory cytokines tumour necrosis factor alpha (TNF-α), interleukin-8 (IL-8), interleukin-4 (IL-4), and interleukin-10 (IL-10), 84 whereas the ventral tail artery of rats exposed to vibrations show a higher expression of interleukin-6 (IL-6), interleukin-1β (IL-1β), and metallothionein 1a (MT-1a). 63
In addition, vibrations at 30 Hz have an effect on the muscle atrophy pathway in myoblasts, which exhibit a reduced expression of myostatin, a key gene involved in muscular tissue loss.64,85 –89 In vivo, a decreased expression of both myostatin and atrogin-1, the other master gene of the atrophy pathway, was observed after 30 Hz-vibration exposure, and a significant phosphorylation of protein kinase B (AKT) was found. 64 Activation of the Akt/mTOR pathway has been shown as necessary and sufficient to induce hypertrophy and to block skeletal muscle atrophy.85,89,90 Therefore, high-frequency vibrations appear to induce hypertrophy in skeletal muscle. 64 The same study revealed an enhanced expression of M-cadherin, 64 which has been shown to be crucial in regulating myoblast alignment and fusion. 91
Furthermore, in 2018, Krajnak et al. 92 found a rise in the transcript number for the genes metallothionen 1a (mt1a), intracellular adhesion molecule-1 (i-cam1), and myeloid leukemia-1 protein (runx) by analysing tissue samples from the ventral tail artery of rats subjected for several days to a 250 Hz-vibration.
A more recent work 84 investigated the effects of vibration on the expression of long non-coding RNA (IncRNA) maternally expressed gene 3 (MEG3) by vascular endothelial cells in vitro. lncRNA MEG3 is involved in the physiological and pathological processes of various vascular diseases and could be considered an effective indicator of vascular structural and functional changes. A vibration-induced decrease in lncRNA MEG3 expression was observed after 1-day stimulation with three different frequencies (63, 200, and 250 Hz), while in the 2-day exposure group, this decrease was evident only with the 63 Hz vibration. Based on these findings, different frequencies and durations of the stimulus seem to differently affect cell expression. However, to date, the association between vibration-induced endothelial cell damage and lncRNA MEG3 remains unclear; therefore, further exploration of its involvement in the process is needed (Table 1).
Main effects of high-frequency stimuli on vascular cells.
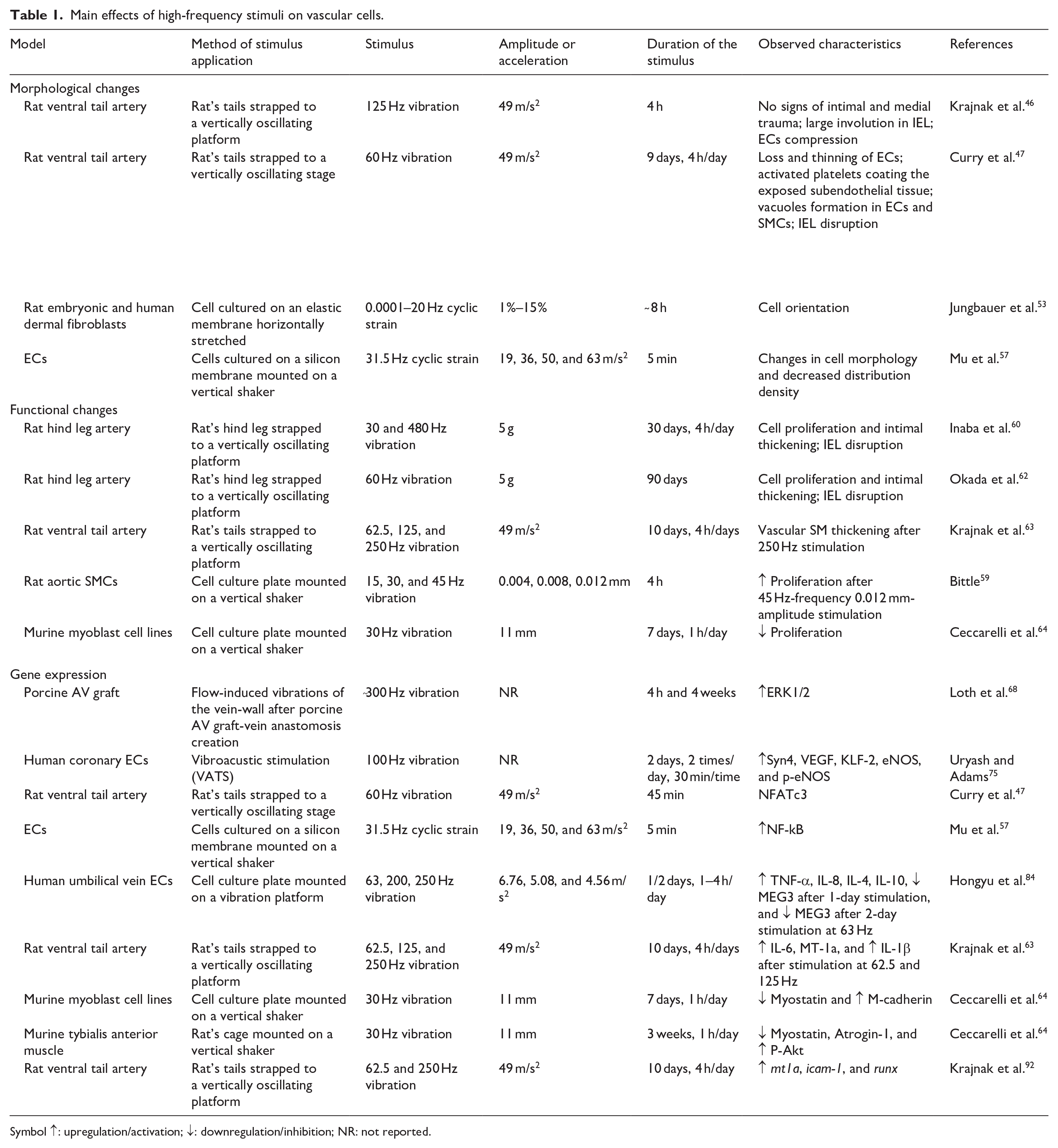
Symbol ↑: upregulation/activation; ↓: downregulation/inhibition; NR: not reported.
Conclusions
In light of all the in vivo and in vitro studies here reported, there is evidence that mechanical vibrations affect cellular phenotypes and gene expression of vascular cells. Specifically, high-frequency vibrations have been shown to induce morphological and functional changes in vascular cells, including alterations in cytoskeletal organization, proliferation, and production of signaling factors. These alterations may contribute to vascular impairment, causing vascular constriction and disruption of the internal elastic lamina, as well as the induction of inflammatory responses, which together might contribute to vascular remodeling and occlusion. Key effectors and signaling pathways, such as ERK1/2, that in SMCs lead to intimal thickening and recruitment of inflammatory cells to damaged vessel sites, Syn4, VEGF, KLF2, ICAM-1, and NFATc3, have been identified in responding to high-frequency vibrations. Furthermore, this mechanical stimulus has been found to influence muscle atrophy pathways, potentially leading to hypertrophic growth of skeletal muscle which could explain stenosis development occurring in some vascular diseases. Whether similar effects develop also in vascular vessel wall subjected to these mechanical stimuli needs further investigation.
It is well-established that disturbances in vascular stresses and strains, whether arising from physiological activities or pathological conditions, can impact the mechanosensors and signaling pathways of endothelial and smooth muscle cells. This review underscores that cellular responses to these cues exhibit variability contingent upon factors like the frequency, duration, and intensity of the stimulus, highlighting the complexity of mechanotransduction processes in vascular cells.
Despite the wealth of evidence suggesting the possible role of high-frequency vibration in vascular damage, several gaps in understanding remain, particularly concerning the molecular mechanisms underlying cellular responses to this stimulus and its implications for vessels disease. Therefore, further research is warranted to elucidate these mechanisms of mechanotransduction and their potential therapeutic implications in vascular pathologies. For instance, studying the effect of in vitro exposure of vascular cells to vibrations at the frequencies observed through computational studies could be useful for the comprehension of the cellular effects triggered by this type of stimulus at molecular and protein level. In addition, also in vivo observation of blood vessels subjected to vibrations could allow the investigation of the contribution of different vascular cells to the biological response. Finally, understanding more in detail the mechanism by which high-frequency vibrations induce vascular changes may open the possibility of developing new pharmacological interventions for vascular diseases.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
