Abstract
To gain insight into how a naturally occurring scaffold composed of extracellular matrix (ECM) proteins provides directional guidance for capillary sprouting, we examined angiogenesis in whole-mount specimens of rat mesentery. Angiogenesis was studied in response to normal maturation, the injection of a mast cell degranulating substance (compound 48/80), and mild wounding. Confocal microscopy of specimens immunolabeled for elastin revealed a network of crosslinked elastic fibers with a density of 140.8 ± 37 mm of fiber/mm2 tissue. Fiber diameters ranged from 180 to 1400 nm, with a mean value of 710 ± 330 nm. Capillary sprouts contained CD31- and OX-43-positive endothelial cells as well as desmin-positive pericytes. During normal maturation, leading endothelial cells and pericytes were in contact and aligned with an elastic fiber in ≃80–90% of all sprouts. In wounding and compound 48/80-treated specimens, in which angiogenesis was markedly increased, leading endothelial cells remained in contact and aligned with elastic fibers in ≃60–80% of all sprouts. These observations indicate that elastic fibers are used for endothelial and pericyte migration during capillary sprouting in rat mesentery. The composition of this elastic fiber matrix may provide important clues for the development of tissue-engineered scaffolds that support and directionally guide angiogenesis.
I
Understanding the structure and composition of an in vivo angiogenesis scaffold may also be beneficial for the field of tissue engineering, in which three-dimensional tissue constructs and implants often fail due to insufficient oxygenation caused by incomplete or absent vascularization. To date, many tissue-engineered constructs have been generated to support neovascularization, either within the construct itself (Richardson et al. 2001; Kidd et al. 2002; Peters et al. 2002) or in the surrounding tissue (Lee et al. 2000; Pinney et al. 2000; Kellar et al. 2001). These include three-dimensional fibroblast cultures (Pinney et al. 2000; Kellar et al. 2001), alginate hydrogels containing angiogenic growth factors (Lee et al. 2000), biodegradable poly (lactideco-glycolide) polymer matrices containing either angiogenic growth factors (Richardson et al. 2001) or endothelial cells (Peters et al. 2002), and implanted porous polytetrafluoroethylene (ePTHE) polymer constructs amended by ECM deposition (Kidd et al. 2002). However, the scaffolds supporting these constructs have not been specifically designed as substrates for directionally guiding the migration of endothelial cells and pericytes. One potential advantage of developing such a scaffold is that the spatial manipulation of angiogenesis could be achieved by altering the orientation and composition of the fibers in the matrix. In part, the present study was undertaken to gain insight into how such a biomimetic scaffold could be designed.
Materials and Methods
Angiogenesis Models
All animal studies were approved by the Animal Research Committee at the University of Virginia and conformed to the American Heart Association Guidelines for the Use of Animals in Research. Angiogenesis was studied in rat mesentery during normal development and in response to both wound healing and the direct application of compound 48/80, a substance that degranulates mast cells. During normal development, microvessel networks develop in previously avascular tissue via capillary sprouting from vessels in the surrounding fat (Hansen-Smith et al. 1994; Ponce and Price 2003; Anderson et al. 2004). In young adult and juvenile animals, microvessel networks are visible at the edges of the mesentery while the center is primarily avascular. Compound 48/80 treated specimens were generated by daily IP injections of the substance dissolved in 0.9% saline (pH 7.2) (Norrby et al. 1986, Ponce and Price 2003; Anderson et al. 2004). Over a 5-day period, the concentration of compound 48/80 was increased daily from 100 μg/ml/100 g bw to 500 μg/ml/100 g bw. Mesenteries were harvested 3 days after the final injection. For the wound-healing stimulus, animals were anesthetized by an IM injection of ketamine (80 mg/kg bw) and xylazine (8 mg/kg bw) and the mesentery was exposed by laparotomy under sterile conditions. Mesenteric tissues were then compressed with a cotton-tipped swab. After this intervention, the abdominal wall and skin were closed with sutures and the animal was allowed to recover. Mesenteric tissues were harvested for immunochemistry and observation 3 days after surgery.
Tissue Harvest and Immunochemistry
Animals were anesthetized as above, sacrificed, and a cannula was placed in the mesenteric artery. Heparinized saline was perfused through the cannula to remove blood from the mesenteric microcirculation. From each animal, several mesenteric windows (wedge-shaped regions of connective tissue bordered by the intestinal wall and the ileal blood vessel pairs) were dissected free, washed in PBS (pH 7.4), dried on gelatin slides, and fixed in 100% MeOH (–20C for 30 min). Slides were then incubated overnight at 4C in 0.1% saponin and 5% normal goat serum in PBS (pH 7.4) containing either polyclonal rabbit anti-elastin (1:100; Chemicon, Temecula, CA) or polyclonal rabbit anti-type I collagen (1:100; Chemicon) and one of three mouse monoclonal antibodies (MAbs). Mouse MAbs were anti-OX-43 antigen (1:100; Serotec, Oxford, UK), biotinylated anti-CD31 (1:200; Serotec and Pharmingen, San Diego, CA), and anti-desmin (1:100; DAKO, Carpinteria, CA). The next day, appropriate secondary antibodies (goat anti-rabbit and anti-mouse IgGs conjugated to either CY2 or CY3 (Jackson Immunoresearch, West Grove, PA) were diluted at a concentration of 1:200 in 0.1% saponin and 5% normal goat serum in PBS (pH 7.4) and applied for 1 hr at room temperature (RT). Biotinylated CD31 was revealed by a 1-hr incubation in 1:1000 CY2- or CY3-conjugated streptavidin (Jackson Immunoresearch) at RT. Because we have shown that the expression of the OX-43 antigen is differentially regulated in some capillary sprouts (Anderson et al. 2004) and OX-43 antigen is also expressed by peritoneal macrophages (Robinson et al. 1986), CD31 labeling was used to confirm and further support the OX-43 results. For double labeling of type I collagen and elastin, specimens were sequentially incubated with polyclonal rabbit anti-elastin (1:100), CY2-conjugated goat anti-rabbit IgG Fab fragments (1:100), polyclonal rabbit antitype I collagen (1:100), and CY3-conjugated goat anti-rabbit IgG (1:200).
Specimen Analysis
Double immunolabeled specimens were examined with a Nikon TE-300 inverted microscope using both oil immersion (×20 and ×60) and air (×10) objectives. For studies documenting the morphology of capillary sprout cells with respect to the underlying elastic fiber matrix, the leading endothelial cell (CD31- or OX-43 positive) or pericyte (desmin-positive) in each clearly visible capillary sprout was observed to determine if it was in contact and aligned with an elastic fiber. The percentage of capillary sprouts with leading endothelial cells or pericytes in this conformation was then recorded. For each cell marker, at least two mesenteric windows were observed per animal. When we analyzed desmin-labeled wound healing and compound 48/80 specimens, we noted that interstitial fibroblasts began to express desmin as they differentiated towards a myofibroblast pheno-type. Because of this, it was often difficult to distinguish between capillary sprout pericytes and interstitial myofibroblasts. Therefore, we restricted the quantitative analysis of pericyte morphology to only the adult and juvenile specimens.
In previous studies, we noted that capillary sprout endothelial cells from highly angiogenic tissues exhibit pheno-typic differences that depend on the position of the sprout within the network (Anderson et al. 2004). To investigate whether the morphology of capillary sprout endothelial cells with respect to the elastic fiber matrix exhibits a similar positional dependence, we performed an additional analysis in which the capillary sprouts from CD31- and elastin-labeled compound 48/80 specimens were divided into two categories. These sprouts were categorized as either invading or introverting. As previously defined (Anderson et al. 2004), invading sprouts grow directly into the avascular region of the mesenteric window, thereby advancing the growing network into the tissue. Introverting sprouts were defined by their characteristic growth into tissue regions bordered on all sides by existing microvessels. Therefore, these sprouts increase microvascular density in tissue regions that have already been vascularized.
For studies examining the density and diameter of the elastic fiber matrix, low (×10)- and high (×180)-magnification confocal images from mesenteric windows of normally developing young adult animals were acquired using a Bio-Rad MicroRadiance confocal scanner attached to the Nikon TE-300 microscope. Elastic fiber densities were determined by thresholding and skeletonizing the low-magnification images using Image J software, which is available for download from the National Institutes of Health (http://rsb.info.nih.gov/ij/). Skeletonized images were used to compute the total number of pixels per image, which, in turn, was converted into the total length of elastic fiber per unit area of mesenteric tissue. Elastic fiber diameters were measured directly from high-magnification confocal images with the use of Image J software. The ×180 magnification images were generated using the ×60 objective and a ×3 electronic zoom on the confocal microscope.
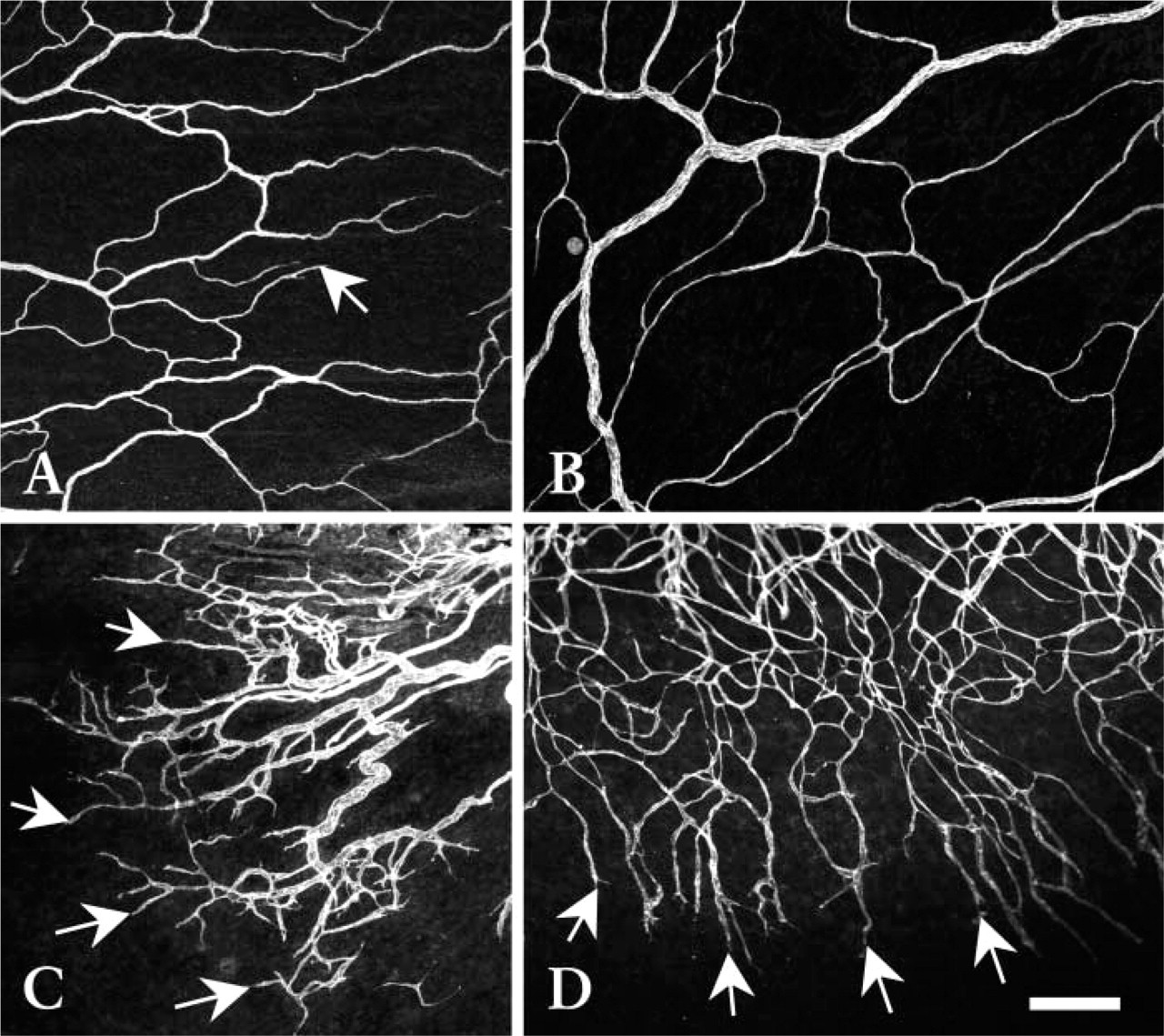
Low-magnification confocal microscopy images of CD31-labeled microvascular networks in rat mesenteric connective tissue windows from juvenile (
Results
Figure 1 depicts representative mesenteric microvascular networks at the time of tissue harvest. As expected, vessel density and capillary sprouting were significantly increased in both wound-healing– (Figure 1C) and compound 48/80–treated mesenteries (Figure 1D) in comparison to both their weight-matched young adult controls (Figure 1B) and the specimens from juvenile animals (Figure 1A). In the wound-healing mesenteries, the arterioles and venules typically had larger diameters and shorter segment lengths compared with the young adult– and compound 48/80–treated specimens. In both the wound-healing– and compound 48/80–treated specimens, capillary loops were abundant after treatment and contributed to a considerable portion of the increase in overall vascular density.
The microguidance of capillary sprouting by the elastic fiber matrix is illustrated in Figures 2–4. Low-magnification images are provided in each of these figures to illustrate the structure of the elastic fiber matrix with respect to the mesenteric microvascular networks and to demonstrate the affinity of the antibodies for the endothelial cells (Figures 2A–2C and 3A–3C) and pericytes/smooth muscle cells (Figure 4A–4C) comprising rat mesenteric microvessels. Higher-magnification confocal images show the influence of the elastic fiber matrix on the morphology of capillary sprout leading endothelial cells (Figures 2F, 3F, and 3I) and pericytes (Figures 4F and 4I). In the majority of capillary sprouts that were invading the avascular tissue space, the leading endothelial cells and pericytes were aligned and in contact with an underlying elastic fiber. In contrast, the endothelial cells leading many short and/or introverting capillary sprouts were more rounded and often were not aligned with an elastic fiber (Figure 3O). In some sprouts, higher-magnification observations revealed that leading endothelial cells extended filopodia that contacted multiple elastic fibers (Figure 3L).
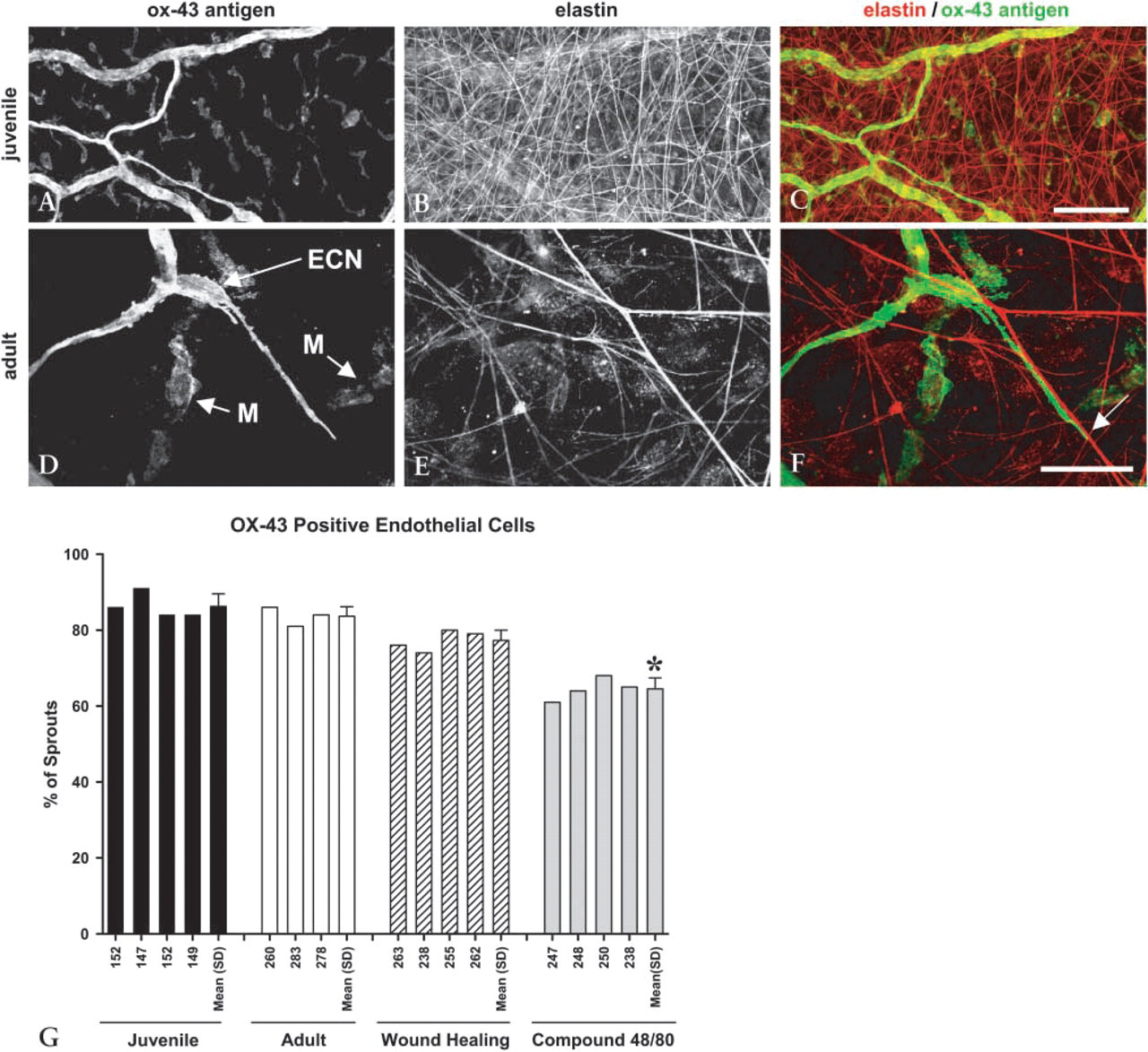
Morphology of OX-43-positive endothelial cells in microvessels and capillary sprout tips. (
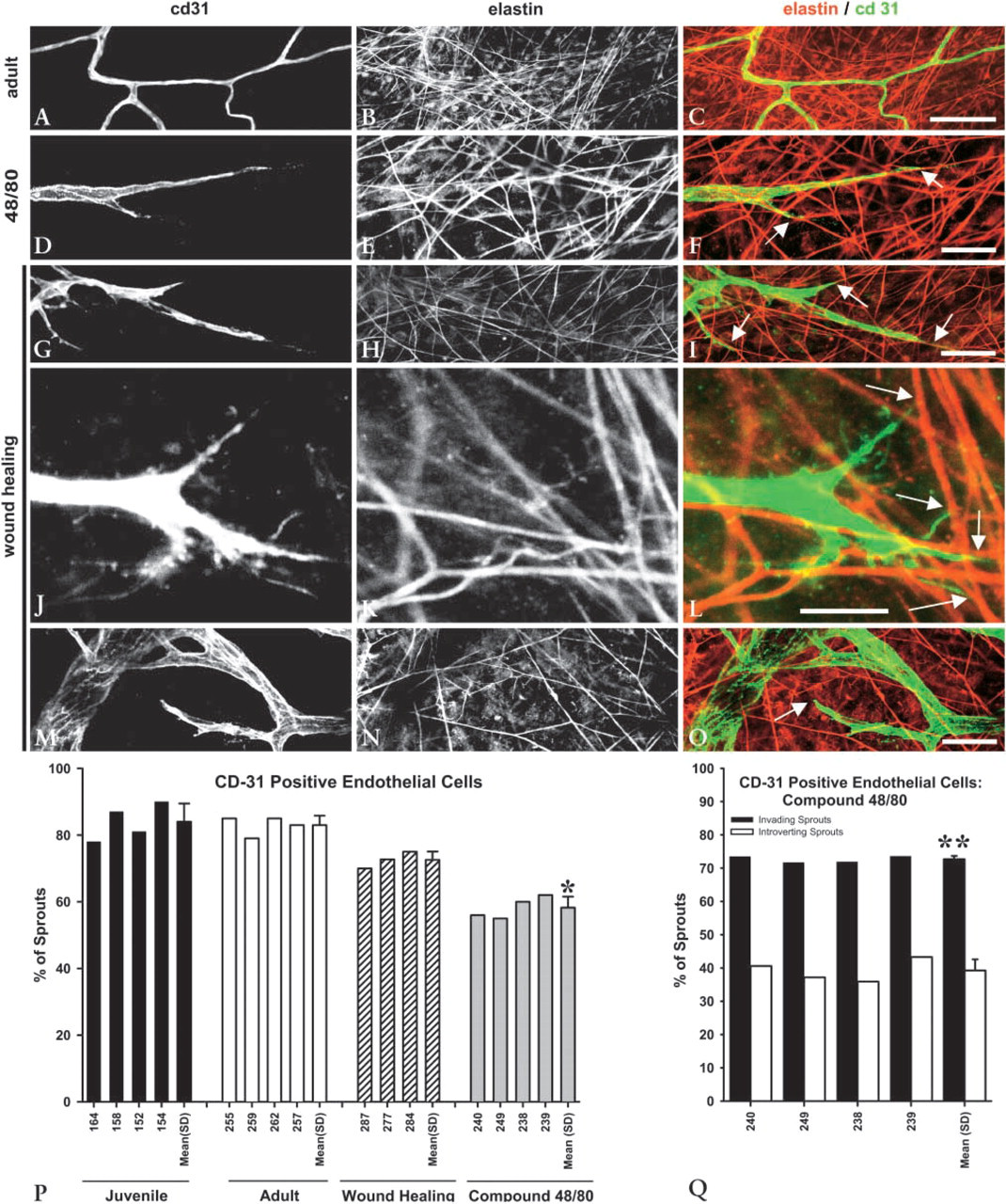
Morphology of CD31-positive endothelial cells in microvessels and capillary sprout tips. (
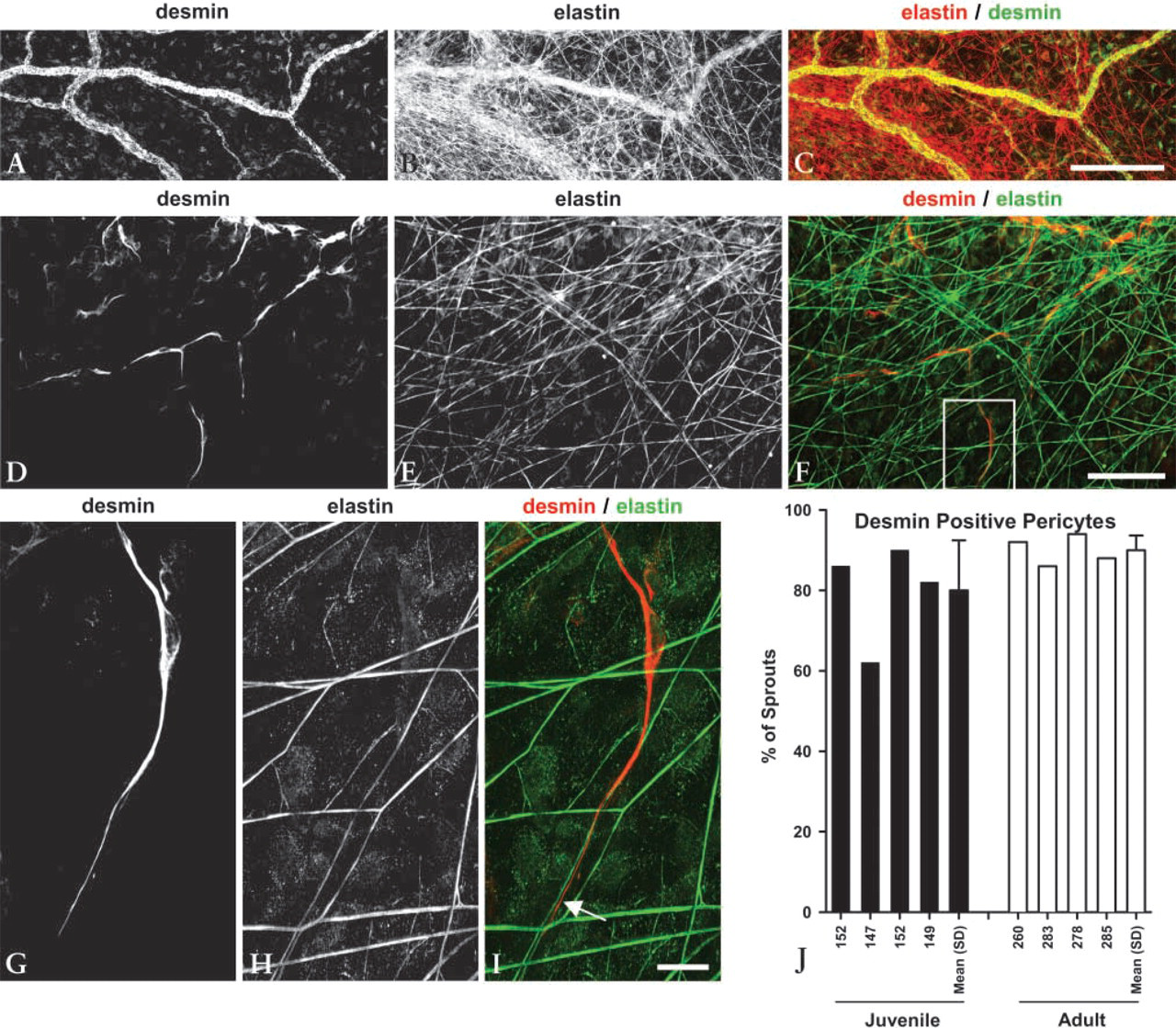
Morphology of desmin-positive pericytes in microvessels and capillary sprout tips. (
Quantification of the contact and alignment of capillary sprout leading endothelial cells with respect to the elastic fiber matrix is presented in Figures 2G, 3P, and 3Q. This same quantity is reported for desmin-positive pericytes in Figure 4J. In each of these graphs, the bars indicate the percentage of capillary sprouts exhibiting the contact and alignment morphology shown in Figures 2F, 3F, 3I, and 4I. Cells exhibiting the morphology shown in Figure 3O are not considered to be in this conformation because, even though it was possible that filopodia from the cell were contacting the fiber, the cell body was not aligned with an elastic fiber. In Figure 2, OX-43 antigen labeling was used to delineate capillary sprout endothelial cells. During normal maturation, ≃80–90% of OX-43 antigen-positive capillary sprout leading endothelial cells were aligned and in contact with an elastic fiber (Figure 2G). This percentage decreased significantly for the compound 48/80–treated specimens but still remained above 60%, thereby indicating that this is the preferred morphology for migrating sprout cells even when angio-genesis is substantially accelerated. The OX-43 data in Figure 2G are supported by Figure 3P, in which CD31 was used as a second marker for endothelial cells. Note that the percentage of capillary sprouts with leading endothelial cells aligned and in contact with elastic fibers is virtually identical for each marker. In Figure 3Q, the compound 48/80 data from Figure 3P are further analyzed by separating the sprouts on the basis of their anatomic position in the network. Here we observed that endothelial cells in capillary sprouts that are invading the avascular tissue space align with and contact elastic fibers far more often (73% of invading sprouts) than the endothelial cells in capillary sprouts that are introverting back into vascularized regions (39% of introverting sprouts). Elastin and CD31 staining was uniform throughout the tissue, indicating that the differences seen in the introverting sprouts were not due to tissue processing and/or staining artifacts. Finally, in Figure 4J, the desmin-labeled pericyte data are shown. Similar to the endothelial cell data, the leading pericyte was aligned and in contact with an elastic fiber in >80% of all capillary sprouts during normal maturation in both the juvenile and young adult animals.
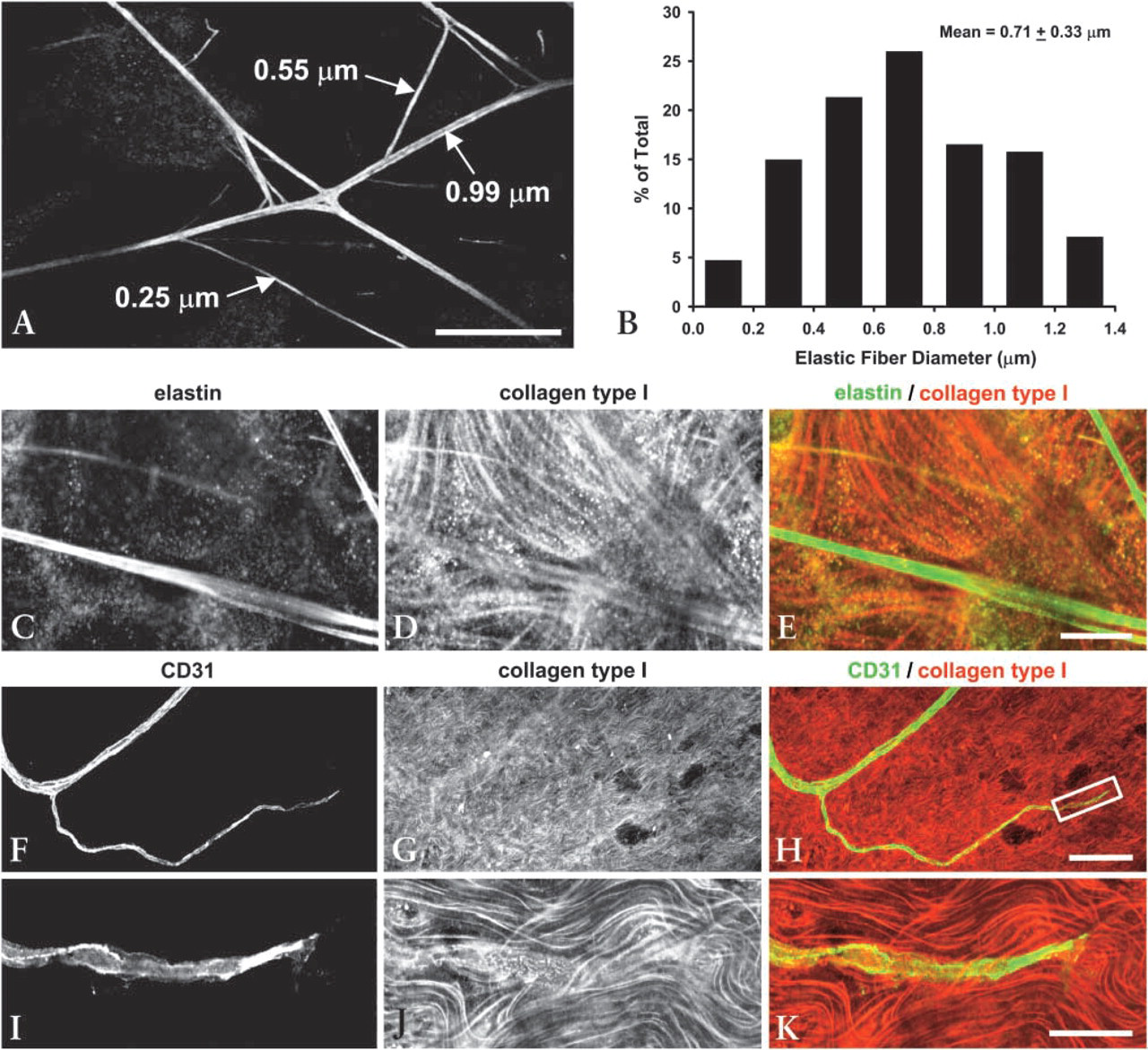
Morphology of elastic and type I collagen fibers in rat mesentery. (
The elastic fiber scaffolds on which capillary sprout cells migrate during normal maturation of the animal in the juvenile phase are characterized in Figures 5A and 5B. Elastic fiber diameters were measured from high-magnification (×180) confocal images (Figure 5A). A histogram of elastic fiber diameters is provided in Figure 5B. Elastic fiber diameters ranged from 0.18 to 1.40 μm, with a mean value of 0.71 μm. When we observed elastic fiber-guided sprouting, we noted that no particular fiber diameter or range of diameters was typically used for migration. The total length of elastic fiber per unit surface area of mesenteric connective tissue was calculated from lower-magnification images (×10) and was found to be 140.8 ± 37 mm of elastic fiber/mm2 tissue. The general morphology of the elastic fiber network with respect to type I collagen fibers is shown in Figures 5C–5E. Parallel type I collagen fibers formed bundles that were woven throughout the tissue and, unlike the almost perfectly straight elastic fibers, exhibited a wavy or buckled morphology. This general morphology is also clearly evident in Figure 5J. In Figures 5F–5K, a CD31-labeled capillary sprout is shown with respect to the resident type I collagen fibers. Unlike the elastic fiber results shown in Figures 2–4, contact guidance of sprout leading cells along type I collagen fibers was not observed.
Discussion
The central finding of this study is that, during angio-genesis in rat mesenteric connective tissue, capillary sprout endothelial cells and pericytes preferentially migrate along resident elastic fibers. The prevalence of this phenomenon was influenced by both the applied angiogenic stimulus and the anatomic position of the sprout within the network. In particular, with compound 48/80 application, the percentage of capillary sprout endothelial cells aligned and in contact with elastic fibers decreased significantly from ≃85% to ≃60% (Figures 2G and 3P). Further analysis of capillary sprouting in these networks revealed that this difference was due to the increased prevalence of capillary sprouts that were introverting back into regions of tissue that were already vascularized (Figure 3Q). In the compound 48/80–treated specimens, only 39% of all introverting sprout leading endothelial cells aligned with elastic fibers. In contrast, 74% of the capillary sprouts that were invading the avascular tissue space were still aligned and in contact with elastic fibers (Figure 3Q). The cells leading many introverting sprouts extended filopodia to elastic fibers but were not yet aligned with the fiber itself. We speculate that this morphology, which is illustrated in Figure 3L, may indicate that the sprouting cells are seeking elastic fibers via filopod extension before changing their direction of migration.
Interestingly, these differences in endothelial cell association with elastic fibers mirror recently observed phenotypic differences between endothelial cells in introverting and invading capillary sprouts (Anderson et al. 2004). These phenotypic differences were revealed by differential expression of the OX-43 antigen. Specifically, invasive capillary sprout endothelial cells typically lack OX-43 antigen expression, whereas introverting sprout endothelial cells express detectable levels of the OX-43 antigen. Whether or not these phenotypic differences are related to elastin fiber attachment is unknown. However, a correlation between endothelial cell phenotype and elastic fiber attachment and alignment exists and would make an interesting topic for future mechanistic studies.
The presence of ECM components, such as laminin, type IV collagen, heparan sulfate proteoglycan, and fibronectin, around capillary sprouts has been shown by other investigators (Jerdan et al. 1991; Sephel et al. 1996; Amselgruber et al. 1999). However, these other studies concluded that the provisional basement membrane was secreted by cells in the capillary sprouts, but not by cells (i.e., fibroblasts) in the surrounding milieu. To our knowledge, the present study is the first to show that capillary sprouts can utilize pre-existing elastic fibers as a substrate for migration during angio-genesis. Fortunately, the manner in which these elastic fibers are produced from elemental components has been well described. Initially, single fibrillin molecules are secreted from a cellular source and self-assemble into polymers in a parallel orientation to form 10-nm microfibrils (Reinhardt et al. 1996). Then secreted tropoelastin is assembled into elastin in the presence of these fibrillin microfibrils (Robb et al. 1999). The elastin-containing microfibrils then bundle to create larger-diameter elastic fibers which, in this study, ranged from ≃100 to 1400 nm in diameter. In addition, it has been shown that microfibril-associated glycoproteins (Fauvel-Lafeve 1999), and the fibrillin homologue known as latent TGFβ-binding protein may also be incorporated into elastic fibers (Raghunath et al. 1998; Sinha et al. 1998).
Given the likelihood that the elastic fibers in rat mesentery are composed of several constituents, a diverse collection of molecular recognition sites may exist for cell binding. Of these potential binding sites, at least two have been studied. First, it has been shown that cells bind directly to the VGVAGP peptide repeat in elastin through a 67-kD non-integrin adhesion molecule known as the elastin-laminin receptor (ELR) (Mecham et al. 1997). Because of the clear abundance of elastin in the fibers of rat mesentery, the ELR is a prime candidate for mediating adhesion. Second, both fibrillin and microfibril-associated glycoprotein-2 contain RGD-binding recognition sites and, as previously documented (Pfaff et al. 1996; Sakamoto et al. 1996; Gibson et al. 1999), cells adhere to these proteins through αv-integrins and the critical role of αv-integrins in angiogenesis has been extensively studied (Brooks et al. 1994; Friedlander et al. 1995). Finally, although our observations suggest that type I collagen fibers do not guide capillary sprouting per se, it is possible that sprouting cells use collagen fibers for support while migrating along elastic fibers. Ultimately, future studies will be needed to determine which adhesion molecules mediate migration in this particular model of angiogenesis.
Our general observations may also be significant for tissue-engineering applications because they demonstrate that sprouting may be guided by discrete elastic fibers comprising a tissue support scaffold. In conjunction with our general observations of type I collagen bundle morphology, the specific measurements of elastic fiber density and diameter provided here may be used to generate biomimetic tissue-engineered scaffolds and constructs that directionally guide the migration of endothelial cells and pericytes within capillary sprouts. Throughout our observations of elastic fiber-guided sprouting, no clear preference of the sprout leading cells for a certain diameter fiber was seen, indicating that both large- and small-diameter elastic fibers were capable of sprout guidance and could be included in such a design. The elastic fibers in such constructs could be directionally orientated to control both the rate and spatial position of a developing microvascular network, essentially providing another level of control over the manner in which tissue-engineered constructs become vascularized. Moreover, the key observation that elastic fibers are capable of guiding pericyte migration may be particularly important because these cells are necessary for stabilizing and maturing microvessels (Lindahl et al. 1997), which is certain to be a critical step in ensuring the functionality of an engineered microvascular network. Although additional knowledge about the molecular interactions between capillary sprout cells and the underlying elastic fiber matrix is still required, the present study provides a foundation for the development of a biomimetic tissue-engineered scaffold for microguiding angiogenesis.
Footnotes
Acknowledgements
Supported by a grant from the National Institutes of Health (RO1 HL66307).
