Abstract
Background:
An increasing number of adult individuals are at risk of vitamin B12 deficiency, either from reduced nutritional intake or impaired gastrointestinal B12 absorption.
Objective:
This study aims to review the current best practices for the diagnosis and treatment of individuals with vitamin B12 deficiency.
Methods:
A narrative literature review of the diagnosis and treatment of vitamin B12 deficiency.
Results:
Prevention and early treatment of B12 deficiency is essential to avoid irreversible neurological consequences. Diagnosis is often difficult due to diverse symptoms, marked differences in diagnostic assays' performance and the unreliability of second-line biomarkers, including holo-transcobalamin, methylmalonic acid and total homocysteine. Reduced dietary intake of B12 requires oral supplementation. In B12 malabsorption, oral supplementation is likely insufficient, and parenteral (i.e. intramuscular) supplementation is preferred. There is no consensus on the optimal long-term management of B12 deficiency with intramuscular therapy. According to the British National Formulary guidelines, many individuals with B12 deficiency due to malabsorption can be managed with 1000 µg intramuscular hydroxocobalamin once every two months after the initial loading. Long-term B12 supplementation is effective and safe, but responses to treatment may vary considerably. Clinical and patient experience strongly suggests that up to 50% of individuals require individualized injection regimens with more frequent administration, ranging from daily or twice weekly to every 2-4 weeks, to remain symptom-free and maintain a normal quality of life. ‘Titration’ of injection frequency based on measuring biomarkers such as serum B12 or MMA should not be practiced. There is currently no evidence to support that oral/sublingual supplementation can safely and effectively replace injections.
Conclusions:
This study highlights the interindividual differences in symptomatology and treatment of people with B12 deficiency. Treatment follows an individualized approach, based on the cause of the deficiency, and tailored to help someone to become and remain symptom-free.
Plain language title
Diagnosis and Treatment of Vitamin B12 Deficiency
Plain language summary
The number of people who are at risk of developing a deficiency of vitamin B12 is steadily increasing. B12 deficiency can develop when people consume too few B12-containing foods of animal origin, or when they develop a form of B12 malabsorption. B12 deficiency can lead to serious complications so prevention and early treatment are essential. Diagnosing B12 deficiency can be challenging: the symptoms vary from patient to patient, and the methods used to measure B12 in the blood, or certain biomarkers associated with B12 metabolism, such as holo-transcobalamin, methylmalonic acid, and total homocysteine are unreliable. When people do not consume enough B12-containing foods, supplementation with B12 tablets is needed. In the case of B12 malabsorption, intramuscular injections of B12 are mandatory. The usual treatment with B12 is starting with injections of 1000 µg hydroxocobalamin twice weekly or on every other day for a period of up to 5 weeks or longer, until all symptoms have disappeared, and thereafter, the frequency of injections is gradually reduced. There is, however, a large group of people who require more frequent administration to become and remain symptom-free: this may range from daily or twice weekly to every 2 to 4 weeks.
Introduction
Vitamin B12 (B12; cobalamin) is an essential water-soluble vitamin which is required for the proper functioning of the nervous system and the development and maturation of blood cells. B12 also has antioxidant effects. It is a cofactor in carboxylic acid interconversions involved in energy metabolism and production within the mitochondria and is essential for DNA synthesis, the methylation cycle, and epigenetic regulation. 1 Humans obtain their B12 by consuming foods of animal origin, such as meat, dairy products, or fortified foods.
Long-term B12 deficiency leads to a variety of symptoms. Historically, B12 deficiency was identified in Addison-Biermer’s disease, a disorder characterized by megaloblastic anemia. 1 In this situation, autoimmune atrophic gastritis associated with reduced or absent intrinsic factor (IF) production leads to B12 malabsorption. Antibodies against IF and/or parietal cells may be detected in the serum of such individuals. This type of anemia was the first condition linked to B12 deficiency, hence many physicians mistakenly believe that B12 deficiency is excluded in patients who are not anemic. The recognized phenotype of B12 deficiency has broadened in recent decades. This can lead to delayed or missed diagnoses, notably in patients presenting solely with neurological symptoms. 2,3 Nowadays, neurologic and neuropsychological or cognitive complaints (Table 1) predominate in many individuals with B12 deficiency, 4 with anemia present in fewer than one-fifth. 5,6 Unfortunately, many clinical symptoms of B12 deficiency are nonspecific, and so not always easily recognized as being associated with a possible deficiency. There is an urgent need for a well-defined (i.e., objective) symptom score and better biomarkers for earlier diagnosis of B12 deficiency and to monitor treatment outcomes. 7 The reasons why some patients mainly present with megaloblastic anemia and others with neurological complaints remain unknown. 8 Earlier research suggests that endogenous formation or intake of cobalamin analogues may play a role in developing a syndrome with more prominent neurological symptoms. 9
The Most Frequently Reported Signs and Symptoms of B12 Deficiency.

Reduced Dietary Intake
Inadequate dietary intake is the most prevalent cause of B12 deficiency worldwide. As foods of animal origin are the main contributors to nutritional sources of B12, vegetarians, vegans, and populations traditionally consuming limited amounts of these food products are at greater risk of developing deficiency. 10 Dietary intake of B12 decreases transitioning from a purely omnivorous diet to a strict plant-based diet, 11 with average daily B12 intake estimated to be approximately 2.1 to 7.2 µg in meat-eaters but only ∼0.4 µg in vegans. 12,13 Food-derived B12 absorption can also become considerably reduced in older individuals, likely due to simple gastric atrophy. 14,15
Impaired B12 Absorption
An individual diagnosed with B12 deficiency who is eating sufficient foods of animal origin has—by definition—inadequate gastrointestinal absorption of B12. 16 As mentioned, the classic presentation of B12 deficiency is megaloblastic anemia caused by pernicious anemia. 17,18 We prefer the use of the term “vitamin B12 deficiency,” as the majority of B12 deficient patients in Western countries do not present with anemia or macrocytosis, 4,19,20 and several other underlying disorders which interfere with B12 absorption may also cause deficiency. 5 These include small bowel diseases (e.g., Crohn’s disease or celiac disease) 21 and gastric or ileal surgery. For example, bariatric gastric-reduction surgery is a major and frequently performed treatment for (morbid) obesity, which may result in diabetes amelioration and improved cardiovascular outcomes. Moreover, decreased production of IF and gastric acid and low intake of B12-containing foods contribute to the development of deficiency. 22 -24 Patients undergoing (partial) gastrectomy, gastric bypass surgery, or small intestinal resections will likely develop B12 deficiency requiring lifelong B12 replacement. Other factors contributing to B12 malabsorption include the use of certain medications such as proton-pump inhibitors, 25,26 H2 receptor blockers, antacids, and metformin. Severe cases of B12 deficiency with extensive neurological abnormalities are also reported in individuals using nitrous oxide as a recreational drug, since it inactivates B12. 27 -29
Familial B12 Deficiency
In contrast to widespread belief, children may also develop B12 deficiency. 30 In recent years, we have counselled several families where the mother and one or more children had established B12 deficiency despite normal nutrition with sufficient dietary B12 intake. In many such families, it could be ascertained that B12 deficiency was present in the mother during pregnancy. Genetic testing in one family revealed the C677 T MTHFR mutation in both the mother and 2 of her affected children (unpublished data). In contrast, genetic testing did not reveal any clue indicating a genetic cause in 3 other families. We hypothesize that deficiency of B12 in the mother during pregnancy may lead to epigenetic alterations in the children, negatively affecting gastrointestinal B12 absorption. A recent study in ileal epithelial cells demonstrated that B12 influences the methylation of genes associated with intestinal barrier function and cell proliferation. 31 Further research is needed to evaluate and better understand the possible transgenerational effects of B12 deficiency on offspring.
Biochemical Diagnosis of B12 Deficiency
Diagnosing B12 deficiency can be challenging, as recently reviewed. 5,17,32 -34 There is no single specific laboratory marker to diagnose or exclude the presence of deficiency in a reliable manner. The most commonly used biomarkers to diagnose B12 deficiency are the serum concentrations of total B12 and—sometimes—methylmalonic acid (MMA). Measurement of serum total homocysteine (tHcy) concentration and holo-transcobalamin (holoTC), the biologically active form of vitamin B12 in blood, may provide additional information. Unfortunately, many diagnoses are still made or dismissed solely based on a single serum B12 measurement.
The diagnostic starting point is an individual seeking a consultation because of complaints (Table 1). Measuring the total serum B12 concentration is usually the first (and often only) diagnostic step. In this situation, a serum B12 concentration below 148 pmol/L 17 indicates B12 deficiency and is generally thought to be sufficient to start supplementation. However, symptoms may also be present in individuals with serum B12 above 148 pmol/L. Deficiency is therefore easily overlooked when total serum B12 is used as a the only status marker. 1,35 -39
Several known factors influence the accuracy of a total serum B12 measurement. Firstly, there is remarkable variability between the results obtained with different commercially available serum B12 assays. Quality control assessments show that the concentration of B12 in a given sample may vary between 240 pmol/L with one assay, and 470 pmol/L with a different assay in another hospital. These quality control assessments have also reported considerable variations between results with the same methodology and assay carried out in different laboratories. Secondly, there is significant day-to-day variation of serum B12 within one individual. For example, a concentration of 160 pmol/L on one day may be 120 pmol/L on another day, and a recent study showed that the variation of serum B12 may be well over 100 pmol/L. 40 Thirdly, some assays may be susceptible to interference through the presence of antibodies to IF, resulting in normal serum B12 concentrations in individuals who are, in fact, B12 deficient. 5,41 Finally, oral B12 supplementation may result in serum B12 concentrations just within or sometimes above the “normal” reference range, often without abolishing symptoms. Unfortunately, this is frequently not recognized in clinical practice, often because vitamin supplement use is not discussed during a consultation.
Holo-Transcobalamin, MMA, and/or Homocysteine
Some studies suggest that measuring holoTC, the biologically active fraction of vitamin B12 in blood, has better diagnostic accuracy than total serum B12. 42,43 However, holoTC also shows a wide variation with indeterminate levels, 44 and the reference values strongly depend on which assay is used. 42
Many authors advocate measuring one, or both, of the additional functional biomarkers MMA and/or tHcy to establish B12 deficiency. They may be helpful, especially in people with borderline serum B12 levels, that is, those in the so-called “grey zone” between 148 and 300 pmol/L. 34,35,44,45 Unfortunately, despite its early use, 46 MMA is not a very sensitive marker of B12 deficiency. One study in Germany showed that 52% of individuals with normal renal function and holoTC concentration below 20 pmol/L—which is considered definite proof of B12 deficiency in almost all available diagnostic algorithms 34,44 —had normal serum MMA, while 9% of people with holoTC above 63 pmol/L had elevated MMA. 47 Others have confirmed the poor sensitivity of MMA. 48 -50 Methylmalonic acid is also elevated with impaired renal function. 51 In addition, recent antibiotic treatment may result in false-normal MMA concentrations. 52 Some authors have suggested replacing serum MMA with urinary MMA measurement, but in one study the number of individuals with zero urinary MMA was 30%, rendering this assay unreliable. 53
Serum tHcy is an alternative functional biomarker of B12 status but is also elevated in folate deficiency, vitamin B6 deficiency, impaired renal function, hypothyroidism, and by certain medications. 54 Thus, elevated serum MMA and/or tHcy may confirm the diagnosis of B12 deficiency, but in people with symptoms, normal concentrations of these biomarkers do not exclude this diagnosis. In a situation where the patient’s symptoms are compatible with a diagnosis of B12 deficiency, but biochemical results cannot confirm this (for instance, in a person taking oral B12 supplementation) a therapeutic trial with B12 supplementation should be considered and discussed with the patient (see later in the section on Treatment).
Genetic Factors Influencing Biomarker Concentrations
In addition to intra-individual differences over time and assay problems, there are also several genetic factors which influence the serum concentrations of B12 and its related biomarkers. A total of 15 genetic polymorphisms or mutations within genes related to the absorption, transport, and metabolism of B12 are known to influence serum B12 concentrations (Figure 1). 55 -57 The best known are polymorphisms in the fucosyltransferase 2 (FUT2) gene, where the FUT2 secretor variant genotype AA is associated with a 10% to 25% higher concentration of total and haptocorrin-bound B12, but not holoTC. 58 Altogether, these single nucleotide polymorphisms (SNPs) may account for considerable variation of serum B12 between individuals. Epidemiological studies have shown a difference of at least 100 pmol/L depending on the number of B12-increasing SNPs in a given individual. 59 A recent study using UK Biobank data suggested that a higher genetically predicted vitamin B12 concentration is associated with a lower chance of being diagnosed with pernicious anemia or B12 deficiency, which may be caused by solely relying on serum B12 concentrations to identify or exclude this disease. 6
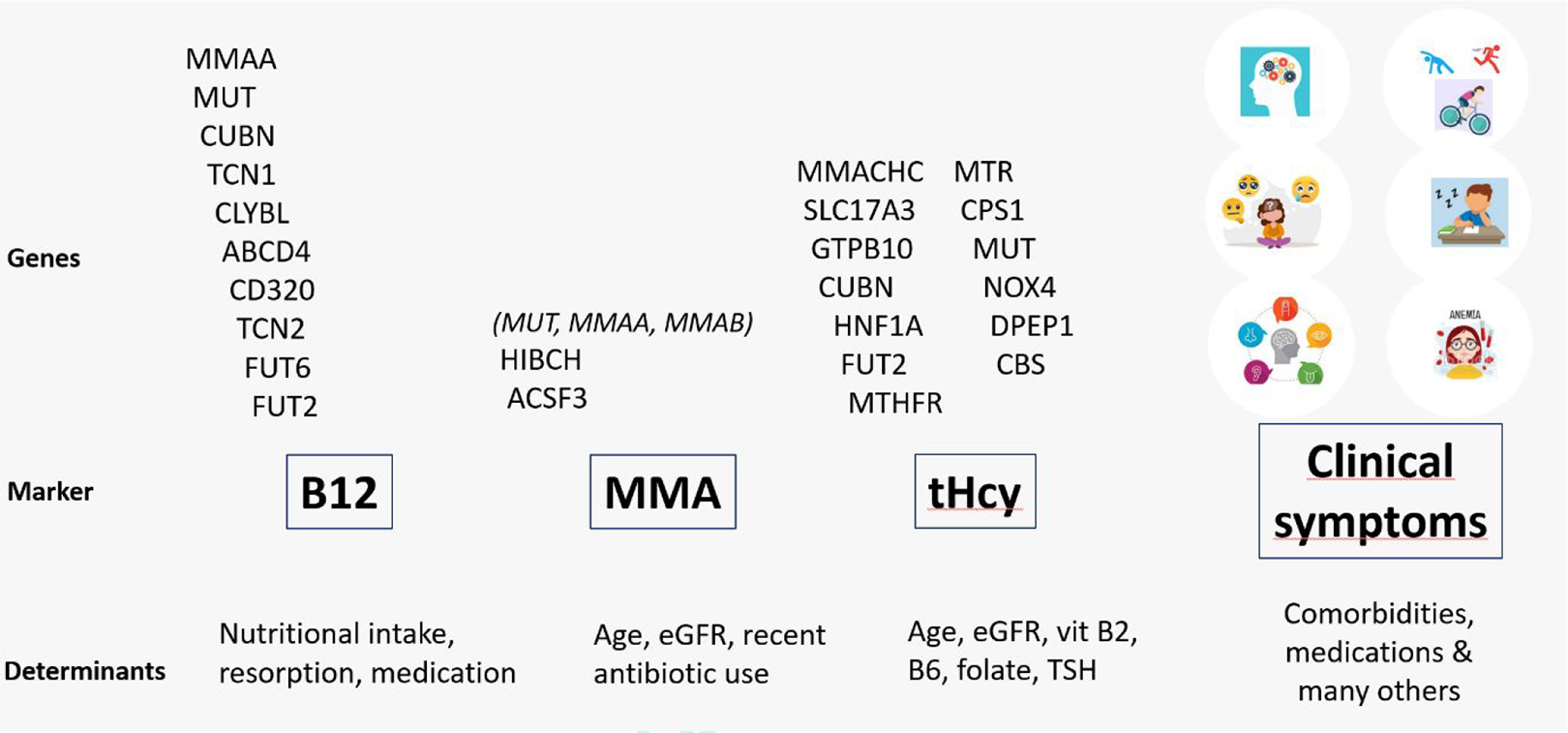
Genetic polymorphisms and other determinants influencing serum concentrations of B12, methylmalonic acid, and total homocysteine, and the presence and severity of clinical symptoms related to B12 deficiency.
Similarly, the results of serum MMA measurements are primarily influenced by genetic polymorphisms in 2 genes, namely HIBCH and ACSF3 (Figure 1). 60,61 For HIBCH rs291466, it was shown that TT homozygotes had a 30% to 40% lower serum MMA concentration than CC homozygotes, and 20% lower than CT heterozygotes. In contrast, this SNP did not affect serum B12 or holoTC concentrations. 61 At least 18 SNPs are associated with serum tHcy concentrations, including several involving folate metabolic pathways, and 3 of them (SNPs in MUT, FUT2, and CUBN) are also associated with serum B12 concentrations. 60
Treatment
The therapeutic goal in people with low dietary intake is to prevent the onset of deficiency and symptoms by means of oral supplementation. In people with established—symptomatic—B12 deficiency, the goal is to reverse the metabolic abnormalities and abolish the clinical symptoms, as recently reviewed. 62
Low Dietary Intake
Healthy adults require an average daily intake of 4 to 7 µg of vitamin B12 to maintain a normal B12 status. 11 In cases of limited B12 intake or dietary insufficiency, oral supplementation is usually sufficient, but it should be taken with food at doses higher than the current 4 µg recommendations. With advancing age, the intestinal absorption of B12 gradually declines, and elderly people need a higher supplemental B12 dose. In people with severe symptoms due to B12 deficiency caused by insufficient dietary intake, parenteral supplementation will replenish stores and abolish the symptoms more rapidly leading to improvement in the clinical situation. Once stabilized, maintenance may be switched to oral supplementation. Daily cyanocobalamin 50 µg tablets are most frequently advised and used. In some countries, preparations containing methylcobalamin and/or adenosylcobalamin are also available, and sometimes heavily promoted. Most compounds are labelled as food supplements, and only a few of them as prescription-only medication. One study in people following a plant-based diet suggests better efficacy of supplementation with cyanocobalamin compared to methylcobalamin. 63 Long-term clinical assessment and laboratory monitoring of B12 status and suitable adjustment of the dose of oral supplementation 64 should be considered. Although several multinutrient combination preparations are available, it is advisable to use these prudently to avoid administration of excessive folate or B6 intake. Notably, excess intake of vitamin B6 can cause or exacerbate neuropathic symptoms, 65,66 and excessive folate intake can negatively affect B12 status. 67
B12 Malabsorption
In most, if not all, cases of symptomatic B12 deficiency caused by malabsorption, B12 should be administered intramuscularly because of low absorption following oral administration due to the underlying disease. 33,68 Importantly, once started, B12 administration should be maintained for life. Suggested treatment schemes differ considerably between countries and, unfortunately, there is very little clinical evidence supporting any one of the treatment regimens over others worldwide. The initial recommended dose is 1000 µg hydroxocobalamin by intramuscular injection twice weekly (Netherlands), or on alternate days (United Kingdom). 62 This frequency should be continued when neurological complaints are present until the symptoms have entirely resolved or do not improve further. This may take several months or even years. Clinical experience suggests that the dose or injection frequency should not be suddenly reduced to a maintenance dose once every 2 months, as too rapid dose reduction may lead to symptom reoccurrence. 5,62 Instead, it is recommended to gradually increase the interval between injections over the course of several months while monitoring symptoms. A recurrence of symptoms suggests halting any further reduction in injection frequency. In general, this personalization of treatment is rarely observed in practice.
In many countries, hydroxocobalamin has replaced cyanocobalamin as the form of vitamin B12 of choice for therapy; it is retained in the body longer than cyanocobalamin and is more available to cells. 69 No systematic studies have compared the long-term efficacy and effectiveness of intramuscular hydroxocobalamin versus other preparations such as cyanocobalamin or methylcobalamin in individuals with symptomatic deficiency. Earlier studies reported considerable differences in pharmacodynamic properties of various forms of B12. 70,71 It is important to highlight that cyanocobalamin is contraindicated in individuals with impaired renal function. 72
Monitoring Outcome and Follow-Up
Treatment with B12 includes monitoring outcome(s), for example, resolution of anemia (if present) and improvements in or disappearance of (neurological) complaints. The blood count will usually normalize within 6 to 8 weeks. It should be noted that coexisting iron deficiency can be present in up to 60% of these patients which will also require appropriate supplementation. 73 Neurological symptoms may take several months or even years to resolve. In some patients, they never fully resolve (see later). Outcome monitoring should be done at regular intervals, as symptoms may reappear when reducing injection frequency. Monitoring should also include serum iron measurements (see earlier), as well as serum folate measurements, as folate deficiency is prevalent in countries without mandatory folic acid food fortification, and both cobalamin and folate are essential for the methylation cycle.
It is very important to highlight that measuring serum biomarkers such as B12 or MMA is neither helpful nor indicated in assessing or monitoring clinical improvement or resolution of symptoms. Biomarkers will normalize more rapidly than it takes for (neurological) symptoms to improve or disappear. Conversely, during relapse, symptoms may reappear without significant changes in biomarkers. “Titration” of injection frequency based on biomarker assessment is of little use and should not be practiced.
Clinical evidence indicates that with a longer duration and more severe B12 deficiency, there is an increased likelihood that neurological symptoms will not entirely disappear. 74 Mild to moderate neurological symptoms including poor memory, impaired concentration, and fatigue often remain even after long-term B12 therapy. 1,75,76
Unfortunately, there is limited evidence regarding the “best” B12 dose and frequency of dosing on long-term health outcomes, and this will vary between individuals. Data from patient surveys indicate that many people with symptomatic B12 deficiency report that their treatment regimen was insufficient for managing symptoms. 2,77 They experience recurrence or worsening of symptoms when the interval between 2 consecutive hydroxocobalamin injections is extended or extended too quickly, and continuation of frequent intramuscular injections of hydroxocobalamin, varying between daily to once every 2 to 4 weeks, 78 may be needed to remain asymptomatic. It is not understood why certain individuals require more frequent B12 treatment. This topic has gained much recent interest, and initial studies evaluating a possible role of the gut microbiome are underway. 79 However, there are possibly a number of other causes. These include competing effects of cobalamin analogue formation in the body, 9 and possibly mutations or epigenetic alterations influencing the transcobalamin receptor. 80,81 An intriguing recent study has demonstrated that (auto)antibodies against the CD320 (transcobalamin) receptor may interfere with the proper uptake of cobalamin into the brain. 82
Long-term studies clearly indicate that treatment with pharmacological doses of B12 and the corresponding increase in serum B12 concentrations do not increase mortality risk. 83 Treatment with parenteral B12 during pregnancy and lactation is safe, and should not be postponed or stopped. 62,84 Furthermore, parenteral, very high-dose hydroxocobalamin treatment has been used and proven to be safe during many decades of therapy of individuals—children and adults—with inborn errors of B12 metabolism. 85
Oral Supplementation or Parenteral Treatment?
Since the 1990s, the use of oral instead of parenteral administration of B12 has been hotly debated. It was suggested that passive absorption of B12 is about 1% to 2% of any oral dose. Hence, the use of sufficiently high doses (2000 µg) could result in up to 10 to 20 micrograms absorbed daily, 73 and this could allow serum B12 concentrations to normalize. However, early studies have shown that passive B12 absorption varies considerably between individuals, and may only be 0.1% to 0.5% in a significant proportion of patients. 86 Unfortunately, there is no feasible and well-described method to study passive B12 absorption in people in daily practice, and low to absent passive absorption may be the reason why many patients with symptomatic B12 deficiency do not respond to oral therapy. Moreover, most studies assessing the effects of oral B12 supplementation focused on normalizing serum B12 concentrations, while the specific goal of treatment is alleviating symptoms. These papers are summarized in the Supplemental Table 1.
The ambiguity surrounding studies comparing oral with intramuscular supplementation is well illustrated by the recently updated Cochrane review. 87 The authors conclude that the published studies only have provided very low-quality evidence that oral B12 supplementation is as effective and safe as intramuscular B12 treatment. Many of the study participants had nutritional deficiencies, a situation in which oral supplementation usually is sufficient (Supplemental Table 1). They advise that further trials should adequately measure pivotal outcomes such as a thorough evaluation of signs and symptoms of B12 deficiency, health-related quality of life, and adverse events. 87 Also, there are no prospective studies demonstrating which individuals with B12 deficiency can be safely transferred from intramuscular therapy to oral B12 supplementation, and many of them are at a considerable risk that their symptoms worsen after such a switch. 62,88
In addition to oral supplementation and parenteral therapy, nasal or sublingual preparations of B12 are available in some countries. Studies on the metabolic effects of such preparations are limited, but their bioavailability is low, 89 and clinical efficacy studies in people with symptomatic B12 deficiency are completely lacking. Some experts advocate subcutaneous injections when intramuscular injections are (relatively) contraindicated, for instance, in people using anticoagulant medications, or in elderly individuals with reduced muscle mass. It should be noted that studies directly comparing intramuscular and subcutaneous administration are also lacking. 90 Anecdotal evidence based on reports from several people with symptomatic B12 deficiency suggests a possible worsening of symptoms when switching from intramuscular to subcutaneous administration, although some physicians report beneficial effects of prolonged subcutaneous administration overall (Dr. A. Klein, MD PhD, personal communication). Nevertheless, in our experience, many individuals with B12 deficiency can be taught to self-administer the intramuscular injections, and this has been shown to improve treatment satisfaction and quality of life, and reduce the burden on GP practices as well as health care costs. 62,88
B12-Responsive Neuropathy
Isolated reports show that serum B12, homocysteine, and MMA levels are unreliable predictors of those neurologic disorders that respond to B12 supplementation, namely “B12-responsive neuropathies.” 91,92 In several countries, the use of hydroxocobalamin to treat neuropathy is a registered indication. Indeed, a recent systematic review concluded that there is evidence for the therapeutic effect of B12 in treating postherpetic neuralgia and painful peripheral neuropathy. 93
A Therapeutic Trial of Vitamin B12 Supplementation?
When a patient has specific symptoms compatible with B12 deficiency, but serum measurements of B12 (and MMA if available) fall within normal reference ranges, expert opinion recommends considering a therapeutic trial with B12 injections. 73 As an example, this can be considered in individuals with neurological or cognitive symptoms highly suggestive of B12 deficiency, but in whom the diagnosis is not clear from laboratory measurements or is obscured by the fact that the individual is already taking oral B12 supplementation, or in individuals with (sensory/painful) neuropathy in whom other causes have been excluded. 92,93 For such a therapeutic trial, we recommend a treatment regimen with the highest possible chance of leading to symptom relief, for example, B12 injections of 1000 µg once to twice weekly, for a period of at least 3 to 6 months. In case of nonresponse, the medication is stopped; in case of a positive response, the treatment is continued for a more extended period, and long-term injection frequency is based on the best possible maintenance of symptom relief as described above.
Conclusions
Long-term B12 supplementation is effective and safe, but responses to treatment may vary considerably. Practical tips for effective pharmacotherapy are summarized in Table 2. In case of limited or insufficient B12 intake, for instance, when adopting a plant-based diet, oral supplementation is needed. We advocate that such a person should take more than the current 4 µg recommendations, especially with age-related diminished B12 absorption. 11 In individuals with established B12 malabsorption, intramuscular injections should be the default therapy, with the dosage of 1000 µg hydroxocobalamin once every 1 to 2 months following initial loading. Clinical and patient experience strongly suggests that up to 50% of individuals, especially those with neurological symptoms, require individualized injection regimens with more frequent administration, ranging from daily or twice weekly to every 2 to 4 weeks, to become and remain symptom-free and maintain a normal quality of life. 62 “Titration” of injection frequency based on measuring biomarkers such as serum B12 or MMA should not be practiced. There is currently no evidence to support that oral/sublingual supplementation can safely and effectively replace injections.
Practical Tips for Effective B12 Pharmacotherapy.

Supplemental Material
Supplemental Material, sj-pdf-1-fnb-10.1177_03795721241229500 - A Brief Overview of the Diagnosis and Treatment of Cobalamin (B12) Deficiency
Supplemental Material, sj-pdf-1-fnb-10.1177_03795721241229500 for A Brief Overview of the Diagnosis and Treatment of Cobalamin (B12) Deficiency by Bruce H. R. Wolffenbuttel, Andrew McCaddon, Kourosh R. Ahmadi and Ralph Green in Food and Nutrition Bulletin
Supplemental Material
Supplemental Material, sj-pdf-2-fnb-10.1177_03795721241229500 - A Brief Overview of the Diagnosis and Treatment of Cobalamin (B12) Deficiency
Supplemental Material, sj-pdf-2-fnb-10.1177_03795721241229500 for A Brief Overview of the Diagnosis and Treatment of Cobalamin (B12) Deficiency by Bruce H. R. Wolffenbuttel, Andrew McCaddon, Kourosh R. Ahmadi and Ralph Green in Food and Nutrition Bulletin
Footnotes
Acknowledgments
BHRW drafted the first version of this manuscript. AM, KRA, and RG contributed to design, analysis, and interpretation; participated in a literature search; discussed its main conclusions; and provided significant additions and critical revisions of the manuscript. All authors approve the manuscript and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
