Abstract
Background:
Food aid is a valuable tool for meeting global nutrition goals, particularly for vulnerable populations of children and reproductive-aged women. On October 21, 2017, the Food Aid Quality Review Project hosted a scientific symposium at the 21st International Congress on Nutrition in Buenos Aires, Argentina, to take stock of what the global community has learned about selected topics in the research literature on food aid used to address malnutrition.
Objective:
This article presents the discussion that took place during the symposium, which was guided by presentations by 6 experts from the field of nutrition, food aid, and humanitarian response.
Conclusion:
The recent upsurge in research on food aid has advanced the collective knowledge of what food aid products and programs work for addressing nutrition, but there is much more to learn. Presentations in this symposium called for further inquiry on (1) different and novel food aid formulations, (2) the cost-effectiveness of products and programs, and (3) market-based approaches to food assistance. Continuing to expand the evidence base on these topics is critical to improving global nutrition programs.
Introduction
Acute and chronic undernutrition in children remain global issues of great public health significance: 23% of the world’s children younger than 5 years—155 million—are stunted, in many cases due to nutrition deprivation in utero or early childhood or both, and 50 million are acutely malnourished. 1 At the same time, conflict and persecution are uprooting more people than ever from their homes, increasing the risk of undernutrition for those affected. 2 Food aid (the donation of food to people in need) has long been an important tool for combating undernutrition among these vulnerable populations, but there is significant scope for increasing the efficiency and cost-effectiveness (CE) of food aid products and programs.
Historically, food aid has served many purposes for a great number of intended outcomes, but there has recently been a focus on how food aid affects the short- and long-term nutrition-related morbidity and mortality of recipients. Food aid activities have expanded beyond the practice of shipping excess raw commodities between nations to offering a basket of foods and “value-added” products—fortified commodities and fortified foods (known as specialized nutritious foods [SNFs], Table 1) that are usually provided in combination with other services. As these activities have evolved, there has been greater political and financial investment in scientific research to understand how to optimize the formulation and programming of SNFs for nutrition-related outcomes.
Different Forms of Food Aid. 3

Abbreviation: SAM, severe acute malnutrition.
A symposium held at the recent 21st International Congress of Nutrition in Buenos Aires brought together expert policymakers, nutrition researchers, and food aid practitioners from around the world to address 3 prominent research questions concerning food aid for nutrition: What formulations of food aid products (ie, nutrients, ingredient profile, macronutrient composition) best improve nutrition outcomes? This question was addressed by: – Dr Kim Michelson, who presented the topic, How does dairy protein perform when it is the principal source of protein in food aid? – Dr Steve Collins, who presented the topic, The efficacy of ready to use therapeutic foods made without milk or animal source protein. – Dr Kathryn G. Dewey, who presented the topic, Rationale and evidence regarding small-quantity lipid-based nutrient supplements for food assistance. What are the relative costs of different food aid products or programs for different nutrition outcomes? This question was addressed by: – Dr Beatrice Lorge Rogers, who presented the topic, Cost-effectiveness of specialized food aid products – what do we know? – Tatyana El-Kour, who presented the topic, “Cost-effectiveness: how can we use what we know?” How can local production of fortified nutritious foods be stimulated to benefit both food aid recipients and self-purchasing consumers? This question was addressed by: – Dr Saskia de Pee, who presented the topic, Scaling up the use of Special Nutritious Foods, going beyond food assistance.
These major topic areas were identified through the Research Engagement on Food Interventions for Nutritional Effectiveness online database of publications and active studies on food aid (http://refinenutrition.org/). This article reports on the topics that were discussed in the symposium and identifies remaining research questions, highlighting the next frontiers in SNF research.
What Formulations of Food Aid Products (ie, Nutrients, Ingredient Profile, Macronutrient Composition) Best Improve Nutrition Outcomes?
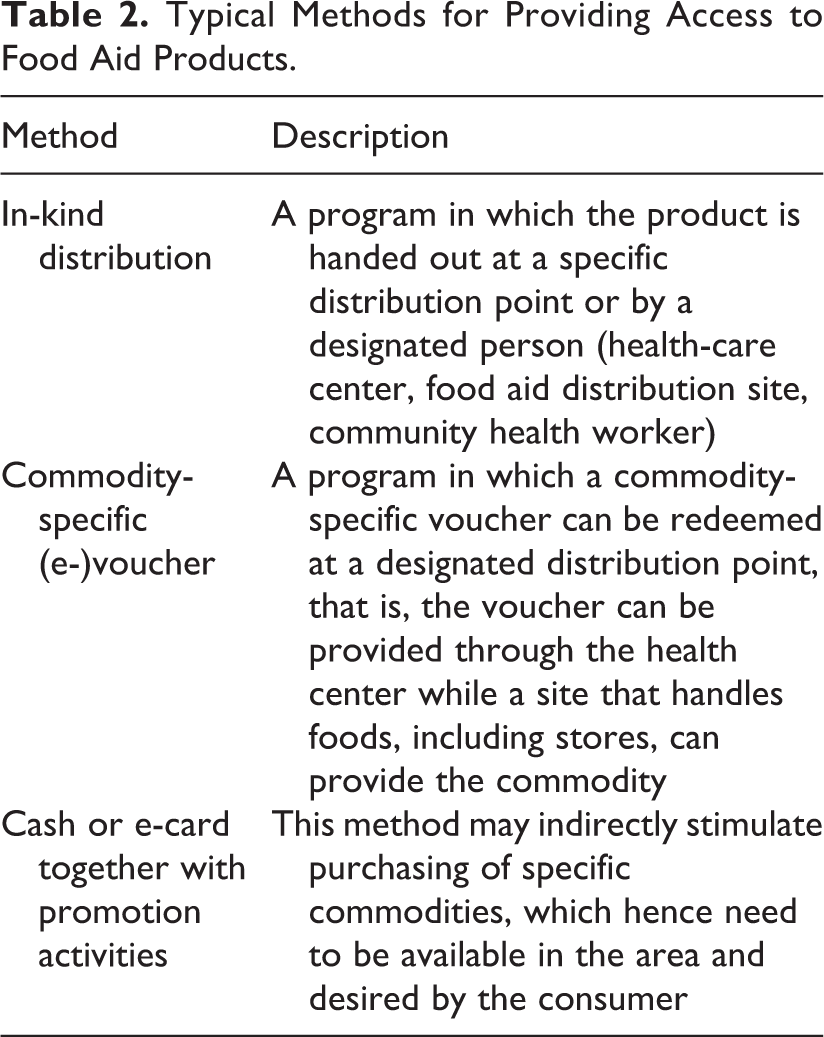
Intake of essential nutrients (including vitamins, minerals, essential amino acids [AAs], and essential fatty acids) is often below required levels among populations who receive food assistance (ie, both in-kind food transfers and other programs that provide the means to acquire food, Table 2) as well as among other populations whose diet has limited diversity due to constraints of availability and affordability. 4 Agencies involved in food assistance have increased their capacity to address these nutrient deficits by developing and distributing SNFs intended to supplement the regular diet for women of childbearing age, mothers, and children. The SNFs, including fortified blended foods (FBFs), micronutrient powders, and lipid-based nutrient supplements (LNS), are being tested for acceptability, stability, cost, as well as nutrient bioavailability and impact on nutritional status and functional outcomes—but the optimal formulations for achieving specific nutritional outcomes remains unknown.
Typical Methods for Providing Access to Food Aid Products.
Using Small-Quantity Lipid-Based Formulations
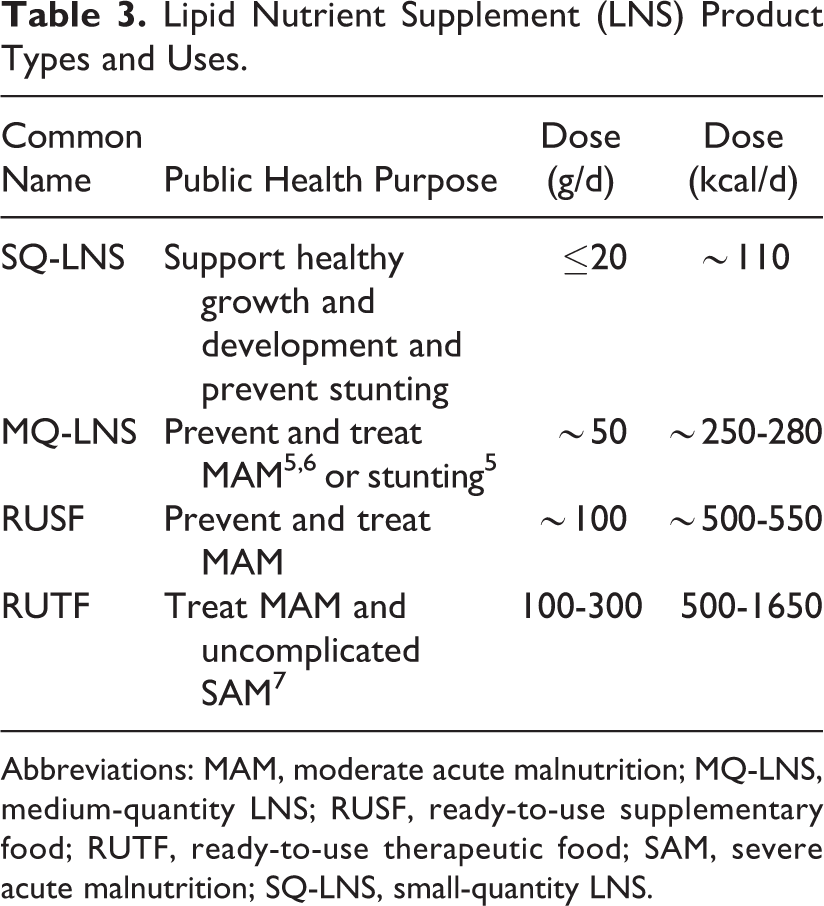
The LNS are a family of SNF that deliver vitamins and minerals, essential fatty acids, protein, and energy in a lipid matrix (Table 3). They can supplement a regular diet in order to meet essential nutrient needs and are increasingly used in development and emergency contexts. 8 To understand how LNS can be used for maximum nutritional impact, studies have evaluated the effects of different quantities for preventing malnutrition among infants and young children and for pregnant and lactating women. Small-quantity versions of LNS (SQ-LNS; ∼110 kcal/d) are made by a variety of manufacturers and can even be made in communities with locally available ingredients. Although there is currently no single “approved” formulation for these products, they are designed with the intention to support healthy growth and development and prevent stunting 9,10 and have been a recent focus of research.
Lipid Nutrient Supplement (LNS) Product Types and Uses.
Abbreviations: MAM, moderate acute malnutrition; MQ-LNS, medium-quantity LNS; RUSF, ready-to-use supplementary food; RUTF, ready-to-use therapeutic food; SAM, severe acute malnutrition; SQ-LNS, small-quantity LNS.
The SQ-LNS have several potential advantages within food assistance programs: (1) The quantity is small, and the product is ready-to-eat by itself or mixed with other foods, so the entire intended daily ration is easily consumed even by infants, ensuring that daily micronutrient needs will be met; (2) the small quantity minimizes transport and storage costs and reduces the likelihood of sharing the supplement with others, by comparison to larger quantity food aid products such as cereal–legume blends; and (3) the “ready-to-use” packaging reduces time spent preparing food. When necessary (eg, widespread food insecurity), provision of SQ-LNS for vulnerable target groups can be coupled with provision of staple foods or cash transfers for the entire household. In humanitarian emergencies, SQ-LNS added to a general food distribution has the potential to improve the nutrient adequacy of rations for infants, young children, and pregnant and lactating women, with the intention of preventing malnutrition in the emergency-affected population. 11
The potential of SQ-LNS to prevent different forms of undernutrition (ie, micronutrient insufficiency, wasting, and stunting) and improve health and nutrition status is still being explored. Through the International Lipid-Based Nutrient Supplements Project, several efficacy and effectiveness trials conducted in Ghana, Malawi, and Burkina Faso have assessed the impact of SQ-LNS on child growth and development. Taken as a whole, and including other studies, the evidence suggests that when SQ-LNS is provided to pregnant women as a supplement to a regular diet, it can promote fetal growth, 9 -11 and SQ-LNS for the mother and/or young child can promote child growth and development and reduce anemia. 12 -17 Although studies have shown heterogeneity in these effects both within and across study populations, this appears to result from contextual factors such as frequent infections and inflammation. 16,18,19
Two systematic reviews 6,20 and protocols describing 2 forthcoming systematic reviews 21,22 assessing the impacts of LNS have been published and address the following topics: (1) LNS of any dose following general formulation guidance to pregnant women, 20 (2) LNS of any dose or formulation to children 6 to 59 months of age with moderate acute malnutrition (MAM), 6 (3) LNS of 20 to 50 g/d (small-to-medium quantity) following general formulation guidance to infants and young children 6 to 23 months of age, 21 and (4) LNS generally providing 200 to 300 g/d to children 6 to 59 months of age with severe acute malnutrition (SAM). 22 These elucidate further (or promise to do so) the effects of LNS in various quantities and formulations on nutrition- and development-related outcomes in stable and emergency settings.
Using Different Protein Ingredients
There is ongoing discussion of whether milk protein is needed in SNFs for preventing and treating undernutrition because it is an expensive ingredient that almost always has to be imported into low-income countries. Indeed, the earliest treatments for undernutrition used reconstituted dried skim milk (DSM), but eventually the high cost was one of the reasons scientists and doctors started to investigate alternative mixtures using only plant-derived proteins. 23 The dialogue about protein is fueled by the fact that the optimal macronutrient and ingredient profile of foods for preventing and treating undernutrition, and sustaining recovery from it, has not been scientifically established.
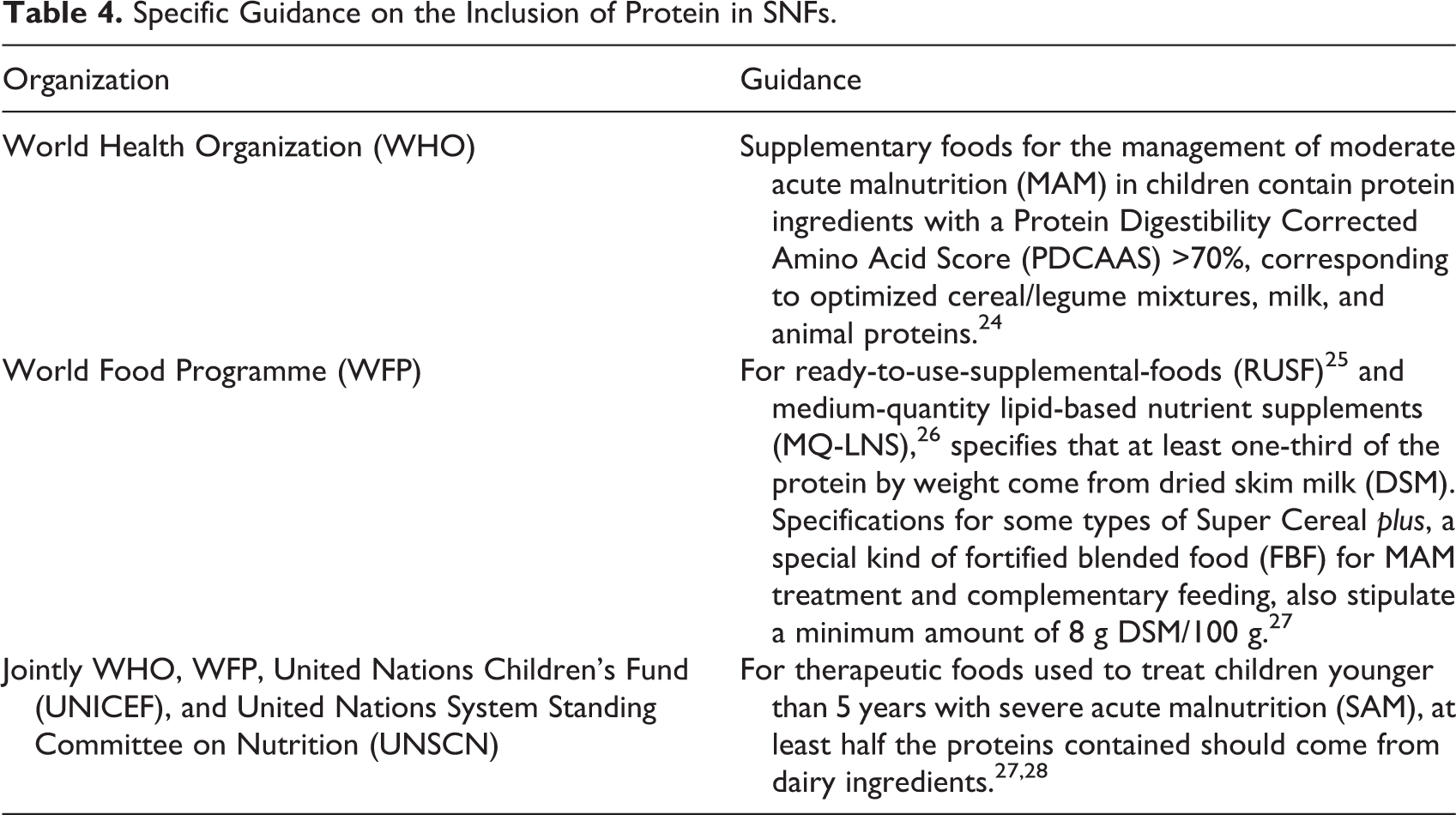
Based on the limited available evidence, normative bodies have provided guidance that generally advises the inclusion of milk ingredients in therapeutic and supplementary SNFs (Table 4). Guidance from the World Health Organization notes that “milk powder” improves the AA profile and bioavailability of calcium and potassium, has stimulating effect on insulin-like growth factor 1 and linear growth, and contains no antinutrients. 24 Other recent work suggests that the high lactose content of some milk products has potentially prebiotic effects and positive effects on growth. 29
Specific Guidance on the Inclusion of Protein in SNFs.
For the treatment of SAM, particularly in children younger than 24 months, lower cost ready-to-use therapeutic food (RUTF) recipes made without the addition of milk powder have been less effective than the standard peanut- and milk-based therapeutic foods. 30 However, this has been challenged with more recent, better-designed investigations. For example, in a 2-arm noninferiority study in Democratic Republic of the Congo, children with SAM were randomized to an RUTF made from a novel soy–maize–sorghum (SMS) formulation containing no milk, and a control group given the standard peanut-DSM formula with 25% DSM. 31 In 6- to 24-month-olds, the recovery rate from the nondairy SMS-RUTF recipe was inferior, but in children ≥24 months, it performed as well as the standard peanut-DSM-RUTF recipe in treating SAM. Following an analysis of plasma AA levels recorded before, during, and after treatment, the SMS-RUTF recipe was enhanced with specific AAs and tested in a second 3-arm randomized, controlled, noninferiority efficacy trial on children 6 to 23 and 24 to 59 months of age with SAM in Malawi. 32 There, children were randomized to receive treatment of 200 kcal/kg weight/d with 1 of 3 recipes: (1) standard peanut-DSM-RUTF formula with 25% DSM; (2) nondairy SMS-RUTF enriched with crystalline AAs; and (3) dairy SMS-RUTF with 9.3% DSM. The results of this trial showed that the AA-enhanced SMS-RUTF recipe was not inferior to the milk-based RUTFs in terms of recovery rates or length of stay in treatment and was superior in treating anemia and restoring body iron stores. Additionally, an increase in body iron stores was observed across the study group that was inversely proportional to the products’ milk powder content. The study’s authors reported that the ingredients for the AA-enhanced SMS-RUTF recipe cost nearly 30% less than the standard peanut-DSM-RUTF formula, which typically costs USD$48 to 69 for each child treated. 33
Recent intervention studies have also examined how milk-based supplemental foods affect weight gain and recovery from MAM. In a randomized intervention study, the effect of different amounts of DSM was examined among children 6 to 23 months old with MAM in Burkina Faso. 34 For 12 weeks, children were provided 500 kcal/d from LNS or corn–soy blend (CSB) containing different quantities of DSM and qualities of soy ingredients. There was no observed overall effect of milk, which authors hypothesize is because 94% of the children were breastfed. Treatment with LNS led to more fat-free tissue gained and higher recovery rates (69%) compared to treatment with CSB (62% recovery). Compared to CSB, LNS also resulted in greater weight gain when both contained soy isolate (rather than de-hulled soy). Another recent randomized study investigated the effects of different supplements among children 6 to 59 months old with MAM in Malawi. 35 For 12 weeks, children were given 75 kcal/kg weight/day milk protein-based ready-to-use supplementary food (RUSF) or soy protein-based RUSF. The milk protein-based product led to significantly greater recovery rates (84% vs 80.5%) and growth markers, but no differences were seen in length gain or time to recovery among those who recovered.
Overall, including dairy ingredients in food aid products intended for children has shown consistent benefits, but (1) these have not been fully characterized, (2) AA-enhanced non-milk SNF formulations promise equivalent nutritional outcomes, and (3) using lower quantities of milk seems desirable from a cost perspective.
What Is the Relative CE of Different Food Aid Products or Programs for Different Nutrition Outcomes?
As far as governance priorities go, public health interventions that achieve high impact at an acceptable cost to taxpayers and donors tend to rank highly. Given that food assistance operations must compete for limited resources, food aid products and programs should evolve with an eye toward demonstrating high impact at acceptable costs through CE evaluations. The CE information can also influence policy decisions around what program designs to implement. For instance, decision-makers who choose not to program a costly product would reconsider if the product were substantially more effective in achieving desired outcomes. In the same way, a slightly less effective product might be deemed appropriate if it were substantially less costly.
The existing evidence base for food assistance CE is sparse, mostly addressing the CE of recovery from SAM and MAM in community-based management of acute malnutrition programs. 36 -46 However, ongoing research led by the Food Aid Quality Review (FAQR) Project is currently investigating CE of multiple different food aid products (FBFs and LNS) in treating MAM and preventing stunting and wasting as components of supplementary feeding programs in Sierra Leone and Burkina Faso. The anticipated results of these studies will be a valuable tool for decision-makers, providing information on the comparative costs of employing different food aid interventions for specific nutrition outcomes.
However, this research has demonstrated that determining CE is not a simple task. To conduct the analyses, researchers need evidence on both costs and effectiveness of comparable outcomes. In the FAQR studies, effectiveness outcomes for each food will be linked with costs per child reached. These costs will consider comprehensive cost components of the overall program, including programmatic costs (eg, cost of the ration needed to achieve the outcome of interest, product transportation, office space for program administration, storage, losses, labor, and delivery costs associated with behavior change communication activities) as well as opportunity costs to program volunteers and caregivers. With this CE model, it quickly becomes clear that every aspect of programming carries importance. Product packaging, for instance, can have insufficient shelf-life and poor resistance to infestation that leads to losses and therefore costs; it can also affect the time required for distribution, influencing staffing costs.
How Can SNFs be Integrated Into Local Markets?
Food assistance provides recipients with access to food in multiple ways, and locally based approaches to preventing and treating undernutrition are considered by many to be a promising new strategy. 47,48 To that end, many studies have investigated the use of food products that are formulated and produced in a geographic area proximal to the recipient population. Linear programming tools help identify how local foods can be combined to create a product that meets international nutrition standards, and are affordable and palatable. 49 -53 In theory, “localized” strategies can benefit local economies by creating employment opportunities or generating income for ingredient suppliers. However, local production tends to be more expensive for various reasons (eg, smaller scale, higher cost of local ingredients, tax on imports, having to rely on international laboratories, less optimized use of equipment, etc), and whether local production translates into lower prices for consumers is yet to be determined.
Still, integrating locally produced SNFs into programming is viewed as a way to scale up demand and supply for both target recipients and self-purchasing consumers. The World Food Programme is investigating this strategy for making SNFs readily accessible on a sustained basis, for example, through 2 coordinated projects in Ghana: local food-based approaches to improve nutrition (LoFAIN), which is focusing on creating demand and increasing the availability of special nutritious foods for children 6 to 23 months old; and Enhanced Nutrition & Value Chains (ENVAC), which is supporting smallholder farmers and food processors to produce good quality SNFs. Rather than a question of supplies or costs, which often disfavor local production, the LoFAIN and ENVAC initiatives are geared toward development and developing local food industry capacity in the longer term, benefitting recipients, self-purchasing consumers, and the local economy. In these cases, the minimal conditions for local production and supply of SNFs are (1) adequate demand from intended consumers and (2) sufficient manufacturing capacity of local manufacturers to supply the market. For a sustainable supply chain to develop, there needs to be sustainable demand, which can be from both self-purchasing consumers and the public sector, whether the government, UN agencies, or nongovernmental organizations. To have sufficient demand, local manufacturers ideally have a portfolio of products for a range of consumer groups. Consumers must also like the product and be prepared to pay for it using their resources, including cash but also opportunity costs (ie, the effort they have to put into obtaining the product). Although markets are being developed, vouchers can be considered in contexts where manufacturers are already supplying suitable products on the market to meet a (growing) demand from different groups of consumers. Development of a government “seal” for nutritional quality and promotion of the “seal” among consumers can help guide their choice for nutritious foods.
Discussion
The lectures in this symposium were a call for further research in the areas of food aid formulations (total caloric delivery, macro- and micronutrient content and bioavailablity, and protein quality and source), CE, and market-based approaches for food security and nutrition programming especially in relation to RUF. If this call is heeded, the potential to feed millions more people and support healthy, well-nourished populations around the world is substantial.
The results of recent studies presented show that even modest supplementation from micronutrient-dense SQ-LNS can lead to positive nutrition outcomes of interest and may have compounding returns when provided to expectant mothers. Further research should assess the effectiveness (including CE) of different products when targeted to mothers and/or children in different contexts—especially those that integrate other strategies to reduce infections/inflammation and promote child development. Other recent research demonstrates that non-milk containing products, but with similar protein quality, have shown encouraging effects on nutritional outcomes with potential cost-savings. This has sparked increasing interest in the effect of protein quality and the need for specific AAs in malnourished children, as well as in optimizing AA quality by combining dairy and plant protein and cereals and legumes. Can adding specific AA (known to be important for growth) to foods with only plant protein match the effects of dairy on growth, development, body composition, and other outcomes in the short and long term? As has been urged by others, 54 the role of milk relative to soy, other plant-based proteins, and AAs merits further study, making sure that any evaluations of non-milk SNFs consider protein quality and antinutrient content.
The presentations also revealed that there is a dearth of published research on the CE of food aid products for short- and long-term nutrition-related outcomes, which poses a major limitation to efficient program funding. Building up the food aid CE literature will help untangle the debates on SNF ingredients and formulations, while also informing which food aid products can be used for which nutrition responses. Donors and governments should fund investigations into more cost-effective RUFs while acknowledging that these are massive research endeavors that require sufficient time and investment to ensure carefully designed studies, rigorous data collection, and appropriate statistical analysis.
Finally, this symposium demonstrated that market-based activities are being seriously investigated as a method for delivering food aid products more effectively, while also making them available for self-purchasing consumers. However, local production is more complex to manage and will need to be deployed from a long-term, development perspective. Monitoring these initiatives for consumer uptake, product acceptance and use, and impact on young children’s diets and nutrient intake will be very important. In addition, more work is needed to identify which nutrition-related outcomes can feasibly be assessed based on market-oriented approaches (eg, increased nutrient intake, specific biological impact). Complete market-based integration of SNFs will depend on certain enabling factors: Local production and commercial sale of SNFs should be encouraged; regulatory mechanisms must be established to ensure a high quality of food products; and local manufacturing will need to be supported with financial and technical investments. There is ample untapped opportunity to work with the private sector food industry to (1) create commercial products that are variations on food aid products, which meet consumer nutritional needs and preferences for taste and affordability; and (2) establish and monitor safety and quality standards, and establish appropriate supply chains. For the (local) food industry, collaborating with the public-sector benefits by improving the nutrition-related health of consumers while expanding market reach.
Conclusions
Overall, food aid represents a global public health good: Basic commodities reliably mitigate large-scale hunger, and SNFs support the survival and health of vulnerable groups. Research efforts have advanced the field substantially, ultimately helping donor states reduce their costs for achieving nutritional impacts—but there is much more to learn. This symposium presented new evidence on operationally relevant food aid research, emphasizing the need for governments, bilateral and multilateral organizations, foundations, research institutions, and the private sector to engage in research activities aimed at improving global nutrition and food security. Funding, directing, or performing research will build existing evidence bases and create others while informing better food security programming for global development and humanitarian response. New research should focus on the most nutritionally appropriate, effective, and cost-effective uses of SNFs (whether lipid-based or not), the efficacy and CE of milk and non-milk protein-based foods, the CE of different food assistance programming options, and the quantifiable public health benefits of linking commercial and food aid activities.
Footnotes
Authors’ Note
Kristine Caiafa, Patrick Webb, Bea Rogers, and Shelley Walton conceived, drafted, critically revised, and gave final approval for this manuscript. Kim Michaelsen, Steve Collins, Kathryn Dewey, Saskia de Pee and Tatyana El-Kour critically revised and gave final approval for the manuscript. All authors agree to be accountable for all aspects of work ensuring integrity and accuracy.
Acknowledgments
The symposium was organized by the Food Aid Quality Review (FAQR), housed at Tufts University’s Gerald J. and Dorothy R. Friedman School of Nutrition Science and Policy, on behalf of the US Agency for International Development’s (USAID) Office of Food for Peace. It was made possible by the generous support of the American people through the support of the USAID and the USAID Office of Food for Peace (FFP) of the Bureau for Democracy, Conflict and Humanitarian Assistance (DCHA), under the terms of Contract AID-OAA-C-16-00020, managed by Tufts University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: U.S. Agency for International Development (USAID) and the USAID Office of Food for Peace (FFP) of the Bureau for Democracy, Conflict and Humanitarian Assistance (DCHA) (AID-OAA-C-16-00020). Kim Michaelsen has received funding from USDEC, ARLA, and Danish Dairy Foundation for research projects including dairy products to malnourished children. Steve Collins, founder and chairman of VALID Nutrition—an Irish charity (CHY 17583), has received some financial and technical support from Ajinomoto Co Inc, as part of a larger collaboration (involving Irish Aid, JICA, and the PRANA Foundation) to develop a non-milk RUTF recipe. Beatrice Rogers has received funding from the US Agency for International Development grant AID-OAA-C-16-00020 to support cost-effectiveness research presented during the symposium. Patrick Webb has received funding from the US Agency for International Development grant AID-OAA-C-16-00020 to support Research Engagement on Food Interventions for Nutritional Effectiveness (REFINE) activities.
