Abstract
Background:
Ready-to-use therapeutic food (RUTF) has been found effective in treating severe acute malnutrition. Vietnam’s National Institute of Nutrition (NIN), the Institut de Recherche pour le Développement (IRD), and UNICEF collaborated to formulate a local RUTF called High-Energy Bar for Integrated Management of Acute Malnutrition (HEBI). RUTF might be useful to address malnutrition in HIV patients.
Objective:
To compare the acceptability of the local RUTF and an imported RUTF among malnourished people with HIV in Vietnam Methods: The acceptability of HEBI and Plumpy’Nut was studied among 80 HIV-positive children and 80 HIV-positive adults. In a crossover design, participants were randomly assigned to receive either Plumpy’Nut or HEBI for 2 weeks and were switched to the other product for the subsequent 2 weeks. A third (control) group of about 40 HIV-positive participants in each study was randomly assigned to receive no RUTF. Nurses took anthropometric measurements weekly, and the subjects or their caregivers monitored daily RUTF intake.
Results:
Children consumed 69% of HEBI and 65% of Plumpy’Nut (p = .13). Adults consumed 91% of HEBI and 81% of Plumpy’Nut (p = .059). Both children (p = .058) and adults (p ≤ .0001) preferred HEBI. Significant gains were observed in percent weight (p = .035), weight-for-age (p = .014), and body mass index (BMI)-for-age (p = .036) in children who received RUTF and in percent weight (p = .017) and BMI (p = .0048) in adults who received RUTF compared with the control groups.
Conclusions:
In this study in Vietnam, both HEBI and Plumpy’Nut were found acceptable by people with HIV.
Keywords
Introduction
Malnutrition contributes to faster progression of HIV to AIDS, and HIV leads to and exacerbates malnutrition. 1 In adults, asymptomatic HIV infection increases resting metabolic rate by approximately 10% and symptomatic HIV infection increases resting metabolic rate by 20% to 30%. 2 Hence, weight loss and malnutrition are common features of HIV, and low body mass index (BMI) at diagnosis of HIV is associated with a significant increase in the risk of mortality. 3 Severe weight loss is one of the strongest indicators associated with morbidity and reduced survival of people living with HIV (PLHIV), 4,5 regardless of antiretroviral therapy (ART). 6 The World Health Organization (WHO) recommends that PLHIV consume more energy than people without HIV to maintain weight, although micronutrient requirements are the same for people with and without HIV. 2
Vietnam has both high chronic undernutrition and increasing rates of HIV infection. In 2012, the prevalence of underweight was 16.2% in women aged 15 to 49 years and 16.2% in children under 5 years of age, and 26.7% of children under 5 years of age were stunted. 7 HIV prevalence was estimated at 0.45% in 2011 8 , but in a population of almost 89 million 9 , this percentage translates into hundreds of thousands of people. The epidemic is largely concentrated among intravenous drug users, sex workers, and men who have sex with men, along with their partners and clients, although prevalence is increasing in the general population. 8
Few interventions have studied the effect of macronutrient supplementation on weight gain 10 and disease progression in PLHIV. Such studies in developing countries are often difficult to compare because of different outcome parameters. Based on current evidence, both macronutrient and micronutrient interventions are likely to be beneficial. The potential health benefits of nutrition interventions include delaying the progression of disease, improving the outcome of opportunistic or concomitant infections, improving the effectiveness of ART, and reducing the side effects of medication. 4 Yet, two recent reviews 11,12 report no impact on most functional outcomes, such as morbidity or mortality, although. BMI was improved in some studies. Most research has been done in sub-Saharan Africa, where the underlying micronutrient deficiencies and nutritional status are likely to be different from those in Asia. Only one study from Thailand reported an improvement in survival in HIV-positive adults, especially in those with CD4 counts < 200. 13
Ready-to-use therapeutic foods (RUTFs) are edible, homogenized, energy-dense, bacteria-resistant, lipid-based foods with added vitamins and minerals that are widely used to treat people with severe acute malnutrition (SAM). These products have been studied extensively and validated as a viable intervention and treatment for SAM. 14 RUTF has been shown to improve the nutritional status of children living with HIV 15 and has been reported as easy to eat by adults with mouth sores or other HIV-related symptoms.
Although RUTF has been found to be a highly effective intervention to treat SAM, the acceptability of such products, and their use according to the prescribed regimen, can vary in populations with SAM. 16 From 2009 onwards, Vietnam’s National Institute of Nutrition (NIN), the Institut de Recherche pour le Développement (IRD), and UNICEF have collaborated to formulate a RUTF adapted to Vietnamese taste using locally available rice, soy, and mung beans with a vitamin and mineral premix. 17 The product was called the High-Energy Bar for Integrated Management of Acute Malnutrition (HEBI). Although HEBI and Plumpy’Nut differ in taste, appearance (HEBI is a soft bar, and Plumpy’Nut is a paste), and packaging, they are similar in energy density and vitamin and mineral content. HEBI also complies with all UNICEF RUTF product specifications, except for trace amounts of two heavy metals (Appendix Table 1). A 2010 study in Vietnam found that both HEBI and Plumpy’Nut were acceptable among HIV-negative, moderately malnourished, preschool-age children. Information was needed, however, on the acceptability of these RUTFs among children and adults with HIV before they could be used reliably with these populations and to develop guidelines and procurement planning for nutrition interventions for PLHIV. IRD and NIN therefore conducted a study to compare the acceptability of Plumpy’Nut and HEBI among 80 HIV-positive children in Hanoi and 80 HIV-positive adults in Ho Chi Minh City. Data were compiled on adherence to a take-home ration, organoleptic properties, and nutritional status separately in children and adults.
Methods
Study Design
Because adherence and preference may differ between children and adults, IRD and NIN conducted two studies simultaneously in 2011, one with children with HIV attending the National Pediatric Hospital in Hanoi and one with underweight adults with HIV attending the Hospital for Tropical Diseases in Ho Chi Minh City. Both studies were randomized crossover trials. Sample size was calculated based on adherence to a targeted 50% consumption of the RUTFs. A difference in adherence of 20 percentage points was regarded as important to detect. Given that the sample size to detect a difference of 20 percentage points in the proportion of subjects who consumed at least 50% of both RUTFs ranged from 49 to 93, a sample size of 80 was selected to balance statistical rigor with cost and feasibility.
For each study, a control group of 40 HIV-positive underweight children and 40 HIV-positive underweight adults was randomly assigned to receive no RUTF, with their anthropometric and morbidity status assessed at baseline and endline. In addition, 80 HIV-positive children and 80 HIV-positive adults were randomly assigned to the order in which they would receive the two RUTFs (HEBI first or Plumpy’Nut first).
Subjects
To be eligible for the study, children had to be confirmed HIV positive, between the ages of 3 and 7 years at the time of enrollment, living in an area accessible to home-based care teams, and with a weight-for-height z-score (WHZ) < 0 at the time of enrollment. A total of 204 children with HIV attending the National Pediatric Hospital in Hanoi were assessed for eligibility in the children’s study. Most children had acquired their HIV infection at birth. The National Pediatric Hospital is a tertiary health service, covering Hanoi and adjacent provinces.
Adult participants had to be confirmed HIV positive, more than 18 years old at the time of enrollment, living in an area accessible to home-based care teams, and with BMI < 20.0. Adult patients at the Hospital for Tropical Diseases in Ho Chi Minh City were initially told about the study during their checkups. Of the 2,572 patients screened, those who met the inclusion criteria had the study explained to them and, if they gave informed written consent, were invited to participate. Most patients lived in Ho Chi Min City, a large city with more than 8 million inhabitants.
Participation
When the participants enrolled in the study, a medical team in each hospital took medical histories and collected baseline weight, height, mid-upper-arm circumference (MUAC), and appetite data. The participants were asked to return to the hospital every week for monitoring, weighing, and MUAC measurement. Height was measured for adults only at enrollment and for children at enrollment and at the end of the study (4 weeks later). The medical teams recorded any side effects from the RUTF (rash, vomiting, diarrhea) and checked morbidity through a recall questionnaire administered during follow-up visits.
Food Products
Each week the children received one bar of HEBI (500 kcal) or one sachet of Plumpy’Nut (also 500 kcal) per day, and adults received two bars of HEBI (1,000 kcal) or two sachets of Plumpy’Nut (1,000 kcal) per day. The composition of HEBI has been described in detail earlier. 17 Each bar or sachet was labeled with a day of the week. Each adult participant or caregiver of a child participant was asked to use the RUTF on the day marked and to take any unused or unopened bars or sachets to the health facility on the next weekly follow-up visit. Participants or caregivers (parents) were given intake forms to take home to record the quantity of RUTF given, eaten, and wasted and/or spilled. The data on the intake forms were totaled over the 2-week period to calculate the percentage of the prescribed dose consumed. At the end of every 2 weeks, a nurse also asked the participant (or caregiver, if a child participant did not understand well) to rate the organoleptic properties of the RUTF. The possible ratings on each of the organoleptic properties (color, smell, taste, texture, ease of swallowing, difficulty of eating) were “dislike,” “so-so,” and “like” After the initial 2-week RUTF treatment, the subjects were crossed over to receive the other RUTF for 2 weeks.
Ethical Approval
The study was nonblinded because the local RUTF was in a bar form and the imported RUTF was in a paste form, making blinding impossible. The Scientific and Ethical Review board of the NIN approved the study.
Data Analysis
The data were analyzed with SAS statistical software, version 9.2. Child and adult data were analyzed separately. For participant baseline characteristics, measures of central tendency (for continuous variables) and spread (for categorical variables) were calculated separately for intervention and comparison groups and compared with each other using a Student’s t-test or chi-square test, as applicable. For children, weight-for-age z-score (WAZ), height-for-age z-score (HAZ), and BMI-for-age z-score (BMIZ) were calculated using the 2006 WHO growth standards and references. 18 Anthropometric changes after consumption of each of the RUTFs were calculated and compared with a paired t-test. Anthropometric changes during the entire study period were compared for the intervention group and comparison group with ANOVA. The participants’ organoleptic assessments of the two products were compared with the Wilcoxon signed-rank test.
Results
Children
The baseline characteristics of the child participants were not statistically different from those of the children in the comparison group (Table 1). A little more than one-half of the children in each group were moderately malnourished, and most were receiving ART.
Baseline Characteristics of Child Participants.
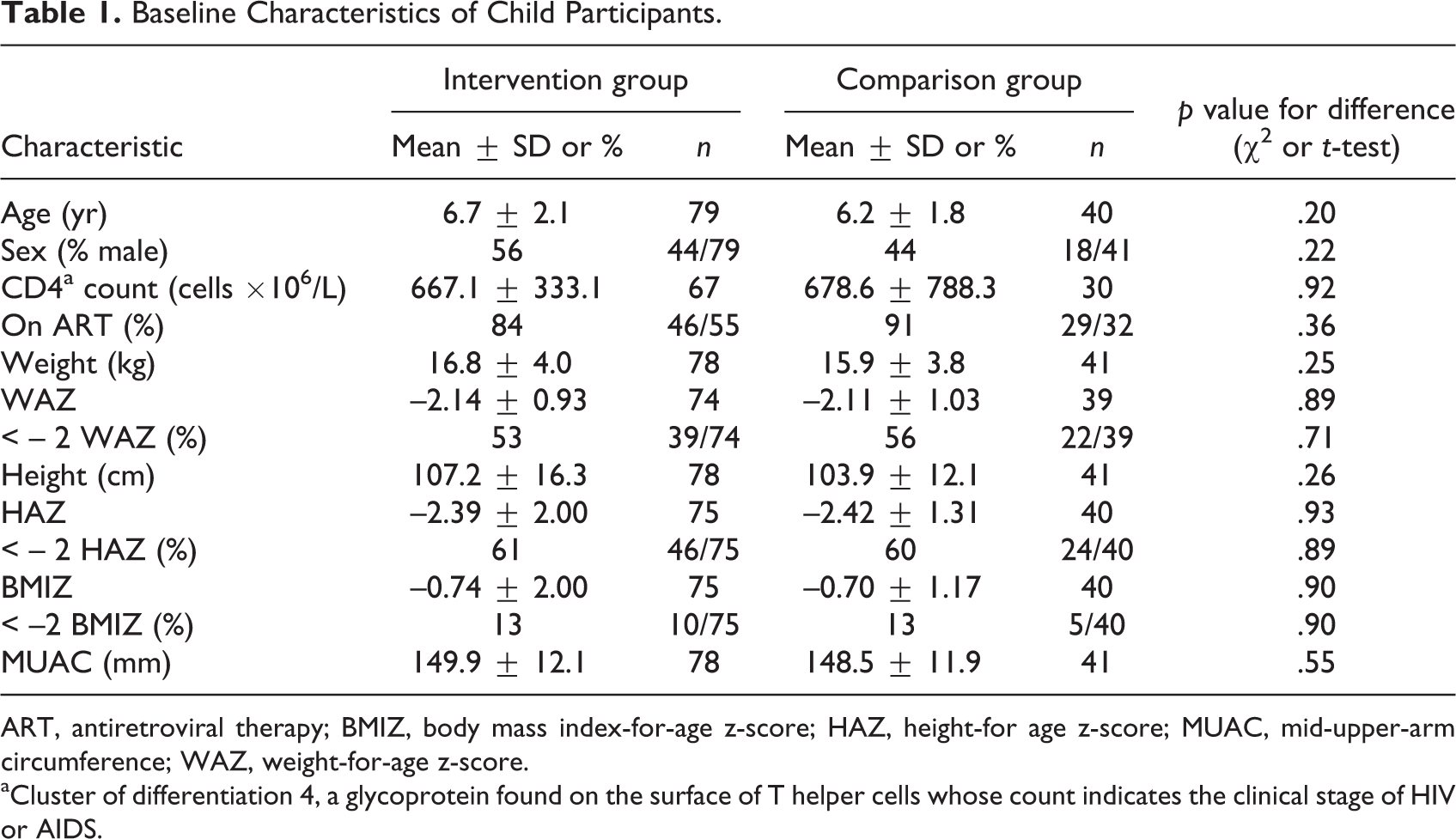
ART, antiretroviral therapy; BMIZ, body mass index-for-age z-score; HAZ, height-for age z-score; MUAC, mid-upper-arm circumference; WAZ, weight-for-age z-score.
aCluster of differentiation 4, a glycoprotein found on the surface of T helper cells whose count indicates the clinical stage of HIV or AIDS.
On average, the children consumed 69% of the HEBI and 65% of the Plumpy’Nut provided in each 2-week period (Appendix Table 2). Both levels were higher than the 50% defined as acceptable. The percent consumption of HEBI did not differ statistically from that of Plumpy’Nut (p = .13). During the second week of the take-home regimen, the children consumed an average of 4.8 servings of HEBI and 4.4 servings of Plumpy’Nut per week (p = .066).
Anthropometric Changes in Child Participants After Consuming Each of the Two RUTFs for 2 Weeks.a
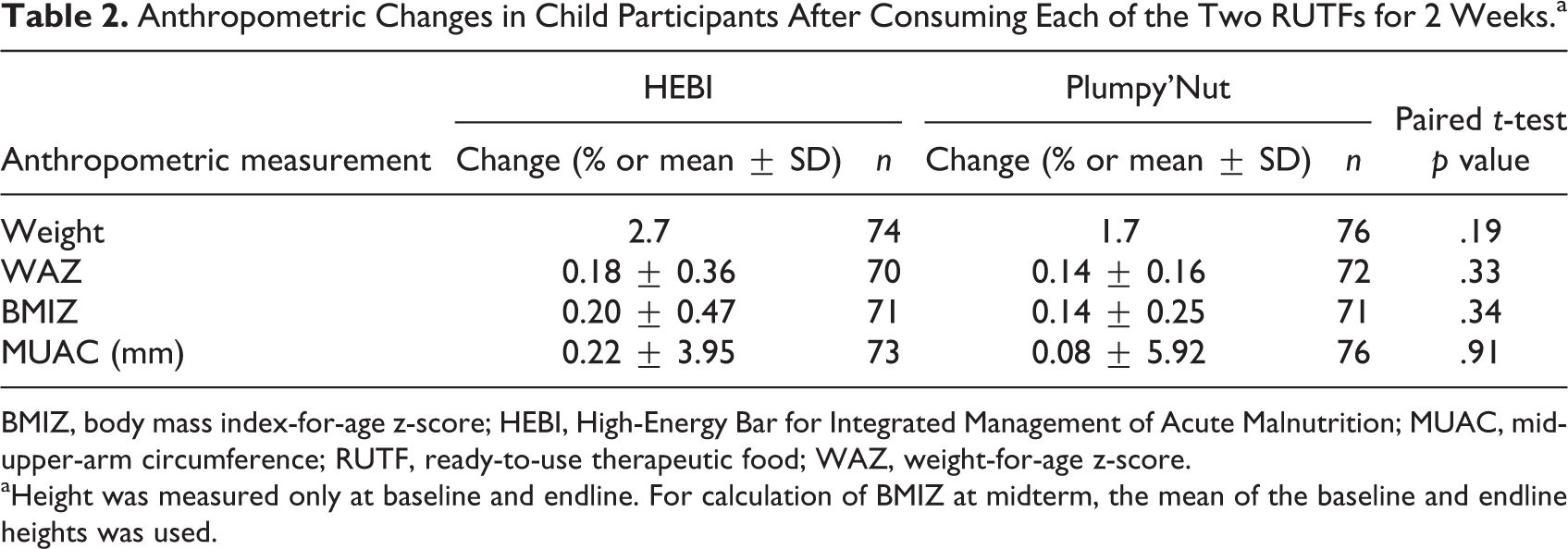
BMIZ, body mass index-for-age z-score; HEBI, High-Energy Bar for Integrated Management of Acute Malnutrition; MUAC, mid-upper-arm circumference; RUTF, ready-to-use therapeutic food; WAZ, weight-for-age z-score.
aHeight was measured only at baseline and endline. For calculation of BMIZ at midterm, the mean of the baseline and endline heights was used.
There were no statistically significant differences between the children’s assessments of the organoleptic properties of both RUTFs. The most frequent rating for each property on a scale of 1 to 3 was 2 (“so-so”). The 51% of children who indicated a preference tended to prefer HEBI (65%) over Plumpy’Nut (35%) (p = .058).
After 4 weeks of consuming the RUTFs, there were no statistically significant differences in anthropometric measurements in the children between the 2 weeks when they consumed HEBI and the 2 weeks when they consumed Plumpy’Nut (Table 2).
On the other hand, the children who consumed the two RUTFs over a 4-week period had significantly greater gains in WAZ (0.32 vs. 0.14, p = .014) and BMIZ (0.35 vs. 0.16, p = .036) than the children who did not receive any RUTF, as well as a significantly greater percentage weight gain (4.4% vs. 2.0%, p = .035) (Table 3).
Anthropometric Changes in Child Participants Over the 4-Week Duration of the Study.
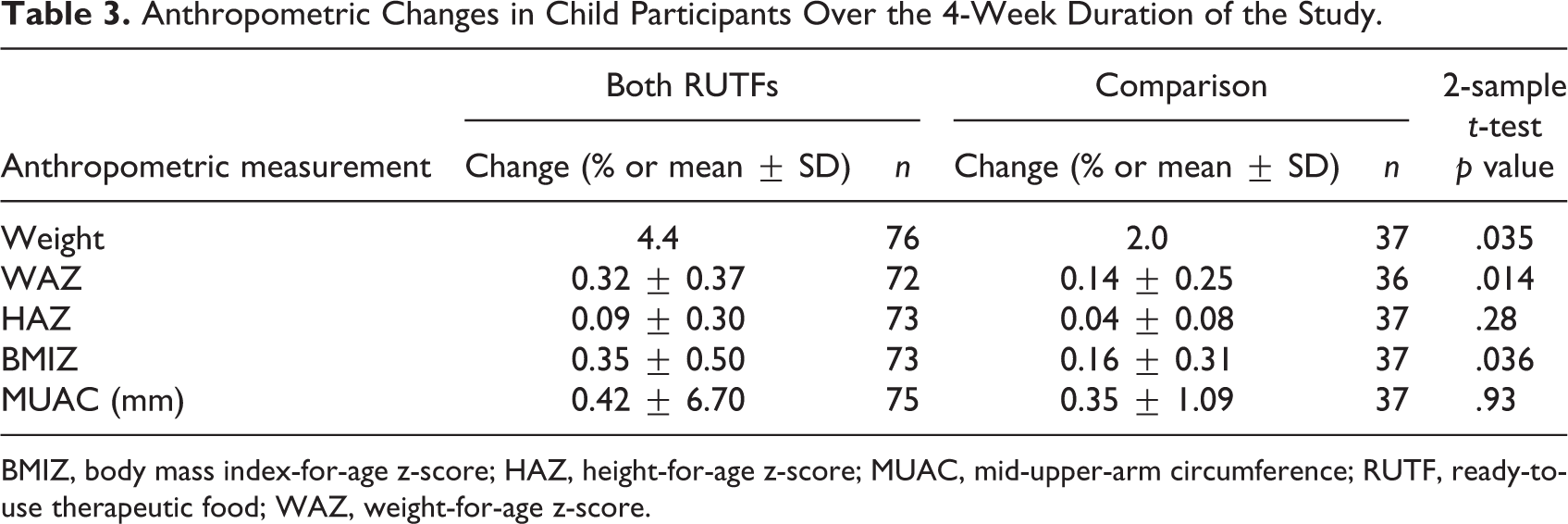
BMIZ, body mass index-for-age z-score; HAZ, height-for-age z-score; MUAC, mid-upper-arm circumference; RUTF, ready-to-use therapeutic food; WAZ, weight-for-age z-score.
Adult Study
The baseline characteristics of the adults in the intervention group also did not differ significantly from those of the adults in the comparison group (Table 4). Almost two-thirds of the participants in each group were male, and all adult participants were receiving ART. Most of the adult participants had BMI < 18.5, and a little more than one-half had MUAC < 230 mm.
Baseline Characteristics of Adult Participants.
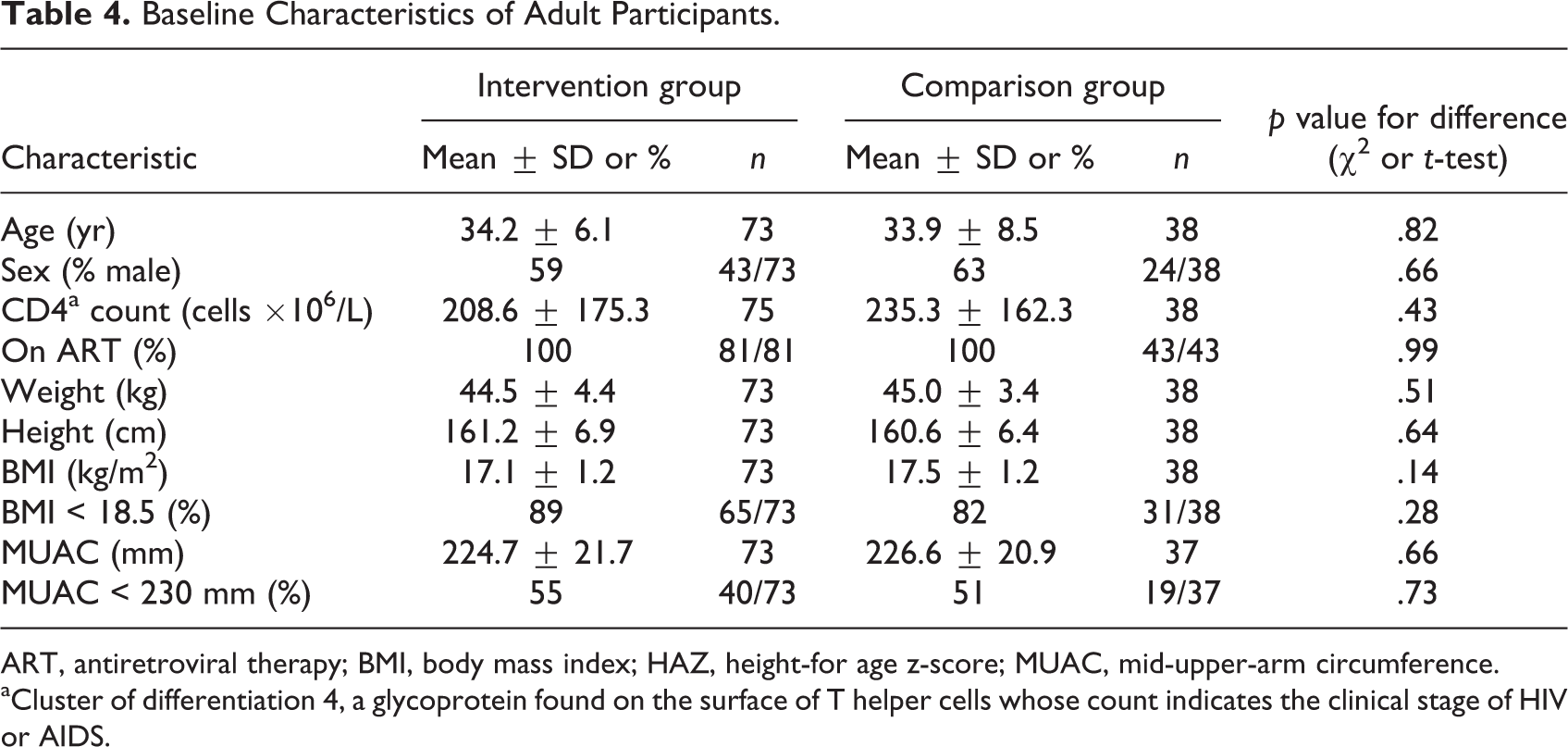
ART, antiretroviral therapy; BMI, body mass index; HAZ, height-for age z-score; MUAC, mid-upper-arm circumference.
aCluster of differentiation 4, a glycoprotein found on the surface of T helper cells whose count indicates the clinical stage of HIV or AIDS.
On average, the adult participants consumed 91% of the HEBI they were provided and 81% of the Plumpy’Nut, a difference that was marginally statistically significant (p = .059) (Appendix Table 3). An interesting trend was noted in consumption of the RUTF from the first week of the regimen to the second week. While the mean number of servings of HEBI consumed tended to increase (nonsignificantly) from week 1 (12.3 servings) to week 2 (12.7) (p = .88), the mean number of servings of Plumpy’Nut consumed decreased significantly from week 1 (11.6 servings) to week 2 (11.0 servings) (p = .019). The difference between the number of servings of HEBI and the number of servings of Plumpy’Nut consumed in week 1 showed a trend toward significance (p = .095), but the difference between the number of servings of the two RUTFs consumed in week 2 became more significant (p = .031).
Most of the adults’ ratings of the organoleptic properties of each RUTF were similar across RUTFs. The only statistically significant difference was that they rated the texture of HEBI more often as “liked” and that of Plumpy’Nut more often as “disliked” (p = .0067, Wilcoxon signed-rank test). They rated the taste of HEBI more often as “liked” and less often as “disliked” than the taste of Plumpy’Nut, but this difference was only marginally significant (p = .086). Most adults expressed an overall preference for HEBI (79%) over Plumpy’Nut (21%) (p ≤ .0001).
Like the children, the adults who consumed HEBI showed no statistically significant differences in anthropometric measurements from those who consumed Plumpy’Nut after 2 weeks of consumption of each RUTF (Table 5).
Anthropometric Changes in Adult Participants After Consuming Each of the Two RUTFs for 2 Weeks.a
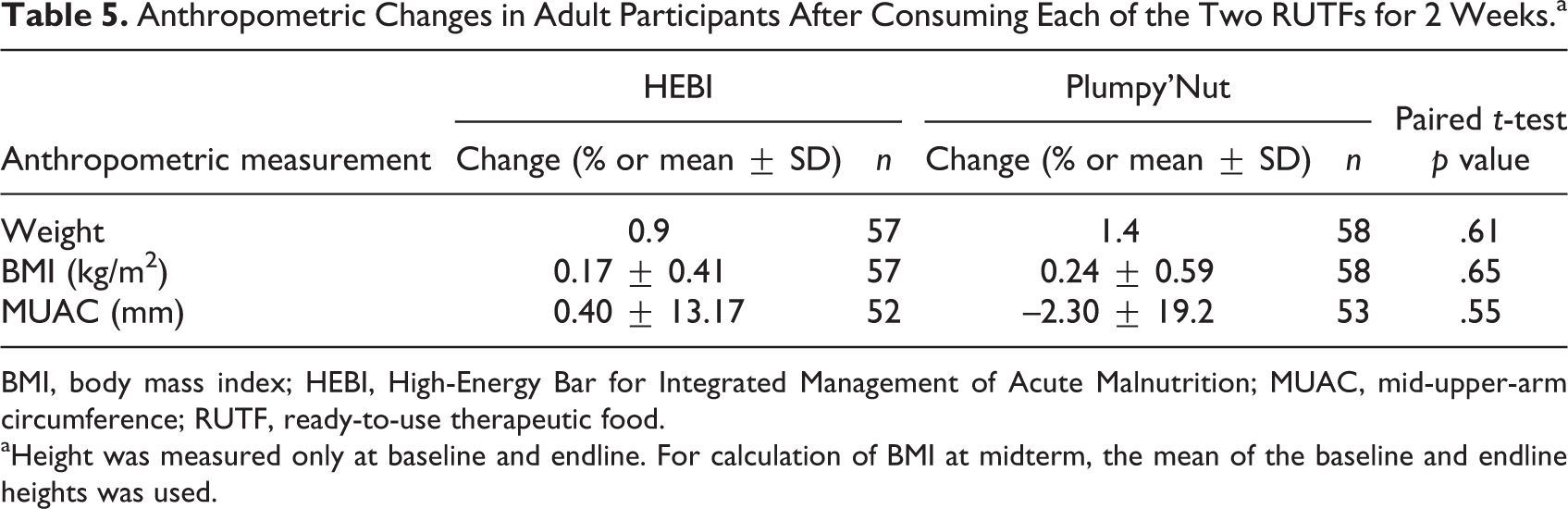
BMI, body mass index; HEBI, High-Energy Bar for Integrated Management of Acute Malnutrition; MUAC, mid-upper-arm circumference; RUTF, ready-to-use therapeutic food.
aHeight was measured only at baseline and endline. For calculation of BMI at midterm, the mean of the baseline and endline heights was used.
There were statistically significant differences, however, in percent weight gain and BMI gain between the adults who consumed the two RUTFs and those who did not consume any RUTF (Table 6). Those who consumed the RUTFs gained an average of 2.3% of their initial weight during the 4 weeks, while the comparison group gained an average of 0.5% (p = .017). This weight gain translated into a significant difference in BMI change but not a significant difference in MUAC change.
Anthropometric Changes in Adult Participants Over the 4-week Duration of the Study.

BMI, body mass index; MUAC, mid-upper-arm circumference; RUTF, ready-to-use therapeutic food.
Discussion
Both the HIV-positive children and the HIV-positive adults in the study consumed more than the 50% of the 2-week take-home regimens of both HEBI and Plumpy’Nut considered acceptable. Children consumed 69% of the HEBI and 65% of the Plumpy’Nut, and only one-half of those who participated in the trial expressed a preference for one product over the other. Of those who did indicate a preference, two-thirds preferred HEBI. The children rated the organoleptic properties of both products similarly. Adults consumed 91% of the HEBI and 81% of the Plumpy’Nut doses, showed a clearer preference than the children for HEBI, and consumed more of it than of the Plumpy’Nut. They tended to rate the organoleptic properties of the two products similarly, except for a significantly greater preference for the texture of HEBI.
The study did not, and was not powered to, find a difference between the two products on anthropometric status. It did, however, show that a 2-week regimen of one RUTF followed by a 2-week regimen of the other RUTF had a statistically significant effect on weight and BMI gain in both children and adults compared with participants who did not receive RUTF.
The study had several limitations. First, because of the different packaging and structure of the products, the participants were not blinded to the treatment they received during each 2-week period. In addition, the Plumpy’Nut was labeled in French and English, according to UNICEF requirements, whereas the local RUTF was labeled in English and Vietnamese. This may have introduced a bias, positive or negative, in participants’ comparative assessment of the products.
A second limitation of the children’s study is that not all of the children were receiving ART, and their anthropometric response to RUTF and assessment of the organoleptic properties of the RUTFs might have differed significantly based on whether or not they were receiving treatment for HIV. However, as the study had a crossover design, and there were no baseline differences between the two crossover groups (data not shown), all subjects consumed both products, minimizing the potential bias in comparing results across products.
Conclusions
Both HEBI and Plumpy’Nut were accepted by the sample of children and adults living with HIV in Vietnam in this study, although both groups preferred HEBI. The producer of HEBI, NINFOOD, is obtaining UNICEF certification to enable international organizations to procure and use the product in their programs. In 2012, UNICEF conducted a General Manufacturing Practices inspection at NINFOOD and accepted the use of the product for the domestic market. Therefore, the Ministry of Health of Vietnam is currently using the product in the national malnutrition program. Plumpy’Nut could be used until the production of HEBI increases to cover the demand, provided that patients and medical providers receive appropriate information and education on its intended purpose and appropriate use.
Footnotes
Acknowledgments
The authors would like to thank the Clinton Health Access Initiative and NIN for donating RUTF; Roger Mathisen of UNICEF/Vietnam for laying the groundwork for the development of HEBI; and Dr. Quang Minh Vo and Dr. Chau Van Vinh Nguyen at the Hospital for Tropical Diseases in Ho Chi Minh City.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Dr. Hop Thi Le, former Director of Vietnam’s National Institute of Nutrition (NIN).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
