Abstract
Background:
Injuries to the deep medial collateral ligament (dMCL) and partial superficial MCL (psMCL) can cause anteromedial rotatory instability; however, the contribution of each these injuries in restraining anteromedial rotatory instability and the effect on the anterior cruciate ligament (ACL) load remain unknown.
Purpose:
To investigate the contributions of the different MCL structures in restraining tibiofemoral motion and to evaluate the load through the ACL after MCL injury, especially after combined dMCL/psMCL injury.
Study Design:
Controlled laboratory study.
Methods:
Sixteen fresh-frozen human cadaveric knees were tested using a 6 degrees of freedom robotic simulator. Tibiofemoral kinematic parameters were recorded at 0°, 30°, 60°, and 90° of knee flexion for the following measurements: 8-N·m valgus rotation, 4-N·m external tibial rotation (ER), 4-N·m internal tibial rotation, and a combined 89-N anterior tibial translation and 4-N·m ER for both anteromedial rotation (AMR) and anteromedial translation (AMT). The kinematic parameters of the 3 different MCL injuries (dMCL; dMCL/psMCL; dMCL/superficial MCL (sMCL)) were recorded and reapplied either in an ACL-deficient joint (load sharing) or before and after cutting the ACL (ACL load). The loads were calculated by applying the principle of superposition.
Results:
The dMCL had the largest effect on reducing the force/torque during ER, AMR, and AMT in extension and the psMCL injury at 30° to 90° of knee flexion (P < .05). In a comparison of the load through the ACL when the MCL was intact, the ACL load increased by 46% and 127% after dMCL injury and combined dMCL/psMCL injury, respectively, at 30° of knee flexion during ER. In valgus rotation, a significant increase in ACL load was seen only at 90° of knee flexion.
Conclusion:
The psMCL injury made the largest contribution to the reduction of net force/torque during AMR/AMT at 30° to 90° of flexion. Concomitant dMCL/psMCL injury increased the ACL load, mainly during ER.
Clinical Relevance:
If a surgical procedure is being considered to treat anteromedial rotatory instability, then the procedure should focus on restoring sMCL function, as injury to this structure causes a major loss of the knee joint’s capacity to restrain AMR/AMT.
Keywords
A medial collateral ligament (MCL) injury can cause valgus and anteromedial rotatory instability (AMRI) as well as changes in external rotation (ER) and internal rotation (IR).2,4,9,16,32,35 During injury, the deep MCL (dMCL) may rupture earlier than the superficial MCL (sMCL) due to the lower strength and shorter length of the dMCL.18,28,35,36 A dMCL injury alone can cause an increase in AMRI without a clinically noticeable increase in valgus rotation (VR).4,9,35 However, this increase in AMRI caused by isolated dMCL may be difficult to discern clinically. Previous work in our laboratory showed that a combined dMCL and partial sMCL (psMCL) injury should be suspected when an increase in both AMRI and VR is present. 4 A psMCL injury can be identified if there is a 1- to 3-mm side-to-side difference in medial gapping on valgus stress radiographs. 17 Due to the frequency of combined dMCL/psMCL injury,21,38 investigation of the biomechanical behavior of this injury pattern is clinically relevant.
Ball et al 2 investigated the way in which the medial structures act to restrain AMRI and found load sharing between the sMCL and dMCL. Those investigators found that the dMCL was most important for restraining ER close to extension and the sMCL was most important in flexion. Previous work in our laboratory showed that a psMCL injury influenced ER and anteromedial rotation (AMR) more than a complete dMCL injury did at 30° to 90° of knee flexion. 4 However, measuring increases in laxity after each MCL injury stage might favor the structure that is cut last. Evaluating the force/torque contributions of the sMCL toward resisting knee motion, even when it is partially injured, could therefore potentially reinforce previous findings.
The MCL and the anterior cruciate ligament (ACL) both work to prevent knee translations/rotations (anterior translation, ER, and IR) when a force/torque is applied to the joint during activities; thus, these ligaments are load sharing.3,31 Therefore, injury to the MCL will result in an increase in the load transmitted through the ACL and other associated structures.3,31 Shapiro et al 31 investigated the load transfer to the ACL during anteromedial translation (AMT) and found an increase in ACL load after a combined dMCL/sMCL injury, compared with the intact knee. More recently, clinical studies have shown that persistent, unaddressed MCL laxity is a risk factor for ACL graft failure.1,33 Following these newer insights, research to better understand the cause and effect of AMRI and possible solutions to AMRI is regaining interest.2,4,5,8,14,16,24,32,35 Akin to how lateral extra-articular procedures reduce the load on the ACL graft during a combined IR and anterior tibial translation (ATT), 22 reducing AMRI might reduce the load on the ACL graft and thereby reduce ACL graft failure rates. New MCL reconstruction techniques are being proposed to better control AMRI in cases of complete dMCL/sMCL injury.5,7,24 The increase of AMRI and subsequent ACL load transfer in partial MCL injuries, however, is of particular interest, as they are often treated nonoperatively.15,19 It remains to be determined whether a medial extra-articular procedure, designed to reduce AMRI, will reduce ACL reconstruction (ACLR) failure rate. Although residual MCL laxity after nonoperative management of MCL injury is impossible to truly replicate in an in vitro setting, a psMCL injury might be the closest to this clinically relevant situation.
The purpose of this study was to investigate the contributions of the dMCL, psMCL, and sMCL to the forces/torques restraining tibiofemoral motion. Furthermore, this study aimed to quantify the ACL load increase after a combined dMCL/psMCL injury. The main hypothesis was that the relative joint reaction force/torque resisting AMRI and VR would decrease more after a high-grade psMCL injury than after a dMCL injury. Furthermore, we anticipated that the loads through the ACL would increase after a dMCL injury during AMR/AMT, but not during VR, and that after combined dMCL/psMCL injury, the loads would increase in both AMR/AMT and VR.
Methods
Sixteen fresh-frozen human cadaveric knee specimens without history of injury, surgery, osteoarthritis, or meniscal pathology were used for this study. Two protocols were used, with 8 specimens in each. In protocol 1, the mean donor age was 60 years (age range, 43-72 years), and mean body mass index was 25 (0 pairs; 5 male, 3 female). In protocol 2, the mean donor age was 60 years (age range, 41-70 years), and mean body mass index was 27 (1 pair; 5 male, 3 female). This study was approved by the institutional research ethics board.
Apparatus and Specimen Preparation
Biomechanical testing was performed with a 6 degrees of freedom joint motion simulator (VIVO; Advanced Mechanical Technologies). Before use, the fresh-frozen specimens were thawed at room temperature over a 24-hour period. The soft tissues were denuded from the proximal femur and distal tibia and fibula, while all tissues within 150 mm of the transepicondylar axis were left intact. The potting and mounting technique we used has been previously described.26,34 Computed tomographic images were used to create 3-dimensional surface models of each specimen’s femur, tibia, and fibula. These were coregistered to the specimens with the help of 2 optical motion trackers (Optotrak; Northern Digital) fixed to the femur and tibia. The femur was positioned on the joint motion simulator using a model-guided, motion capture–aided alignment technique. 26 To minimize unwanted secondary motions, we oriented the knee flexion axis concentric with the mechanical flexion axis of the machine and centered the joint mediolaterally. With the knee in extension, a purely compressive load (100 N) was applied across the joint parallel to the long axis of the tibia while all other degrees of freedom were unconstrained. The resulting pose was defined as the reference pose; all joint kinematic parameters were measured relative to this pose by the joint motion simulator using Grood and Suntay coordinate conventions. 10
Biomechanical Testing
Before data collection and to reduce tissue hysteresis, we preconditioned each specimen by repeating the entire loading sequence for 5 test cycles. Tibiofemoral kinematic parameters were measured with the joint flexion angle fixed at 0°, 30°, 60°, and 90° under a compression load of 10 N. Valgus laxity was measured using an 8-N·m VR torque. ER and IR laxity were measured using a 4-N·m rotation torque. Anteromedial laxity was measured after first applying an anterior-directed force (89 N) and then adding an ER torque (4 N·m) to the tibia. The maximal AMT referencing the center of the joint and the maximal tibial ER (AMR) were recorded. The forces used were in line with the forces used in previous studies investigating medial knee biomechanics.2,9,29,35 Specimen-specific kinematic measurements derived from the laxity tests were recorded at select dissection stages (accuracy of 0.1 mm and 0.1°) such that they could be reapplied to the joints as prescribed displacements/rotations to measure changes in joint reaction forces/torques and soft tissue contributions via the principle of superposition.
Dissection Sequences
Two distinct dissection protocols were used (Figure 1). Protocol 1 was used to determine the load sharing between the different MCL structures in the ACL-deficient knee by measuring how much the joint reaction force/torque decreased at each dissection stage, under prescribed (ACL-deficient) kinematics. Protocol 2 was used to quantify the loads transferred to the ACL after sequential MCL cuts.
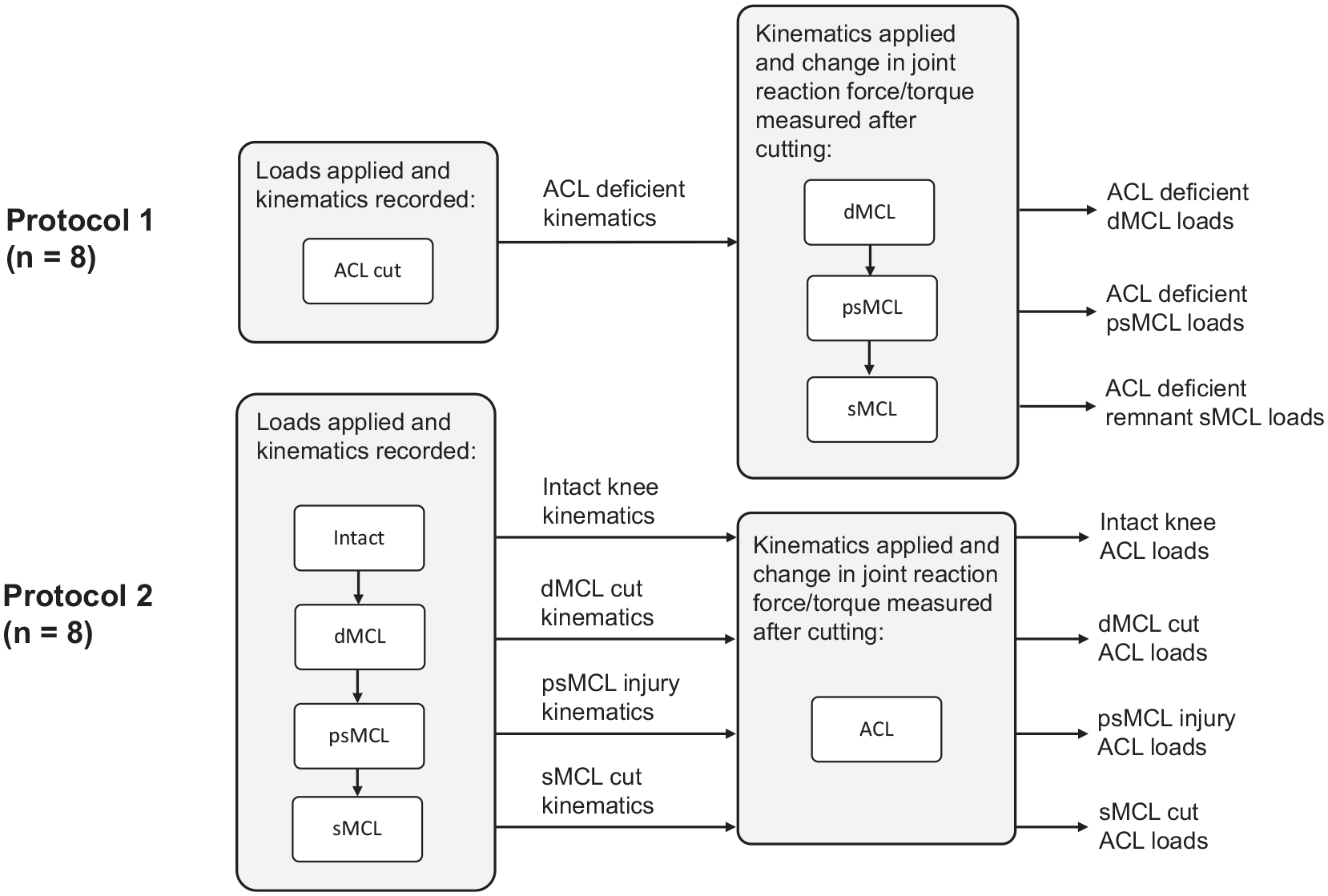
Data collection from both protocols: protocol 1 to determine the load sharing between the different medial collateral ligament (MCL) structures and protocol 2 to quantify the anterior cruciate ligament (ACL) load transfer after MCL injury. dMCL, deep MCL; psMCL, partial superficial MCL; sMCL, superficial MCL.
During the dissections, the specimens remained mounted on the joint motion simulator. The exception was dissection and cutting of the ACL, as this process required removal of the specimen to permit the transpatellar approach that was used for subperiosteal sharp dissection of the ACL from its femoral attachment. 23 The fixtures used were indexed to allow the specimen to be removed and reinstalled at the same position on the machine. Therefore, any changes in load were due to the (intentional) surgical alterations of the joint, which we wanted to measure. The dMCL was approached by incising the anteromedial fascia anterior to the sMCL at the level of the joint.18,35 A tissue plane was developed between the sMCL and dMCL. The dMCL cut was made by cutting the meniscofemoral and meniscotibial divisions of the dMCL.18,35 For the psMCL injury, a 10-N·m VR was applied with the knee at 30° of flexion. Two coordinates, 10 mm proximal and distal to the joint line, were defined using the motion capture system to measure the opening of the medial compartment. The sMCL was “pie-crusted” with an 18-gauge needle on its femoral side until the inducible medial compartment opening increased by 2.0 ± 0.5 mm. 3 The 2.0 ± 0.5–mm medial compartment opening represented a partial injury of the sMCL, as a full sMCL injury would cause a 2.8-mm increase in medial opening under 10-Nm VR torque. 17 To create the full sMCL injury, we completely transected the sMCL below the medial joint line, proximal to the proximal tibial sMCL insertion. During the dissection, care was taken to not damage the anterior arm of the posterior oblique ligament (POL). Injury to the POL and posteromedial joint capsule can cause posteromedial instability26,29 and therefore lies outside the scope of this study. All surgical procedures were conducted by the same trained orthopaedic fellows (T.V. and W.B.) supervised by the senior fellowship-trained orthopaedic surgeon (A.G.) to reduce interobserver error.
For specimens in protocol 1, we recorded the motions of the ACL-deficient joints in response to the applied loads and the resulting laxity limits reached. The motions were then precisely reapplied to the joints, and the forces/torques required to reach the same positions were recorded. These same ACL-deficient motions were applied at each MCL dissection stage, and the changes in the force/torque required were recorded. For specimens in protocol 2, the principle of superposition was used to measure the forces/torques transmitted through the ACL, using the joint load changes in force/torque. The laxity limits of the joints were determined during the first 4 cutting stages (intact, dMCL, psMCL, and sMCL), such that unique motion profiles were associated with each stage. With the ACL still intact, the joints were driven to those same positions (motions prescribed), and the forces/torques required were recorded. The ACL was then cut, the same motions were prescribed, and the new forces/torques were recorded (Figure 1). The loads through the ACL at each of those 4 cutting stages were therefore just the differences in the joint forces/torques between the paired sets of measurements for each motion. The joint-level loads were used because specific tissue-level loads require more assumptions and therefore are more prone to potential errors. During the reapplication of motions, the joint motion simulator operated in displacement control with an accuracy of ±0.1 mm in translation and ±0.1° in rotation. The joint reaction forces were measured at 500 samples per second through a loadcell located in the tibial actuator. Each test was repeated sequentially for a total of 5 cycles. The mean of the second, third, and fourth tests was used as the final output. Data from the first and fifth cycles were not used, in order to limit possible beginning- and end-of-cycle behavior effects.
Statistical Analysis
Statistical analysis was performed using commercial software (GraphPad Prism 9; Dotmatics).
A 2-factor (flexion angle and cutting state) repeated-measures analysis of variance was performed to compare the applied force/torque in each MCL cut state between the different states. A post hoc Bonferroni correction was used to correct for multiple comparisons.
To evaluate the change in force/torque through the ACL, we performed a 2-factor (flexion angle and cutting state) repeated-measures analysis of variance to compare each state with all of the previous states. A post hoc Bonferroni correction was used to correct for multiple comparisons.
A post hoc power analysis, based on the ACL load data for AMR, found that a 0.2-N·m change in ACL load could be detected (after Bonferroni correction) with a power of 80% and 95% confidence using 8 specimens. Statistical significance was set at P < .05 (adjusted P value after Bonferroni correction) for all analyses.
Results
The medial compartment opening increased by 2.0 ± 0.1 mm and 1.9 ± 0.2 mm after the psMCL injury in protocols 1 and 2, respectively.
Anteromedial Rotation
The torque required to reach the AMR limit that was established for the ACL-deficient knee decreased significantly after subsequent progressive release of the medial structures (dMCL and then psMCL followed by complete sMCL). This was true for all knee positions when compared with the ACL-deficient state. In extension, the largest reduction in torque, compared with the previous state, was seen after the dMCL cut. At 30° to 90° of knee flexion, the largest reduction in torque was seen after the psMCL injury. Cutting the remnant of the sMCL caused a significant reduction in torque compared with the dMCL cut state, but not the previous psMCL state, in knee flexion (Figure 2A).
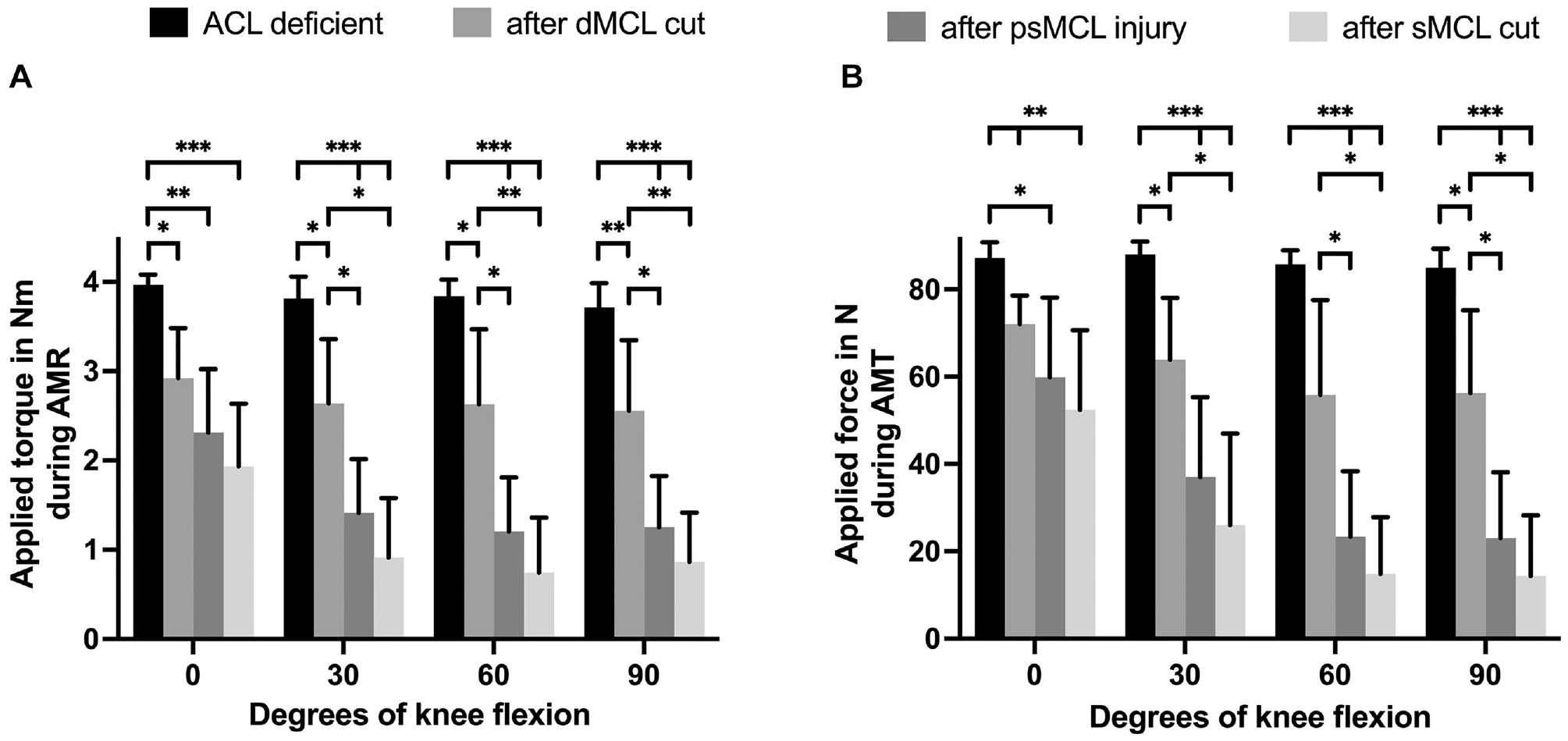
Data from protocol 1 showing the reduction in joint reaction torque during (A) anteromedial rotation (AMR) and (B) anteromedial translation (AMT) after progressive cutting of the different medial collateral ligament (MCL) structures in an anterior cruciate ligament (ACL)–deficient knee for each knee flexion angle. Results are expressed as mean ± SD. Statistically significant differences are indicated (*P < .05; **P < .01; ***P < .001). dMCL, deep MCL; psMCL, partial superficial MCL; sMCL, superficial MCL.
The changes in mean torque through the ACL during AMR, at each flexion angle after each MCL cutting stage, were not statistically significant (Figure 3A).
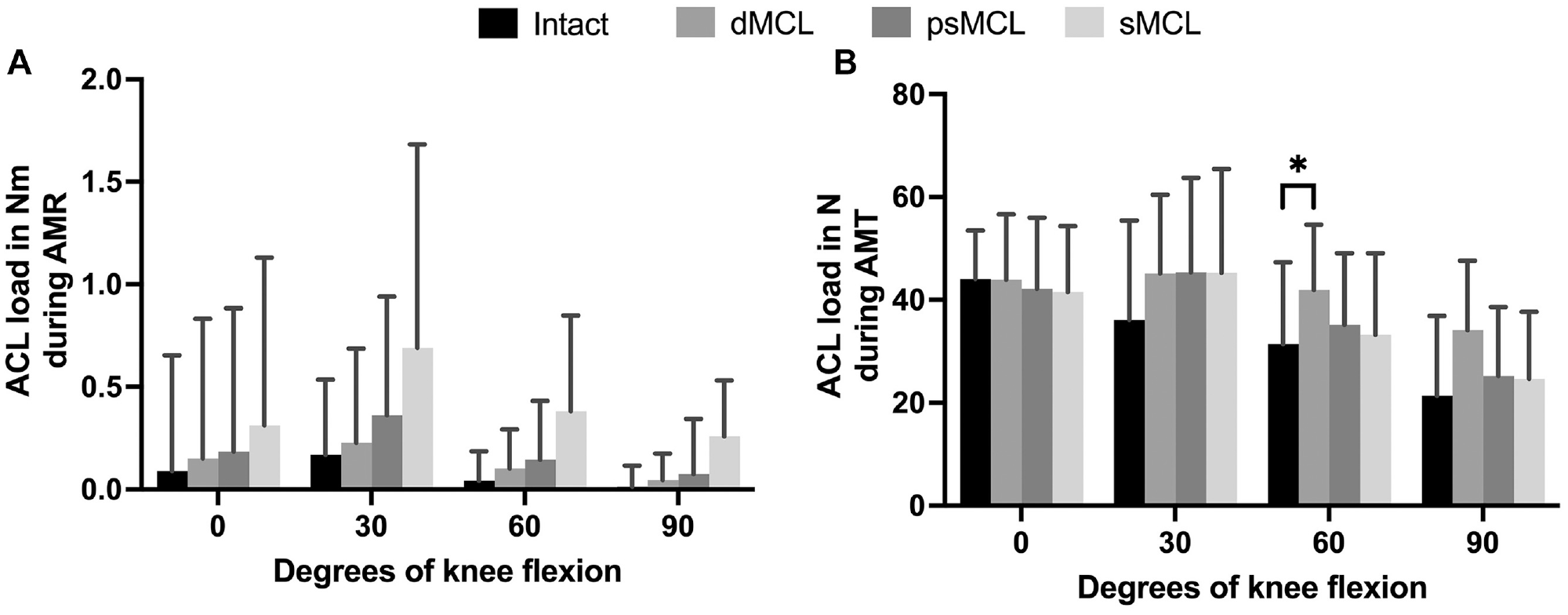
Data from protocol 2 showing the load transfer to the anterior cruciate ligament (ACL) during (A) anteromedial rotation (AMR) and (B) anteromedial translation (AMT) for each knee flexion angle. Results are expressed as mean ± SD. Statistically significant difference is indicated (*P < .05). dMCL, deep medial collateral ligament; psMCL, partial superficial medial collateral ligament; sMCL, superficial medial collateral ligament.
Anteromedial Translation
The force required to reach the AMT limit that was established for the ACL-deficient knee decreased significantly after subsequent progressive release of the medial structures (dMCL and then psMCL followed by complete sMCL). This finding was true for all knee positions when compared with the ACL-deficient state except for the dMCL at 60° of knee flexion. In extension, the largest reduction in force, compared with the previous state, was seen after the dMCL cut. At 30° to 90° of knee flexion, the largest reduction in force was seen after the psMCL injury. Cutting the remnant of the sMCL caused a significant reduction in force compared with the dMCL cut state, but not the previous state, in knee flexion (Figure 2B).
After the dMCL was cut, in comparison with the intact state, a significantly greater amount of the force was transmitted through the ACL at 60° of knee flexion (Δ10 ± 3 N). No significant difference was noticed between all other stages (Figure 3B).
External Rotation
The torque required to reach the ER limit that was established for the ACL-deficient knee decreased significantly after subsequent progressive release of the medial structures (dMCL and then psMCL followed by complete sMCL). This finding was true for all knee positions when compared with the ACL-deficient state. In extension, the largest reduction in torque, compared with the previous state, was seen after the dMCL cut. At 30° to 90° of knee flexion, the largest reduction in torque was seen after the psMCL injury. Cutting the remnant of the sMCL caused a significant reduction in torque compared with the dMCL cut state, but not the previous psMCL state, in all knee positions (Figure 4A).
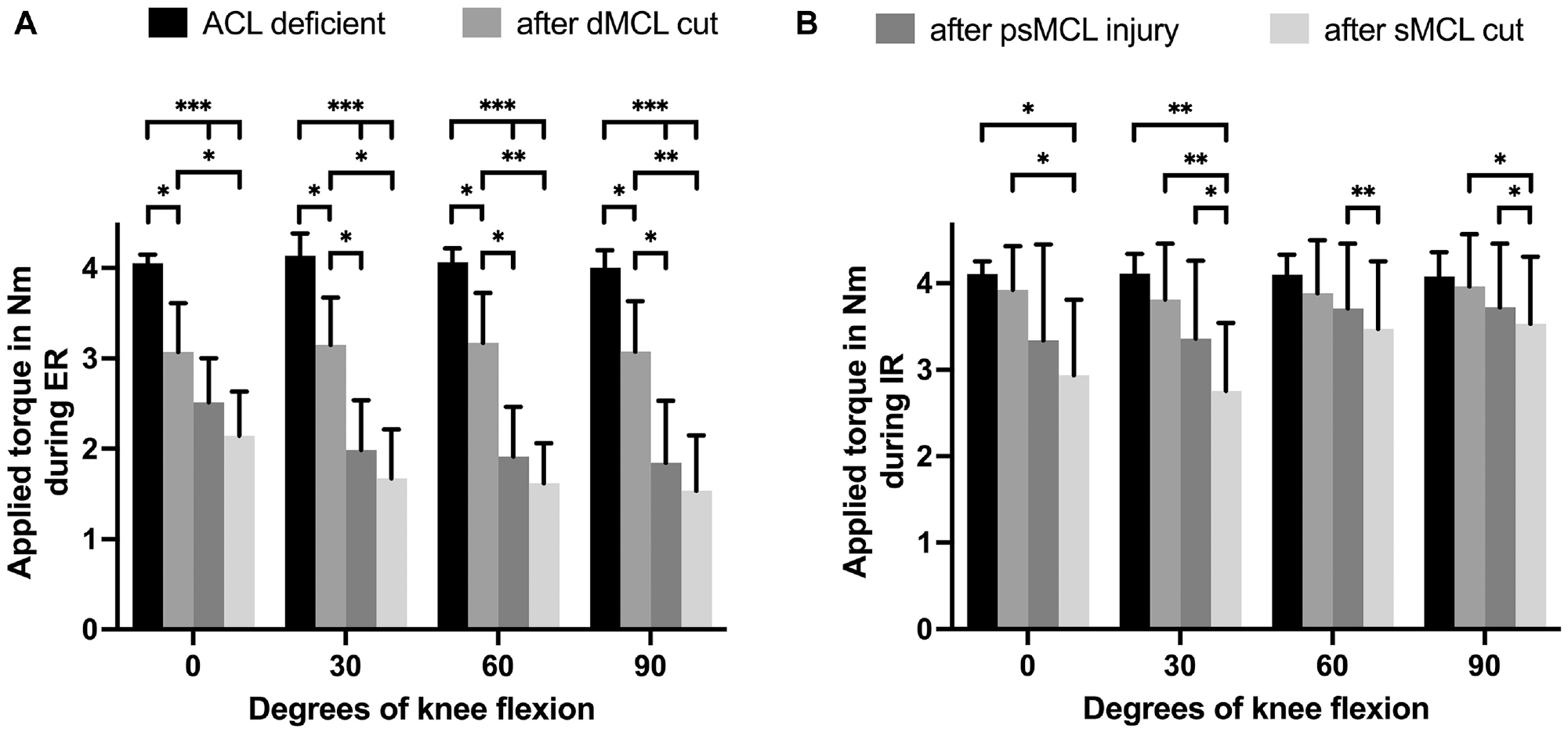
Data from protocol 1 showing the reduction in joint reaction torque during (A) external rotation (ER) and (B) internal rotation (IR) after progressive cutting of the different medial collateral ligament (MCL) structures in an anterior cruciate ligament (ACL)–deficient knee for each knee flexion angle. Results are expressed as mean ± SD. Statistically significant differences are indicated (*P < .05; **P < .01; ***P < .001). dMCL, deep MCL; psMCL, partial superficial MCL; sMCL, superficial MCL.
The torque through the ACL during ER increased after the dMCL cut at 30° of knee flexion (Δ0.1 ± 0.02 N·m). Further significant increases were seen after the psMCL and sMCL injuries, compared with the previous stage, at 90° of knee flexion (Δ0.2 ± 0.06 and Δ0.1 ± 0.03 N·m, respectively). In a combined dMCL/sMCL injury, the ACL load increased compared with a dMCL injury alone at 60° and 90° of knee flexion (Δ0.4 ± 0.1 and Δ0.4 ± 0.05 N·m, respectively). Compared with the intact state, the torque through the ACL increased significantly at 30°, 60°, and 90° after psMCL injury and at 0°, 60°, and 90° of knee flexion after the sMCL cut (Figure 5A).
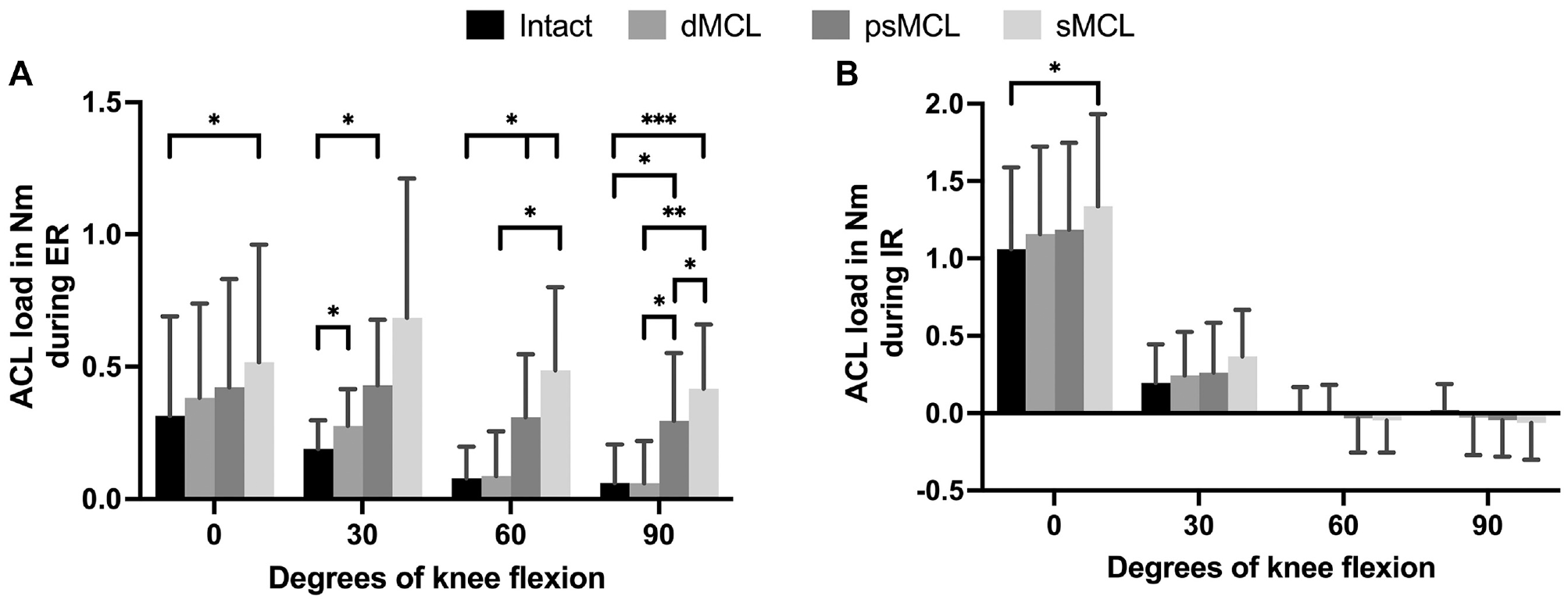
Data from protocol 2 showing the load transfer to the anterior cruciate ligament (ACL) during (A) external rotation (ER) and (B) internal rotation (IR). Results are expressed as mean ± SD. Statistically significant differences are indicated (*P < .05; **P < .01; ***P < .001). dMCL, deep medial collateral ligament; psMCL, partial superficial medial collateral ligament; sMCL, superficial medial collateral ligament.
Internal Rotation
The torque required to reach the IR limit that was established for the ACL-deficient knee decreased significantly at 0° and 30° of knee flexion, when compared with the ACL-deficient state, after cutting of the sMCL remnant (Figure 4B).
After the sMCL was cut, in comparison with the intact state, a significantly greater amount of the torque was transmitted through the ACL in knee extension (Δ0.3 ± 0.07 N·m) (Figure 5B).
Valgus Rotation
The torque required to reach the VR limit that was established for the ACL-deficient knee decreased significantly after the psMCL and sMCL remnant cutting stages (P < .001) but not after the dMCL cut. This was true for all knee positions. The largest reduction in torque was seen after the psMCL injury (Figure 6).
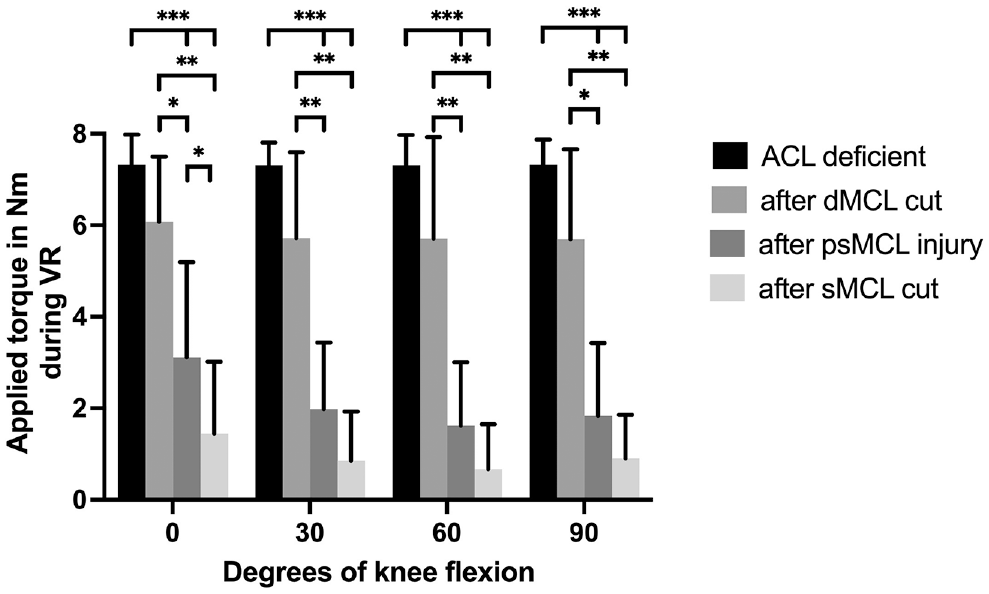
Data from protocol 1 showing the reduction in joint reaction torque during valgus rotation (VR) after progressive cutting of the different medial collateral ligament (MCL) structures in an anterior cruciate ligament (ACL)–deficient knee for each knee flexion angle. Results are expressed as mean ± SD. Statistically significant differences are indicated (*P < .05; **P < .01; ***P < .001). dMCL, deep MCL; psMCL, partial superficial MCL; sMCL, superficial MCL.
After the sMCL was cut, in comparison with the dMCL cut state, a significantly greater amount of the torque was transmitted through the ACL at 90° of knee flexion (Δ1.4 ± 0.4 N·m; P = .0497) (Figure 7).
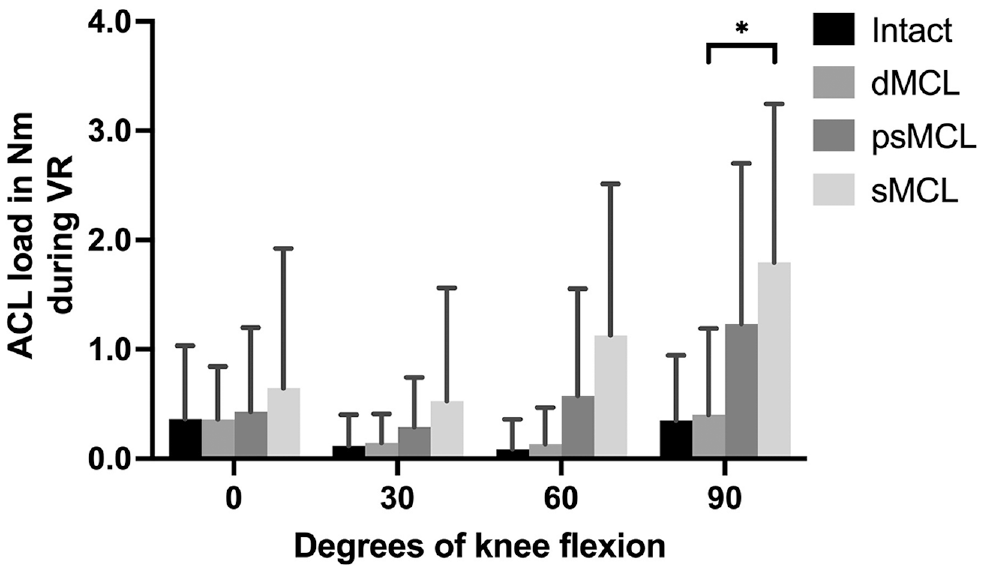
Data from protocol 2 showing the load transfer to the anterior cruciate ligament (ACL) during valgus rotation (VR). Results are expressed as mean ± SD. Statistically significant difference is indicated (*P < .05). dMCL, deep medial collateral ligament; psMCL, partial superficial medial collateral ligament; sMCL, superficial medial collateral ligament.
Discussion
The main findings of this study were that for AMRI, the dMCL injury had the largest contribution in reducing the force/torque in extension and the psMCL in knee flexion in an ACL-deficient knee. For VR, the psMCL injury had the largest contribution to the reduction in torque at all knee positions. The torque through the ACL increased, mainly in knee flexion, after the different MCL cutting states for ER, but not during AMR. In VR, a significant increase was seen only at 90° of knee flexion. Finally, for AMT, the force through the ACL increased after dMCL injury at 60°; however, it did not differ in all other states. Together, these results could only partially confirm the study hypothesis.
Debate remains in the literature about whether the dMCL or sMCL plays the most important role in controlling AMRI.2,35 Previous work found that psMCL injury had a greater influence on AMR and ER than did dMCL injury in 30° to 90° of knee flexion but not in knee extension. 4 The current study used the reductions of force/torque instead of changes in kinematics to calculate contributions of the MCL substructures. In agreement with our previous work, the dMCL contribution was larger in extension, and the reduction after psMCL injury was larger in flexion. The most important new finding, however, was that the contributions to the reductions in force/torque after dMCL or psMCL injury for ER, AMR, and AMT were relatively close to each other. These results suggest that in the combined dMCL/psMCL injury created in this study, both structures played a near equal role in restraining AMRI. Ball et al 2 found that ER was mostly restrained by the dMCL from 0° to 30° of knee flexion; however, Wierer et al 35 reported that the sMCL was the most important structure in restraining AMR, ER, and AMT. The finding in the current study lies between the findings from these recent studies; however, comparing the studies is difficult because we created a psMCL injury. That said, an injury to the psMCL can result in an important loss of its capacity to restrain AMRI. In patients with a higher grade of AMRI, where valgus laxity also exists, it is therefore likely that at least a psMCL injury is present.
Shapiro et al 31 were the first to show that an ACL load increases after dMCL/sMCL injury during VR, ER, and AMT, which increases in knee flexion as measured up to 45° of knee flexion. Furthermore, Battaglia et al 3 showed an ACL load increase in VR, IR, ER, and ATT in extension and 30° of knee flexion after a partial MCL and complete dMCL/sMCL injury. In the present study, the load in the ACL was observed to increase depending on the MCL cutting stage at 0° to 90° of knee flexion for ER. During AMR and in all but 1 comparison for VR, the results did not show a statistically significant difference. However, an in-depth analysis and interpretation of the clinical relevance of the results are as important as solely focusing on the statistically significant changes. 20 We found a trend showing an increase of ACL load for AMR and VR. For VR, the ACL load increased with the knee flexion angle, whereas for ER and AMR, the ACL load was highest at 30° of knee flexion. The nonsignificant results could be related to the high variance in force/torque through the ACL in the used specimens, indicating a lack of precision and hence reducing statistical power. Despite the lack of statistical confirmation and the trend in the results, showing a potential clinically significant increase in force/torque through the ACL depending on the MCL cutting stage in VR, ER, and AMR,3,31 these results should not be ignored but instead interpreted with appropriate caution.
It is difficult to compare the resulting ACL loads after the different joint rotations (AMR, ER, IR, and VR) with those of previous studies in the literature, as they reported the ACL tension in newtons and not as a contribution to joint restraint in newton-meters.3,31 Although the loads used in the current study are in line with those in other biomechanical in vitro MCL studies,2,9,29,35 the loads applied were small, which inherently resulted in low ACL loads. That being said, the relative load increase should not be underestimated. In ER at 30° of knee flexion, the ACL load increased by 46% after the dMCL cut alone and by 127% in the combined dMCL/psMCL injury, both compared with the intact state. Furthermore, after the dMCL/sMCL cut at 90° of knee flexion, the ACL load increased by 593% compared with the intact state. However, these relative increases are expected to be lower in vivo, where the higher joint compression force and the acting muscles co-influence the ACL load. 6
The force through the ACL during AMT increased only at 60° of knee flexion after the dMCL cut. After the psMCL and sMCL were cut, the ACL load was at a similar level as in the intact knee in all knee positions. This seems counterintuitive because the sMCL is a major restraint to AMT, particularly in flexion.2,35 Furthermore, in the literature, the ACL load increased during AMT and ATT after MCL injury, measured at up to 45° of knee flexion.3,31 After an in-depth analysis of the raw data, it remains unclear why the ACL load did not increase after sMCL injury. This finding could be related to the sequence of load application: first ATT and then ER. In contrast, this finding could be due to a change in the rotational axis seen after MCL injury.14,16 The rotational axis might not change much after cutting of the dMCL alone; however, after subsequent psMCL injury, the center of rotation shifts more toward the lateral compartment.14,16 After this, other knee ligaments and capsular structures, like the anteromedial capsule or posterolateral corner, may play a more important role in load sharing. Unfortunately, potential load increases in these other structures were not captured in the current study and are purely speculative.
Registry data have shown that non–surgically managed MCL injuries increase ACL revision rates. 33 In agreement, Ahn and Lee 1 showed that the odds of ACLR failure were higher if a concomitant grade 1 or 2 MCL injury was present at the time of ACL injury. An isolated dMCL injury or, what may be a more clinically relevant injury pattern, a dMCL/psMCL injury may represent these non–surgically managed MCL injuries. The present study showed that an isolated dMCL injury could cause an increase in the ACL load during ER and AMT in certain flexion angles. The combined dMCL/psMCL injury caused a significant further increase on the ACL load during ER. This in vitro biomechanical study therefore supports previous clinical studies; a concomitant incomplete MCL injury increases the ACL load, which explains why an MCL injury is a risk factor for ACLR failure.1,33
In patients with low-grade AMRI due to an isolated dMCL injury, dMCL reconstruction can be discussed. This, however, would mainly apply to high-demand athletes and in specific sports where symptomatic dMCL laxity can be a problem, such as alpine skiing or soccer. 27 With a higher grade of AMRI, it is very likely that a combined dMCL/psMCL injury is present, which can be determined by a concomitant increase in VR. 4 This can be confirmed through the use of valgus stress radiography, where a 1- to 3-mm side-to-side difference in medial gapping would signify a psMCL injury. 17 To reduce ACLR failure rates in cases with medial laxity due to a combined dMCL/sMCL injury, it is important that the medial reconstruction is focused on restoring the sMCL, as it bears a larger share of the net joint load compared with the dMCL and has a greater influence on the ACL load, especially in ER. It may be difficult to fully restore the sMCL function with a minor extra-articular procedure.5,24,25
This study has a number of limitations that must be discussed. First, it uses the principle of superposition to measure the MCL load sharing and ACL load. Although this is not a direct measurement, it is often used in the peer-reviewed literature2,3,11,26,30 because it has some advantages over a direct measurement. The tested structures are not altered by placing force transducers 37 ; further, testing is not limited to specific ligament bundles 30 and allows for measurements of ligament force where transducers cannot be placed, in structures such as the dMCL. 37 One of the assumptions required to apply the principle of superposition, described by Guenther et al, 12 is that there is no interaction between separately measured structures. For the MCL load-sharing measures in the current study, there could have been a physical interaction between the injured and uninjured sMCL bundles; therefore, psMCL contributions may be inaccurate. Schafer et al 30 found that the effect on the loads caused by physical interaction of the adjacent soft tissues, in their setup between the sMCL and POL, was minimal. Although those authors investigated a different situation, their study provides some evidence that the effect due to physical interaction could be limited. 30 We tried to mitigate this violation by adding comparisons between the sMCL and dMCL states, which do not violate the principle, and so the reader has more liberty in interpreting the psMCL injury state. For the ACL load transfer, we applied superposition to measure the force/torque contribution of the ACL, not of the dMCL, psMCL, or sMCL. We therefore did not violate the assumptions of superposition for ACL force by making a psMCL injury because we reported the ACL load changes as a result of these accumulated injuries. Admittedly, the surgical approach to reach the ACL may introduce some damage that was not measured; however, we believe the possible influence is negligible due to the minimally invasive transpatellar approach, which includes a 2- to 3-cm incision, with subsequent closure of all layers (joint capsule, subcutaneous layer, and skin).
A second limitation involves the large spectrum of different types of psMCL injuries in vivo. The psMCL created in this study did not necessarily cause the same ACL load increase or show the same reduction in force/torque contribution as every in vivo psMCL injury. However, the designed technique to create a psMCL injury was reproducible and thereby helped to keep the effect of a psMCL injury consistent. Third, the loads applied were not like those used during functional activities but were similar to those used during clinical manual laxity examinations. Higher loads, however, have the potential to stretch the remaining intact ligaments leading to uncontrolled increases in joint laxity, which may alter the measured ligament contribution and ACL load. The loads used were in line with those used in previous biomechanical studies on the MCL.2,3,29,35 Fourth, the contributions of the surrounding muscles, which might increase or decrease ACL load or reduce AMRI, were not considered.6,13 It therefore remains difficult to extrapolate these findings to the in vivo forces on the ACL during activities of daily living or sporting activities.
Conclusion
The psMCL injury made the largest contribution to the reduction of net force/torque during AMR/AMT at 30° to 90° of flexion. Concomitant dMCL/psMCL injury increased the ACL load, mainly during ER. If a surgical procedure is being considered to treat AMRI, then the procedure should focus on restoring the sMCL function because injury to this ligament causes a major loss of the knee joint’s capacity to restrain AMR/AMT.
Footnotes
Submitted July 29, 2023; accepted March 10, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was supported by Smith & Nephew, in the form of a research grant that funded the presented research; Ossur, in the form of an educational grant for our research fellowship position; the Natural Sciences and Engineering Research Council Discovery: RGPIN-2018-05693; the Ontario Early Researcher Award ER18-14-197; and the Canadian Foundation for Innovation JELF and Ontario Research Fund–Research Infrastructure, 38141. A.G. has received consulting fees from Smith & Nephew; is on the advisory board and holds stock in Spring Loaded Technology; and holds stock in Precision OS, LinkX Robotics, and Ostesys Robotics. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
