Abstract
Background:
Injuries to the medial collateral ligament (MCL), specifically the deep MCL (dMCL) and superficial MCL (sMCL), are both reported to be factors in anteromedial rotatory instability (AMRI); however, a partial sMCL (psMCL) injury is often present, the effect of which on AMRI is unknown.
Purpose:
To investigate the effect of a dMCL injury with or without a psMCL injury on knee joint laxity.
Study Design:
Controlled laboratory study.
Methods:
Sixteen fresh-frozen human cadaveric knees were tested using a 6 degrees of freedom robotic simulator. The anterior cruciate ligament (ACL) was cut first and last in protocols 1 and 2, respectively. The dMCL was cut completely, followed by an intermediary psMCL injury state before the sMCL was completely sectioned. Tibiofemoral kinematics were measured at 0°, 30°, 60°, and 90° of knee flexion for the following measurements: 8 N·m of valgus rotation (VR), 4 N·m of external tibial rotation, 4 N·m of internal tibial rotation, and combined 89 N of anterior tibial translation and 4 N·m of external tibial rotation for both anteromedial rotation (AMR) and anteromedial translation. The differences between subsequent states, as well as differences with respect to the intact state, were analyzed.
Results:
In an ACL-intact or -deficient joint, a combined dMCL and psMCL injury increased external tibial rotation and VR compared with the intact state at all angles. A significant increase in AMR was seen in the ACL-intact knee after this combined injury. Cutting the dMCL alone showed lower mean increases in AMR compared with the psMCL injury, which were significant only when the ACL was intact in knee flexion. Moreover, cutting the dMCL had no effect on VR. The ACL was the most important structure in controlling anteromedial translation, followed by the psMCL or dMCL depending on the knee flexion angle.
Conclusion:
A dMCL injury alone may produce a small increase in AMRI but not in VR. A combined dMCL and psMCL injury caused an increase in AMRI and VR.
Clinical Relevance:
In clinical practice, if an increase in AMRI at 30° and 90° of knee flexion is seen together with some increase in VR, a combined dMCL and psMCL injury should be suspected.
Keywords
The medial collateral ligament (MCL) is a commonly injured knee structure and accounts for up to 20% of all knee ligament injuries.11,16,23,28 An MCL injury is commonly seen in patients with an anterior cruciate ligament (ACL) tear.11,16,23,28 Most MCL injuries, with or without concomitant ACL injury, have historically been treated nonoperatively.9,15 After healing, some knee joints exhibit residual valgus and rotational laxity in the form of anteromedial rotatory instability (AMRI).8,22 Residual medial laxity may increase the load on the ACL graft after reconstruction.1,3,21 The revision rate after ACL reconstruction has been noted to be higher with concomitant nonsurgically treated MCL tears than in ACL reconstruction without concomitant MCL injury.1,23
Several biomechanical studies have investigated the role of the superficial MCL (sMCL) and deep MCL (dMCL) in controlling knee laxity.2,4,20,25,27 Typically, each structure is sequentially cut to examine the corresponding effect on knee laxity.2,4,25 In clinical practice, however, it is more common to see partial injuries, such as intrasubstance strains or partial femoral or tibial avulsions.11,16,23,28 Patients with an incomplete medial-side injury, or partially healed injury, commonly have an increase in valgus laxity with or without AMRI.8,15 Because the sMCL is the primary restraint to valgus stress, these patients likely have a partial sMCL (psMCL) injury.8,15,28 Which structure plays the primary role in controlling AMRI in such medial injuries is still under discussion.2,25
The purpose of the current study was to investigate the role of a combined dMCL and psMCL injury on knee joint laxity. The main hypothesis was that dMCL injury would cause an increase in AMRI without causing an increase in valgus rotation (VR) but additional psMCL injury would cause an increase in both AMRI and VR.
Methods
Sixteen fresh-frozen human cadaveric knee specimens without history of injury, surgery, osteoarthritis, or meniscal pathology were used for this study. This study was approved by the institutional research ethics board.
Apparatus and Specimen Preparation
Biomechanical testing was performed with a 6 degrees of freedom joint motion simulator (VIVO; Advanced Mechanical Technologies). Each specimen was thawed at room temperature over a 24-hour period before testing. Soft tissues were denuded from the proximal femur and distal tibia or fibula, leaving intact all tissues within 150 mm of the transepicondylar axis. Our techniques for potting and mounting tissues onto the VIVO have been described previously.18,24 Two optical motion trackers (Optotrak; Northern Digital) were fixed to the femur and tibia. Three-dimensional surface models of each specimen’s femur and its tibia or fibula, previously segmented and reconstructed from computed tomography images (3-Dimensional Slicer; http://www.slicer.org/), were coregistered to the specimens. A model-guided, motion capture–aided alignment technique was used to position the femur onto the joint motion simulator, such that the knee flexion axis was concentric with the mechanical flexion axis of the machine and centered mediolaterally, which minimizes unwanted secondary motions, 18 and the tibial shaft was vertically oriented in the frontal and sagittal planes. With the specimen in full extension, a compressive load (100 N) was applied across the joint parallel to the long axis of the tibia while all other degrees of freedom were unconstrained. The resulting position was defined as the reference position; all joint kinematics were measured relative to this pose by the joint motion simulator using coordinate conventions from Grood et al. 5
Biomechanical Testing
Before data collection, we preconditioned each specimen to reduce tissue hysteresis by performing the entire loading sequence for 5 test cycles. Tibiofemoral kinematics were measured with the joint flexion angle fixed at 0°, 30°, 60°, and 90° under a compression load of 10 N. Valgus laxity was measured in response to an 8-N·m VR torque. External rotation (ER) and internal rotation laxity were measured using a 4-N·m rotation torque. AMRI was evaluated based on the maximum anterior translation (anteromedial translation; AMT) referencing the center of the joint and the maximum tibial ER (anteromedial rotation; AMR) measured after first applying an anteriorly directed force (89 N) and then adding an ER torque (4 N·m) to the tibia. The forces used were in line with those used in previous studies investigating medial knee biomechanics.2,4,20,25 Each load was applied and removed over a 30-second period, and kinematic parameters were measured using sensors embedded in the actuators of the joint motion simulator (accurate to 0.1 mm and 0.1°) and recorded at 500 samples per second. The resulting maximum achievable rotations or displacements were measured relative to their corresponding neutral positions. Each test was repeated 5 times. The mean of the second, third, and fourth tests was used as the final laxity output. Data from the first and fifth cycles were discarded to limit possible beginning- and end-of-test effects.
Dissection Sequences
Two distinct dissection protocols were used. The ACL was cut first or last, depending on the protocol, to evaluate the effect of the different MCL injury patterns on AMRI in an ACL-intact or -deficient joint. The dMCL was always cut before performing a psMCL, for 3 reasons: (1) during knee injury, the dMCL is suggested to rupture first due its lower strength and stiffness as well as its shorter length compared with the sMCL8,19,20,25,26; (2) keeping the order of MCL cuts consistent in protocols 1 and 2 aids comparisons between both protocols; (3) during our pilot tests (n = 4), it was not possible to create a psMCL injury without damaging the dMCL. Protocol 1 included 8 specimens (mean age, 60 years [range, 27-72 years]; mean body mass index, 25; 0 pairs; 5 male, 3 female), and protocol 2 included 8 specimens (mean age, 60 years [range, 54-70 years]; mean body mass index, 27; 1 pair; 5 male, 3 female) (Figure 1).
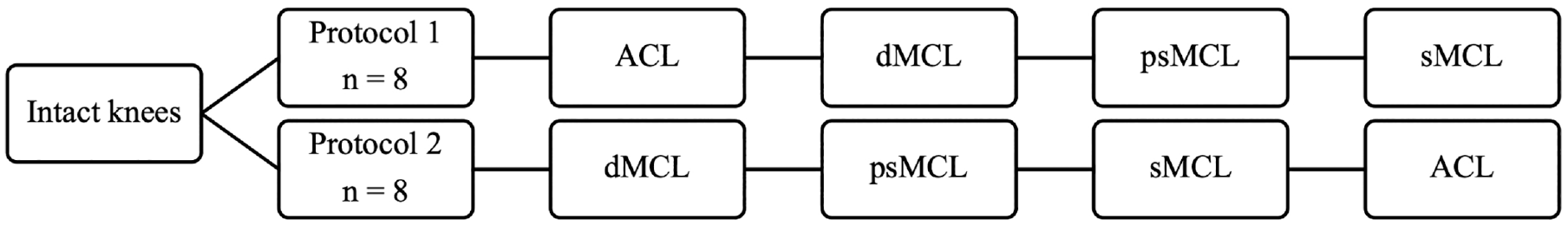
The 2 cutting protocols (n = 8 samples for each protocol). ACL, anterior cruciate ligament; dMCL, deep medial collateral ligament; psMCL, partial superficial medial collateral ligament; sMCL, superficial medial collateral ligament.
The sequential cutting was done with the specimen mounted on the joint motion simulator, except for the dissection and cutting of the ACL, which required removal to permit a transpatellar approach that was used for subperiosteal sharp dissection of the ACL from its femoral attachment. 17 Our joint motion simulator features custom indexing fixtures that permit this: Half of the fixtures remain securely connected to the specimen, half remain securely connected to the machine, and the 2 halves include indexing features so they can disconnect from each other and reconnect repeatably. The dMCL was approached via a longitudinal incision of the anteromedial fascia just anterior to the sMCL at the level of the joint line.14,25 After the tissue plane between the dMCL and sMCL was developed, the meniscofemoral and meniscotibial divisions of the dMCL were cut.14,25 To perform a psMCL cut, we positioned the knee in 30° of flexion and applied a 10-N·m VR torque. The motion capture system was used to define coordinates approximately 10 mm proximal and distal to the joint line, which were references for measuring the opening of the medial compartment. On the basis of previous work that demonstrated a 2.8-mm increase in medial opening with a complete sMCL injury in response to a 10-N·m VR torque, we performed a selective release (“pie crusting”) of the sMCL using an 18-gauge needle until there was a medial compartment opening of 2.0 ± 0.5 mm.3,13 The release was performed anterior to the posterior oblique ligament (POL) at the level of, or proximal to, the joint line. A complete sMCL cut was made by cutting the sMCL below the medial joint line, proximal to the proximal tibial sMCL attachment. This was extended posteriorly to the anterior margin of the anterior arm of the POL. Injury to the POL and posteromedial joint capsule can cause increased VR and internal rotation laxity in extension (posteromedial instability),18,20 which lies outside the scope of this study, and so care was taken to ensure that these structures were not damaged. All surgical procedures were conducted by 2 trained orthopaedic fellows (W.B. and T.V.) supervised by the senior fellowship-trained orthopaedic surgeon (A.G.) to reduce interobserver error.
Statistical Analysis
Statistical analysis was performed using commercial software (GraphPad Prism 9; Dotmatics). A 2-factor (flexion angle and cutting state) mixed-effects analysis was performed to compare laxities at each cutting state with the previous state as well as with the intact state. A post hoc Bonferroni correction was used to correct for multiple comparisons when comparing estimated means. Sample sizes were chosen such that changes in laxities as small as 1° between states could be detected with 80% power and 95% confidence. 20 The kinematic results are given as mean ± SD. Statistical significance was set at P < .05 (adjusted P value after Bonferroni correction).
Results
The inducible medial compartment opening increased by 2 ± 0.1 mm and 1.9 ± 0.2 mm after the psMCL injury in protocols 1 and 2, respectively. Two specimens exceeded the range limit of the joint motion simulator in protocol 1, which resulted in missing kinematic data points (14 of 320 in these cutting states). In the results below, the differences in kinematics are always compared with the previous state, except when stated otherwise.
Anteromedial Rotation
In protocol 1, only the psMCL injury caused a significant increase in AMR at 30° of knee flexion (Δ2.7°± 0.5°, P = .010).
In an ACL-intact knee, AMR increased significantly with subsequent progressive release of the medial structures (dMCL and then psMCL, followed by complete sMCL), except for the dMCL in extension. Finally, cutting the ACL led to a significant decrease of AMR in 60° and 90° of flexion (Δ4.7°± 0.9°, P = .006, and Δ5.0°± 0.7°, P = .001, respectively). The results for AMR are reported in Figure 2.
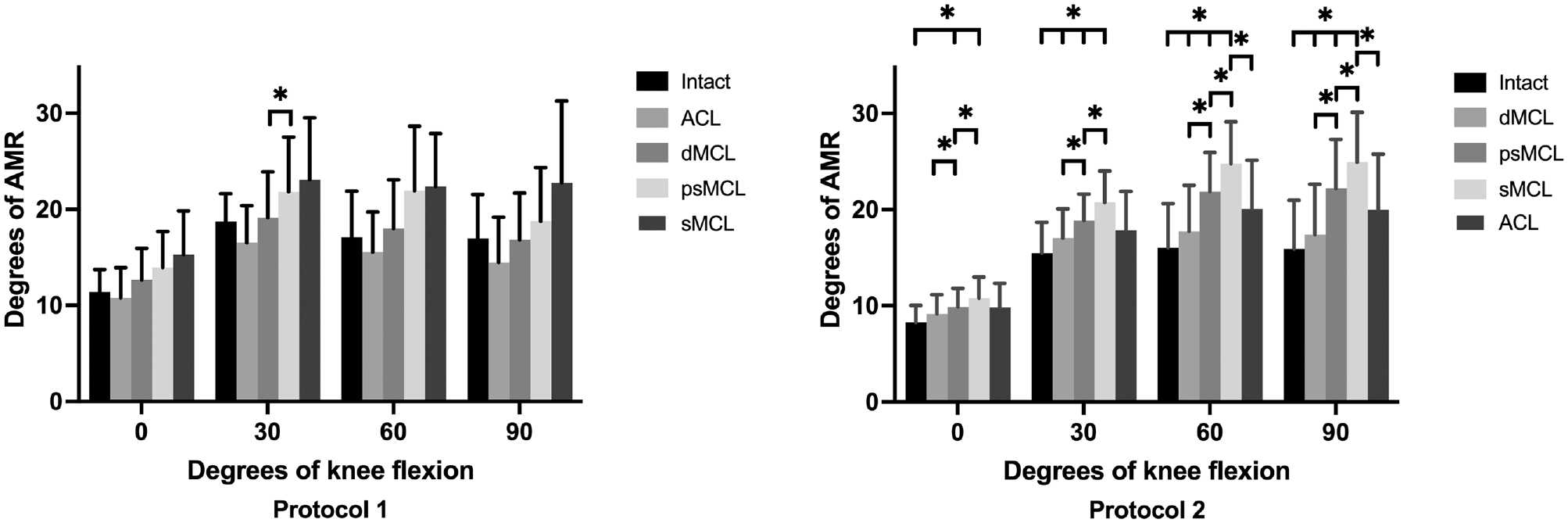
Anteromedial rotation (AMR) for each flexion angle in all knee states for protocols 1 and 2. Results are given in degrees as mean ± SD. *Statistically significant differences compared with the previous states as well as with the intact state. ACL, anterior cruciate ligament; dMCL, deep medial collateral ligament; psMCL, partial superficial medial collateral ligament; sMCL, superficial medial collateral ligament.
Anteromedial Translation
The largest increases in AMT were seen after the ACL was cut in both protocols. In protocol 1, dMCL sectioning caused a significant increase in AMT at 0° and 30° of knee flexion (Δ0.7 ± 0.1 mm, P = .001, and Δ1.7 ± 0.4 mm, P = .036, respectively). At 30° to 90° of knee flexion, the ACL, dMCL, psMCL, and sMCL cutting states differed significantly from the intact state (P < .05).
In an ACL-intact knee, AMT increased significantly after the psMCL injury at all knee flexion angles (0°, Δ0.2 ± 0.05 mm, P = .015; 30°, Δ0.7 ± 0.1 mm, P = .006; 60°, Δ1.0 ± 0.2 mm, P = .008; and 90°, Δ1.0 ± 0.2 mm, P = .022). The results for AMT are reported in Figure 3.
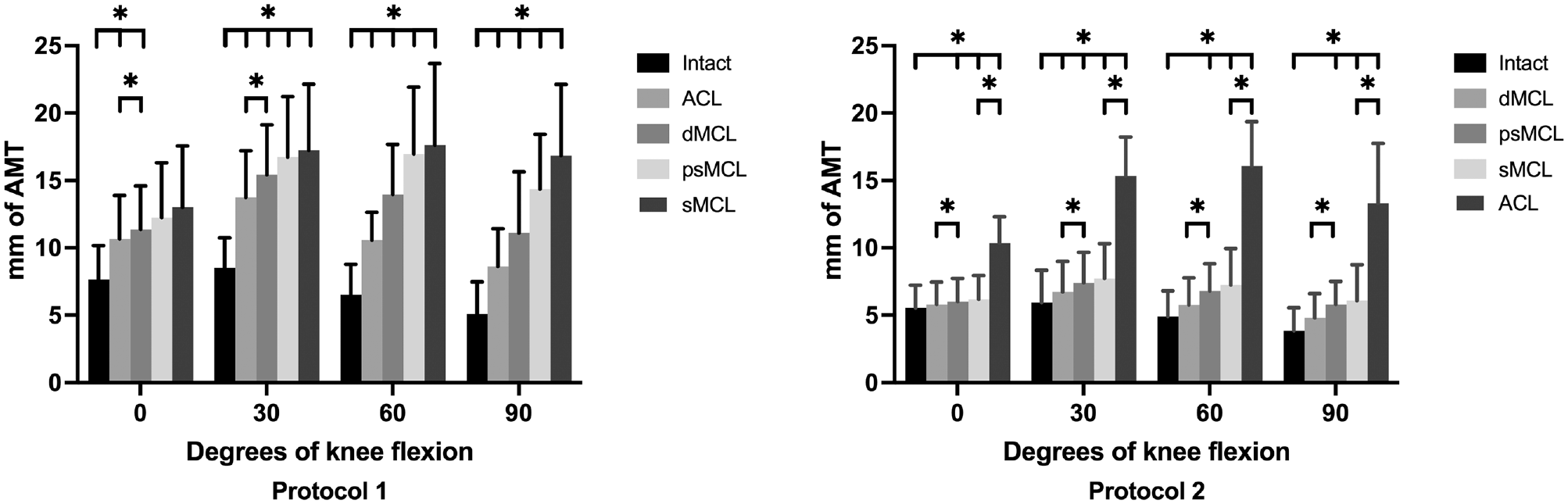
Anteromedial translation (AMT) for each flexion angle in all knee states for protocols 1 and 2. Results are given in millimeters as mean ± SD. *Statistically significant differences compared with the previous states as well as the intact state. ACL, anterior cruciate ligament; dMCL, deep medial collateral ligament; psMCL, partial superficial medial collateral ligament; sMCL, superficial medial collateral ligament.
External Rotation
In protocol 1, cutting the dMCL resulted in a significant increase of ER compared with the intact state (P < .05) but not compared with the previous ACL cut state (P > .05). Subsequent psMCL injury caused a significant increase in ER compared with the previous state as well as the intact state at all knee flexion angles except at 0° (compared with previous state) (P < .05).
With the ACL intact, ER significantly increased at each subsequent MCL cutting stage (dMCL cut, psMCL injury, and sMCL cut) at all flexion angles except for after the sMCL cut at 30° of flexion (P < .05). The results for ER for protocols 1 and 2 are reported in Figure 4.
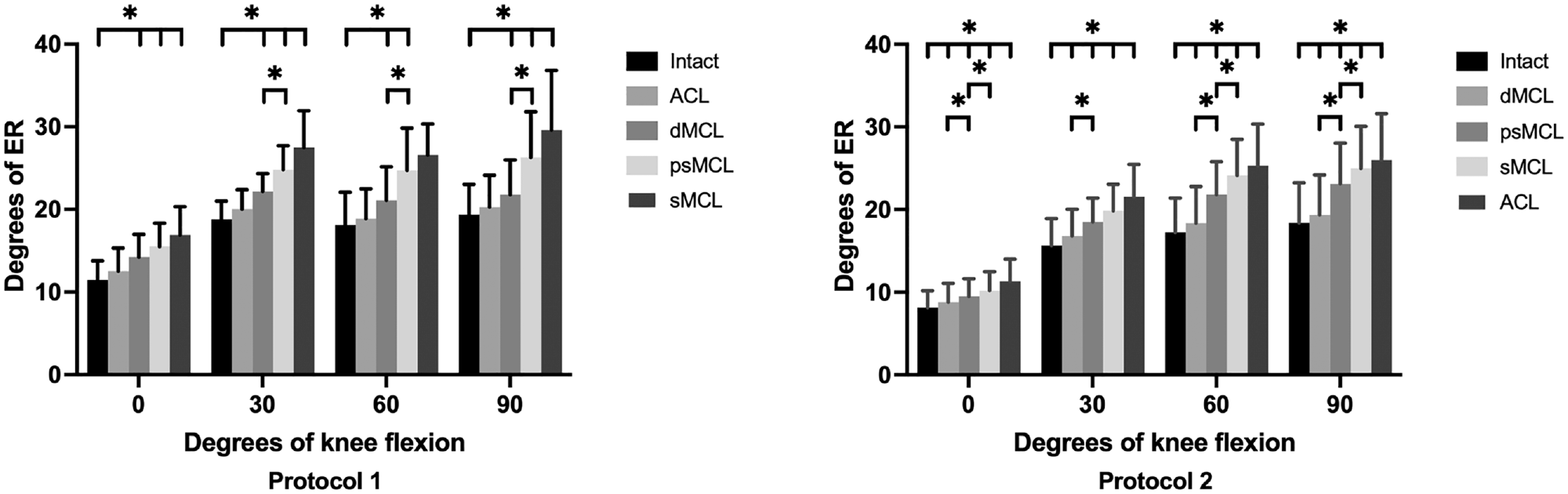
External rotation (ER) for each flexion angle in all knee states for protocols 1 and 2. Results are given in degrees as mean ± SD. *Statistically significant differences compared with the previous states as well as the intact state. ACL, anterior cruciate ligament; dMCL, deep medial collateral ligament; psMCL, partial superficial medial collateral ligament; sMCL, superficial medial collateral ligament.
Internal Rotation
A significant increase was seen in protocol 1 at 90° of knee flexion after psMCL injury (Δ0.6°± 0.1°, P = .036). In the ACL-intact knee (protocol 2), increases were observed after psMCL injury at 60° and 90° of knee flexion (Δ1.2°± 0.2°, P = .008, and Δ1.0°± 0.2°, P = .007, respectively). Subsequent cutting of the sMCL and ACL increased internal rotation at 60° of knee flexion (Δ0.8°± 0.2°, P = .022) and 0° of knee flexion (Δ2.5°± 0.4°, P = .006), respectively. The results for internal rotation for protocols 1 and 2 are reported in Figure 5.
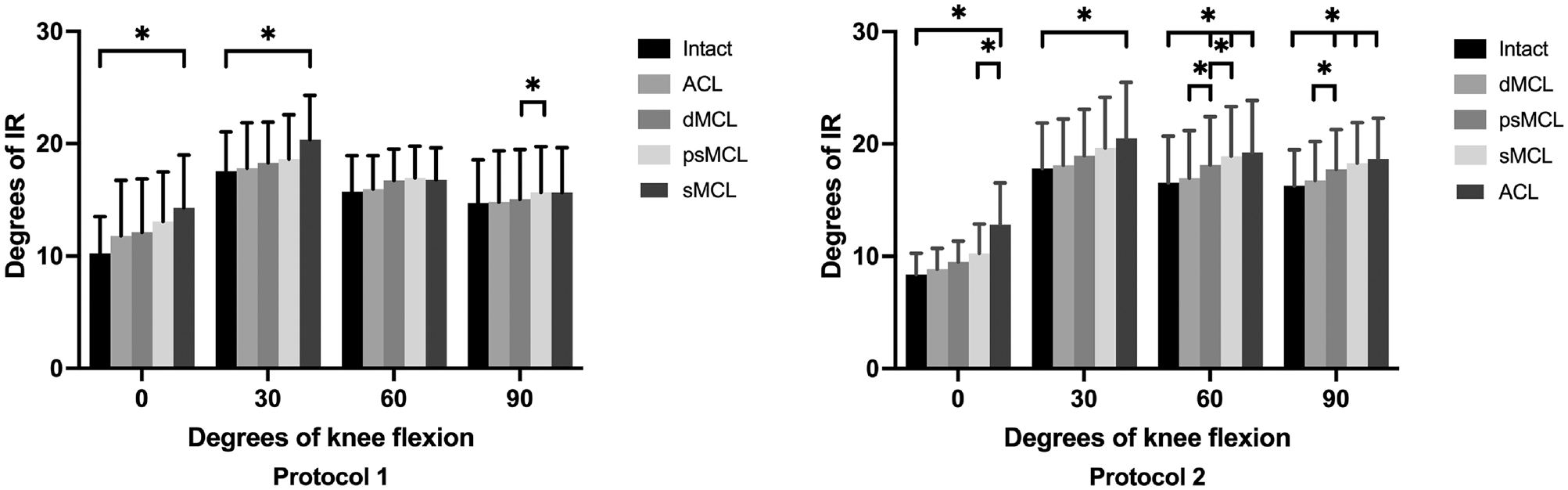
Internal rotation (IR) for each flexion angle in all knee states for protocols 1 and 2. Results are given in degrees as mean ± SD. *Statistically significant differences compared with the previous states as well as the intact state. ACL, anterior cruciate ligament; dMCL, deep medial collateral ligament; psMCL, partial superficial medial collateral ligament; sMCL, superficial medial collateral ligament.
Valgus Rotation
In protocol 1, cutting the ACL and dMCL while leaving the sMCL intact did not increase VR compared with either the previous state or the intact state (P > .05). VR increased significantly after both the psMCL and the subsequent complete sMCL injuries at all angles except at 90° for the psMCL injury (P < .05).
In an ACL-intact knee, cutting the dMCL did not cause an increase in VR. A psMCL injury caused significant increases in VR at all angles (0°, Δ1.1°± 0.2°, P < .001; 30°, Δ1.8°± 0.3°, P = .005; 60°, Δ2.1°± 0.4°, P = .005; 90°, Δ2.3°± 0.3°, P = .002). Cutting the ACL increased VR at all angles except at 30° (0°, Δ0.6°± 0.1°, P = .02; 60°, Δ2.7°± 0.5°, P = .006; 90°, Δ4.0°± 0.3°, P = .03). The results for VR across protocols 1 and 2 are reported in Figure 6.
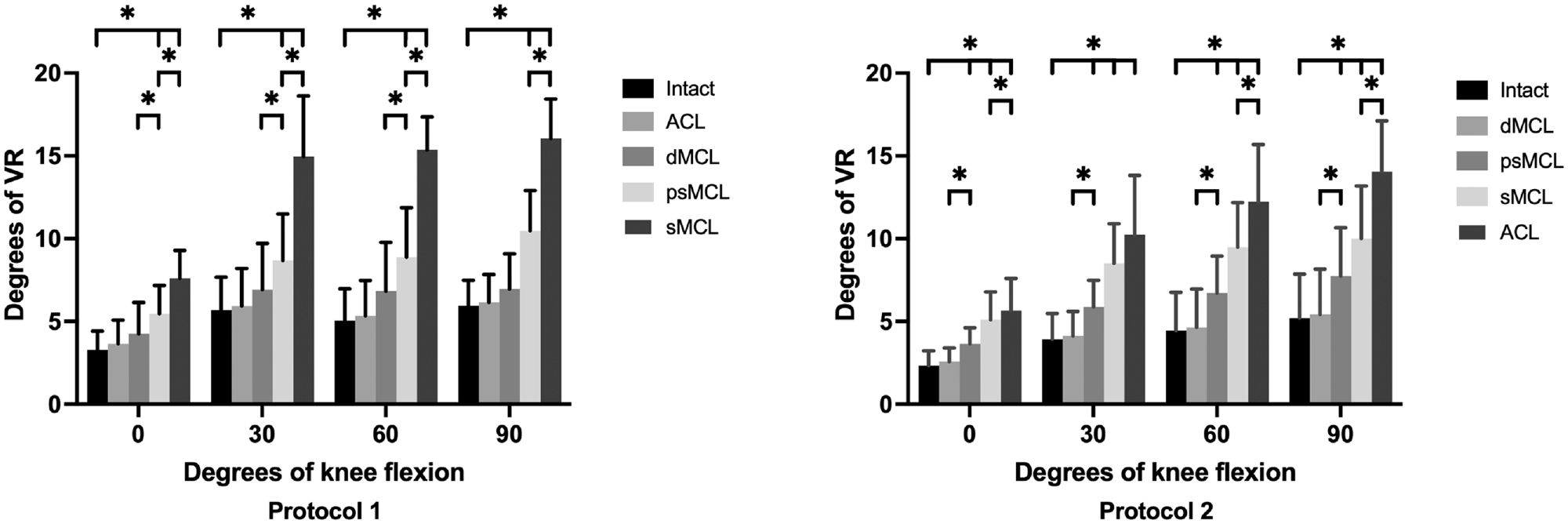
Valgus rotation (VR) for each flexion angle in all knee states for protocols 1 and 2. Results are given in degrees as mean ± SD. *Statistically significant differences compared with the previous states as well as the intact state. ACL, anterior cruciate ligament; dMCL, deep medial collateral ligament; psMCL, partial superficial medial collateral ligament; sMCL, superficial medial collateral ligament.
Discussion
The main findings of this study were that a combined dMCL and psMCL injury, in both an intact knee and an ACL-deficient knee, increased AMT, ER, and VR compared with the intact state at all angles, except in extension for AMT (protocol 1). A significant increase in AMR was also seen in the ACL-intact knee after this combined injury. Cutting the dMCL alone showed lower mean increases in AMR compared with the psMCL injury, which were only significant when the ACL was intact in 30° to 90° of knee flexion. Moreover, cutting the dMCL had no effect on VR. The ACL was the most important structure in controlling AMT, followed by the psMCL or dMCL depending on the knee flexion angle. Together, these results confirm the main hypothesis.
A challenge in biomechanical studies is to create injuries that are similar to those observed in clinical practice. Some authors have suggested that the dMCL ruptures first due its lower strength and stiffness as well as its shorter length compared with the sMCL.8,19,20,25,26 The sMCL is stronger as a structure overall, but it does not function as a single cordlike structure.19,26 Its broad, flat structure has different bundles of fibers, with each having different length change characteristics depending on the angle of knee flexion.12,14,27,29 Consequently, different fibers are taut depending on the knee position, making it more prone to partial injuries.19,26,29 Because the sMCL is the primary restraint for ER, AMR, and VR, at most flexion angles the dMCL is protected if the injury mechanism involves these forces.2,4,25,29 A recent study by Willinger et al 28 investigating ACL injuries with concomitant low-grade MCL injuries showed that a psMCL injury can occur without dMCL injury. Moreover, a solitary dMCL injury was uncommon, as dMCL injury was mostly seen with concomitant sMCL injury. 28 Therefore, a dMCL injury in isolation may not be the most common cause of low-grade AMRI.2,25 Consistent with other studies, we found that cutting the dMCL produced no significant increase in VR laxity.4,20,25 These data suggest that AMRI might be more likely caused by a combined dMCL and psMCL injury, which also causes an increase in valgus laxity that can be seen clinically. 28 The current study demonstrated that this combined injury pattern caused a significant increase in ER, AMR, and VR compared with the previous state as well as with the intact state for most flexion angles.
To the best of our knowledge, this is the first study to report on in vitro kinematic changes after a psMCL injury that was carefully measured to result in 2 ± 0.5 mm of VR laxity. The definition of a psMCL injury is critical to interpret biomechanical cutting studies. The biomechanical study by Battaglia et al 3 reported on kinematic changes after what the investigators defined as a psMCL injury. However, they used a 5-mm increase in the inducible medial compartment opening in 30° of knee flexion, referring to the International Knee Documentation Committee (IKDC) grades, to define a psMCL injury.3,6 MCL injuries have been historically graded according to the IKDC form, which is based on a non–scientifically validated, clinically applied stress and subjective assessment: grade I, 3 to 5 mm; grade II, 6 to 10 mm; and grade III, >10 mm. 6 The grade is defined by the difference in the induced medial compartment opening created by a valgus stress at 0° and 30° of flexion of the injured knee compared with the healthy contralateral knee. 6 However, in a cadaveric study using radiographs to determine side-to-side differences in medial compartment gapping, LaPrade et al 13 showed that a complete sMCL injury caused an increase of medial compartment opening of 2.8 mm under 10 N·m of valgus load at 20° of knee flexion. The results of the Battaglia et al. study, using the IKDC thresholds to define a psMCL injury, should therefore be interpreted with caution because these thresholds may not be representative of a true psMCL injury.3,13
Debate remains in the literature about whether the dMCL or sMCL is the primary restraint to ER, AMR, and AMT.2,20,25 A recent in vitro cutting study using a 6 degrees of freedom robotic setup to measure joint laxity found that the sMCL was the most important restraint to AMRI. 25 We had a similar finding: In extension and with an intact ACL, a psMCL injury caused a small but significant increase in AMR, whereas isolated division of the dMCL did not have a significant effect. Furthermore, the psMCL injury had a greater influence on ER and AMR compared with the dMCL at 30° and especially 60° and 90° of knee flexion. Therefore, although the importance of a complete sMCL injury was previously known, we found that even a psMCL injury has a larger effect on ER and AMR than does a dMCL injury. 25 AMR decreased after ACL injury, which is consistent with the results reported by Wierer et al. 25 This might be due to shifts in the rotational axis from the center of the knee joint to the medial or lateral compartment in an MCL-intact or -deficient knee, respectively.10,22 We found that cutting the ACL resulted in the largest increase in AMT in both cutting protocols. The second greatest increase that was statistically significant varied between either cutting the dMCL or after psMCL injury, depending on the protocol and knee position. Wierer et al found that cutting the sMCL led to larger increases in AMT than cutting the dMCL at all knee flexion angles. In agreement with previous studies, we found that the sMCL was the primary restraint to VR2-4,20,25; however, we also showed that a psMCL injury can increase both valgus laxity and AMRI more compared with a dMCL injury, particularly in the flexed knee.
Classification systems are important to stratify injuries to guide clinicians in their treatment as well as for documentation and reporting.6,25 However, the most commonly used grading scale, the IKDC scale, grades a medial injury solely depending on the amount of medial opening and therefore completely ignores the amount of AMRI. 6 Recently, Wierer et al 25 described an anteromedial grading system in which they propose 3 grades, depending on the increase in VR, AMR, and AMT. This is a good first step toward an improved medial-side injury grading system. The authors of the current study believe that eventually, the amount of VR laxity and AMRI should be decoupled in the grading system. Furthermore, it might be better to use a different naming system so there is no confusion with the old IKDC classification. 6 From a clinician’s standpoint, it is important to acknowledge that an increase in the anteromedial drawer and ER test (Slocum test), 22 especially in 30° of knee flexion, can be due to a dMCL injury alone. However, when there is a higher grade of AMRI in 30° and 90° of knee flexion, a combined dMCL and psMCL injury is likely present, which can be confirmed by the concomitant increase in VR.
The present study is not without limitations. First, the sMCL is a complex ligament, with different length-changing characteristics depending on the anatomic location.12,14 Therefore, the psMCL injury created in this study does not necessarily cause the same kinematic effect as every partial sMCL injury in vivo. However, the technique was designed to be reproducible between the different specimens, thereby keeping the testing consistent. Furthermore, the kinematic results must be interpreted as “time zero” after injury because in vivo biological healing of the MCL will cause changes in kinematics over time. Second, the loads applied were similar to those used during clinical manual laxity examinations and not like those used during functional activities. However, this was needed to avoid potential stretching of the remaining ligaments, which could lead to further uncontrolled increases in joint laxity. The loads used in this study are similar to those used in previous biomechanical studies on the MCL.2,3,20,25 Third, the contribution of the surrounding muscles on knee joint stability was not assessed. Specifically, the semimembranosus muscle is a known dynamic stabilizer of AMRI in the MCL-deficient knee. 7 Unfortunately, the in vitro setup in this study did not allow us to account for dynamic restraints. Fourth, 2 specimens exceeded the range limit of the joint motion simulator in protocol 1, which resulted in missing kinematic data points (14 of 320 in these cutting states). The missing data were mitigated by applying a mixed-model statistical analysis.
Conclusion
The dMCL injury alone may produce a small increase in AMRI but not in VR. A combined dMCL and psMCL injury caused an increase in AMRI and VR. In clinical practice, if an increase in AMRI at 30° and 90° of knee flexion is seen together with some increase in VR, a combined dMCL and psMCL injury should be suspected.
Footnotes
Submitted July 29, 2023; accepted February 14, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: Support was provided by Smith & Nephew, in the form of a research grant that funded the presented research; Ossur, in the form of an educational grant for our research fellowship position; the Natural Sciences and Engineering Research Council (NSERC) Discovery: RGPIN-2018-05693; the Ontario Early Researcher Award ER18-14-197; and the Canadian Foundation for Innovation JELF and Ontario Research Fund–Research Infrastructure, 38141. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
