Abstract
Background:
Tranexamic acid (TXA) is widely used across surgical specialties to reduce perioperative bleeding. It has been shown to be effective in spinal surgery and lower limb arthroplasty. Among all languages, there are no systematic reviews or meta-analyses investigating its clinical effectiveness for all types of shoulder surgery.
Purpose:
To investigate the clinical effectiveness of TXA in all types of shoulder surgery, including open and arthroscopic procedures. To investigate the effect of TXA on bleeding and non–bleeding-related outcomes.
Study Design:
Systematic review and meta-analysis.
Methods:
A protocol for the study was designed and registered with PROSPERO (CRD42020185482). The literature search included the MEDLINE, Embase, PsycINFO, and Cochrane Library databases. All randomized controlled trials evaluating the use of TXA against placebo, in all types of shoulder surgery, were included. Assessments were undertaken for risk of bias and certainty of evidence. The primary outcome was total blood loss. Secondary outcomes included those not directly related to bleeding. Data from comparable outcomes were pooled and analyzed quantitatively or descriptively, as appropriate.
Results:
Eight randomized controlled trials were included in the systematic review, and data from 7 of these studies were pooled in the meta-analysis. Pooled analysis demonstrated a significant reduction in 2 of 3 outcomes measuring perioperative bleeding with TXA compared with controls: estimated total blood loss (mean difference, −209.66 mL; 95% CI, −389.11 to −30.21; P = .02) and postoperative blood loss as measured by drain output (mean difference, −84.8 mL; 95% CI, −140.04 to −29.56; P = .003). Hemoglobin reduction was reduced but not statistically significant (mean difference, –0.33 g/dL; 95% CI −0.69 to 0.03; P = .07). This result became significant with sensitivity analysis excluding arthroscopic procedures.
Conclusion:
This systematic review and meta-analysis indicated that TXA was effective in reducing blood loss in shoulder surgery. Larger randomized controlled trials with low risk of bias for specific surgical shoulder procedures are required.
Clinical Relevance:
TXA can be used across shoulder surgery to reduced perioperative blood loss. The use of TXA may have other beneficial features, including reduced postoperative pain and reduced operative time.
Perioperative bleeding is an important consideration when undergoing surgery. Strategies to minimize perioperative blood loss have been developed as part of enhanced recovery protocols in surgery.10,26 Excessive bleeding during surgery may influence length of stay, postoperative pain, swelling, and wound healing.9,35 The rate of blood transfusion after shoulder surgery is relatively low.6,33 However, there is significant increased morbidity associated with whole blood transfusion.15,21 This may include hemolytic reactions, acute lung injury, circulatory overload, and disease transmission.3,14 The use of a tourniquet is not possible in shoulder surgery. Development of other strategies must be utilized to reduce bleeding perioperatively. 32
Tranexamic acid (TXA) is a synthetic derivative of the amino acid lysine. It is an antifibrinolytic agent that binds reversibly to plasminogen, inhibiting its conversion to plasmin. It stabilizes formed clots by preventing fibrin degradation.7,28,30 The use of TXA in orthopaedic surgery has gained popularity because of its clinical efficacy at reducing blood loss. Its use gained popularity in situations where excessive bleeding may be life threatening, such as high-energy trauma. CRASH-2, a large multinational randomized placebo-controlled trial, investigated the clinical effectiveness of TXA on death, vascular-occlusive events, and blood transfusion in 20,000 patients with significant hemorrhage after high-energy trauma. 4 It concluded that the early administration of TXA in these patients reduced the risk of death from hemorrhage. It also found no increased risk of vascular-occlusive events. Other studies have demonstrated safe use of TXA, without an increase in thromboembolic events.8,38,39
Since this landmark clinical trial, TXA has been used effectively across many surgical specialties to reduce blood loss.7,30 In orthopaedics, the use of TXA was initially explored in spinal surgery and lower limb arthroplasty. Recent systematic reviews and meta-analyses have demonstrated its effectiveness in reducing blood loss and transfusion rates.13,18,27 Clinical trials investigating its use in shoulder arthroplasty have also shown favorable results.2,17,22,23,36,41 The first randomized controlled trial (RCT) evaluating the use of TXA in shoulder arthroplasty, by Gillespie et al, 12 was published in 2015. Several RCTs have since been undertaken investigating the effect of TXA on perioperative bleeding in other types of shoulder surgery.5,11,19,25,31,37,40 These have included trials evaluating its use in open procedures as well as arthroscopic procedures. A mutual limitation in all these RCTs has been sample size. The sample size ranges between 54 and 111, likely leading to underpowered studies and possible type II error. A meta-analysis, permitting pooling of data and quantitative synthesis of results, may help provide clarity.
The aim of this systematic review and meta-analysis is to investigate the clinical effectiveness of TXA in shoulder surgery. The primary outcome examined was total blood loss (mL). Other outcomes measuring perioperative bleeding included postoperative blood loss as measured by drain output (mL) and hemoglobin reduction (g/dL). Secondary outcomes included non–bleeding-related outcomes: patient-reported outcome measures, operative time (minutes), hospital length of stay (days), adverse events, and number of blood transfusions.
Methods
This study is reported according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement. 29 A protocol for the study was designed and registered with PROSPERO (CRD42020185482).
Eligibility Criteria
Only RCTs were included. All other trial designs were excluded. Clinical trials of humans of any age undergoing any type of surgery (open or arthroscopic techniques) to the shoulder girdle were included. The intervention of interest was intraoperative use of TXA. All individual methods of delivery, timing, and dose of TXA were included. The comparator was any form of placebo or no treatment. The primary outcome was total blood loss. Other outcomes measuring perioperative bleeding were postoperative blood loss via drain output and reduction in serum hemoglobin. Secondary outcomes were those not directly related to bleeding. These consisted of patient-reported outcome measures, operative time, hospital length of stay, incidence of adverse events, and number of blood transfusions.
Information Sources and Search Strategy
The search strategy involved using the following bibliographic databases: MEDLINE, Embase, PsycINFO, and the Cochrane Library. No restrictions were placed on publication date or language. The Cochrane Group RCT filters for each database were utilized to increase sensitivity and maximize precision in the search strategy. Search terms are cited in Appendix 1 (available in the online version of this article). References from published systematic reviews investigating the same or similar topic were manually searched for eligible studies. The PROSPERO database was searched, which revealed no ongoing or recently completed systematic reviews on this topic. The literature search was repeated before submission, on June 22, 2020, to ensure that no additional studies had been published since the original search was performed.
Study Selection and Data Extraction
All literature search results were combined and collected in Microsoft Excel. Duplicate articles were removed manually. Two independent reviewers (A.W.H., K.H.T.) then screened the titles and abstracts for eligibility. Both reviewers completed this process independently, and a consensus was required before full-text review. Full-text review of all articles meeting the eligibility criteria then determined final inclusion.
Data extraction involved the same 2 independent reviewers. One reviewer extracted the data using a standardized pro forma. The second reviewer checked the extracted data for inaccuracies. Any disagreement in data extraction was resolved by discussion and involvement of a third reviewer (M.S.R.) as necessary. Attempts to contact corresponding authors for studies regarding unclear or missing data or for any additional details required were made via email. Microsoft Excel was used for data capture and RevMan Version 5.3 (Cochrane Collaboration) for data management.
Data extraction included study design, patient cohort, study characteristics, TXA dose/method of delivery, control group intervention, primary outcome measures, and any secondary outcome measures. Means and standard deviations were extracted for all outcome measures. Where the mean or standard deviation was not reported, the corresponding author was contacted to request raw data.
Assessment of Risk of Bias
Two reviewers assessed risk of bias independently (A.W.H., K.H.T.). The Risk of Bias Tool (Version 2.0; Cochrane Collaboration) was used to assess each trial for possible bias. 34 Using this tool, bias was categorized into 5 domains. Each domain was assigned a level of risk of bias (high risk, low risk, or some concerns). Signaling questions within each domain guided interpretation of bias. The tool generated an overall risk of bias for each study. Each study was also assessed using the Jadad Scale as a supplementary method for assessing bias. 20 The Jadad Scale has a maximum of 5 points. Up to 2 points were given for randomization: 1 point for mentioning randomization and an additional point if the method of randomization was appropriate. Up to 2 points were given for blinding: 1 point for mentioning blinding and an additional point if the method of blinding was appropriate. A final point was given if an account of all patients involved in the trial was reported.
To assess for risk of bias across studies, publication bias was assessed by using a funnel plot of studies investigating the primary outcome. Selective reporting within studies may also affect the cumulative evidence when pooling data. This was evaluated by reviewing the trial protocols or trial registrations for each study. If available, the predefined outcomes and statistical analysis plans were compared with what was analyzed and reported in the published article.
Data Synthesis and Statistical Analysis
Data synthesis and analysis were performed in 2 categories based on the outcome measures reported. The 2 categories were outcomes related to bleeding, including the primary outcome (total blood loss) and key secondary outcomes not directly related to bleeding. With a paucity of level 1 RCTs examining the use of TXA in shoulder surgery, data from a minimum of 3 studies were synthesized for each outcome. Data were synthesized only if the method in which the outcome was recorded was comparable. Outcomes with continuous variables, such as blood loss and hemoglobin reduction, were summarized by using mean differences and inverse variance statistical analysis. All analyses were performed using a random effects model given the expected heterogeneity across the studies. Heterogeneity was quantified using the I2 value and the χ2 test for heterogeneity. I2 values were interpreted as described in the Cochrane Handbook: 0% to 40% might not be important; 30% to 60% may represent moderate heterogeneity; 50% to 90% may represent substantial heterogeneity; and 75% to 100% represents considerable heterogeneity. Data related to an outcome reported in <3 studies, without sufficient study power, were not synthesized and were only reported descriptively. The inclusion of all surgical procedures involving the shoulder meant that a variety of outcomes was expected. Given the low rate of transfusion and mortality in shoulder surgery, we expected data for some outcomes to include too few events for pooled statistical analysis, such as rates of blood transfusion and other adverse events. In this case, data were pooled and reported descriptively.
Determining Certainty of Findings
Description of the strength of the body of evidence provided and confidence in the cumulative estimate was assessed using the GRADE (Grading of Recommendations Assessment, Development and Evaluation) approach.1,16,24 In this approach, RCTs all start out as providing evidence of high certainty. The certainty can then be reduced because of risk of bias, inconsistency, indirectness, imprecision, and publication bias. A large magnitude of effect, dose-response gradient, or effect of plausible residual confounding can increase the level of certainty. Each outcome in this case was thus determined as “very low,”“low,”“moderate,” or “high” certainty.
Results
Study Selection
The literature search of Embase, MEDLINE, PsychINFO, and Cochrane Library databases identified 57 records. After removal of duplicates, 28 unique articles were then screened: 18 were excluded after title and abstract screening, and 10 were selected for full-text review. Of these 10 records, 8 were eligible for inclusion as part of the systematic review. Seven were eligible for inclusion in the meta-analysis.5,11,12,19,25,31,37 The study selection process, with reasons for exclusion, is available in the PRISMA flowchart (Figure 1).
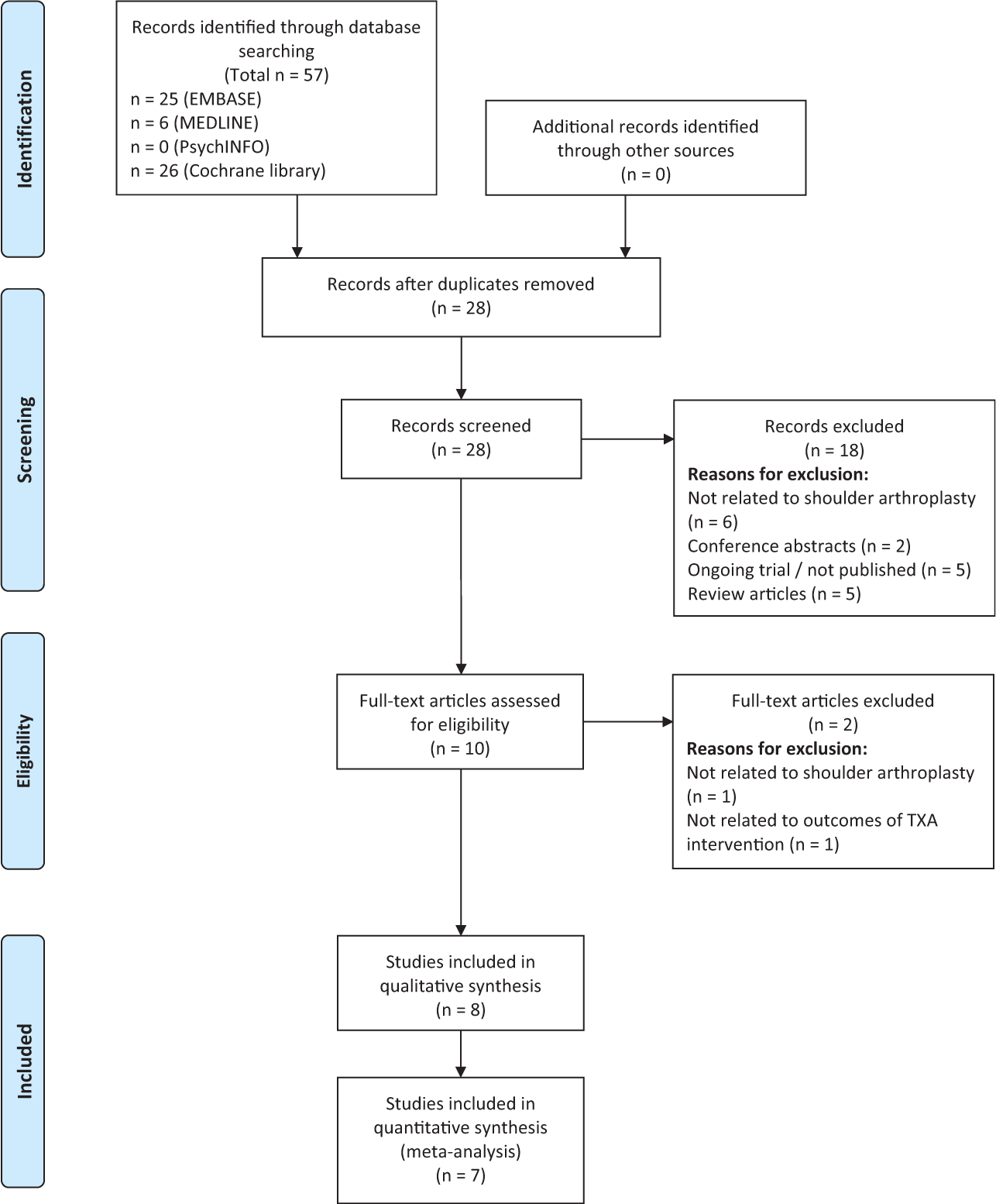
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram for included studies. TXA, tranexamic acid.
Study Characteristics
Trials were published between 2015 and 2020. Individual characteristics are reported in Table 1. Only RCTs were included. Of these, 3 were conducted in the United States,5,12,37 1 in Ireland, 19 1 in Austria, 31 1 in China, 11 1 in South Korea, 40 and 1 in Taiwan. 25 Sample sizes ranged from 54 to 111, with 708 patients (406 received TXA, 302 received placebo). Three studies comprised patients undergoing anatomic or reverse total shoulder arthroplasty (RTSA); 2 studies, RTSA only; 1 study, open Latarjet surgery; and 2 studies, arthroscopic rotator cuff repair. Seven studies compared TXA intervention against placebo. One study compared administration methods of TXA without placebo.
Included Studies and Study Characteristics a
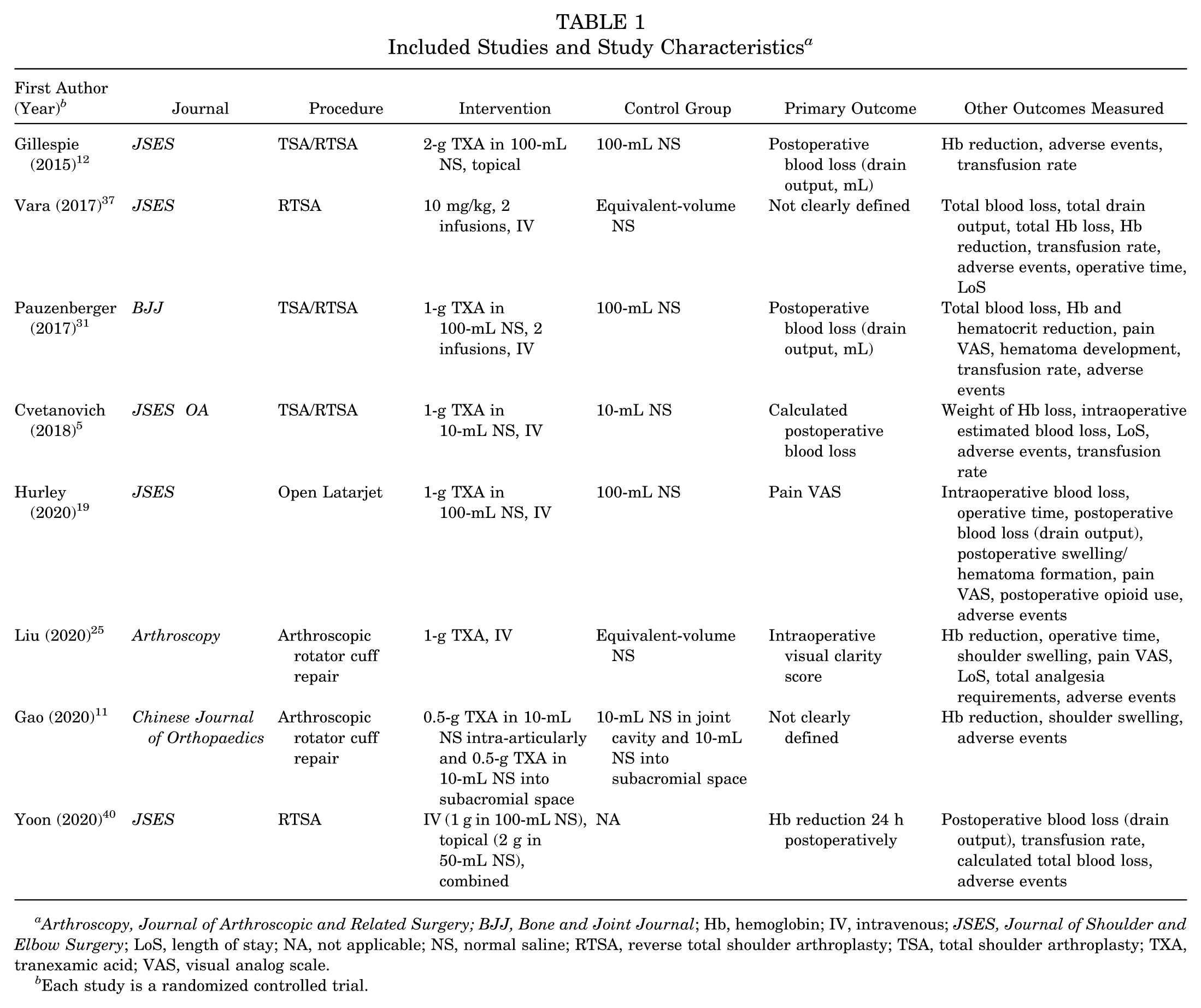
Arthroscopy, Journal of Arthroscopic and Related Surgery; BJJ, Bone and Joint Journal; Hb, hemoglobin; IV, intravenous; JSES, Journal of Shoulder and Elbow Surgery; LoS, length of stay; NA, not applicable; NS, normal saline; RTSA, reverse total shoulder arthroplasty; TSA, total shoulder arthroplasty; TXA, tranexamic acid; VAS, visual analog scale.
Each study is a randomized controlled trial.
Risk of Bias Within Studies
One study was assessed as having a low risk of bias overall 37 and 2 studies, a high risk of bias.19,31 Of these, the randomization and blinding process was assessed as having a high risk of bias. In these trials, the anesthetist was informed of the patient’s group allocation; therefore, blinding of others in the operating room could not be guaranteed. Significant risk of bias may be present when the allocation is not concealed from all staff involved in the operating room, when the intervention or control intervention is administered. One of these trials failed to report on outcome data for all participants randomized, resulting in a high risk of bias. Five trials were assessed to have some concerns of bias overall. “Selection of the reported result” domain was assessed as consistently having some concerns of bias. This was due to the lack of a clear predefined statistical analysis plan finalized before commencement of the trial.
Risk of Bias Across Studies
Substantial heterogeneity was found among studies when analyzing bleeding outcomes: total blood loss (I2 = 81%), postoperative blood loss as measured by drain output (I2 = 88%), and hemoglobin reduction (I2 = 68%). This was expected and likely due to differing surgical procedures and TXA dose/administration. In contrast, the heterogeneity in terms of non–bleeding-related outcomes was low: postoperative pain (19%), operative time (0%), and length of stay (40%) (Figure 2). Using a funnel plot of studies measuring the primary outcome (total blood loss), no clear evidence of publication bias was found (Appendix 2, available online). Based on the small number of studies (n = 4), publication bias cannot be excluded definitively.
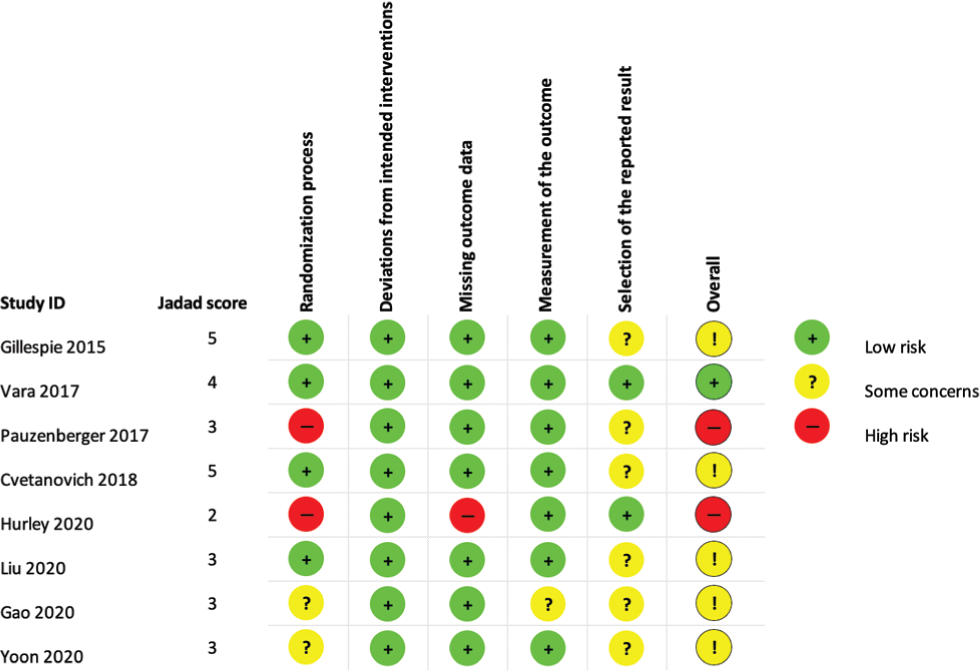
Results of risk-of-bias assessments for included studies based on the Jadad Scale and the Risk of Bias Tool (Version 2.0; Cochrane Collaboration).
Results of Individual Studies
Seven studies in the systematic review were RCTs comparing TXA with placebo. All of these studies compared various doses and administration methods of TXA with a similar volume and administration of normal saline. Gillespie et al 12 found that 2 g of topical TXA significantly reduced postoperative blood loss and hemoglobin reduction in patients undergoing total shoulder arthroplasty (TSA) or RTSA. No transfusion requirements or perioperative complications were seen in either group. Vara et al 37 found that two 10-mg/kg intravenous (IV) TXA transfusions decreased the drop in hemoglobin, postoperative blood loss, and total blood loss in patients undergoing RTSA. Transfusion rates did not differ significantly between groups (5.7% for TXA vs 14.3% for placebo). Pauzenberger et al 31 reported that two 1-g IV TXA transfusions reduced the drop in hemoglobin, postoperative blood loss, and total blood loss in patients undergoing TSA or RTSA. This study noted lower, but not statistically significant, pain scores in the TXA group. No blood transfusions or perioperative complications were seen in either group. Cvetanovich et al 5 reported that 1-g IV TXA transfusion significantly reduced total blood loss in patients undergoing TSA or RTSA. No blood transfusions were administered in either group. One postoperative deep vein thrombosis was observed in the placebo group. Hurley et al 19 investigated TXA use during the open Latarjet procedure. They reported that 1-g IV TXA transfusion significantly reduced postoperative blood loss, as well as day 1 postoperative pain scores. Two studies investigated TXA use in shoulder arthroscopy. Liu et al 25 found no significant benefit of TXA in reducing hemoglobin or total blood loss. Significant improvement in intraoperative visual clarity scores and postoperative pain scores was noted in the TXA group. Gao et al 11 observed no significant reduction in hemoglobin, postoperative bleeding, or shoulder swelling between groups. No thromboembolic complications were seen in the 2 studies investigating TXA use in arthroscopic shoulder surgery.
One study 40 in the systematic review was an RCT comparing the method of administration of TXA in patients undergoing RTSA. This study found no significant difference in postoperative blood loss, hemoglobin reduction, or total blood loss when comparing topical, IV, or combined administration routes. Although the IV group had the highest incidence of transfusion (n = 4), the difference between groups was not statistically significant.
Synthesis of Results
A meta-analysis was performed evaluating bleeding-related and non–bleeding-related outcomes by pooling data from at least 3 studies. Only studies with comparable outcome measures were pooled. One study is not in any meta-analysis because it compared administration methods of TXA rather than the use of TXA versus placebo. 40
Bleeding-Related Outcomes
The administration of TXA resulted in a decrease in blood loss as measured by estimated total blood loss, postoperative blood loss (drain output), and reduction in hemoglobin.
Estimated Total Blood Loss
Total blood loss, based on intra- and postoperative blood loss measurements, was recorded in 4 studies (169 patients in TXA group, 167 in placebo group). The pooled mean difference was −209.66 mL (95% CI, −389.11 to −30.21; P = .02; I2 = 81%), indicating a significant reduction when using TXA (Figure 3).
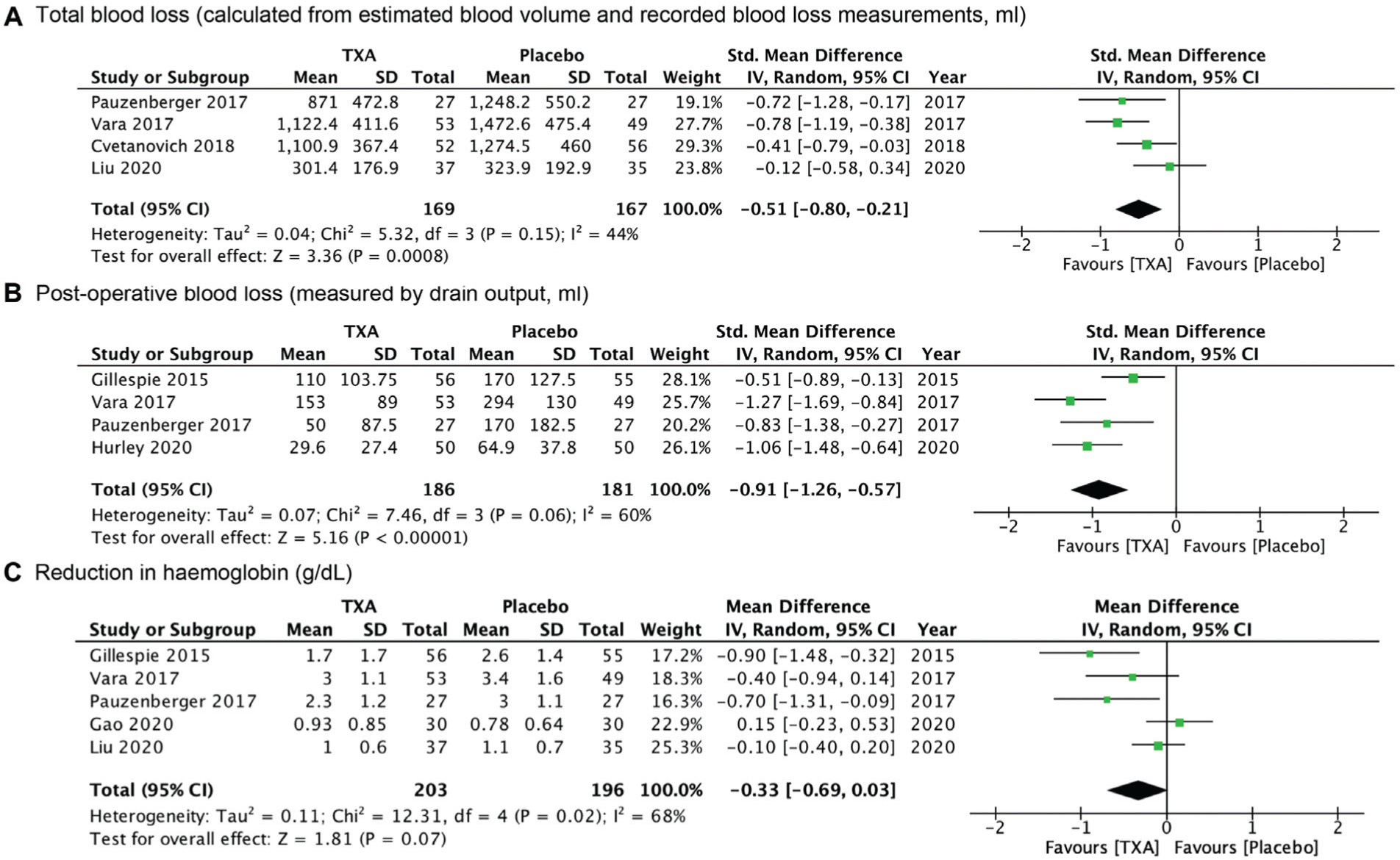
Forest plots of bleeding-related outcomes: (A) total blood loss (mL), (B) pooled change in drain output (mL), and (C) reduction in hemoglobin (g/dL) with tranexamic acid (TXA) as compared with placebo. IV, inverse variance.
Postoperative Blood Loss as Measured by Drain Output
Of the studies, 4 measured postoperative blood loss via drain output at 24 hours postoperation (186 patients in TXA group, 181 patients in placebo group).12,19,31,37 A significant reduction in postoperative blood loss was observed with a pooled mean difference of −84.8 mL (95% CI, −140.04 to −29.56; P = .003; I2 = 88%) (Figure 3).
Reduction in Hemoglobin
Five studies compared the change in hemoglobin from preoperative level to day 1 postoperative level (203 patients in TXA group, 196 in placebo group). This revealed a difference in favor of TXA with a pooled mean difference of −0.33 g/dL (95% CI, –0.69 to 0.03; P = .07; I2 = 68%) (Figure 3). A sensitivity analysis was performed to assess hemoglobin reduction in studies with open surgical procedures only. Exclusion of the 2 studies involving arthroscopic procedures produced only a statistically significant hemoglobin reduction, with substantially decreased heterogeneity (mean difference, −0.65; 95% CI, −0.98 to −0.32; P = .0001; I2 = 0%). This result is likely not clinically significant.
Non–bleeding-related Outcomes
Postoperative Pain
Postoperative pain was measured using the visual analog scale in 3 studies (114 patients in TXA group, 112 patients in placebo group). The pooled mean difference was −1.06 (95% CI, −1.48 to −0.65; P < .00001; I2 = 19%), revealing a significant reduction in postoperative pain day 1 (Figure 4).
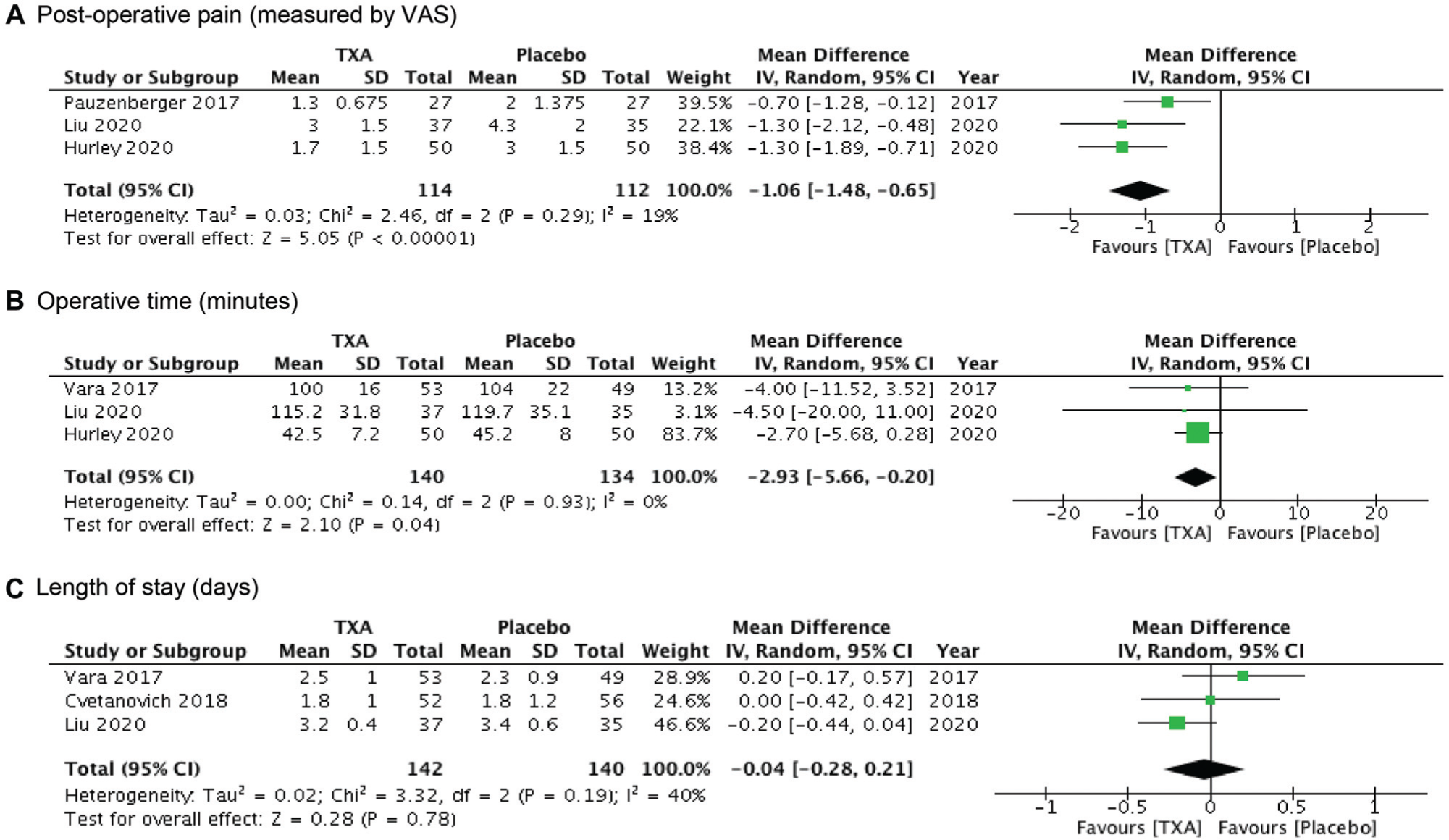
Forest plots for non–bleeding-related outcomes: (A) pooled change in postoperative pain (visual analog scale [VAS]), (B) operative time (minutes), and (C) length of stay (days) with tranexamic acid (TXA) as compared with placebo. IV, inverse variance.
Operative Time
Three studies measured operative time in minutes in 274 patients (140 patients in TXA group, 134 patients in placebo group). The pooled mean difference was −2.93 minutes, which is not clinically significant (95% CI, −5.66 to −0.20; P = .04; I2 = 0%) (Figure 4).
Length of Stay
Three studies reported hospital length of stay in days (142 patients in TXA group, 140 patients in placebo group). No significant difference between the groups was noted (mean difference, −0.04 days; 95% CI, −0.28 to 0.21; P = .78; I2 = 40%) (Figure 4).
Adverse Events
Five studies recorded the need for blood transfusion. Three of these studies reported no blood transfusion requirements at all. The transfusion rate was 3.15%, with 15 of 476 participants requiring a blood transfusion. Four of these studies compared the use of TXA against the use of placebo. Three patients required a transfusion in the TXA group (3/188; transfusion rate, 1.56%) versus 7 patients in the placebo group (7/187; transfusion rate, 3.74%). A study comparing method of TXA delivery reported 5 transfusion events (4 in the IV group and 1 in the topical group). The event rate for blood transfusion across all studies was low and precluded statistical analysis.
All studies recorded the incidence of varying complications or adverse events. One patient receiving TXA had a minor complication described as a syncopal fall. There were no thromboembolic complications recorded in the TXA group in any trial. Four patients in the control group, who did not receive TXA, reported complications: an allergic skin reaction, a non–ST-elevated myocardial infarction, a deep vein thrombosis, and a surgical revision. The rate of blood transfusions or complications in either group was low.
Certainty of Meta-analysis Findings
The quality of evidence was rated for all outcomes using the GRADE approach (Table 2). The highest level of certainty for any outcome considered was “moderate” for total blood loss and operative time. All other outcomes were assessed as either low or very low. The GRADE score of 3 outcomes was upgraded because of a large effect size. This was indicated by a test for overall effect Z score >2. A Z score of 1 is equivalent to 1 SD; a Z score >2 was chosen, as 2 SDs away from the mean is widely accepted as significant. Outcomes related to bleeding were assessed as higher importance than the non–bleeding-related outcomes. Postoperative blood loss and total blood loss outcomes were assessed as “critical.”
Findings and GRADE Evidence Profiles of Randomized Controlled Trials a
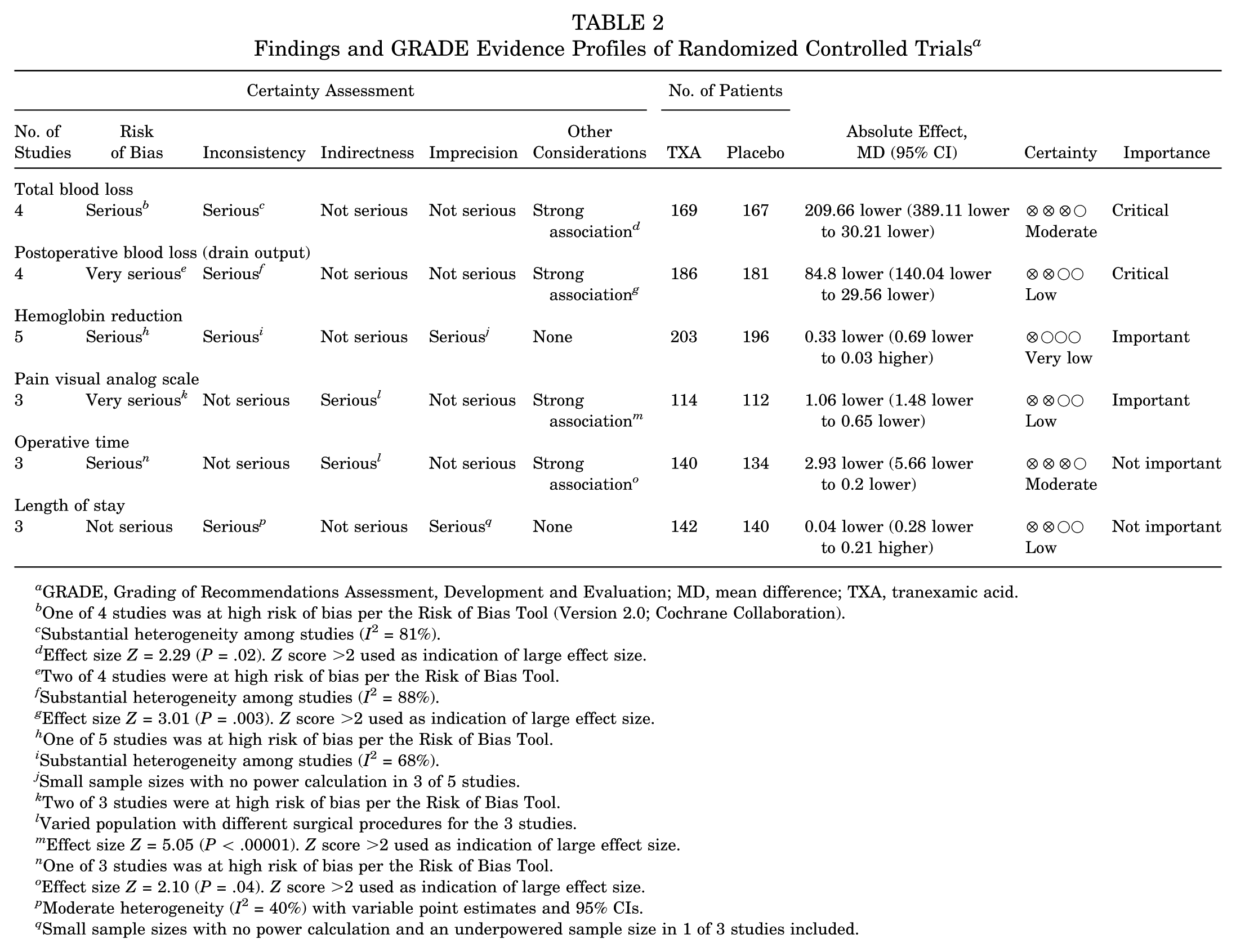
GRADE, Grading of Recommendations Assessment, Development and Evaluation; MD, mean difference; TXA, tranexamic acid.
One of 4 studies was at high risk of bias per the Risk of Bias Tool (Version 2.0; Cochrane Collaboration).
Substantial heterogeneity among studies (I2 = 81%).
Effect size Z = 2.29 (P = .02). Z score >2 used as indication of large effect size.
Two of 4 studies were at high risk of bias per the Risk of Bias Tool.
Substantial heterogeneity among studies (I2 = 88%).
Effect size Z = 3.01 (P = .003). Z score >2 used as indication of large effect size.
One of 5 studies was at high risk of bias per the Risk of Bias Tool.
Substantial heterogeneity among studies (I2 = 68%).
Small sample sizes with no power calculation in 3 of 5 studies.
Two of 3 studies were at high risk of bias per the Risk of Bias Tool.
Varied population with different surgical procedures for the 3 studies.
Effect size Z = 5.05 (P < .00001). Z score >2 used as indication of large effect size.
One of 3 studies was at high risk of bias per the Risk of Bias Tool.
Effect size Z = 2.10 (P = .04). Z score >2 used as indication of large effect size.
Moderate heterogeneity (I2 = 40%) with variable point estimates and 95% CIs.
Small sample sizes with no power calculation and an underpowered sample size in 1 of 3 studies included.
Discussion
The results of this systematic review and meta-analysis reveal a significant reduction in perioperative blood loss with the use of TXA in shoulder surgery when compared with placebo. The primary outcome of total blood loss, measured by several methods, including total blood loss (in mL), drain output (in mL), and hemoglobin reduction (in g/dL), demonstrated significant benefit in the TXA group. Hemoglobin reduction was statistically significant only when studies relating to arthroscopic procedures were removed. This result was not clinically significant. For non–bleeding-related outcomes, a significant reduction in postoperative pain with the use of TXA was noted. Operative time was reduced in the TXA group; however, this may not be clinically significant. Complication rates were very low, including the need for blood transfusion, and no thromboembolic complications were associated with TXA use.
The results of this study are comparable with those of other systematic reviews and meta-analyses in the literature regarding the use of TXA in shoulder surgery. Kuo et al 23 examined the use of TXA in TSA and RTSA. They found an estimated total blood loss reduction of 249 mL, a hemoglobin reduction of 0.64 g/dL, and a drain output reduction of 95 mL. Box et al 2 investigated TXA administration for anatomic TSA and RTSA. They observed a reduction in estimated total blood loss (242 mL), hemoglobin reduction (0.70 g/dL), and drain output (114 mL). The results of the current meta-analysis produced similar results, but the values are lower. Hemoglobin reduction was also found not to be statistically significant with the inclusion of arthroscopic procedures. The inclusion of arthroscopic procedures, where blood loss volume is expected to be minimal, would likely explain these differences to the findings.
There was dissonance between systematic reviews and meta-analyses on the reduction of blood transfusions with use of TXA in shoulder surgery. Three meta-analyses reported the rate of blood transfusion to be lower with the use of TXA.17,23,36 Three meta-analyses noted no statistically significant reduction in rate of blood transfusion with TXA administration.2,22,41 Unlike that in lower limb arthroplasty or major trauma surgery, the rate of blood transfusion after shoulder surgery was very low and precluded robust statistical analysis in this meta-analysis.
Three meta-analyses investigated differences in operative time and hospital length of stay.23,36,41 All 3 studies found no significant difference. This meta-analysis did reveal a small but significant reduction in operative time. This difference is due to the inclusion of other surgical procedures of the shoulder, including the Latarjet and arthroscopic procedures. The potential benefit of reduced bleeding from TXA use in arthroscopic procedures differs from that of open surgery. In arthroscopic shoulder surgery, TXA may improve visual clarity, while improvements in other bleeding-related outcomes, such as total blood loss, may be negligible. This is due to the minimal amount of bleeding that occurs in shoulder arthroscopy as compared with open procedures.
He et al 17 reported a negative correlation between the dose of TXA and blood loss (via drain output). The optimal dose/administration method of TXA is yet to be determined in shoulder surgery and needs investigation.
A major strength of this systematic review and meta-analysis is that it provides a comprehensive review of the literature from large databases and includes very recent publications. In addition, the search was not limited by language (1 study from China was included). The robust assessment of bias is a further strength of this review. Bias was assessed within individual studies using the Risk of Bias Tool and the Jadad Scale. Bias was also assessed across studies, with comparable outcomes, using the GRADE method.
This study has several limitations. The heterogeneity among studies was significant. Studies investigated TXA use in a variety of surgical procedures relating to the shoulder. We included all studies to increase statistical power and maximize sample sizes by pooling results. Increased numbers of RCTs investigating the use of TXA in arthroscopy are required to review this topic separately. The overall benefit of TXA use is still applicable in all surgical procedures; however, the benefits may differ per the need to reduce bleeding for given operations.
The dosing regimen and route of administration of TXA differed among studies. The efficacy of TXA based on its route of administration has been reviewed in studies involving lower limb arthroplasty, with contradictory results. Yoon et al 40 performed the first trial comparing the effects of TXA, when used in shoulder arthroplasty, among routes of administration: IV, topical, and combined. They concluded that blood loss did not differ among TXA administration methods. Further research is required to compare the administration methods of TXA as well as the dosing regimen. A dose-response may add weight to the certainty of evidence provided and should be investigated. Another limitation of this study is the small number of RCTs, each with a small sample size. This raises the potential of type II error. Event rates of blood transfusion and adverse events, including thromboembolic events, were very low. This meant that we were unable to perform a meta-analysis for these outcomes.
The results of this study complement the current literature regarding the use of TXA in shoulder surgery. It highlights the efficacy of TXA in reducing blood loss. However, there remains a paucity of high-quality RCTs in this area. For increased confidence, future research should aim for large RCTs with low risk of bias. To reduce a common source of assessment bias, an independent pharmacist should prepare TXA and placebo doses in identical syringes to allow effective allocation concealment and blinding of all staff members involved in the patients’ care. Trials should be registered in a trial registry. Trial protocols with a predefined statistical analysis plan should be published in advance of trial commencement to reduce potential reporting bias. The primary outcomes may differ by the procedure examined. Blood loss is likely more prominent and critical in arthroplasty, whereas visual clarity or operative time may be better suited to arthroscopic procedures.
Conclusion
This systematic review and meta-analysis indicated that TXA was effective in reducing blood loss in shoulder surgery. Larger RCTs with low risk of bias are required for specific surgical shoulder procedures. The use of TXA may have other beneficial features, such as reduced postoperative pain and reduced operative time.
Supplemental Material
sj-pdf-1-ajs-10.1177_0363546520981679 – Supplemental material for Clinical Effectiveness of Intraoperative Tranexamic Acid Use in Shoulder Surgery: A Systematic Review and Meta-analysis
Supplemental material, sj-pdf-1-ajs-10.1177_0363546520981679 for Clinical Effectiveness of Intraoperative Tranexamic Acid Use in Shoulder Surgery: A Systematic Review and Meta-analysis by Alexander W. Hartland, Kar H. Teoh and Mustafa S. Rashid in The American Journal of Sports Medicine
Footnotes
Submitted June 28, 2020; accepted September 1, 2020.
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
An online CME course associated with this article is available for 1 AMA PRA Category 1 Credit™ at ![]() . In accordance with the standards of the Accreditation Council for Continuing Medical Education (ACCME), it is the policy of The American Orthopaedic Society for Sports Medicine that authors, editors, and planners disclose to the learners all financial relationships during the past 12 months with any commercial interest (A ‘commercial interest’ is any entity producing, marketing, re-selling, or distributing health care goods or services consumed by, or used on, patients). Any and all disclosures are provided in the online journal CME area which is provided to all participants before they actually take the CME activity. In accordance with AOSSM policy, authors, editors, and planners’ participation in this educational activity will be predicated upon timely submission and review of AOSSM disclosure. Noncompliance will result in an author/editor or planner to be stricken from participating in this CME activity.
. In accordance with the standards of the Accreditation Council for Continuing Medical Education (ACCME), it is the policy of The American Orthopaedic Society for Sports Medicine that authors, editors, and planners disclose to the learners all financial relationships during the past 12 months with any commercial interest (A ‘commercial interest’ is any entity producing, marketing, re-selling, or distributing health care goods or services consumed by, or used on, patients). Any and all disclosures are provided in the online journal CME area which is provided to all participants before they actually take the CME activity. In accordance with AOSSM policy, authors, editors, and planners’ participation in this educational activity will be predicated upon timely submission and review of AOSSM disclosure. Noncompliance will result in an author/editor or planner to be stricken from participating in this CME activity.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
