Abstract
Introduction
Tranexamic Acid (TXA) has been associated with improved range of motion (ROM) in knee arthroplasty literature. The primary aim of this study is to assess the association between perioperative TXA in Total Shoulder Arthroplasty (TSA) and ROM up to 12-months postoperatively. The authors hypothesized there would be no difference between groups.
Methods
A single-institution retrospective cohort study of patients who underwent TSA between 2012–2022 was performed. Shoulder ROM was recorded pre-operatively, 3-months, 6-months, and 9–12-months postoperatively. Secondary outcomes including blood loss, transfusions, pain scores, and 90-day complications were collected. Statistical analyses were performed using STATA/SE.
Results
A total of 551 TSAs were included. Pre-operative elevation was greater in the TXA group (90.1° vs 74.0°, p < .001). Elevation was significantly greater at 3, 6, and 9–12 months postoperatively in the TXA group (106.7°, 124.7°, 129.4°) compared to controls (95.0°, 109.3°, 117.1°) (p < .006). When propensity match analysis was performed to control for preoperative ROM, elevation was significantly greater at 6 months in the TXA group (p = .001).
Conclusion
Patients who received perioperative TXA had significantly improved elevation at 6-months following TSA compared to controls. The results suggest there may be a link between postoperative ROM and TXA in TSA.
Keywords
Introduction
The incidence of Total Shoulder Arthroplasty (TSA) has increased significantly since 2000, with approximately 100 000 TSA procedures now performed yearly. 1 This number is increasing at a significantly higher rate than lower extremity joint arthroplasty, which signals a need for prompt updates to the existing TSA literature. 2 Both Reverse and Anatomic TSA are common in the United States and have demonstrated favorable outcomes.3,4 Tranexamic acid (TXA) is an antifibrinolytic agent that has demonstrated effectiveness in reducing bleeding in major Orthopaedic surgeries. 5 Because of its low risk of complication and potential upside, TXA has been extensively investigated in lower extremity joint arthroplasty literature. Repeatedly, it has been shown to decrease blood loss and transfusion rates following Total Hip and Total Knee Arthroplasty (TKA).5–10
While much of the enthusiasm for TXA in Orthopaedic Surgery revolves around the lower extremity literature, its effects are less delineated in open shoulder surgery, including Anatomic and Reverse TSA. The available literature suggests TXA may reduce blood loss in TSA, although many of these studies have not demonstrated this relationship with statistical significance and transfusion rates are consistently reported as unchanged.11–13 It is possible that blood loss in the upper extremity is less to begin with, and therefore TXA may have no significant impact on intraoperative blood loss or postoperative transfusion rates. It is also possible that a link has not yet been demonstrated given there is less literature on the subject. Many of the studies that do exist on the topic may be underpowered given the relative infrequency of the procedure versus TKA and THA. 2
Beyond intraoperative blood loss and transfusion rates, previous studies have investigated secondary benefits to patients who received perioperative TXA in lower extremity joint arthroplasty. Publications in TKA literature have documented improved range of motion (ROM) and better ambulation in patients who received TXA.14–18 It is unknown whether perioperative TXA has an impact on ROM following open shoulder surgery. Herein, we seek to define the impact of TXA on various TSA outcomes. The primary outcome of interest is postoperative ROM in the first year following surgery between patients who received perioperative TXA versus those who did not. Secondarily, the authors aimed to characterize the effect of TXA on estimated blood loss (EBL), transfusion rates, postoperative pain scores, need for physical therapy after 6-months, and 90-day complication rates after TSA. The authors hypothesized that there would be no difference regarding primary or secondary outcomes between patients who received perioperative TXA and controls.
Materials and Methods
Study Design
We performed a single-institution retrospective cohort study of patients who underwent TSA from January first, 2012, to January first, 2022. The study was approved by the institution's Institutional Review Board under IRB#22-166. Informed consent was waived in accordance with the institution's policies. All procedures were performed by fellowship-trained shoulder/elbow or upper extremity surgeons at one institution. Data were collected via chart review and compiled in a secure database with patient identifiers removed for confidentiality. Chart review included operative reports, progress notes, discharge summaries, emergency department notes, outpatient clinic notes, and physical therapy notes.
Patient Selection
Patients were considered eligible if they underwent Anatomic, Reverse, or Hemi-arthroplasty within the data collection period. All revision procedures were excluded from the study. Those with missing information, including postoperative ROM values, were also excluded (Figure 1). Patients who had bilateral TSA performed within the study's time period only had their first arthroplasty recorded and the second was excluded. Primary demographic data including age, sex, race, ethnicity, height, weight, and body mass index (BMI) were recorded. Patients’ cardiovascular and diabetic comorbidities, as well as preoperative American Society of Anesthesiologist (ASA) scores, were recorded. Cardiovascular comorbidities were defined as diagnoses of hypertension and/or coronary artery disease. Administration of perioperative TXA was identified and recorded based on anesthesia documentation.

Flow Diagram Representing Cohort Identification and Exclusion Criteria.
Outcome Measures
The primary outcome was postoperative ROM at 3, 6, and 9–12 months after surgery. When available, ROM values were recorded according to Orthopedic Surgeon clinic notes. In the event of incomplete data or missing physician notes, ROM measurements were recorded from physical therapy notes. If no data were available after this process, the patient was excluded from the study (Figure 1). Secondary outcomes included EBL documented in operative reports, postoperative blood transfusions, prescription for physical therapy after 6-months, pain analog scores at 6-months, and 90-day complications. A pain score greater or equal to 5/10 was considered significant pain. 90-day complications were defined as ipsilateral shoulder-related events leading to emergency room visits, readmittance or reoperation. Complications were collected via emergency department notes, inpatient history and physical notes, outpatient clinic notes, and operative reports.
Statistical Analysis
All data were descriptively analyzed. Continuous variables were reported as mean ± SD or median (IQR) and categorical variables were expressed as frequency (percentage), respectively. To compare TXA and control cohorts, a two-sample t-test or Mann-Whitney U test and Fisher's exact test or Pearson's Chi-square test were used for continuous variables and categorical variables as appropriate. In addition, a propensity score analysis was performed for elevation at 3-months, 6-months, and 9–12-months. Propensity scores for receiving TXA were estimated as the predicted probabilities from a logistic regression model with the following covariates: age, gender, race, BMI, pre-operative elevation, and type of surgery. A two-tailed p-value < .05 was considered statistically significant. All statistical analyses were performed using STATA/SE software version 18.0 (StataCorp LP, Texas, USA).
Results
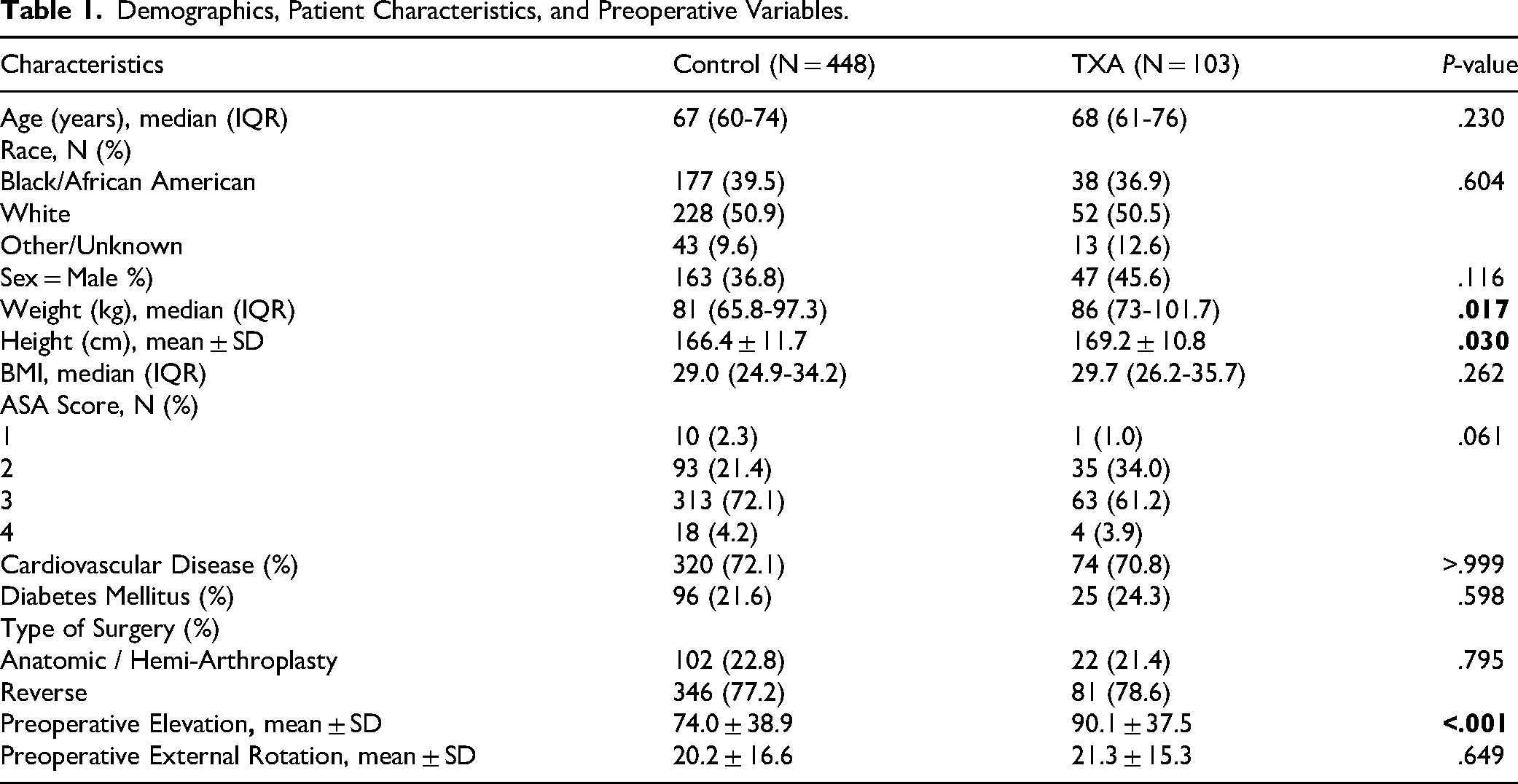
Demographic data and patient characteristics are summarized in Table 1. 551 shoulders fit criteria and were included in the analysis (Figure 1). There were no significant differences regarding patient age, sex, race, BMI, or prevalence of diabetes mellitus or cardiovascular disease between TXA and control cohorts. Pre-operative elevation was greater in the TXA group (90.1° vs 74.0°, p < .001). Pre-operative external rotation was not significantly different between groups (20.2° vs 21.3°, p = .65). Postoperative variables are described in Table 2. Forward elevation was significantly greater at 3, 6, and 9–12 months postoperatively in the TXA group (106.7°, 124.7°, 129.4°, respectively) than in the control group (95.0°, 109.3°, 117.1°, respectively) (p < .006). External rotation was greater at 6- and 9-months in the TXA group (31.6° and 34.6°, respectively) compared to the control group (23.1° and 22.8°, respectively); however, this association did not reach statistical significance (p = .067-.068). EBL, transfusion rates, postoperative pain scores, physical therapy prescriptions after 6-months, and 90-day complication rates between TXA and control groups were not significantly different (Table 2). After propensity-score matching, there was a significant difference between TXA and controls in elevation at 6-months with an average of 11.0° [95% confidence intervals (CI): 4.6°, 17.5°], p = .001 (Table 3).
Demographics, Patient Characteristics, and Preoperative Variables.
Postoperative Variables by TXA Given.

Propensity-Matched Analysis for Elevation.

Discussion
Available literature suggests TXA may decrease postoperative swelling and promote early ROM following lower extremity arthroplasty.15–18 The present study is the first we are aware of to assess the association between TXA administration and ROM in TSA. The authors hypothesized that forward elevation and external rotation would be unchanged in patients who did and did not receive perioperative TXA. This null hypothesis was rejected by the findings of the study. When preoperative range of motion was controlled, elevation was significantly increased at 6-months following surgery in those who received TXA. The TXA cohort also demonstrated increased external rotation at 6- and 9–12 months postoperatively, although this association narrowly missed statistical significance. There was not enough documentation available in our patient cohorts’ EMR to assess internal rotation or glenohumeral abduction.
The data of the present study suggests that TXA administration may be associated with improved ROM during the rehabilitative course following TSA. Interestingly, an association between TXA and ROM has been demonstrated in rotator cuff repair. In a blinded randomized control trial, Mackenzie et al 19 demonstrated significantly improved forward elevation (159° vs 144°, p = .08), internal rotation (8.0° vs 6.1°, p = .004), and external rotation (58° vs 45°, p = .003) at 6-months follow up after arthroscopic rotator cuff repair in patients who were given TXA versus placebo. In the same study, there was a reduced rate of secondary adhesive capsulitis at 6-months in the TXA group versus placebo, although this association fell just short of statistical significance (p = .066). The authors hypothesized that TXA may reduce the inflammatory effect of hematoma in the subacromial space and limit scar and adhesion formation, subsequently increasing ROM after surgery. They also posit that a reduced intraarticular hematoma burden may decrease capsular pain and allow earlier and more effective postoperative ROM. 19
In the present study, pain ≥5/10 on a visual analog scale was not significantly different between the TXA group and controls at 6-months follow up. We do not possess early postoperative pain data. It is known that early postoperative hemarthrosis in total joint arthroplasty is associated with elevated pain in the immediate preoperative period. 20 Therefore it is plausible that reduced postoperative pain afforded by TXA may have contributed to favorable early motion in the present study. Liu et al 21 demonstrated patients who were administered TXA perioperatively had significantly lower immediate postoperative visual analog pain scores compared to placebo after shoulder arthroscopy (3.0 ± 1.5 vs 4.3 ± 2.0, p = .009). In their study, postoperative analgesic usage was also significantly lower in the TXA group versus placebo (9.6 ± 9.7 morphine milligram equivalent vs 14.7 ± 13.4 morphine milligram equivalent, p = .037). Mackenzie et al 19 reported improved visual analog pain scores as long as eight-weeks postoperatively after rotator cuff repair in TXA patients versus placebo (1.7 ± 0.5 vs 2.3 ± 0.6, p = .044). It is certainly possible that reduced pain in the postoperative period may contribute to successful early physical therapy after shoulder surgery, eventually amounting to significant differences in elevation at the 6-month mark.
Similar trends have been reported in lower extremity arthroplasty literature. Serrano Mateo et al demonstrated that topical application of TXA in TKA was associated with improved early functional recovery 6-weeks postoperatively measured by KSS scores (p < .001). 22 Dorweiler et. al. demonstrated significant differences in knee extension, flexion, and total arc ROM at postoperative days 1–3 between TXA and control patients. 15 Grosso et. al. 18 reported that patients who received TXA perioperatively ambulated 20% greater total distance than controls in the first five PT sessions following TKA (p < .001). Naylor et al found that increased discharge ROM after TKA was associated with improved knee ROM at both 8-weeks and 1-year follow up (p = .013).23,24 In a prospective randomized control trial, Wang et. al., 17 observed increased knee flexion 6-weeks status post TKA in their TXA group versus placebo (111.5° vs 103.5°, p = .007). A mechanism underlying improved ROM after perioperative TXA administration still needs to be elucidated. The most hypothesized mechanism in the TKA literature is reduced postoperative hemarthrosis formation.15,17 This may allow more efficient early mobilization. Additional mechanisms to explain this link include reduced postoperative pain and decreased scar/adhesion formation.19,21
The secondary aim of the present study was to assess the association between TXA and EBL, transfusion rates, postoperative pain, physical therapy after 6-months, and complications requiring ED visits, readmissions, or reoperation. No significant differences were identified between groups for any secondary outcome measure. This is consistent with previously published literature assessing TXA in TSA, which bolsters the reliability of our findings as a whole. Indeed, in a prospective, double-blinded randomized controlled trial, Cvetanovich et al 13 found no difference in EBL, length of stay, transfusion rates, thromboembolic events, or reoperation/readmission after TSA between their TXA group and control group. In a separate randomized controlled trial, Gillipsie et al 12 reported no significant differences in EBL, transfusion rate, or perioperative complications after TSA between their TXA group and controls. Finally, Vara et al 11 conducted a randomized control trial that demonstrated significant decreases in total blood loss, drain output, total hemoglobin loss, and postoperative-day-one transfusion rates in those who received TXA; however, overall transfusion rates, surgical site complications, and systemic complications did not differ between TXA and placebo groups. Rojas et al 25 conducted a systematic review and meta-analysis confirming no evidence of an association between TXA and transfusion rates, hematoma formation, or infection rates in TSA in the available current literature. Our findings are consistent with this data and add to the existing body of literature to support a different relationship between TXA and shoulder arthroplasty versus the relationship observed in lower extremity arthroplasty, where TXA has been shown to decrease many of the above variables. 6
The present study has important limitations. Inherently, a retrospective study design is predisposed to selection biases. These biases make it challenging to control for all potential confounding variables and establish causation. Next, and specific to the present study, the primary outcome variable was significantly different between groups preoperatively. The TXA group had significantly greater elevation at baseline versus controls. This may be explained by increasing institutional use of TXA for open shoulder surgery over the course of the study's time period. Patients who had TSA performed later in the study period were more likely to have received TXA and may have also had less significant preoperative shoulder arthrosis given TSA's rise in popularity over the last decade. The authors performed a propensity-matched analysis to account for preoperative differences between groups which did reveal important insights detailed throughout our report. Another limitation is that ROM was recorded by multiple providers in the electronic medical record. When surgeon clinic notes were unavailable or incomplete, physical therapy notes were utilized. This certainly may have introduced the risk of inter-observer variability in motion assessment. It's been shown that inter-observer reliability in the assessment of shoulder ROM has a correlation coefficient ranging from 0.77–0.93. This agreement is considered moderate-excellent, however any inter-observer variability should be considered when interpreting the study's results. 26 Lastly, while the present study does demonstrate a statistically significant difference in ROM following TXA administration at 6-months postoperatively, there are no functional outcomes included in our report. This data would help clarify the clinical benefit of improved ROM after TXA administration. Future prospective studies are required to further assess the relationship between TXA and postoperative motion in multiple planes and correlate improved ROM with patient-reported outcomes to further support the routine use of perioperative TXA in shoulder arthroplasty.
Conclusion
Patients who underwent TSA who received perioperative TXA had improved external rotation up to one year postoperatively, narrowly missing statistical significance (p = .067-.068). Propensity-matched analysis revealed that patients who received perioperative TXA had significantly improved elevation at 6-months following TSA versus those who did not receive TXA (p = .001). The results of the current study suggest there may be a link between postoperative ROM and TXA in TSA.
Footnotes
Ethical Considerations
Approved by institution's Institutional Review Board under IRB#22-166
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
