Abstract
Background
Photophobia is experienced by most individuals with migraine, and it is frequently the most bothersome symptom other than headache. Light is not perceived uniformly; spectral composition and intensity influence discomfort and can exacerbate symptoms. While monochromatic green light shows therapeutic potential, it appears visually unnatural and is impractical for daily use. This study aimed to determine whether spectrally tailored white light, more visually acceptable in everyday environments, reduces light sensitivity among those with migraine.
Methods
Twenty adults (18–44 years) with episodic migraine completed two research sessions: one during an acute migraine attack and one between migraine attacks. Participants were exposed to four spectrally distinct white light conditions (red-, green-, cyan-, and blue-enriched) in a randomized order delivered at ten intensity levels (50–590 lux). After each exposure, visual discomfort was rated on a 0–10 scale. Pupil size was measured using a pupillometer. Repeated-measures ANOVAs examined the effects of light spectrum, intensity, and migraine state on visual discomfort and pupil response.
Results
Migraine attack status significantly increased visual discomfort (
Conclusion
Spectral composition, light intensity, and migraine state jointly influence both subjective and physiological responses to light. Across lighting spectra, lower light intensity was consistently associated with reduced visual discomfort, indicating greater visual comfort at lower intensities. These results identify green-enriched white light at lower intensities as a visually acceptable, migraine-conscious lighting option with potential for real-world application.
Introduction
Migraine is a leading cause of disability and impaired quality of life.1,2 Migraine limits work, household responsibilities, social participation, and relationships.3–5 One of migraine's most frequent and disabling symptoms is photophobia, defined as discomfort or pain in response to light.6,7 Nearly all individuals experience photophobia during migraine attacks, with about half identifying it as their most bothersome symptom other than headache. 6 For many, sensitivity to light also lingers between attacks.8,9 Light is also consistently identified as a common migraine attack trigger. 10 These relationships between migraine, lighting, and photophobia make indoor lighting environments particularly relevant when considering migraine management.
Recent research has advanced our understanding of the physiological mechanisms linking light to migraine-related pain.11,12 Studies show that light activates trigeminal nucleus caudalis neurons via intraocular neurovascular mechanisms. 13 Intrinsically photosensitive retinal ganglion cells (ipRGCs), involved in non-image-forming vision, relay light signals to the posterior thalamus and then to somatosensory and visual cortices, regions associated with pain processing, vision, and sensory integration.14,15 These findings help explain the acute pain and discomfort that light can cause in people with migraine.
Building on this neurophysiological foundation, several studies have explored the effects of specific wavelengths of light on migraine symptoms. Research suggests that blue (∼420 nm) and red (∼640 nm) light intensify headache pain, whereas green light (∼540 nm) may reduce it.16–18 Thresholds for visual discomfort are higher with green light compared to other colors. These findings have informed the development of light-based interventions, such as narrow-band green light therapy, for migraine relief. 17 Most prior studies investigating light-based interventions for migraine have relied on monochromatic green light delivered in dim condition, often during acute attacks.17,19 For instance, Lipton et al.17 tested a narrow-band green light lamp used exclusively during migraine episodes in darkened rooms, while Martin et al.19 implemented green LED strips emitting in 4–100 lux as a migraine relief strategy in a home setting.
Though these studies showed reductions in headache symptoms, their lighting conditions, visually unnatural, dim, and green in color, are not suitable for everyday environments where people need to read, work, or interact socially. In addition, laboratory protocols often involved direct retinal exposure through devices like the Ganzfeld Color Dome, which fail to represent the ambient lighting typical of real-world indoor spaces. 16
While green light may cause less discomfort than blue, amber or red wavelengths, it lacks the visual neutrality and aesthetic acceptability of white light, which better supports task performance and comfort.20,21 Humans evolved under broad-spectrum daylight, and prolonged exposure to non-white colored light can contribute to visual fatigue and reduced productivity.22,23 Thus, there is a need for studies that evaluate lighting for individuals with migraine under realistic spectral and environmental conditions that reflect daily life. Few studies have investigated how spectral composition and intensity of white light affect light sensitivity and visual discomfort. To the authors knowledge, no study has assessed how these responses vary across patients with migraine, migraine states (during vs. between attacks), or included physiological markers like pupil response alongside subjective ratings. This study addresses these gaps by evaluating how white lights enriched with different spectral peaks (green, blue, cyan, red) and intensities (ranging from 50 to 590 lux) affect subjective discomfort and pupil size, a physiological marker of light sensitivity, in individuals with migraine.24,25 We assessed responses both during and between migraine attacks to capture the dynamic nature of photophobia across the migraine cycle. By simulating real-world ambient lighting conditions and using visually neutral white light, rather than monochromatic stimuli, we aimed to identify lighting conditions that reduce visual discomfort and light sensitivity while remaining visually acceptable for everyday environments. We hypothesized that light sensitivity and visual discomfort are significantly influenced by both spectral content and intensity, with greater effects during migraine attacks.
Methods
Study design
This was a prospective four-condition randomized crossover study conducted with 20 participants. This study is reported in accordance with the STROBE guidelines for a prospective observational investigation. The study was performed in accordance with the Declaration of Helsinki and was authorized by the Institutional Review Board (IRB) at Arizona State University (STUDY00018233). All research participants completed and signed an informed consent document. Each participant completed a screening session and, if eligible, took part in two research sessions, each lasting up to one hour. One session occurred between migraine attacks (when the participant was headache-free), and the other occurred during a migraine attack. When arriving for the no migraine attack research visit, participants reported when their last headache occurred.
Participants
Participants were individuals between the ages of 18 and 45 who met the International Classification of Headache Disorders, 3rd edition (2018) diagnostic criteria for migraine, with or without aura. 32 All participants reported photosensitivity during migraine attacks, experienced between four and 14 headache days per month, and reported at least three migraine attacks per month. Participants were required to be fluent in English and have no history of ocular disease such as glaucoma, macular degeneration, retinal degenerative diseases, or blindness, as assessed by self-reported medical history. Eligible individuals also needed to demonstrate willingness to attend one of the study visits during an untreated migraine attack. An untreated attack was defined as one in which the participant had not taken any “as-needed” medication, prior to the testing session. Participants were permitted to take their medication immediately following the session. Recruitment was conducted using methods found to be effective in prior studies, including word-of-mouth referrals, email advertisements, and flyer distribution.
Screening research visit
During the screening session, participants completed questionnaires to assess photophobia, migraine characteristics, and psychological health. These included questionnaires about participant demographics and migraine characteristics, the Utah Photophobia Symptom Impact Scale (UPSIS-12), the Visual Light Sensitivity Questionnaire-8 (VLSQ-8), the Patient Health Questionnaire-8 (PHQ-8), and the Generalized Anxiety Disorder-7 (GAD-7).33–36
The UPSIS-12 is a 12-item scale measuring the severity and functional impact of photophobia, with each item rated from 0 (“not at all”) to 5 (“very much”). Scores were summed (range: 0–60), with higher scores indicating greater interference from light sensitivity. The VLSQ-8 assesses the frequency, symptoms, severity, and daily impact of light sensitivity using eight items rated from 1 (“never”) to 5 (“always”). A composite score was calculated by averaging item responses, with higher scores reflecting greater photosensitivity.
Psychological symptoms were assessed using two validated scales. The GAD-7 includes seven items measuring anxiety severity, rated on a 0–3 scale. The PHQ-8 includes eight items assessing depressive symptoms over the past two weeks, also rated on a 0–3 scale. In both measures, item responses were summed to produce total scores, with higher scores indicating greater symptom severity.
Participants also received instructions for their upcoming lab visits, including refraining from wearing blue-blocking or tinted glasses or contact lenses during the experimental sessions, while clear, non-tinted eyewear was permitted, and avoiding the use of acute migraine/pain medications prior to the session scheduled during an active migraine attack.
Experimental sessions
Experimental sessions were conducted in a controlled laboratory environment within a 10 × 9 ft room located at the Arizona State University Novus Building in Tempe, AZ. The objective of the sessions was to assess visual discomfort levels and light sensitivity under different spectral properties and various intensities of white light. Four spectral conditions were tested: red-enriched white light (LWR; λ_peak = 640 nm), green-enriched white light (LWG; λ_peak = 540 nm), cyan-enriched white light (LWC; λ_peak = 480 nm), and blue-enriched white light (LWB; λ_peak = 440 nm). Lighting conditions were generated using ten custom-built direct/indirect LED systems (75% uplight) positioned along the room perimeter to produce a glare-free, uniform ambient illumination comparable to that of a well-lit indoor environment. Each participant experienced all conditions in two sessions: once when migraine attack-free and headache-free and once during a migraine attack.
At the beginning of each session, participants were seated in a dimly lit room (<10 lux, neutral white) for a 10-min light adaptation period and asked to complete a day-of-visit headache questionnaire to evaluate the attack phenotype. This was followed by a 5-min dark adaptation phase in complete darkness. Subsequently, one of the four light conditions was presented, beginning at 50 ± 10 lux corneal illuminance and increasing in intensity every 40 s (consisting of a 15-s fade-in and a 25-s hold), up to a maximum of 590 ± 10 lux. Light intensities progressed through 10 discrete levels for each spectral condition. Participants were instructed to maintain a forward gaze and to rate their visual discomfort after each lighting scene using a numeric scale from 0 (comfortable) to 10 (very uncomfortable). Ratings were provided verbally to avoid participant interaction with devices, and responses were recorded by research personnel present in the experiment room. The sequence was terminated either when the participant reported reaching their maximum discomfort level or when the maximum light intensity was reached. Pupil size was measured by research personnel at the end of exposure to each lighting condition (LWR, LWB, LWC, LWG) using a NeuroOptic PLR-3000 pupilometer. Pupil size was not measured at each individual light-intensity level; instead, measurements were obtained at the end of the exposure period, corresponding to the highest tolerable light intensity. If a participant reached their maximum level of visual discomfort before completing the full exposure duration and could not continue, pupil size was measured at that earlier time point, corresponding to the highest intensity tolerated by the participant. All pupil size values are reported in millimeters (mm). Pupil size served as an objective indicator of light sensitivity, with smaller pupil diameters indicating greater sensitivity to the specific spectral condition tested. Each participant completed all four spectral conditions during each session, presented in randomized order determined using a computer-generated random sequence. A 3-to-5-min dark adaptation period was provided between conditions, with additional time permitted upon participant request (Figure 1).
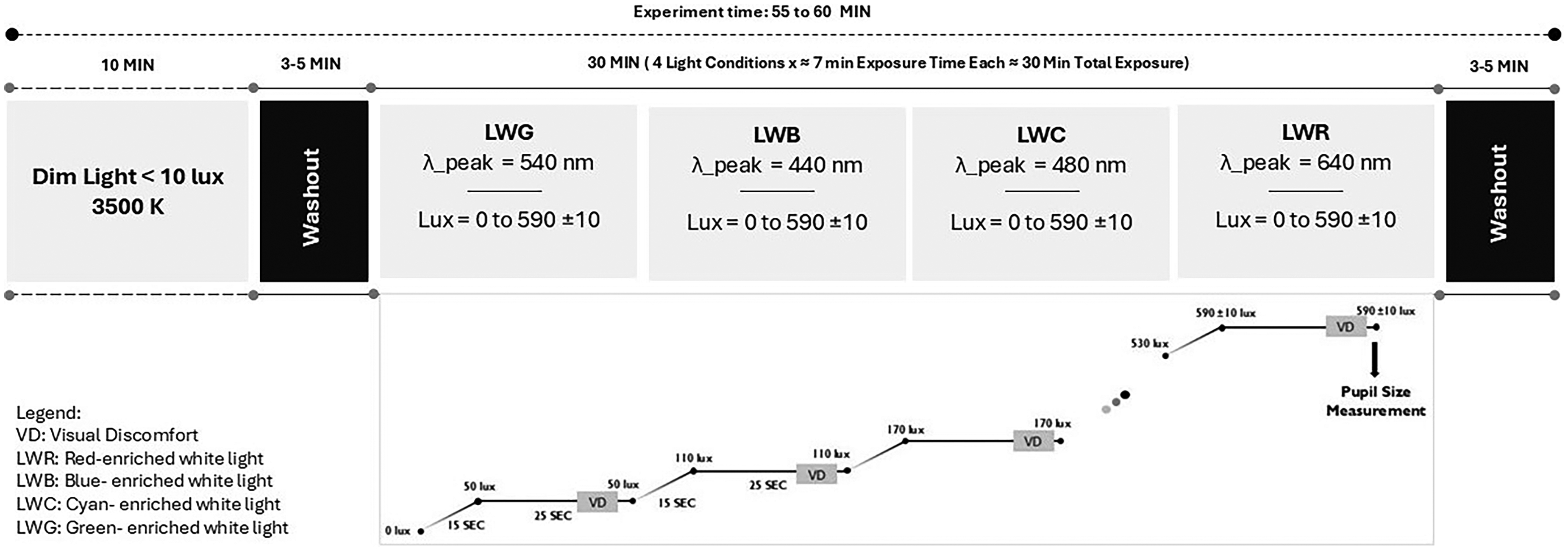
Study design and research procedures: participants were exposed to all lighting conditions in a randomized order, each following the same exposure sequence shown in the image. Each condition began at 0 lux and increased stepwise, with a 15-s fade-in to reach the next intensity level, followed by a 25-s hold at that level. Visual discomfort ratings were recorded at the end of each 25-s hold. Pupil size measurements were taken at the end of exposure to each lighting condition.
Power and statistical analysis
To detect significant differences between visual discomfort ratings for the four lighting conditions, a sample size of twenty participants was determined to be sufficient with a power of 0.85, a two-sided alpha of 0.05, a moderate Cohens f effect size of 0.3, and an assumed correlation among the repeated measures of 0.5. G* Power 3.1 was used for sample size calculations.
All statistical analyses were performed in IBM SPSS Statistics (Version 27) using repeated-measures ANOVA to examine the effects of headache status, lighting spectrum, and light intensity on visual discomfort levels and pupil size. The within-subject factors included migraine status (migraine attack vs. free from migraine attack), lighting spectrum (red-, green-, blue-, and cyan-enriched white light), and light intensity (10 levels from 50 to 590 lux). Assumptions of sphericity were tested using Mauchly's test, and Greenhouse-Geisser corrections were applied when violations were identified. Normality of the dependent variables was evaluated through inspection of residual distributions. Because all factors were within-subjects, homogeneity of variance between groups (Levene's test) was not applicable.
For significant main effects, Bonferroni-adjusted post hoc pairwise comparisons were conducted where appropriate. Effect sizes are reported using partial eta squared (η2). Pearson correlation coefficients were computed to assess the relationship between pupil size and subjective discomfort. A significance threshold of p < .05 was used for all tests.
Results
Participant characteristics
A detailed summary of participant demographics, migraine characteristics, and screening questionnaire results is presented in Table 1. Twenty participants aged 18–45 years completed the study. Mean age (n = 19; data missing from one participant) was 27.2 years (SD 7.3), 75% were female (n = 15), and 25% were male (n = 5). The majority identified as Not Hispanic or Latino (85%), 10% as Hispanic or Latino, and 5% undisclosed. Racially, the cohort included 70% White, 25% Asian, and 5% Black or African American.
Participant characteristics (N = 20).

Participants reported an average of 7.3 headache days per month (SD = 2.62;). Eight participants (40%) had migraine with aura and 12 (60%) had migraine without aura. During the migraine attack free visit, participants reported that their most recent migraine attack occurred on average 3.05 days prior (SD = 1.96). For the visit occurring during an untreated migraine attack, 85% of participants (n = 17) met criteria for having a migraine attack and 15% (n = 3) met criteria for a probable migraine attack.
All participants reported sensitivity to light and sound during migraine attacks. Photophobia impact was assessed using the UPSIS-12, with total scores averaging 25.95 (SD = 7.21). Responses revealed considerable variability in how light sensitivity interfered with daily activities. On average, participants rated the severity of their light sensitivity as moderate with a mean of 3.30, (SD = 0.98). Strong light was perceived as moderately unpleasant during headache-free periods with an average of 2.15(SD = 1.31), but markedly unpleasant during migraine attacks with an average of 4.50(SD = 0.61). Participants also reported a substantial increase in light sensitivity during headache compared to non-headache states (mean = 3.95, SD = 1.10). Light sensitivity impacted several functional domains. It often interfered with watching television (mean = 2.30, SD = 1.13), driving (mean = 1.90, SD = 1.29), riding in a car (mean = 1.35, SD = 1.04), and using a computer screen (mean = 2.10, SD = 1.29). Its impact on completing housework or working outside the home was somewhat lower (mean = 1.40, SD = 1.23). Additionally, participants reported that strong light frequently triggered headache episodes (mean = 3.00, SD = 1.17). Visual sensitivity was also assessed using the VLSQ-8. The overall mean score across all items was 3.09 (SD = 0.50), indicating a moderate degree of photosensitivity among participants. Participants reported moderate frequency of light sensitivity in daily life (mean = 3.20, SD = 0.82) and a comparable level of symptom intensity, such as glare or visual disturbances (mean = 3.29, SD = 0.91). Light sensitivity frequently impacted participants’ ability to engage in visual tasks such as reading, watching television, or using a computer (mean = 3.19, SD = 0.75). The severity of the worst light sensitivity experienced in the past month was also rated as moderate (mean = 3.43, SD = 0.81). Behavioral adaptation, such as wearing sunglasses indoors or on cloudy days, was less common (mean = 2.24, SD = 1.22), suggesting variability in coping strategies among participants.
The effect of headache status, lighting spectrum, and lighting intensity on visual discomfort
Assumptions for the repeated-measures ANOVA were evaluated prior to interpreting the findings. Mauchly's test indicated that the assumption of sphericity was violated for several within-subject factors, including lighting spectrum (χ2 (5) = 17.27,
The analysis revealed that migraine status had a significant effect on visual discomfort,
The spectrum of white light also had a significant effect on visual discomfort ratings,

Mean visual discomfort levels across lighting spectra (a) between migraine attacks and (b) during migraine attacks. Green-enriched white light (LWG) was associated with significantly lower visual discomfort compared to red- (LWR), blue- (LWB), and cyan-enriched (LWC) white light in both conditions. Brackets indicate significant Bonferroni-adjusted post-hoc pairwise comparisons based on the main effect of lighting spectrum averaged across migraine status and across all lighting intensity levels (*p < .001).
Light intensity had a stronger effect,
There was a significant interaction between migraine status and light intensity,
The interaction between white light spectrum and intensity was also significant,
Impact of migraine attack status, lighting spectrum, and lighting intensity of visual discomfort ratings.

In contrast, there was no significant interaction between migraine status and lighting spectrum,

Mean visual discomfort ratings across lighting spectra and light intensity levels (a) between migraine attacks and (b) during migraine attacks. Visual discomfort increased with increasing light intensity across all lighting spectra and was higher during migraine attacks. Green-enriched white light (LWG) was associated with lower visual discomfort compared to red- (LWR), blue- (LWB), and cyan-enriched (LWC) white light across intensities (main effect of light spectrum). Statistical results for main and interaction effects are reported in Table 2.
The effect of white light spectrum and headache status on pupil size
Pupil size was analyzed as an objective physiological indicator of visual sensitivity to light, with smaller pupil diameters interpreted as reflecting higher sensitivity. A repeated-measures ANOVA was conducted to examine the effects of white light spectrum and migraine status on average pupil size. The analysis revealed a significant effect of white light spectrum on pupil size,
Bonferroni-corrected post hoc pairwise comparisons associated with this main effect showed that in the migraine attack-free condition, pupil size was significantly larger under green-enriched (

Mean pupil size (mm) across lighting spectra (a) between migraine attacks and (b) during migraine attacks. Brackets indicate statistically significant Bonferroni-corrected post hoc pairwise comparisons (*p < .05). During migraine attacks, green-enriched white light (LWG) was associated with significantly larger pupil size compared to blue- and cyan-enriched white light, and red-enriched white light was associated with larger pupil size compared to blue-enriched white light. Between migraine attacks, pupil size was significantly larger under green-, red-, and cyan-enriched white light compared to blue-enriched white light. Larger pupil size reflects lower physiological light sensitivity.
There was no significant main effect of migraine attack status,
These findings indicate that pupil size varied significantly as a function of lighting spectrum, while no significant main effect of migraine attack status was observed. As an objective measure of light sensitivity, pupil size was consistently smaller under blue-enriched and cyan-enriched white lighting conditions and larger under green-enriched white lighting. Figure 4(a) and Figure 4(b) illustrate average pupil size by lighting spectrum across migraine-free and migraine attack conditions, respectively.
Associations between pupil size and subjective discomfort across lighting conditions
To examine the relationship between physiological and subjective responses to light, a series of Pearson correlation analyses were conducted between pupil size (objective measure) and self-reported visual discomfort level (subjective measure) under each lighting spectrum, separately for migraine attack and migraine attack free conditions. Given that smaller pupil size reflects heightened physiological sensitivity to light, negative correlations were expected if discomfort and pupil constriction were aligned.
A significant negative correlation was observed under green-enriched white light during migraine attacks (

Relationship between mean pupil size (objective measure) and mean visual discomfort level (subjective measure) across white light spectra (a) between migraine attacks and (b) during migraine attacks. Pearson correlation analyses were conducted separately for each lighting spectrum. The reported Pearson correlation reflects the green-enriched white light condition during migraine attacks only, for which a significant negative association between pupil size and visual discomfort was observed (r = −0.46, p = .043). No significant correlations were observed for other spectra or in the migraine-free condition (all p > .05).
Discussion
This study examined how white light spectrum, intensity, and migraine state jointly influence subjective visual discomfort and objective physiological responses. Overall, migraine attacks significantly increased visual discomfort, with light intensity exerting the strongest effect across conditions. Importantly, spectral composition also played a meaningful role: green-enriched white light consistently elicited lower discomfort ratings and larger pupil sizes, whereas blue- and cyan-enriched white light were associated with greater discomfort and smaller pupil size, indicating greater light sensitivity. These spectral effects were observed both during and between migraine attacks and became more pronounced at higher light intensities. Together, these findings indicate that spectrally tuned white light, particularly green-enriched profiles, may reduce light sensitivity under everyday indoor lighting conditions while remaining visually comfortable compared to monochromatic colored light.
Foundational studies have uncovered the neurophysiological mechanisms underlying light sensitivity, with intrinsically photosensitive retinal ganglion cells (ipRGCs) projecting to pain-regulating brain regions. 14 Building on this work, a series of studies demonstrated that green light is less likely to exacerbate migraine symptoms compared to other colors.16,18 These studies laid the groundwork for therapeutic applications of green light and shaped the development of targeted lighting interventions for migraine. Our study was designed to build on these insights while addressing key limitations in prior research.
Unlike previous studies that exposed participants to monochromatic, saturated lights in dim settings or directly into the eyes through devices like the Ganzfeld Color Dome, our design simulated everyday indoor lighting by using white light enriched with different spectral peaks (red, green, cyan, and blue).16,18 This ambient lighting, paired with gradual intensity changes and a wide illuminance range (50 to 590 lux), allowed us to better approximate naturalistic lighting conditions found in classrooms, offices, or healthcare environments.
Prior studies have demonstrated the benefits of green light for migraine relief, but under conditions not suited for everyday use. In a prior study, researchers tested a narrow-band green light-emitting lamp during acute attacks in darkened rooms, using very dim light (∼50–100 lux) for symptom relief. 17 Similarly, another study exposed participants to 4–100 lux green LED strips for short daily periods in dim environments. 19 While both studies reported reductions in headache symptoms, the low intensities and lighting context limit their relevance to standard indoor lighting applications.
Our findings reinforce and extend previous work. Green-enriched white light was rated as the most visually comfortable and caused the least physiological sensitivity, while blue- and cyan-enriched white lights elicited the highest discomfort ratings and the greatest pupil constriction, a marker of light sensitivity.24,26–27 These results align with prior research showing that short-wavelength light is more aversive, whereas green light is generally more tolerable. Similar to green light, which activates cone-driven pathways to a lesser degree than other spectrums, green-enriched white light also peaks around 540 nm while appearing visually white. 16 This may explain its relatively positive effects, providing a more natural and comfortable lighting experience. Importantly, these benefits were observed under everyday white-light conditions and across intensities representative of typical indoor environments. Another contribution of this study is the inclusion of data both during and between migraine attacks, which allowed us to assess how spectral composition and intensity interact with dynamic changes in migraine state. Furthermore, by integrating pupil size as an objective physiological marker alongside discomfort ratings, we strengthened the reliability of our findings and confirmed that pupil response aligns with perceived discomfort across lighting conditions.
All participants in this study reported experiencing photophobia, as reflected in their UPSIS and VLSQ-8 scores (Table 1), with light sensitivity increasing substantially during migraine attacks compared to non-migraine attack states; future work should examine whether similar spectral effects emerge in individuals with lower baseline light sensitivity. Although prior studies have shown that even brief light exposure can provoke migraine symptoms, there is a growing need to explore how people with migraine respond to lighting over longer durations. 28 A field-based study provided valuable insights into the long-term benefits of green light, and future work should extend this approach using white light with controlled spectral profiles. 19
Our study was limited by a modest sample size, limited demographic diversity, and the exclusion of individuals with chronic migraine, which may constrain generalizability. In addition, exposures were brief and conducted in a controlled laboratory environment rather than in real-world settings. Accordingly, causal inferences and direct clinical recommendations should be made cautiously. Rather than serving as a standalone therapeutic approach, these findings are best interpreted as informing both environmental design considerations and future clinical research, highlighting how spectrally tuned white light may be incorporated into migraine-conscious indoor environments. Future studies should assess longer-term exposure to spectrally tuned white light under naturalistic conditions and examine its effects on headache intensity, other migraine symptoms, cognitive performance, sleep quality, and emotional wellbeing. These future studies should further investigate quantitative and qualitative outcomes to better delineate the potential impact of spectrally tuned white light on participant wellbeing. In the current study, reductions in visual discomfort under green-enriched white light, compared to blue-enriched white light, hovered around 1.5 units on the 11-point numeric rating scale of visual discomfort, corresponding to an approximately 30% or greater reduction in visual discomfort between migraine attacks and an approximately 20% or greater reduction in visual discomfort during migraine attacks. The clinical relevance of visual discomfort change of this magnitude warrants careful consideration. In chronic pain research, reductions of approximately 1–2 points on a 10-point numeric rating scale have been identified as representing minimally clinically important differences, with relative reductions of approximately 15–30% considered clinically meaningful and ≥30% often interpreted as reflecting moderate improvement.29–31 Within this context, the reductions observed in the present study fall within thresholds for clinical relevance. Importantly, spectrally tuning ambient white light is not intended as a standalone migraine treatment, but rather as an environmental modification aimed at reducing symptom exacerbation. In settings where individuals with migraine must tolerate lighting for their work, education, healthcare, or daily functioning, even modest reductions in discomfort are likely to be considered meaningful. However, future studies incorporating qualitative assessments will be important to further delineate how individuals perceive the meaningfulness of these differences.
Conclusion
This study demonstrates that spectral composition, light intensity, and migraine attack status significantly associate with visual discomfort and light sensitivity among those with migraine. Green-enriched white light consistently produced the lowest discomfort and least physiological reactivity, even at high intensities appropriate for visual tasks. By preserving the look and function of natural white light while incorporating the beneficial aspects of green wavelengths, this study offers a new direction for designing migraine-conscious lighting environments that are not only therapeutic, but also practical, visually acceptable, and performance-supportive. These findings lay the groundwork for translating laboratory research into real-world lighting solutions that accommodate the needs of individuals living with migraine.
Article highlights
Among those with migraine, visual discomfort and light sensitivity are associated with light spectral composition, light intensity, and migraine attack status. Green-enriched white light is the most visually comfortable and causes the least physiological sensitivity, while blue- and cyan-enriched white lights elicit the highest discomfort ratings and the greatest pupil constriction.
Footnotes
Acknowledgements
The authors gratefully acknowledge all participants for their involvement in the experimental sessions. The authors wish to express their appreciation to Jennifer K. Hallof, M.S.N., R.N., for her valuable contributions to the study design and for ensuring its suitability for those with migraine. The authors also thank Mohammed Alrahyani, PhD candidate at Arizona State University, for his assistance in preparing the laboratory setting.
Ethical considerations
The study was approved by the Institutional Review Board (IRB) of Arizona State University (Approval No. STUDY00018233) and was conducted in accordance with the ethical standards of the Declaration of Helsinki.
Consent to participate
All research participants completed and signed an informed consent document.
Author contributions
MF conducted all experiments and data collection, performed data curation, analysis, and visualization. NS and MF and prepared the original manuscript draft. NS and TJS conceived the study, developed the design and methodology, and secured funding. All authors contributed to writing and editing the manuscript and approved the final version. The corresponding author confirms that all listed authors meet authorship criteria and that no individuals meeting the criteria have been omitted.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by support from National Headache Foundation and the Arizona State University Herberger Institute Research Building Investment grant.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MF declares no competing interests. Within the prior 24 months, NS has held stock options in Allevalux and BeyondLynk. Her institution received research grants on her behalf from Flinn Foundation and National Headache Foundation. Within the prior 24 months, TJS received personal compensation for consulting with Lundbeck and Salvia. His employer received compensation for his consulting activities with AbbVie. He received royalties from UpToDate, held stock options in Nocira. He holds equity in and is a co-founder of Allevalux. His institution received research grants on his behalf from American Heart Association, Flinn Foundation, Henry Jackson Foundation, National Headache Foundation, National Institutes of Health, Patient Centered Outcomes Research Institute, Pfizer, Spark Neuro, and United States Department of Defense. NS and TJS have a patent pending through Arizona State University for an optimized white light for managing migraine and photophobia.
Data availability statement
The data that support the findings of this study are available on reasonable request from the corresponding author.
