Abstract
This review is a tribute to Professor Jes Olesen involvement in the genetics of migraine and tension-type headache as it is coming up to his 60th birthday.
Migraine
Migraine with aura (MA) and migraine without aura (MO) are clinically distinguishable by the presence or absence of aura symptoms prior to the pain phase (1). The aura symptoms occur simultaneously with reduced regional cerebral blood flow (2), whereas regional cerebral blood flow is normal in MO (3). In the pain phase dilatation of the middle cerebral artery has been shown in both MO and MA (4, 5). These results indicate that the pathogenesis is different in the early phase of MO and MA, while it may be similar later in the pain phase. The existence of these data and the classification of the International Headache Society (6) were important prior to the first genetic epidemiological survey of migraine. A review of the genetic literature revealed that no firm conclusion could be drawn from previous family and twin studies due to methodological shortcomings (7), while a pilot family study suggested it would be feasible to make a genetic epidemiological survey (8).
This paper is a tribute to professor Jes Olesen and his involvement in the genetics of migraine and tension-type headache.
Genetic epidemiological survey
Forty-year-old probands were included. They were found among 4000 representative people from the general population. First degree relatives of probands were blindly interviewed with regard to the diagnostic status of the proband. The interviews were conducted by a physician. Figure 1 shows the relative risk of MO and MA among first-degree relatives (9). Compared with the general population, first-degree relatives of probands with MO had a 1.9-fold significantly increased risk of MO and a 1.4-fold increased risk of MA. Spouses had a 1.5-fold increased risk of MO (not shown in Fig. 1). First degree relative of probands with MA had a 3.8-fold increased risk of MA and no increased risk of MO, while spouses of probands had no increased risk of MA. The results indicate that MO and MA are distinct disorders.
Compared with the general population, first-degree relatives of probands with migrainous disorder type MO had an increased risk of MO, but it was less than the risk among first-degree relatives of probands with MO (10). Thus, it is likely that migrainous disorder type MO is genuine MO in some people and in others it is not. First-degree relatives of probands with MO associated with head trauma had no increased risk of MO, which makes it likely that a head trauma can cause MO (11). A population-based twin study included 1013 monozygotic (MZ) and 1667 same-gender dizygotic (DZ) twin pairs (12). The twins were blindly interviewed with regard to the diagnostic status of the co-twin. The interviews were conducted by two physicians. Table 1 shows the number of concordant and discordant twin pairs (13, 14). The pairwise concordance rate was significantly higher among MZ than DZ twin pairs (MO P < 0.05 and MA P < 0.001).
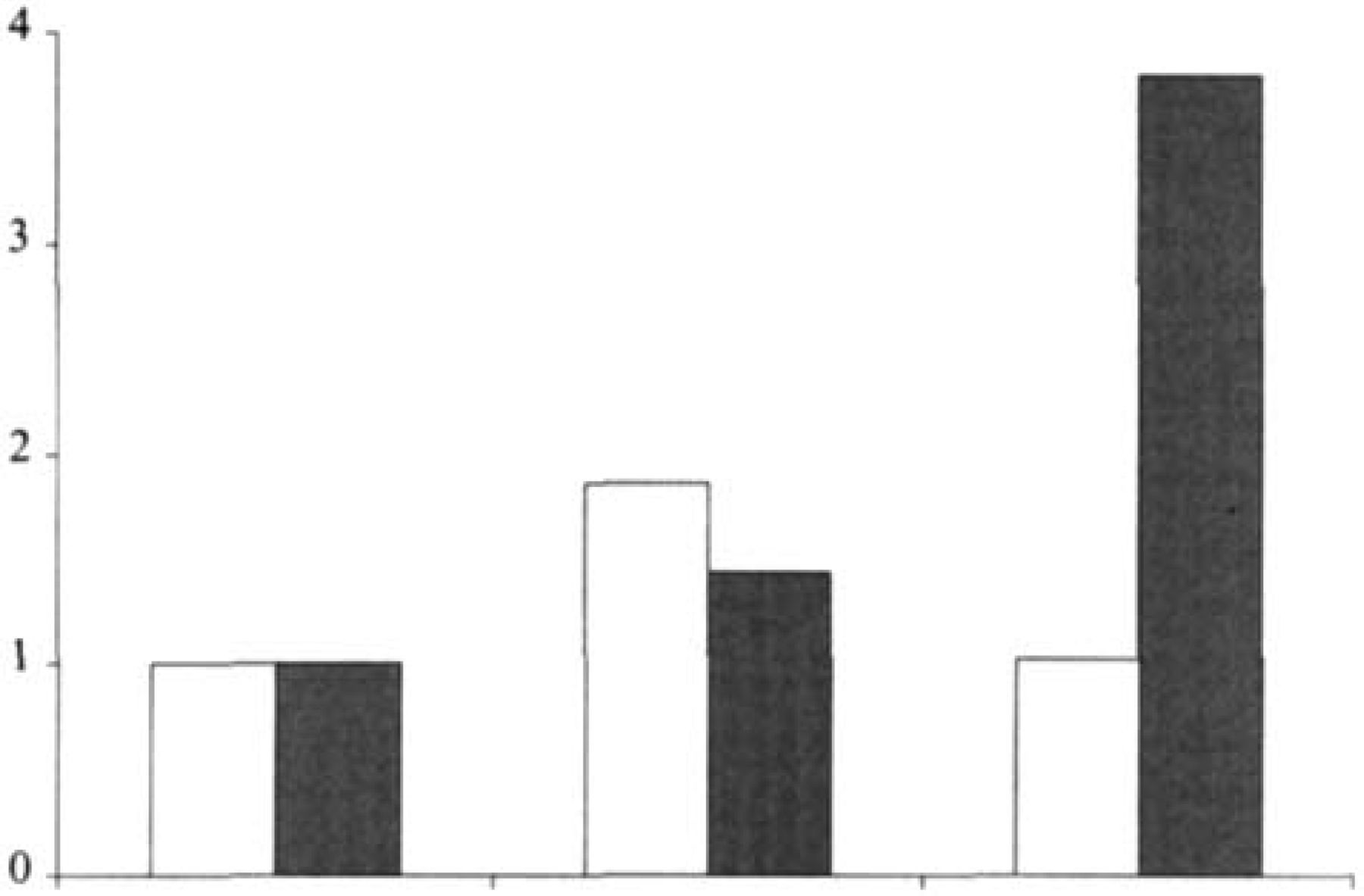
The relative risk of migraine without aura (white columns) and migraine with aura (hatched columns) among first-degree relatives of probands with migraine without aura and migraine with aura. The risk of migraine in the general population is reflected in the two first columns (9).
The genetic epidemiological surveys indicate that both MO and MA are caused by a combination of genetic and environmental factors.
Mode of inheritance
A complex segregation analysis of 126 families with MO and 127 families with MA from the general population indicated that both MO and MA have multifactorial inheritance without generational differences (15). The twin data (Table 1) were analysed with a structural equation model using the MX program. The model that combined additive genetic and individual-specific environmental effects gave the best fit for both MO and MA (16, 17). This is compatible with multifactorial inheritance. The results do not exclude that some families have a mitochondrial or Mendelian pattern of inheritance, i.e. genetic heterogeneity, since neither the complex segregation analysis nor the structural equation modelling can detect it. For that reason high-risk families with MA were studied (18). The 31 nuclear families consisted of an affected and an unaffected parent and at least one affected and one unaffected child. The nuclear families were expanded with other first-degree relatives, as well as second-degree relatives, in case the first-degree relatives were affected. An analysis of the families indicated multifactorial inheritance. Considering the high prevalence, a single gene is not likely to cause either MO nor MA, since the gene has to be more common than any known disease-causing gene.
Molecular genetics
The serotonin2C(5-HT2C) receptor has been implicated along with other components of the 5-HT system in the pathophysiology and pharmacotherapy of migraine. To investigate whether the 5-HT2C receptor gene contributes to the risk of migraine an association study of the allelic variation at codon 23 (Cys or Ser) of the 5-HT2C receptor gene was performed (19). The study indicated that this allelic variation did not contribute to the genetic predisposition of either MO nor MA. Another association study investigated the role of allelic variation of the human serotonin transporter gene (HSERT) (20). The intron 2 variable number tandem repeat (VNTR) polymorphic region of the HSERT was investigated. The MO group had an over-representation of individuals with the genotype carrying two 12 repeat (STin2.12) and a reduction in individuals with genotypes containing one 10 repeat (STin2.10) compared with people who had never had migraine. The MA group showed a similar pattern, but also a trend towards increase in genotypes containing the nine repeat allele of the VNTR (STin2.9). Genotypes containing this allele were found in 6.4% of the MA group compared with 2.3% of people who had never had migraine. The group with co-occurrence of MO and MA had a different pattern of overall allele frequency distribution, reflecting a reduction in genotypes containing the STin2.10 allele and a shift both to STin2.9 carriers and to STin2.12 homozygosity. These results support the view that susceptibility to MO and MA has a genetic component, mat these disorders are distinct and that genetic susceptibility may in some cases be associated with a locus at or near the serotonin transporter gene.
Tension-type headache
Until recently a genetic factor had not been suspected in tension-type headache. The high prevalence of episodic tension-type headache causes a positive family history simply by chance in >99% of families with an affected proband and four first-degree relatives. One or both parents will be affected by chance in >94% of the families. Episodic tension-type headache is most likely a heterogeneous disorder. The uniform symptomatology makes it likely that nociceptive mechanisms are shared but can be activated by different mechanisms. It may be caused by multiple genes in a concerted action with environmental factors or it may be non-genetic.
Only a single genetic epidemiological study has investigated the familial aggregation of chronic tension-type headache (21, 22). It included 122 probands from a headache clinic meeting the criteria of the International Headache Society for chronic tension-type headache (6). The probands first-degree relatives and spouses were interviewed by a neurological resident. The risk of familial occurrence was assessed by estimating the relative risk. Compared with the general population, first-degree relatives had a 3.1-fold significantly increased risk of chronic tension-type headache, while spouses had no increased risk of chronic tension-type headache. This result indicates the importance of genetic factors. A complex segregation analysis further emphasized the result by suggesting that chronic tension-type headache has multifactorial inheritance (23).

