Abstract
Background
Physical activity (PA) has been associated with reduced migraine burden, but patients with frequent or comorbid migraine often report difficulty sustaining regular activity. We examined the association between minimal PA and migraine-related outcomes, and explored whether a disease burden threshold limits PA engagement.
Methods
We analyzed data from 550 participants with migraine in the Negev Migraine Cohort in southern Israel who completed a questionnaire assessing PA (≥2 h/week), migraine-related disability (MIDAS), depressive symptoms (DASS-21), and psychosocial functioning. Between-group comparisons and multivariable linear regression models were conducted to assess the independent associations of PA with key outcomes, adjusting for demographic characteristics. We also explored the probability of engaging in PA by migraine frequency, and conducted unsupervised clustering based on migraine days, depressive symptoms, and PA status to identify patient profiles.
Results
Physically active participants (46%) reported significantly fewer migraine days per month (median 3.0 vs. 5.0), lower use of triptans per month (mean 2.6 vs. 4.1 pills), and reduced migraine-related disability (median MIDAS score 26.0 vs. 36.0). They also reported fewer days of presenteeism per month (median 3.5 vs. 5.0). In addition, they experienced less impairment in overall life satisfaction (mean 4.49 vs. 5.14). In fully adjusted models, PA remained independently associated with reduced disability (β = −0.14, 95% CI −0.28 to −0.1) and improved life satisfaction (β = −0.46, 95% CI −0.9 to −0.1). Probability modeling showed that individuals with more than three migraine days per month had less than 50% likelihood of meeting PA targets. Clustering analysis identified three subgroups: a high-burden and low-activity group, an intermediate group, and an active and well-functioning group.
Conclusions
Minimal PA was associated with lower migraine-related disability and better life satisfaction. The identification of an activity engagement threshold and distinct patient clusters suggests a staged care model where pharmacologic stabilization enables PA, which may itself serve as a marker of recovery.
This is a visual representation of the abstract.
Introduction
Migraine care remains a challenge even in the CGRP pathway era. 1 Monoclonal antibodies that target the CGRP pathway and gepants are expensive, and health care systems struggle to provide access to as many migraine patients as possible, while many remain without treatment. 2 Even when access is available, a subset of patients experiences an incomplete response, 3 and comorbidities, both mental (depression, anxiety) and physical (fibromyalgia, primary pain syndromes, irritable bowel syndrome) may still affect overall well-being 4 despite these advances. 5
In recent years, physical activity (PA) has gained attention as a potential means to reduce migraine burden. Epidemiological studies show that low PA or prolonged sedentary behavior correlates with higher migraine frequency, while habitual PA is associated with fewer and milder episodes.6–11 Clinical trials have demonstrated that moderate aerobic exercise, performed two to three times per week, can reduce migraine days by 30–50%, with efficacy comparable to that of some preventive pharmacotherapies and minimal side effects12,13 Meta-analyses confirm that exercise reduces migraine frequency, severity, and duration, and enhances quality of life.14–16
Depression, a common migraine comorbidity, may also be improved by PA. Meta-analyses show a moderate but consistent antidepressant effect of regular PA. 17 Cross-sectional and population-based studies further indicate that inactive migraine patients report poorer health-related quality of life, particularly when depression is present.18,19
The association between PA and migraine severity is likely bidirectional; PA can improve migraine, while migraine and its comorbidities may limit the ability to be active. More active patients generally report better functioning, whereas those with higher migraine burden, depression, or burnout are often the least able to exercise. This cycle of symptoms and inactivity may, in turn, worsen outcomes.20,21
This study aims to examine the association between engagement in a minimal level of PA and migraine severity, mental health, and psychosocial burden. It also seeks to identify the threshold of migraine severity at which patients still engage in PA, suggesting when activity may become a feasible intervention.
Methods
Study setting
This observational, population-based study was conducted in the Negev region of southern Israel, which is home to approximately 8.2% of the national population. The region includes a diverse demographic composition, with around 75% Jewish and 25% Bedouin residents. Healthcare in the Negev is primarily provided by Clalit Health Services (CHS), the largest health maintenance organization (HMO) in Israel, serving approximately 67% of the region's 730,000 residents through a network of community-based primary care clinics. The Israeli healthcare system is based on universal coverage through four non-profit HMOs, with compulsory enrollment under the National Health Insurance Law.
Study design
This study is part of the SMILE project (Southern Israel Migraine Impact on Life Experiences), which aims to assess the multidimensional impact of migraine on life experiences. Participants were drawn from the Negev Migraine Cohort (NMC), a population-based retrospective cohort of adult CHS members (aged 18 years or older) identified between 2000 and 2018 using physician-assigned ICD-9 codes for migraine and/or prescription claims for triptans. While International Headache Society (IHS) criteria were not directly applied within the survey, migraine diagnoses in routine clinical practice are generally made in accordance with IHS definitions.
In 2021, we employed stratified random sampling to recruit participants with migraine from the NMC for an online survey examining the lived experience of migraine. A total of 1475 migraine patients were approached, resulting in the recruitment of 675 migraine participants (46% response rate). Participants completed the survey via a secure online platform operated by an external survey company. Recruitment methodology and cohort characteristics have been described previously. The study's general design and recruitment process were previously reported.22,23
Data collection
The study questionnaire incorporated both validated instruments and custom-developed items based on themes identified through prior focus groups with patients from the NMC. The study questionnaire also included a question regarding migraine status to confirm the study group of each participant. None of the participants reported a diagnosis that contradicted the physician-assigned diagnosis. The questionnaire included the following sections:
Demographics and Self-Rated Health: Participants reported age, sex, ethnicity, marital status, number of children, educational attainment, socioeconomic index (derived from residential clinic assignment), and PA (binary yes/no). Depression, Anxiety, and Stress Scale (DASS-21): A validated 21-item self-report instrument assessing depression, anxiety, and stress, with seven items per domain. Each item is rated on a 4-point scale. The DASS-21 has demonstrated good internal consistency across populations (Cronbach's alpha 0.77–0.91).
24
Migraine Disability Assessment (MIDAS): A widely used 5-item questionnaire assessing migraine-related disability across work, household, and social domains over the past three months. Scores range from 0 to 450, with higher scores indicating more severe disability. The MIDAS has shown strong reliability (Cronbach's alpha 0.79–0.89).
25
Employment status: This form gathered Self-reported information about the participant's employment status, working hours, workplace commuting time, desire to work from home, and tenure at the workplace. Burnout : (Maslach Burnout Inventory General Survey, MBI-GS): Occupational burnout was assessed using the MBI-GS, a validated 16-item self-report questionnaire measuring three dimensions: emotional exhaustion, cynicism, and professional efficacy. Items are rated on a 7-point Likert scale, with higher scores indicating greater burnout. Migraine Impact on Life Domains Questionnaire: A custom, non-validated instrument developed for this study, designed to assess the subjective impact of migraine across eight life domains: romantic relationships, parenting, family life, housework, work productivity, academic functioning, career planning, and leisure. Respondents rated the impact of migraine in each domain on a 7-point Likert scale, from 1 (“minimal impact”) to 7 (“severe impact”). An item assessing overall life satisfaction was used as a global indicator of perceived migraine-related impact on well-being. Although not formally validated as a standalone scale, this item has strong face validity and captures a widely used construct in quality-of-life research.
Statistical analysis
The main exposure of interest in this study was PA, defined as reporting regular engagement in PA and at least two hours per week. The ≥2 h per week modest threshold was chosen to avoid an unrealistic or demanding definition, ensuring that even patients with substantial migraine burden could be classified as active. By adopting such a non-ambitious cutoff, we aimed to capture a broad range of participants who engage in even minimal PA, while still allowing meaningful differentiation in outcomes. Primary and secondary outcomes included migraine-related disability (MIDAS), depressive symptoms (DASS-21), and additional patient-reported outcomes related to quality of life, psychological burden, and functioning.
Descriptive statistics were used to summarize participant characteristics. Normally distributed continuous variables were summarized using means and standard deviations, while non-normally distributed variables were reported as medians and interquartile ranges. Categorical variables were expressed as frequencies and percentages. Between-group comparisons (physically active vs. inactive) were conducted using t-tests, Mann-Whitney U tests, chi-square tests, or Fisher's exact tests, as appropriate. Standardized mean differences (SMDs) were calculated to assess group imbalances.
Univariate analyses were followed by multivariable linear regression models for each outcome. Two models were specified for each outcome: (1) unadjusted and (2) adjusted for demographics (age, sex, marital status, number of children, ethnicity, and socioeconomic status). A directed acyclic graph (DAG) was constructed to guide covariate selection and control for confounding (Supplementary Figure 1). Covariates with SMD > 0.1 or identified as confounders by the DAG were included in multivariable models. Results are presented as β coefficients with 95% confidence intervals and p-values. A forest plot was constructed to summarize the estimated associations across models and outcomes visually.
To explore the threshold of migraine severity beyond which PA becomes unlikely, we fitted a logistic regression model with regular PA (≥2 h/week) as the binary outcome and number of migraine days per month (MIDAS-A) as the continuous predictor. Predicted probabilities were then calculated across the range of values from 0 to 30 days, and a probability curve was plotted. We examined the graph to identify a potential inflection point below which PA is more likely to occur.
To evaluate distinct patterns of migraine burden, psychological distress, and PA, we performed an unsupervised clustering analysis. We applied k-means clustering using three standardized input variables: number of migraine days per month (MIDAS A), depression symptom score (range 0–3), and a binary indicator of regular PA (≥2 h/week). The number of clusters (k = 3) was selected a priori based on clinical reasoning to identify meaningful subgroups ranging from high to low burden. To compare clusters, we generated a descriptive summary table. For all analyses, a 2-sided P value <0.05 was to be considered statistically significant. All analyses were performed using R version 4.4.1 (R Foundation for Statistical Computing, Vienna, Austria) in the RStudio 2024.04.2 environment (Posit PBC, Boston, MA, USA). The study was approved by the Soroka Medical Center Institutional Review Board. The study was conducted and reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational studies.
Results
A total of 550 participants were included in the analysis, of whom 253 (46%) were classified as physically active, defined as engaging in at least two hours of sport per week and reporting regular activity. As shown in Table 1, active individuals were more likely to have post-secondary education (51.4% vs. 36.0%, SMD = 0.86) and a higher socioeconomic status (mean SEI 10.8 vs. 7.2, SMD = 0.29). They rated their health slightly higher (mean 7.59 vs. 7.38). No meaningful differences were observed in age (median 44 vs. 42), gender, marital status, or number of children.
Demographic characteristics, migraine-related variables, and patient-reported outcome measures (PROMs).
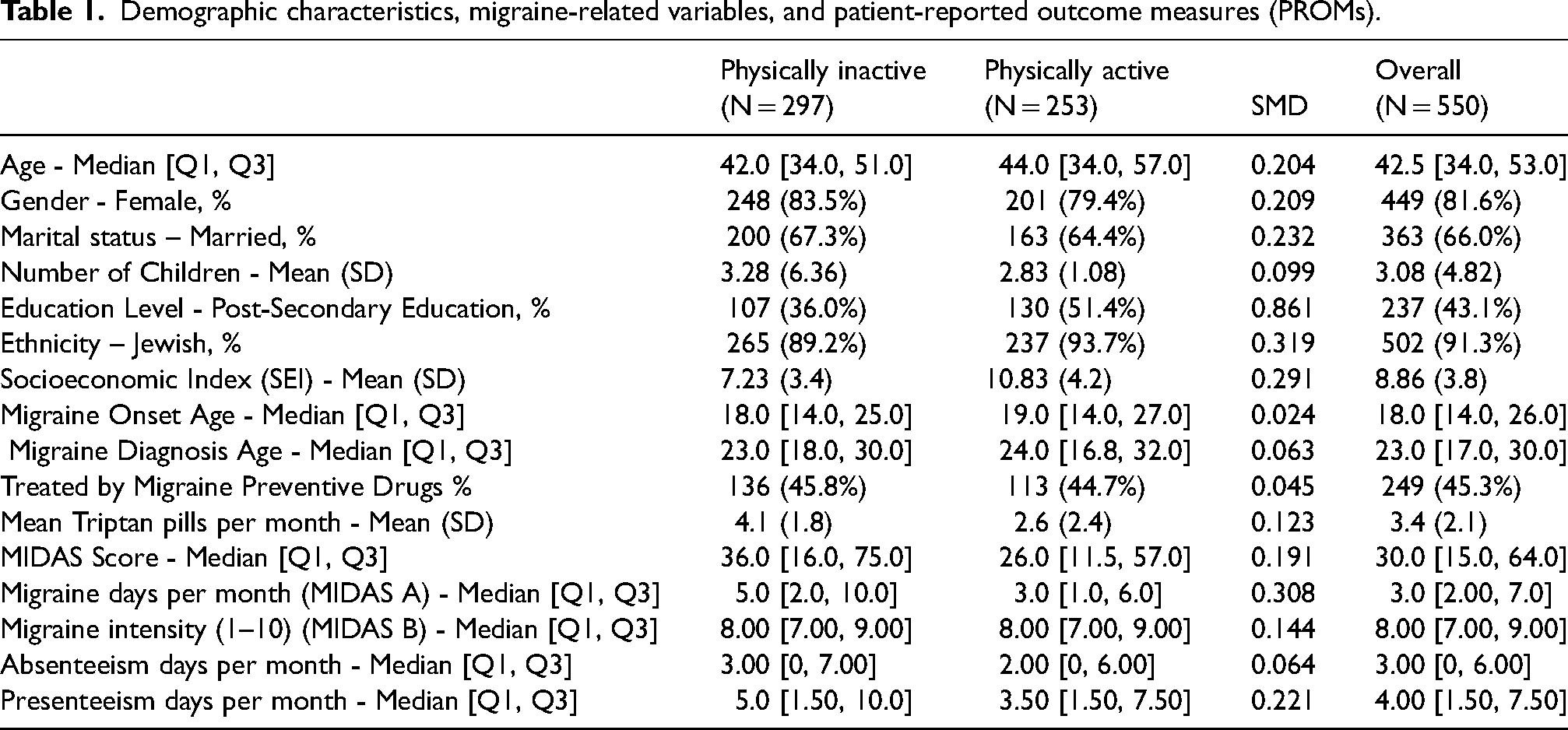
Table 1 also presents migraine-related characteristics and work outcomes. Compared to inactive participants, those who were physically active reported fewer migraine days per month (median 3.0 vs. 5.0), lower monthly use of triptans (2.6 vs. 4.1 pills), and lower migraine-related disability (median MIDAS score 26.0 vs. 36.0). Active participants also reported fewer days of presenteeism (median 3.5 vs. 5.0), and absenteeism and migraine intensity were similar between groups.
Table 2 presents mental health symptoms and psychosocial burden among active and inactive migraine patients. Physically active individuals reported lower perceived impact of migraine on overall life satisfaction (mean 4.49 vs. 5.14), and lower burnout scores (mean 3.29 vs. 3.64). Depression symptoms were lower among active participants (mean 0.78 vs. 0.94), as were anxiety and stress levels.
Mental health symptoms and psychosocial burden among active and inactive migraine patients.

Multivariable models (Table 3) fully accounting for demographic covariates (age, gender, marital status, number of children, ethnicity, and socioeconomic index) PA was significantly associated with reduced migraine-related disability (β −14, 95% CI −28 to −1.1, p = 0.034) and with less reported impact of migraine on life satisfaction (β −0.46, 95% CI −0.9 to −0.1, p = 0.040), but not with depression and burnout.
Multivariable analysis of migraine-related characteristics and patient-reported outcome measures (PROMs) by Physical Activity Status.

CI = Confidence Interval
*Beta represents the effect of being Physically Active Vs. Inactive on the model outcome.
*The Demog adjustment includes age, gender, marital status, number of children, ethnicity, and socioeconomic index.
These findings are illustrated in Figure 1, which presents a forest plot of crude and adjusted β coefficients, along with their confidence intervals, for each outcome across the three modeling stages. The visual trend supports the consistent association between PA and lower MIDAS score, with effect sizes diminishing but remaining significant after accounting for key confounders.

Multivariable analysis –forest plot showing the association between physical activity and migraine-related outcomes.
A logistic regression model revealed a negative linear association between migraine days/month and the likelihood of engaging in PA. As illustrated in Supplementary Figure 2, migraine patients with fewer monthly migraine days had a higher predicted probability of engaging in at least two hours of PA per week. Specifically, subjects suffering from 3 migraine days per month had a predicted probability exceeding 50% of being physically active, whereas participants reporting more than 3 days fell below this threshold.
Clustering analysis revealed three distinct participant groups. Cluster 1, labeled the Trapped Group (N = 127), was characterized by high migraine burden (median 10 monthly migraine days, MIDAS score 49), high depressive symptoms (mean = 2.09), and no engagement in regular PA (0%). This group also reported the highest levels of burnout and the lowest life satisfaction. Cluster 2, referred to as functioning but sedentary (N = 217), showed intermediate levels of disease severity (MIDAS median = 30), lower depression (mean = 0.64), and low rates of regular PA (21.7%). Functional outcomes in this group, including burnout, work-family conflict, and life satisfaction, fell between the other two clusters. Cluster 3, termed the Active & Functional group (N = 206), comprised participants with the lowest migraine disability (MIDAS median = 25), lowest depression (mean = 0.53), and the highest rate of PA (100%). This group also demonstrated more favorable psychosocial profiles, including lower burnout, less work-family conflict, and higher life satisfaction (Figure 2 and Supplementary Table 1).

Cluster profiles by health and psychological measures.
Discussion
In this cohort, participants reporting at least two hours per week of PA tended to have lower migraine-related disability. They reported better overall life satisfaction compared to their inactive peers, even after adjusting for demographic variables. Associations with depression and burnout were weaker and did not remain statistically significant once these covariates were considered, suggesting that differences in mental health may partly reflect underlying disease burden and sociodemographic variation rather than PA itself.
The probability of reporting regular PA declined once individuals exceeded approximately three migraine days per month, suggesting an activity engagement threshold beyond which sustained PA is less likely. Clustering analysis further illustrated this by delineating three distinct subgroups, “Trapped,” “Functioning but sedentary,” and “Active & functional”, which differed systematically in migraine burden, depressive symptoms, and activity levels. While directionality cannot be inferred, these profiles underscore that at higher levels of migraine frequency and psychosocial burden, participation in PA becomes increasingly unlikely.
These findings are consistent with prior evidence showing that active individuals with migraine often report lower disability and improved quality of life,26–28 as well as with the inverse associations between PA, migraine occurrence, and occupational stress. 29 Randomized trials and meta-analyses indicate that aerobic and multimodal exercise can reduce migraine frequency and intensity, though adherence and effect sizes vary.30,31 Reports linking PA to improved mental health are more inconsistent.32–34 In our cohort, differences in depression attenuated after adjustment, suggesting these associations may be mediated by overall burden rather than PA alone. Notably, the observed activity engagement threshold has not been previously reported and aligns with recent IHS position, which defines optimal migraine control as fewer than four headache episodes per month, an objective that is also commonly used to guide the initiation and evaluation of preventive therapy. 35
Proposed mechanisms by which PA may influence migraine include improvements in cerebrovascular function, modulation of nitric oxide signaling, reductions in systemic and neuroinflammatory activity, and alterations in central pain-processing pathways. Importantly, these mechanisms may become biologically relevant only after partial symptom stabilization allows patients to engage in regular activity. In addition to direct effects, PA has been shown to influence migraine-related outcomes through indirect pathways, including improvements in sleep regulation, 36 reductions in stress-related immune activation, 29 and modulation of mood processing. 37 Emerging evidence suggests that aerobic exercise and habitual PA can modulate inflammatory profiles in migraine patients, including changes in cytokine balance, and stress-related immune pathways, which have been linked to both migraine prevention and improvements in psychological well-being.38–40
Within this context, our results underscore a central paradox: while physically active individuals consistently report better functioning and well-being, those most burdened by migraine, depression, and burnout are often the least capable of engaging in activity. This highlights a bidirectional cycle, in which disease severity and psychological distress limit opportunities for exercise, and the absence of regular activity may in turn aggravate both migraine outcomes and mental health, as illustrated in Figure 3.

Conceptual illustration of the vicious cycle linking physical inactivity, migraine severity, and mental health, and their impact on occupational burnout, work-family conflict, and overall life satisfaction.
To break this cycle, we suggest the Pharmacology-Enabled Physical Activity (PEPA) framework, positioning pharmacologic treatment not as an alternative but as a means of lowering disease burden to a level that enables PA, which may then help consolidate broader functional and psychosocial gains. This staged approach is supported by recent evidence showing superior clinical responses to prophylactic anti-CGRP treatment among physically active patients, 41 as well as increases in daily step counts among treatment responders. 42
Clinically, this suggests that PA should not be delivered as uniform advice but integrated as a second-phase intervention once symptom control is achieved. Importantly, engagement in PA after pharmacologic stabilization may also serve as a marker of improvement, providing a pragmatic indicator for when cautious de-escalation of treatment might be considered. At a population level, PA represents a low-cost and scalable strategy with potential benefits across disability, functioning, and work participation. Recognizing it as part of a staged approach to care may also inform health-system guidelines and reimbursement models, particularly if engagement can be validated as a marker of recovery.
This study has several limitations. First, PA was measured by self-report and categorized dichotomously; we did not record specific modalities or intensity, which raises the possibility of exposure misclassification. Second, all outcomes were self-reported, which can introduce common-method bias. Finally, the cross-sectional design precludes causal inference; bidirectional associations among migraine, activity, and mental health cannot be ruled out. Future studies should test whether pharmacologic stabilization enables PA uptake and whether subsequent engagement predicts durable improvements, ideally through longitudinal or interventional designs with standardized outcome measures.
Conclusions
In this cohort, PA was associated with lower migraine-related disability and greater life satisfaction. An activity engagement threshold of about three migraine days per month emerged, beyond which PA was less likely. These findings suggest that PA may function best as a second-phase intervention following pharmacologic stabilization. That successful engagement could serve as a marker of improvement and a rationale for treatment de-escalation. Although causality cannot be inferred, the results support a staged approach to migraine care that integrates pharmacology with lifestyle strategies to improve long-term outcomes.
Public Health relevance
In this cohort, PA was associated with lower migraine-related disability and greater life satisfaction.
An activity engagement threshold of about three migraine days per month emerged, beyond which PA was less likely.
These findings suggest that PA may function best as a second-phase intervention following pharmacologic stabilization.
That successful engagement could serve as a marker of improvement and a rationale for treatment de-escalation.
The results support a staged approach to migraine care that integrates pharmacology with lifestyle strategies to improve long-term outcomes.
Supplemental Material
sj-docx-1-cep-10.1177_03331024261426569 - Supplemental material for Physical activity in migraine: Identifying an engagement threshold and patient clusters in a population-based cohort
Supplemental material, sj-docx-1-cep-10.1177_03331024261426569 for Physical activity in migraine: Identifying an engagement threshold and patient clusters in a population-based cohort by Ido Peles, Shaked Sharvit, Victor Novack and Gal Ifergane in Cephalalgia
Supplemental Material
sj-docx-2-cep-10.1177_03331024261426569 - Supplemental material for Physical activity in migraine: Identifying an engagement threshold and patient clusters in a population-based cohort
Supplemental material, sj-docx-2-cep-10.1177_03331024261426569 for Physical activity in migraine: Identifying an engagement threshold and patient clusters in a population-based cohort by Ido Peles, Shaked Sharvit, Victor Novack and Gal Ifergane in Cephalalgia
Footnotes
Acknowledgements
The study investigators received funding from Teva to conduct the study. Teva had no input on the study design, execution, data collection, analysis, interpretation of results, or preparation of the manuscript.
Author contributions
Ido Peles: Data analysis and manuscript preparation. Shaked Sharvit: focus group interviews, development of study questionnaires, and participation in study conceptualization. Victor Novack: Design and data analysis. Gal Ifergane: Ideation and conceptualization, study design, data analysis, manuscript preparation.
Consent to participate
The Israeli healthcare system is based on universal coverage through four non-profit HMOs, with compulsory enrollment under the National Health Insurance Law.
Consent for publishing
All authors have read and approved the final version of the manuscript and agree to its publication.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: o Gal Ifergane received consulting fees and honorariums from Teva, Novartis, Eli Lilli, Pfizer, and AbbVie. o Gal Ifergane received research support from Teva and Pfizer.
Ethical considerations
The study was approved by the Helsinki committee of Soroka Medical Center.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Teva Pharmaceutical Industries.
Open practices
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
