Abstract
Background
Classical trigeminal neuralgia (CTN) is a severe chronic pain disorder. While microvascular decompression (MVD) is an effective treatment, outcomes are variable, and preoperative predictive biomarkers are needed. Furthermore, the relationship between brain microstructural alterations and neurotransmitter systems in CTN is not well understood. This study aimed to characterize these alterations and assess their prognostic value for MVD.
Methods
We evaluated microstructural alterations in both white matter (WM) and gray matter (GM) in 101 CTN patients and 84 healthy controls (HCs) using Neurite Orientation Dispersion and Density Imaging (NODDI). Tract-Based Spatial Statistics (TBSS) and Gray Matter-Based Spatial Statistics (GBSS) were used to identify microstructural differences. Spatial correlations between WM and GM alterations and neurotransmitter distributions were analyzed using JuSpace. Univariate and multivariate logistic regression models with bootstrap validation were employed to identify predictors of pain recurrence after MVD in 58 patients who underwent the surgery.
Results
Compared to HCs, patients with CTN exhibited widespread microstructural disruptions in both WM and GM. These alterations, characterized by neurite injury and elevated free water, were spatially correlated with distributions of key neurotransmitter systems including serotonergic, dopaminergic, and GABAergic pathways. Critically, preoperative free-water accumulation in a specific GM cluster was identified as an independent predictor of MVD failure (AUC = 0.847). This neuroimaging biomarker provided a specificity of 97.92% for predicting poor surgical outcome.
Conclusion
Our findings indicate neurite injury in both WM and GM of CTN patients that is linked to specific neurotransmitter systems. Critically, preoperative free-water accumulation in GM emerges as a specific neuroimaging biomarker for predicting MVD failure, potentially guiding personalized therapeutic strategies.
This is a visual representation of the abstract.
Keywords
Introduction
Classical trigeminal neuralgia (CTN) is a severe and debilitating chronic pain disorder characterized by recurrent, paroxysmal shock-like pain in the trigeminal nerve distribution (1). It is often caused by neurovascular compression (NVC) at the root entry zone of the trigeminal nerve (2). Microvascular decompression (MVD) is the primary surgical intervention aimed at relieving this compression (3). Although MVD is an effective intervention for trigeminal neuralgia, a substantial proportion of patients still experience surgical failure (4). This suggests that even after peripheral compression is alleviated, persistent pain in some CTN patients may be attributable to central neuroplasticity and maladaptive changes within the pain matrix, a concept supported by neuroimaging evidence (5). However, findings regarding these central alterations are inconsistent, with some studies reporting correlations with clinical features or outcomes while others do not (6). The neurobiological basis of these alterations and the factors predicting surgical outcomes remain incompletely understood.
While conventional neuroimaging has revealed macrostructural changes in CTN, its sensitivity for detecting subtle microstructural and neurochemical disruptions is limited (7). Advanced diffusion MRI techniques, such as neurite orientation dispersion and density imaging (NODDI), allow for a more nuanced investigation of microstructural integrity by modeling specific biological features like neurite density index (NDI) and orientation dispersion index (ODI) (8). Furthermore, the interplay between structural reorganization and imbalances in neurotransmitter systems, which are critical in pain processing and modulation, is yet to be systematically explored in CTN.
To address these gaps, we employed a multimodal neuroimaging approach. First, we used NODDI to characterize microstructural alterations in both white matter (WM) and gray matter (GM) in a large cohort of CTN patients compared to healthy controls (HCs). Second, we investigated the spatial correlation between these changes and the distributions of major neurotransmitter systems using the JuSpace toolbox (9). Finally, we sought to identify preoperative diffusion MRI biomarkers predictive of pain recurrence after MVD surgery.
We hypothesized that CTN patients would demonstrate extensive microstructural disruptions within the central pain network, spatially associated with specific neurotransmitter systems. Moreover, we hypothesized that preoperative microstructural indices would predict long-term surgical outcomes, offering a potential tool for improving patient selection and personalizing treatment strategies.
Methods
Participants
We recruited 101 patients with CTN and 84 age- and sex-matched HCs between February 2024 and February 2025. All patients were diagnosed according to the International Classification of Headache Disorders (ICHD-III) by two independent neurologists (10). Inclusion criteria for patients were: age ≥ 18 years; unilateral CTN with a disease duration ≥ 6 months; paroxysmal electric shock-like pain with identifiable trigger points; absence of significant sensory deficits. Exclusion criteria included: persistent pain; history of cranial surgery (including MVD or other CTN-related procedures); other chronic pain or neuropsychiatric disorders; metal implants; abnormal MRI findings, including severe WM lesions (Fazekas grade III), multiple sclerosis or space-occupying lesions suggestive of secondary trigeminal neuralgia (STN); left-handedness; and contraindications to MRI (11,12). Demographic and clinical data were recorded. Pain intensity was assessed using the Visual Analog Scale (VAS), ranging from 0 (no pain) to 10 (the most severe pain imaginable). The VAS score was defined as the mean of the worst daily pain intensity ratings during paroxysms over the preceding 7 days. Additionally, mental status of all participants was assessed using the Self-Rating Anxiety Scale (SAS) and the Self-Rating Depression Scale (SDS) (13). 58 patients subsequently underwent MVD surgery. Postoperative outcomes (follow-up: 7–29 months) were classified using the Barrow Neurological Institute (BNI) pain intensity scale: complete relief (Grade I: no pain without medication), partial relief (Grades II–III: occasional pain controlled without or with medication) and patients with persistent pain (Grades IV–V) (14). Postoperative outcomes were categorized as BNI I in 48 cases (82.76%) and BNI II–III in 10 cases (17.24%). The inclusion flowchart is shown in Figure 1.

Flowchart of CTN patients enrollment and inclusion. CTN, classical trigeminal neuralgia; MVD, microvascular decompression.
This study was approved by the Ethics Committee of Lanzhou University Second Hospital (approval No. 2024A-1408). According to the Declaration of Helsinki, written consent was obtained from every participant after a detailed explanation of the study.
Image acquisition
All MRI data were acquired on a 3.0 T MRI scanner (Ingenia CX, Philips Healthcare, the Netherlands) with a 32-channel head-neck coil. During scanning, all participants were instructed to remain awake, keep their eyes closed, relax, and refrain from any active thinking. Earplugs and foam padding minimized noise and head motion. The protocol included:
NODDI: time of repetition (TR) = 5000 ms; time of echo (TE) = 102 ms; spatial resolution = 2 × 2 × 2 mm3; diffusion directions at b = 1000 s/mm2 (20 directions), b = 1800s/mm2 (40 directions), and b = 2500 s/mm2 (60 directions). An additional b = 0 s/mm2 image with reverse phase encoding for distortion correction. 3D Fast Imaging Employing Steady-State Acquisition (FIESTA): To visualize NVC with high spatial resolution, the following parameters were used: TR = 5.1 ms; TE = 1.9 ms; flip angle = 60°; matrix size = 320 × 288; field of view (FOV) = 200 × 170 mm2; slice thickness = 1 mm; slice spacing = 0 mm; number of slices = 50; and spatial resolution = 0.63 × 0.59 × 1 mm3. 3D T1-Weighted Imaging (3D T1WI): For anatomical reference and exclusion of secondary causes, the following parameters were used: TR = 5.9 ms; TE = 3.7 ms; flip angle = 8°; FOV = 256 × 256 mm2; voxel size = 1 × 1 × 1 mm3.
Image processing and analysis
All MRI images were independently analyzed by two experienced radiologists who were blinded to the clinical data.
Preprocessing
Diffusion data were preprocessed using the FMRIB Software Library (FSL, version 6.0.1, https://www.fmrib.ox.ac.uk/fsl) (15). First, the images underwent correction for eddy current distortions and rigid-body head motion to reduce image artifacts and motion-related interference. Subsequently, skull stripping was performed on the b0 images using FSL's Brain Extraction Tool (BET) to remove non-brain tissue (16)
Diffusion model fitting and spatial statistics
The preprocessed diffusion data were first used to derive DTI metrics (fractional anisotropy [FA], mean diffusivity [MD], axial diffusivity [AD], and radial diffusivity [RD]) using the dtifit tool in FSL. Subsequently, the CUDA Diffusion Modelling Toolbox (cuDIMOT, https://users.fmrib.ox.ac.uk/∼moisesf/cudimot/) was employed for NODDI model fitting to generate parameter maps, including the NDI (reflecting neurite density), ODI (reflecting axonal branching complexity), and the fraction of isotropic diffusion (FISO; representing free water content and potentially indicating edema or inflammation) (17)
Voxel-wise analyses of WM and GM microstructural alterations were performed using Tract-Based Spatial Statistics (TBSS) and Gray Matter-Based Spatial Statistics (GBSS), respectively. The detailed preprocessing and statistical pipelines for TBSS and GBSS were described in the Supplementary Method S1.
Neurotransmitter mapping
Spatial correlations between microstructural alterations and neurotransmitter systems were explored using the JuSpace toolbox (https://github.com/juryxy/JuSpace). This analysis utilized preprocessed neurotransmitter maps derived from PET/SPECT data of independent healthy cohorts, registered and normalized to MNI space (9). We aimed to determine whether the spatial pattern of alterations in CTN patients resembled the distribution of maps for: the serotonergic system (5-hydroxytryptamine 1A [5-HT1a], 5-hydroxytryptamine 1B [5-HT1b], 5-hydroxytryptamine 2A [5-HT2a], and 5-hydroxytryptamine 4 [5-HT4], serotonin transporter [SERT]); the dopaminergic system (D1, D2, dopamine transporter [DAT], and 6-[18F]fluoro-L-dopa [FDOPA]); the cholinergic system (vesicular acetylcholine transporter [VAChT]); the glutamatergic system (metabotropic glutamate receptor 5 [mGluR5] and N-methyl-D-aspartate [NMDA]); the GABAergic system (gamma-aminobutyric acid type A [GABAa]); the endocannabinoid system (cannabinoid 1 [CB1]); the opioid system (µ-opioid [MU] and kappa opioid [KappaOp]); the noradrenergic system (noradrenaline transporter [NAT]); and other relevant maps (cerebral blood flow [CBF]) (18–23). Significant clusters from TBSS/GBSS group comparisons (z-transformed relative to HCs) served as masks. Spearman correlation coefficients (Fisher's z-transformed) were computed between these patient z-transformed maps and each neurotransmitter map. The distribution of observed Fisher's z-transformed individual correlation coefficients was tested for significant deviation from zero by calculating exact permutation-based p-values in the JuSpace, which involved 10,000 orthogonal permutations randomly assigning group labels.
Spatial autocorrelation was accounted for by computing partial correlation coefficients between the significant clusters and each neurotransmitter map. Additionally, local GM probability maps, estimated from the SPM12 TPM.nii template, were included as a voxel-wise covariate to adjust for tissue composition effects. The standard atlas used for registering TBSS results was the JHU-ICBM-tracts-maxprob-thr25-1 mm.nii, while the m_labels_Neuromorphometrics.nii atlas was applied for GBSS result registration (23). All figures were generated using OriginPro (version 2024).
Statistical analysis
Group comparisons
Categorical variables were assessed for group differences using the chi-square test. The normality of continuous variables was evaluated using the Shapiro-Wilk test. Normally distributed continuous variables were expressed as mean ± standard deviation (SD) and compared between CTN patients and HCs using independent samples t-tests. Non-normally distributed variables are summarized as median and interquartile range (IQR) and compared between groups using the Mann–Whitney U test. Group comparisons between the TBSS/GBSS results of CTN and HCs were performed using a general linear model (GLM), with age, sex, and years of education as covariates. To control for multiple comparison errors, threshold-free cluster enhancement (TFCE) and 5000 permutation tests were applied for multiple testing correction in each contrast. Significant voxels were determined by controlling the family-wise error (FWE) rate at a corrected p < 0.050.
Clinical correlation analysis
Pearson correlation analysis (for normally distributed data) and Spearman correlation analysis (for non-normally distributed data) were used to explore the relationships between MRI measurements and neuropsychological scores.
Association between clinical features and microstructural indices
To explore the relationship between brain microstructure and clinical presentation, subgroup analyses were performed based on four clinical variables.
NVC Grade: Grade I (vessel-nerve contact without indentation), Grade II (contact with visible indentation), and Grade III (contact causing nerve displacement or deformation). Carbamazepine/oxcarbazepine usage: Patients were dichotomized into “users” or “non-users” based on preoperative use of carbamazepine or oxcarbazepine. Of the cohort, 60 patients were treated with these medications. An additional 8 patients received only traditional Chinese medicine (TCM, including herbs or acupuncture). Since 4 patients received both anticonvulsant and TCM therapies, and this subgroup was small. Therefore, all patients using carbamazepine/oxcarbazepine (n = 64) were classified as “users” group, while others (n = 27) were classified as “non-users” group. Vasculometabolic comorbidities: Within the patient cohort, 12 patients had hypertension, 4 had diabetes, and 2 had cardiovascular disease, with one patient having both hypertension and diabetes. Based on this clinical profile, patients were categorized into a “present” group if they had at least one of these specified conditions, while all others comprised the “absent” group. Daily pain attack frequency:1–5 attacks /day, 6–10 attacks /day, 11–20 attacks /day, ≥21 attacks /day.
For each diffusion metric, inter-subgroup comparisons used the Mann–Whitney U test (binary variables) or Kruskal–Wallis H test (ordinal variables), with post-hoc Dunn's tests when the overall test remained significant after multiple comparison correction.
Prediction of surgical outcome
For the 58 patients who underwent MVD, univariate and multivariate logistic regression models were developed to predict poor surgical outcomes. Candidate predictors included both imaging metrics (mean values extracted from significant TBSS/GBSS microstructural maps) and key clinical features (NVC grade, carbamazepine use, vasculometabolic comorbidity, and pain attack frequency). Given the limited sample size, bootstrap validation with 1000 iterations was applied to enhance the robustness and generalizability of the model. The discriminative performance of the final multivariate model was evaluated using the area under the receiver operating characteristic (ROC) curve (AUC), implemented in MedCalc (version 22.0) (24)
All statistical analyses were performed using SPSS (version 27.0) and GraphPad Prism (version 9.0). For analyses involving multiple comparisons, the false discovery rate (FDR) was controlled using the Benjamini-Hochberg procedure. Effects with a pFDR value < 0.050 were considered statistically significant.
Results
Demographic and clinical characteristics
Among the initially enrolled 101 patients, five were excluded due to persistent pain, two due to a history of MVD, one due to discomfort preventing MRI completion, and two due to a cerebellopontine angle lesion. Consequently, a total of 91 patients with CTN (51 females, 40 males; mean age 57.65 ± 11.79 years) and 84 age- and sex-matched HCs (48 females, 36 males; mean age 54.85 ± 8.09 years) were enrolled. The disease duration ranged from 6 to 360 months (median, 58 months). No significant differences were found in demographic characteristics (age, sex, education level) between the groups (all p > 0.050). however, patients with CTN had significantly higher SDS and SAS scores than HCs (p < 0.001) (Table 1).
Demographic and clinical characteristics of CTN patients and HCs.

CTN, classical trigeminal neuralgia; HCs, healthy controls; IQR, interquartile range; NVC, neurovascular compression; SAS, self-rating anxiety scal ;SDS, self-rating depression scale;VAS, visual analog scale.
WM microstructural alterations
Compared to HCs, patients with CTN showed widespread WM alterations. TBSS analysis revealed significant decreases in FA, NDI, along with increases in MD, AD, RD, ODI, and FISO (all pTFCE < 0.050). These changes were located in major fiber tracts, including the anterior thalamic radiations (ATR), corticospinal tracts (CST), cingulum, superior/inferior longitudinal fasciculi (SLF/ILF), and uncinate fasciculi (UF) bilaterally. The peak MD increase was in the right ATR and CST. These results suggest widespread microstructural damage and increased free water in the WM of patients with CTN (Table S1, Figure 2(a)). No significant correlations between VAS, SDS, or SAS scores and these WM alterations survived FDR correction (Table S2, Figure S1).

Widespread microstructural alterations in both WM and GM of CTN patients. a. TBSS results demonstrate widespread white matter microstructural alterations in patients with CTN compared to HCs, overlaid on the mean WM skeleton (green). Significant decreases in FA and NDI are shown in blue, while significant increases in MD, AD, RD, ODI, and FISO are indicated in red. b. GBSS results show regions of significant microstructural differences between patients and HCs, projected onto the mean gray matter skeleton (green). Clusters with decreased FA are depicted in blue, and those with increased FISO are shown in red. AD, axial diffusivity; COR, coronal plane; FA, fractional anisotropy; FISO, fraction of isotropic water; GBSS, gray matter-based spatial statistics; GM, gray matter; HCs, healthy controls; MD, mean diffusivity; NDI, neurite density index; ODI, orientation dispersion index; RD, radial diffusivity; SAG, sagittal plane; TBSS, Tract-Based Spatial Statistics;TRA, transverse plane; WM, white matter. pFWE, p-value corrected for Family-Wise Error.
GM microstructural alterations
GBSS analysis revealed significant GM changes. We observed decreased FA in clusters within the parahippocampal gyrus (PHG), insular cortex (INS), and a large cluster extending from the precentral gyrus (PreG) to the occipital pole. Concurrently, increased FISO was found in the posterior cingulate cortex (PCC), precuneus, fusiform gyrus, and lingual gyrus (LG) (Table S3, Figure 2(b)). Clinical correlation analyses revealed that higher SDS scores correlated with elevated FISO cluster2 (rs = 0.26, pFDR=0.039) (Table S2, Figure S2).
Association between microstructural alterations and clinical features
The comprehensive results of all subgroup comparisons were detailed in Table S4. After FDR correction, only one association remained significant: TBSS_MD1 varied significantly across NVC grades (pFDR = 0.048). Post-hoc comparisons indicated that TBSS_MD1 values were significantly higher in Grade III than in Grade I (pFDR = 0.009) (Table S5).
Correlation with neurotransmitter systems
We observed significant spatial correlations between neuroimaging markers of microstructural integrity and the distributions of various neurotransmitter systems (Table S6, Figure 3). Regarding WM alterations, lower TBSS_NDI showed negative spatial correlations with multiple neurotransmission maps, including serotonergic (5-HT1a, 5-HT1b, 5-HT2a, 5-HT4), cannabinoid (CB1), dopaminergic (D1, FDOPA), cholinergic (VAChT), GABAergic (GABAa), glutamatergic (mGluR5), and opioid (MU, KappaOp) systems, as well as with CBF (all Fisher's z < 0). Increased TBSS_FISO was specifically negatively correlated with 5-HT1b and 5-HT2a (all Fisher's z < 0). For GM alterations, lower GBSS_FA was negatively associated with 5-HT1b (Fisher's z = -0.36) but positively associated with dopaminergic (D1, D2, DAT, FDOPA), serotonergic (5-HT1a, 5-HT4, SERT), cholinergic (VAChT), noradrenergic (NAT), and glutamatergic (NMDA) markers (all Fisher's z > 0). Increased GBSS_FISO was negatively coupled with 5-HT1b, GABAa, and CBF (all Fisher's z < 0), but positively corresponded with the dopaminergic (D1, D2, DAT, FDOPA), serotonergic (5-HT1a, SERT), and cholinergic (VAChT) systems (all Fisher's z > 0).

Radial bar plot visualization of spatial correlations between microstructural alterations and neurotransmitter systems. a. Correlations between white matter alterations and neurotransmitter systems. b. Correlations between gray matter alterations and neurotransmitter systems. 5-HT1a, 5-hydroxytryptamine 1A; 5-HT1b, 5-hydroxytryptamine 1B; 5-HT2a, 5-hydroxytryptamine 2A; 5-HT4, 5-hydroxytryptamine 4; CB1, cannabinoid 1; CBF, cerebral blood flow; DAT, dopamine transporter; FDOPA, 6-[18F]fluoro-L-dopa; GABAa, gamma-aminobutyric acid type A; KappaOp, kappa opioid; mGluR5, metabotropic glutamate receptor 5; MU, µ-opioid; NAT, noradrenaline transporter; NMDA, N-methyl-D-aspartate; SERT, serotonin transporter; VAChT, vesicular acetylcholine transporter. *False discovery rate (FDR)-corrected p < 0.05.
Preoperative microstructural alterations predict surgical outcome
At follow-up, pain recurred in 10 of the 58 patients (17.24%) who underwent MVD. In univariate analysis, severe neurovascular conflict (NVC Grade III vs. Grade I) was associated with a significantly lower likelihood of recurrence (pBootstrap < 0.001). Among neuroimaging metrics, preoperative elevation of TBSS_AD (pBootstrap=0.018), GBSS_FISO1 (pBootstrap=0.013), GBSS_FISO2 (pBootstrap=0.009) and GBSS_FISO3 (pBootstrap=0.003) were each significantly associated with a higher recurrence risk. Multivariate analysis confirmed free-water accumulation in a specific gray matter cluster (GBSS_FISO3) as the sole independent predictor of surgical failure (pBootstrap = 0.006) (Table 2). This biomarker demonstrated excellent discriminatory power (AUC = 0.847; Figure 4). A preoperative GBSS_FISO3 value > 0.491 identified patients with a poor prognosis, with a specificity of 97.92% (95% CI, 88.90–99.90%) and a sensitivity of 60.00% (95% CI, 26.20–87.80%).
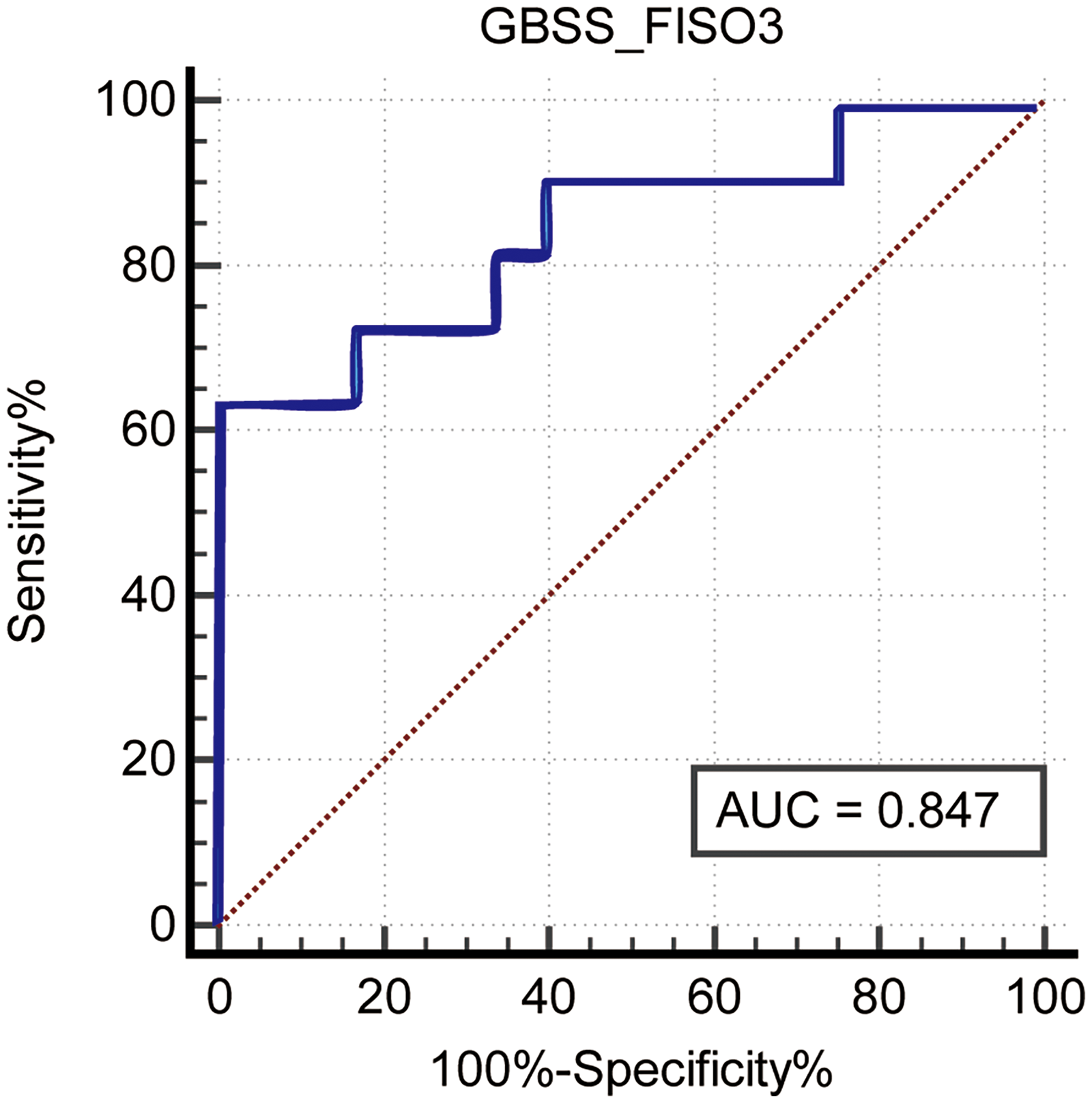
ROC curve for the predictive performance of biomarker GBSS_FISO3. AUC, area under the curve; ROC, receiver operating characteristic.
Univariate and multivariate logistic regression analyses of preoperative imaging metrics for predicting surgical recurrence.

AD, axial diffusivity; CI, confidence interval; FA, fractional anisotropy; FISO, fraction of isotropic water; GBSS, gray-matter-based spatial statistics; MD, mean diffusivity; NDI, neurite density index; ODI, orientation dispersion index; RD, radial diffusivity; TBSS, Tract-Based Spatial Statistics.
A positive coefficient indicates an increased risk of recurrence. The multivariate model includes all variables with p < 0.05 from univariate analysis.
Discussion
The present study utilized advanced multimodal neuroimaging to comprehensively investigate the microstructural and neurochemical underpinnings of CTN and to identify a novel predictor for surgical outcomes. Our principal findings reveal three key insights: first, CTN is associated with extensive microstructural disruptions, characterized by neurite injury and free water accumulation, spanning both WM and GM regions within the central pain matrix. Second, these structural alterations exhibit a specific spatial correlation with the distributions of key neurotransmitter systems involved in pain processing and modulation. Third, and most critically, preoperative free-water accumulation in GM (GBSS_FISO3) emerges as a robust and independent neuroimaging biomarker predicting poor long-term response to MVD.
Our NODDI findings provide a more biologically specific characterization of microstructural pathology in CTN than conventional DTI metrics. The widespread reductions in FA and NDI across multiple WM tracts suggest a coherent pattern of axonal degeneration and compromised microstructural integrity (25). This pattern likely reflects morphological changes in activated microglia and astrocytes, including cellular hypertrophy and process proliferation, which alter local tissue architecture and subsequently influence both diffusion anisotropy and molecular hindrance (26). Concurrently, the observed increase in FISO is indicative of extracellular edema, a recognized imaging hallmark of neuroinflammation or glial activation (27). This process may be driven by proinflammatory cytokines such as tumor necrosis factor-α and interleukin-1β, which can disrupt blood-brain barrier integrity and promote pathological fluid extravasation (28). Crucially, we extended this analysis to GM using GBSS, revealing similar microstructural deficits in key regions of the sensory-discriminative and affective-motivational pain pathways, including the thalamus, insula, and cingulate cortex. Together, the concurrence of elevated free water and altered diffusivity across both WM and GM strongly suggests a shared underlying pathophysiology, potentially driven by widespread neuroinflammatory processes and glial activation, which may represent a central mechanism bridging structural damage, neurochemical dysregulation, and clinical symptoms in CTN. Furthermore, our clinical subgroup analyses revealed that more severe NVC was associated with elevated TBSS_MD1. This correlation likely reflects more pronounced axonal injury and microstructural disorganization under greater mechanical stress. (29)
Despite extensive brain-wide microstructural alterations, only the GBSS_FISO2 exhibited a significant positive correlation with depression scores after multiple comparisons correction. This specific association may highlight the role of free-water accumulation in the high comorbidity of affective disorders in CTN (30). More broadly, the general dissociation between the magnitude of macrostructural change and clinical symptom severity suggests that subjective pain and distress may not be directly encoded in static diffusion metrics but are likely mediated by dynamic functional reorganizations or neurochemical adaptations within the affected networks.
The JuSpace analysis offers a pioneering link between macroscale imaging findings and microscale neurochemistry (31). The microstructural defects we discovered in CTN show significant spatial correlations with the distributions of serotonergic, dopaminergic, GABAergic, and glutamatergic systems.
Both the serotonergic and noradrenergic systems play fundamental roles in descending pain inhibition, and their dysregulation may contribute to the persistent and paroxysmal pain characteristic of trigeminal neuralgia (32,33). However, emerging evidence suggests that in certain pathophysiological contexts, the descending serotonergic system may undergo a functional shift—transitioning from a protective “pain suppressor” to a facilitatory “pain promoter” (34). This paradoxical mechanism may explain the observed negative correlations between specific serotonergic markers and microstructural alterations in our study, highlighting a complex, potentially maladaptive, neurochemical reorganization in trigeminal neuralgia. Similarly, alterations in dopaminergic signaling, which plays a complex role in pain perception and modulation, and an imbalance between excitatory (glutamatergic) and inhibitory (GABAergic) neurotransmission could contribute to central sensitization and hyperexcitability (35,36). This integrative approach suggests that the observed microstructural damage may colocalize with or disrupt specific neurotransmitter circuits, providing a more holistic pathophysiological model of CTN.
The most translatable finding of our study is the identification of GBSS_FISO3 as a powerful independent predictor of MVD outcome. Although whole-brain WM alterations in CTN have been previously documented, and microstructural metrics at the NVC site have been proposed as potential predictors, (37,38). our work specifically focuses on the predictive value of whole-brain WM and GM microstructural changes for MVD outcomes (39,40). Importantly, our results highlight that increased free-water content in specific GM areas, reflective of underlying neuroinflammation or gliosis, is a critical factor portending a suboptimal surgical response. This suggests that a subset of patients may present with a more progressive central disease phenotype, where peripheral decompression is insufficient to reverse established maladaptive central changes (41). Consequently, preoperative FISO mapping could serve as a valuable clinical tool for prognostic stratification, guiding neurosurgeons and patients in personalized decision-making. Patients identified as high-risk for recurrence based on this biomarker might be candidates for earlier intervention, adjunct medical therapies, or more comprehensive pain management strategies.
Several limitations should be considered. First, while JuSpace provides compelling spatial correlations, it is an indirect method and cannot confirm causal or direct neurochemical changes. Second, the cross-sectional design of our main case-control analysis limits our ability to infer whether the observed microstructural alterations are a cause or a consequence of chronic pain. Longitudinal studies tracking patients from disease onset would be valuable in elucidating the temporal evolution of these changes. Third, our logistic regression analysis is exploratory in nature, and the limited patient cohort size from a single center limits the generalizability of our findings. Fourth, and relatedly, the identified predictive biomarker requires external validation in independent, multi-center cohorts to confirm its clinical utility and robustness. Future research should integrate longitudinal neuroimaging with direct neurochemical assessments such as positron emission tomography (PET) and the analysis of biofluid biomarkers from cerebrospinal fluid and peripheral blood to elucidate the temporal dynamics and molecular mechanisms linking neuroinflammation, glial activation, and treatment resistance.
In conclusion, our study provides robust evidence that CTN involves widespread neurite injury and free-water accumulation in the central pain network, which is spatially linked to major neurotransmitter systems. We move beyond mere description to offer a clinically actionable biomarker: preoperative GM free-water accumulation predicts MVD failure with high accuracy. This finding not only advances our pathophysiological understanding of CTN but also paves the way for imaging-guided personalized medicine in the surgical management of this debilitating condition.
Clinical implications
Our study reveals widespread microstructural alterations in both gray and white matter across the central pain network of classical trigeminal neuralgia.
We demonstrate that these microstructural changes spatially correlate with key neurotransmitter systems, suggesting linked pathophysiological mechanisms.
We identify preoperative free-water in gray matter (GBSS_FISO3) as a predictive biomarker for surgical outcome, offering a tool for clinical prognosis.
Supplemental Material
sj-docx-1-cep-10.1177_03331024261423444 - Supplemental material for Predictive value of preoperative white and gray matter microstructural alterations for microvascular decompression outcome in classical trigeminal neuralgia
Supplemental material, sj-docx-1-cep-10.1177_03331024261423444 for Predictive value of preoperative white and gray matter microstructural alterations for microvascular decompression outcome in classical trigeminal neuralgia by Jingqi Jiang, Danyang Wang, Shu Cui, Pengfei Zhang, Jun Wang, Wenjing Huang, Zheng Cheng, Fei Jia, Zhuo Wang, Zhengbo Lan, Yanming Han, Yanli Jiang, Guangyao Liu and Jing Zhang in Cephalalgia
Footnotes
Author contributions
Jingqi Jiang: Conceptualization, Data Curation, Formal Analysis, Writing – Original Draft.
Danyang Wang: Methodology, Formal Analysis, Investigation, Writing – Original Draft.
Shu Cui: Software, Formal Analysis, Writing – Original Draft.
Pengfei Zhang: Methodology, Investigation, Validation.
Jun Wang: Methodology, Resources, Funding Acquisition.
Wenjing Huang: Visualization, Software.
Zheng Cheng: Visualization, Software.
Fei Jia: Investigation, Validation.
Zhuo Wang: Investigation, Validation.
Zhengbo Lan: Resources, Data Curation.
Yanming Han: Resources, Data Curation.
Yanli Jiang: Writing – Review & Editing, Project Administration.
Guangyao Liu: Writing – Review & Editing, Project Administration.
Jing Zhang: Conceptualization, Supervision, Funding Acquisition.
Consent to participate
Written consent was obtained from every participant after a detailed explanation of the study.
Consent for publishing
All authors have read and approved the final version of the manuscript and consent to its submission to Cephalalgia for publication. The work described is original and has not been published previously, nor is it under consideration for publication elsewhere.
Data availability statement
Raw data inquiries are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
This study was approved by the Ethics Committee of Lanzhou University Second Hospital (approval No. 2024A-1408).
Funding
This work was supported by Gansu Provincial Key Research and Development Program: No. 23YFFA0041. Gansu Provincial Science and Technology Program Project: No. 21JR7RA438. Cuiying Scientific and Technology Innovation Program of Lanzhou University Second Hospital: No. CY2024-YB-B04.
Open practices
Not applicable.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
