Abstract
Vascular compression is a well-established cause of cranial nerve neuralgic syndromes. A unique case is presented that demonstrates that vascular compression may be a possible cause of occipital neuralgia. A 48-year-old woman with refractory left occipital neuralgia revealed on magnetic resonance imaging and computed tomographic imaging of the upper cervical spine an atypically low loop of the left posterior inferior cerebellar artery (PICA), clearly indenting the dorsal upper cervical roots. During surgery, the PICA loop was interdigitated with the C1 and C2 dorsal roots. Microvascular decompression alone has never been described for occipital neuralgia, despite the strong clinical correlation in this case. Therefore, both sectioning the dorsal roots of C2 and microvascular decompression of the PICA loop were performed. Postoperatively, the patient experienced complete cure of her neuralgia. Vascular compression as a cause of refractory occipital neuralgia should be considered when assessing surgical options.
Introduction
Occipital neuralgia is an uncommon cause of headache described in 1821 by Beruto and Ramos. It is a specific syndrome of paroxysmal severe lightening-like sharp headache in the distribution of the great or lesser occipital nerve. This paroxysmal jabbing pain may be associated with aching in the greater or lesser occipital nerve distributions between paroxysms. Often there is tenderness over the nerve and sometimes there can be diminished sensation or dyaesthesia in the distribution of the nerve. The pain is temporarily relieved by local block (1). Although most cases of occipital neuralgia are idiopathic, they may be related to specific causes such as trauma, prior skull base surgery, rheumatoid arthritis, nerve entrapment by hypertrophied atlantoepistrophic (C1–C2) ligament, compression by an anomalous ectatic vertebral artery, or degenerative C1–C2 arthrosis (2, 3). A wide variety of treatments have been tried, including cervical collars, transcutaneous nerve stimulation, analgesics and antimigraine drugs, occipital nerve block with or without glucocorticoid injection, chemical or radio-frequency occipital nerve ablation, atlantoaxial arthrodesis, and dorsal cervical rhizotomy, all with variable degrees of success.
We present a case of a patient with occipital neuralgia refractory to all forms of conservative treatment. This patient's findings were unique in that they are the first, to our knowledge, to reveal posterior inferior cerebellar artery (PICA) compression of the upper cervical nerve roots. Following microvascular decompression and cervical root sectioning, the patient was cured of her neuralgic pain.
Case report
History
A 48-year-old, left-handed female with a history of depression and occasional benign headache presented with a 17-month history of left occipital headache. The pain originated at the occipito-cervical junction and radiated to the vertex. It was a sharp pain that was constant in nature with exacerbating factors such as neck flexion and leftward deviation. At times, it was associated with nausea and photosensitivity. Her initial diagnosis was that of occipital neuralgia with superimposed migraine features.
During her evaluation, she had been placed on >15 medications that included antiemetics, opioids, non-steroidal anti-inflammatory drugs (NSAIDs), antiepileptics, steroids, anticholinergics, β-blockers, triptans and others unknown to her. She had received three separate occipital nerve blocks with only brief (days) palliation. Radio-frequency ablation of the greater and lesser occipital nerves had been attempted twice with minimal effect. During this time, she had also sought the assistance of a chiropractor and acupuncturist. On the day of presentation, her headache had remained unchanged in severity. Furthermore, she was wearing a transdermal narcotic patch and taking high doses of oral narcotics.
Examination
Intracranial and cervical magnetic resonance imaging (MRI) was obtained and was negative for haemorrhage, mass effect or cervical disk abnormality. The cervical foramina were open and there were no compressive lesions on the cervical cord. On the T2-weighted sequence, an anomalous, low PICA was observed on the left. The distal loop extended to the level of the lamina of the first cervical vertebrae (Fig. 1). A subsequent computed tomographic myelogram confirmed the findings of the MRI (Fig. 2), indicating that the left PICA was anomalous and that it extended to the level of the upper cervical nerve roots.
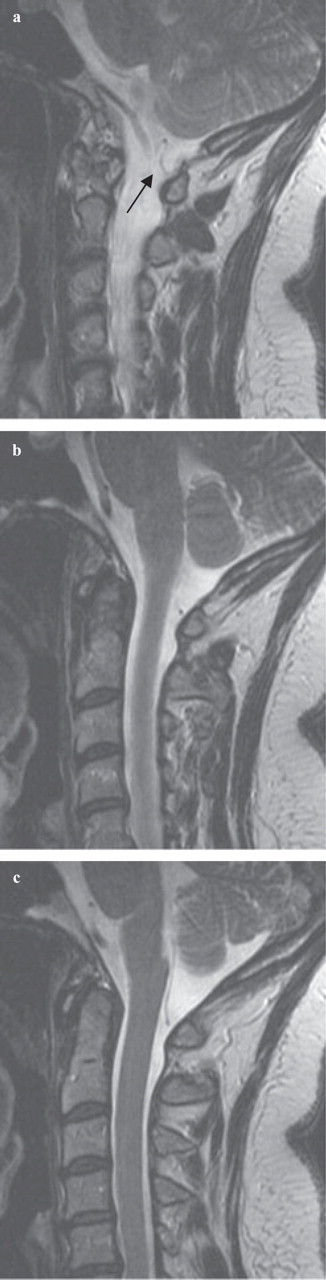
Sagittal, fast-spin echo T2-weighted magnetic resonance images of the occipito-cervical junction showing the anomalous course of the left posterior inferior cerebellar artery (arrow). (a–c) Sequential images from the patient's left to right.

Coronal, reformatted computed tomography-myelogram images of the occipito-cervical junction showing the tortuous and caudal course of the posterior inferior cerebellar artery as it nears the left lamina of C1 (arrow). (a–c) Sequential imaging from posterior to anterior.
Treatment
The patient was taken to the operating room, where a suboccipital craniectomy and left C1 hemilaminectomy were performed. A curvilinear incision was made in the dura and it was reflected using silk suture. Through the arachnoid layer, the anomalous PICA could be viewed as it coursed near the C1 and C2 nerve roots (Fig. 3). The PICA was positioned dorsal to C2 yet ventral to C1. After identification of the upper cervical roots, the C1 and C2 nerve roots were sectioned. A small piece of polyvinyl chloride was then placed between the PICA and the brainstem. The dura was closed primarily and the case was completed without complication.

Intraoperative photographs of the microvascular decompression. (a) Elevation of the C1 nerve root as it courses over posterior inferior cerebellar artery (PICA). (b) Elevation of the C2 nerve root as it rests ventral to PICA. (c) The C1 and C2 nerve roots after sectioning. (d) The end of the decompression with both C1 and C2 nerve roots sectioned as well as polyvinyl chloride placement between PICA and the brainstem. ∗PICA.
Postoperative course
The patient awoke with incisional pain typical for her procedure. During her 2-day hospitalization, she did not experience any neuralgic pain. At 5 months' follow-up, she had alleviation of her neuralgic pain, although a severe holocephalic headache occurred intermittently.
Discussion
Occipital neuralgia is a pain syndrome characterized by periodic lancinating pain involving the occipital nerve complex. Generally, the pain starts at the nape of the neck and radiates upward over the vertex (1). Temporal and retro-orbital pain has also been reported with patients suffering from occipital neuralgia. Likewise, patients who experience occipital neuralgia often experience trigeminal neuralgia (2). This diagnosis must be distinguished from cervicogenic headache due to an occipital referred pain from other sources, such as the atlantoaxial or upper zygoapophyseal joints. Cervicogenic headache is diagnosed in the setting of imaging consistent with a valid cause of headache, such as tumour, fracture, infection, or inflammatory arthritis, none of which was identified in this patient (1).
The source of pain in occipital neuralgia is thought to emanate from the greater and lesser occipital nerves. The greater occipital nerve is formed by the medial branch of dorsal ramus of C2. It also receives a branch from the medial branch of the dorsal ramus of C3. The suboccipital nerve may also lend a branch via the dorsal ramus of C1. The occipital nerve innervates the skin of the back of the scalp and as far cephalad as the vertex.
The medical treatment of occipital neuralgia has taken many forms, including antiepileptics, antidepressants, tricyclic amines, triptans, narcotics, steroids and NSAIDs. Radio-frequency ablation and occipital nerve blocks have also been utilized with mixed results. As conservative measures fail, cervical rhizotomy has often been the definitive treatment (4).
Surgical treatment of occipital neuralgia is generally in the setting of typical neurovascular anatomy. We are aware of only one previous report in the literature where the neuralgic pain was thought to be due to vascular compression. Watanabe et al. have presented a case of occipital neuralgia in the setting of bilateral anomalous vertebral arteries (5). In their report, the vertebral arteries entered the spinal canal between the atlas and axis, leading to spinal cord compression. The surgical treatment was elevation of the vertebral arteries with subsequent cure of the patient's neck pain.
Our patient presented with convincing symptoms of occipital neuralgia in the setting of polypharmacy and migraine headaches. The neuroimaging and operative findings were convincing for a compression-related neuralgia as has been reported in other types of neuralgia. Though the imaging and surgical findings were convincing, there was reluctance to perform only a microvascular decompression given that all other attempts to control her occipital neuralgia had failed. Surgical cure was achieved by a C1 and C2 rhizotomy as well as microvascular decompression of the brainstem.
The idea was first conceptualized by W. Dandy, and subsequently the first series by Gardner et al. appeared (6). Jannetta popularized the procedure and has produced the largest microvascular decompression series in the world for trigeminal neuralgia, with excellent results. The notion of microvascular decompression for neuralgic pain is not a new one (7, 8). In his series, the superior cerebellar artery was the most common vascular structure impinging upon those structures. Since the initial reports, microvascular decompression has been used for treatment of hemifacial spasm, tinnitus, vertigo, hypertension and glossopharyngeal neuralgia. It is an attractive hypothesis in this patient also, and suggests that a vascular compressive aetiology should be sought in refractory cases of occipital neuralgia.
Conclusions
We have suggested that occipital neuralgia may on occasion be produced by PICA compression of the upper cervical nerve roots. In occipital neuralgia patients for whom conservative treatment modalities fail, a vascular compressive cause should be sought and surgical therapy offered accordingly.
