Abstract
Background
Migraine is a heterogeneous disorder marked by recurrent attacks and highly variable treatment outcomes, and it continues to impose a substantial personal and societal burden worldwide, underscoring the need for innovative and accessible therapeutic approaches. Although pharmacological options have expanded in recent years, many patients still experience incomplete response or limited access to specialist care. In this context, digital and virtual technologies—including mobile health applications, remote electrical neuromodulation (REN), virtual reality (VR), biofeedback, and internet-based behavioral therapy—have emerged as promising non-pharmacological tools to enhance self-management and improve clinical outcomes.
Methods
We conducted a systematic review of randomized and quasi-randomized controlled trials (RCTs) evaluating digital or virtual interventions for migraine prevention or acute treatment. Searches were performed in MEDLINE and Embase (via PubMed and Ovid®) from database inception to September 4, 2025, following PRISMA guidelines (PROSPERO CRD420251078125). Eligible studies included adults with migraine diagnosed according to the International Classification of Headache Disorders (ICHD). Data were extracted on intervention type, outcomes, adherence, and adverse events. Risk of bias was assessed using the Cochrane RoB 2 tool.
Results
Twelve studies met inclusion criteria, mean age across studies ranged from 35 to 47 years and interventions included digital behavioral therapy (42%), REN (17%), VR (17%), biofeedback (17%), and telemedicine (8%). REN and VR-based interventions demonstrated significant reductions in pain intensity and acute medication use compared with controls (p < 0.01), while sEMG-digital biofeedback was associated with significant improvements in migraine-related disability (p = 0.03) and quality of life (p = 0.003). Behavioral and app-based interventions showed high feasibility but modest effects on headache frequency. No serious adverse events were reported. Risk of bias was judged to be low in only one study, with some concerns identified in two, while the remaining nine were considered at high risk of bias, largely driven by open-label study designs and incomplete outcome data.
Conclusions
Digital and virtual therapeutics represent a rapidly evolving and clinically promising frontier in migraine care. REN and VR modalities yield reproducible short-term benefits, while digital behavioral tools enhance feasibility, patient engagement, and continuity of care. Future large-scale, blinded RCTs with standardized endpoints are warranted to confirm efficacy, establish long-term outcomes, and define the role of digital health within integrated, precision-based migraine management.
Trial Registration
PROSPERO, CRD420251078125.
This is a visual representation of the abstract.
Introduction
Affecting more than one billion people across the globe, migraine stands as one of the most prevalent and disabling neurological conditions, imposing a heavy personal and societal burden that extends far beyond the pain itself (1,2). Despite major advances in pharmacological treatment—ranging from triptans and gepants to monoclonal antibodies targeting the calcitonin gene-related peptide (CGRP) pathway—many patients continue to experience suboptimal outcomes, treatment-related side effects, or barriers to access specialized care (3,4). In parallel, the chronic and fluctuating nature of migraine, together with its strong behavioral and psychosocial components, highlights the need for integrated management strategies that extend beyond medication (5).
In recent years, the digital health revolution has offered new opportunities to address these unmet needs (6). A growing body of evidence supports the use of digital and virtual interventions, including mobile applications, telemedicine, biofeedback, cognitive-behavioral therapy (CBT) delivered online, and virtual reality (VR)–based approaches, as innovative tools to support migraine prevention and acute management (7,8). Digital and virtual interventions are defined as technology-enabled, non-pharmacological treatments that actively deliver therapeutic content or modulation, rather than tools limited to symptom monitoring or clinical communication alone. These technologies can provide real-time monitoring, personalized feedback, and behavioral guidance, promoting self-management and adherence while reducing healthcare costs and geographical inequities.
Furthermore, regular physical activity is an established non-pharmacological strategy in migraine management. In this context, digital approaches such as telecoaching may support adherence and behavioral change, highlighting the broader potential of digital health interventions in migraine self-management (9,10).
Despite the growing use of digital and virtual interventions for migraine, evidence from randomized controlled trials (RCTs) remains fragmented and heterogeneous. Most published studies are small, heterogeneous in design, and differ widely in intervention type, delivery mode, and outcome measures (11). Moreover, digital interventions for migraine range from purely educational or monitoring applications to interactive and immersive therapies that actively modulate pain perception or stress response, making direct comparison challenging (12). Given the need for robust and reliable evidence on clinical efficacy, this review focuses specifically on RCTs. Therefore, this systematic review aims to provide an updated synthesis of RCTs evaluating the efficacy and feasibility of digital and virtual interventions for migraine.
Methods
Search strategy
A systematic search was conducted to identify clinical trials investigating digital interventions for migraine. Two major databases—MEDLINE and Embase—were searched via the PubMed and Ovid® platforms, respectively. This review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (13). The study protocol was prospectively registered at the PROSPERO international systematic review registry (CRD420251078125). Clinical trials published from database inception to September 1, 2025, that evaluated the effectiveness of digital non-pharmacological interventions on migraine-related outcomes were included. The final search was completed on September 4, 2025. The PubMed and Ovid® searches used the following terms: “Migraine Disorders” OR migraine AND “digital” OR “mobile application” OR “smartphone” OR “Virtual Reality” OR “telemedicine” OR “biofeedback” AND “digital”.
Eligibility criteria (PICOS framework)
To ensure a clear and consistent assessment aligned with the research objectives, predefined inclusion and exclusion criteria were applied (Table 1) and structured according to the PICOS framework (Population, Intervention, Comparator, Outcomes, and Study design).
Inclusion and exclusion criteria.

Population: Eligible studies included adults (≥18 years) diagnosed with episodic or chronic migraine, with or without aura, according to the International Classification of Headache Disorders (ICHD), third or earlier editions (14). Studies were excluded if they included other primary headache disorders (e.g., tension-type headache or cluster headache) without providing a separate efficacy analysis for migraine, or if they involved secondary headache disorders (e.g., headache attributed to traumatic brain injury or substance withdrawal.
Intervention: Studies were required to evaluate a digital interventional component, including but not limited to smartphone-based control, mobile application use, or integration with virtual reality. Conventional neuromodulation or biofeedback interventions without a digital element, as well as digital tools used solely for diagnostic purposes or symptom recording without an interventional component, were excluded.
Comparator: Eligible studies included a control condition consisting of sham or placebo digital interventions, or standard care, including pharmacological and/or educational intervention.
Outcomes. Studies were required to report migraine-related clinical outcomes, including at least one of the following: migraine/headache frequency, pain intensity, migraine-related disability, acute medication use, or quality of life measures.
Study Design: Only RCTs and quasi-RCTs—where participant allocation followed a systematic but not fully random process (e.g., by birth date or admission day)—were included to ensure methodological rigor. Quasi-RCTs were considered because, despite lacking full randomization, they maintain key features of controlled allocation and intervention, allowing for meaningful comparison of outcomes. Open-label trials, observational or diagnostic studies, review articles, and non–peer-reviewed or unpublished registry trials were excluded. Only English-language publications were considered.
Data collection and analysis
Study selection was performed a priori by three independent reviewers (CA, BR, JG). Two reviewers (BR, JG) independently extracted and categorized data from all eligible studies, and a third reviewer (CA) verified the results. The same reviewers subsequently conducted the analysis. Extracted data included the type of digital intervention (virtual reality, mobile applications, digital neuromodulation or biofeedback, digital cognitive behavioral therapy, and telemedicine), study population characteristics (sample size, mean age, and proportion of female participants), study duration, and efficacy outcomes. The latter encompassed migraine or headache frequency, pain intensity measured by the Visual Analog Scale (VAS) or Numeric Rating Scale (NRS), use of rescue medication, and measures of disability, including the Migraine Disability Assessment (MIDAS) and the Headache Impact Test-6 (HIT-6). Quality of life was evaluated where applicable, most commonly using the Migraine-Specific Quality of Life Questionnaire (MSQ). In addition, dropout rates and adverse events were systematically recorded.
Risk of bias assessment
The methodological quality of the included trials was assessed independently by two reviewers (CA, JG) using the Cochrane Risk of Bias 2 (RoB 2) tool (15). Studies were evaluated across five domains: the randomization process, deviations from intended interventions, completeness of outcome data, accuracy of outcome measurement, and selection of reported results. Each domain was rated as having low risk, some concerns, or high risk of bias, and an overall risk-of-bias judgment was assigned to each study.
Results
Search results
A total of 1488 studies were identified through our systematic search strategy. After removal of duplicates, 1062 studies were screened, of which 65 were deemed potentially eligible and sought for full-text retrieval. Upon detailed screening, 12 studies met the inclusion criteria and were included in the review (Figure 1). Table 1 presents an overview of the characteristics of included studies. All included studies predominantly involved female participants, ranging from 66% to exclusively female samples, reflecting the higher prevalence of migraine in women. In terms of migraine subtype, five studies (42%) did not specify whether participants had chronic or episodic migraine, whereas two studies (17%) focused exclusively on episodic and one on chronic migraine (8%). Four studies (33%) explicitly reported inclusion of both subtypes. The mean age across studies ranged from 35 to 47 years. All studies confirmed migraine diagnosis according to the International Classification of Headache Disorders (ICHD) criteria. Of the 12 studies, 11 (92%) were RCTs and one was a quasi-randomized controlled trial. Four studies were explicitly reported as pilot trials with relatively small sample sizes (29–139 participants).

PRISMA 2020 flow diagram a. The search on PubMed yielded 392 journal articles, while a search on Ovid® identified 1096 journal articles.
Intervention types
Digital (cognitive) behavioral therapy
The majority of studies (42%; 5/12) investigated digital (cognitive) behavioral therapy interventions, delivered either by smartphone application or internet-based programs. Among these, two studies focused specifically on progressive muscle relaxation (PMR) delivered via a smartphone application (RELAXaHEAD) (16,17). In both trials, participants were randomized to receive either the PMR-enabled application or a control version without PMR content. While both studies primarily assessed feasibility and user acceptance as their main outcomes, migraine-related disability was evaluated as a secondary outcome measure. Another digital health application (M-sense Migräne), which includes a comprehensive headache diary, trigger analysis, medication tracking, and access to non-pharmacological modules such as PMR, autogenic training, and chatbot-guided educational content, was evaluated in 477 participants with either episodic or chronic migraine (18). Participants were randomized to receive either full or limited access to the app for 12 weeks, with the primary outcome being the reduction in monthly migraine days.
Two recent studies examined internet-based cognitive behavioral therapy (iCBT) in 421 and 29 migraine patients, respectively (19,20). In both trials, the iCBT programs emphasized psychoeducation, self-monitoring, and trigger management, and were compared against usual medical care or a waiting-list control condition. The larger study employed a self-guided format and focused primarily on the reduction in monthly headache days as its primary endpoint. In contrast, the smaller pilot trial implemented therapist-supported sessions, primarily assessing acceptability and feasibility, with quality of life and migraine frequency evaluated as an additional outcome.
Digital neuromodulation
Two studies (17%) evaluated smartphone-controlled remote electrical neuromodulation (REN) using the Nerivio device—a wearable, battery-operated stimulator managed via a mobile application that allows digital adjustment of stimulation intensity and automatic session tracking. The first trial, including 252 participants, evaluated REN for acute migraine treatment, with the primary outcome being change in pain intensity two hours after a migraine attack (21). The second trial(22) explored the preventive effects of REN in 248 migraine sufferers, who self-administered either active or placebo stimulation every other day over an eight week period. The primary outcome was the reduction in monthly migraine days, while secondary outcomes included changes in headache days and acute medication use.
Virtual reality applications
Two studies (17%; 2/12) evaluated virtual reality (VR) applications. A recent quasi-randomized trial involving 140 participants compared VR-assisted intravenous (IV) dexketoprofen infusion with standard IV treatment alone for the acute management of migraine in the emergency department (23). Participants in the VR group were immersed in a calming virtual environment during the IV analgesic infusion, and change in pain intensity was assessed at multiple time points post-treatment. In contrast (24), investigated the preventive effects of VR-based heart rate variability (HRV) biofeedback in 50 adults with chronic migraine over a 12-week period, examining changes in mean monthly headache days and rescue medication use.
Digital biofeedback
Two studies (17%) investigated digital biofeedback interventions for migraine management. One recent study employed a surface electromyography (sEMG)-based wearable device with surface electrodes for 6 weeks, providing real-time visual feedback via a tablet application to facilitate neuromuscular relaxation (25). In this study, 84 patients with episodic migraine were randomized to either at-home digital biofeedback or treatment as usual. Another study explored app-based HRV biofeedback in 52 participants, using an earlobe sensor to deliver real-time feedback through a smartphone app (26). While the first study focused on the effects of digital biofeedback on migraine-related disability and frequency, the second study primarily assessed the feasibility and acceptability of the intervention, with quality of life as the primary clinical outcome.
Telemedicine
Friedman et al. conducted a small, randomized trial involving 45 participants with migraine, of whom 96% were female. Participants were randomized to receive either telemedicine or in-person consultations over one year. The primary outcome was treatment adherence, while secondary outcomes included measures of migraine-related disability, headache frequency, and pain intensity (27).
Outcome analysis
Frequency
Eight studies (67%) assessed headache or migraine frequency as a primary, secondary or additional outcome. Among these, only one demonstrated a significant effect compared to the control group: smartphone-based REN resulted in a mean reduction of 4.0 migraine days per month, compared to 1.3 days with placebo (p < 0.001) after a 12-week intervention (22). In contrast, iCBT, smartphone-delivered PMR, digital health application and digital biofeedback interventions did not produce a statistically significant reduction in migraine frequency compared to control (17,18,20,24,25). Notably, the largest study of digital behavioral therapy by Pach et al. found no significant difference between the intervention and control groups in the intention-to-treat analysis, although the per-protocol analysis demonstrated a significant reduction of 2.1 migraine days from baseline to the 12-week follow-up (p = 0.023) (18). Similarly, there was no difference in the mean number of headache days between video-based telemedicine and in-office treatment (27).
Intensity
Four studies (33%) assessed pain intensity as a measure of clinical efficacy, including two conducted in a preventive setting and two in an acute setting. Across all studies, pain intensity was assessed via Visual Analog Scale (VAS). Smartphone-based REN, applied for 30–45 min at the onset of a migraine attack, proved more effective than sham stimulation in achieving reduction in pain intensity at two hours (66.7% vs. 38.8%, p < 0.001) (21). Similarly, participants who received VR-assisted treatment (IV analgesic + VR) reported significant reduction in pain intensity at 15, 30, 60, and 120 min following treatment of an acute migraine attack, compared to those receiving IV medication alone (all p < 0.001) (23). In contrast, no significant reductions in pain intensity were observed in telemedicine (27) or iCBT intervention (20).
Acute medication use
Five studies (42%) evaluated changes in analgesic use, including two conducted in acute and three in preventive treatment settings. VR-assisted therapy led to a significant reduction in the need for additional rescue medication compared to the control group (4.6% vs. 27.3%, p < 0.001) (23), whereas REN showed no significant impact on rescue medication use when used as acute treatment (21).
A biofeedback-based VR device for migraine prevention resulted in a 65% reduction in total analgesic use, compared to a 35% reduction in the control group (p < 0.01) (24). Similarly, REN used for prevention of migraine significantly reduced the number of rescue medication days compared to the control group (1.4 vs. 3.5, p < 0.001) (22). In contrast, a digital health app had no significant effect on the use of triptans or non-specific acute medications (18).
Migraine-related disability
Most of the studies (75%; 9/12) tracked changes in migraine-related disability throughout the course of the study. Migraine-related disability was assessed using either the MIDAS and/or HIT-6 scales, or both. Among these, only digital sEMG-based biofeedback demonstrated a significant reduction in HIT-6 by 3.02 points compared to usual care (p = 0.03) (25). In contrast, interventions such as HRV biofeedback, VR, digital CBT, digital health apps, telemedicine, and smartphone-based REN did not show significant improvements in migraine-related disability (18,20,22,24,26,27).
Quality of life
Four studies assessed quality of life using the Migraine-Specific Quality of Life Questionnaire (MSQ). Only the digital sEMG-based biofeedback intervention resulted in a significant improvement in MSQ scores (84.6 ± 18.7 to 92.8 ± 15.5, p = 0.003), with no similar change observed in the control group (23). In contrast, digital (cognitive) behavioral therapy, digital biofeedback and smartphone-based REN showed no significant effects on quality of life (19,22,25,26).
Safety and adherence
Across all reviewed trials, no significant differences in adverse events were observed between the intervention and control groups. Adherence to digital interventions varied substantially across studies: digital health apps (18) and digital biofeedback (25,26) reported very low dropout rates (0–4%), whereas digital CBT showed considerably higher dropout rates, ranging from 28% to 48% (19,20).
Study quality
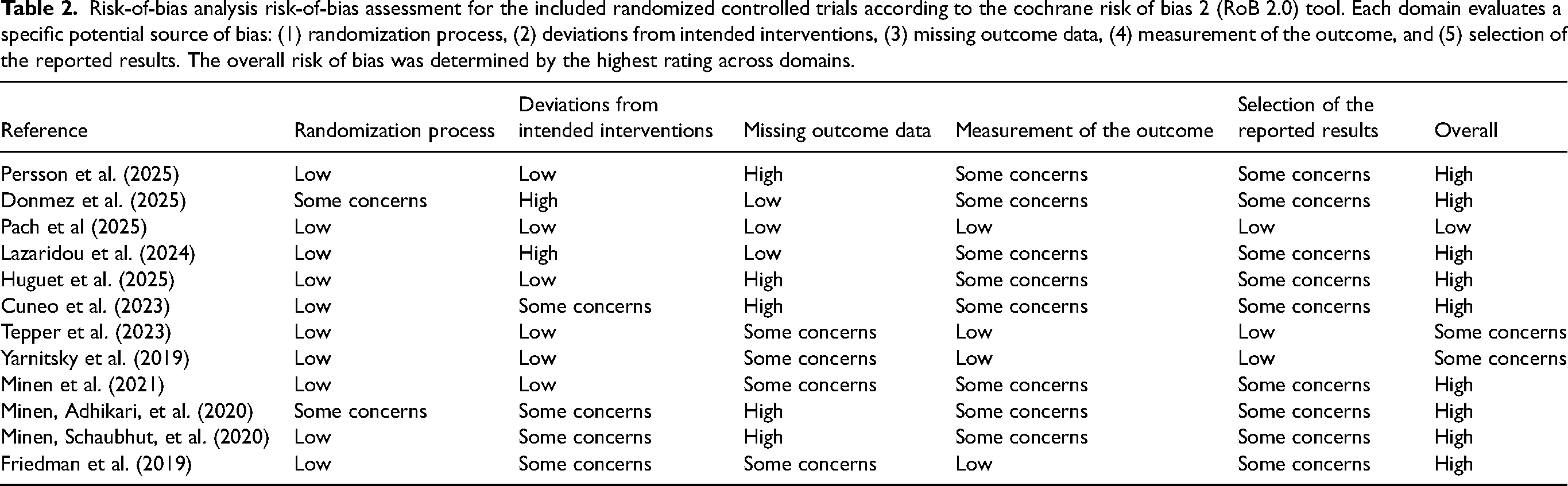
Table 2 summarizes the results of the risk-of-bias assessment. Most studies demonstrated a low risk of bias in the randomization process (83%; 10/12), while the remaining studies were rated as having some concerns (17%; 2/12). Half of the studies (6/12) were judged to have a low risk of bias due to deviations from the intended interventions, whereas four studies (33%) had some concerns and two (17%) were rated as high risk. For missing outcome data, one-third of the studies (4/12) were rated as high risk, one-third as having some concerns (4/12), and one-third as low risk (4/12). In relation to measurement of the outcome, the majority of studies (67%; 8/12) were rated as having some concerns, while the remaining four (33%) were judged to have low risk. Most studies (75%; 9/12) were rated as having some concerns in the selection of reported results, and only three studies (25%) were rated as low risk. Overall, only one study was rated as having a low risk of bias (18), two studies as having some concerns (21,22), and the remaining studies as high risk.
Risk-of-bias analysis risk-of-bias assessment for the included randomized controlled trials according to the cochrane risk of bias 2 (RoB 2.0) tool. Each domain evaluates a specific potential source of bias: (1) randomization process, (2) deviations from intended interventions, (3) missing outcome data, (4) measurement of the outcome, and (5) selection of the reported results. The overall risk of bias was determined by the highest rating across domains.
Discussion
This systematic review provides an updated synthesis of RCTs assessing the efficacy and feasibility of digital and virtual interventions for migraine. The included studies encompass a wide variety of modalities—ranging from smartphone-based behavioral programs and digital biofeedback to VR and REN—illustrating the rapid expansion and diversification of digital therapeutics in headache medicine.
Interpretation of main findings
Overall, digital and virtual interventions demonstrated encouraging clinical and feasibility results, though with variable magnitudes of effect across modalities. Among them, REN and VR-assisted approaches showed the most consistent benefits, particularly in reducing pain intensity and acute medication use, in line with previous studies supporting the efficacy of non-invasive neuromodulation in migraine management (28). Previous systematic reviews have shown that digital interventions for migraine are generally feasible and acceptable, but provided limited and heterogeneous evidence on clinical efficacy. In particular, Noser et al. reported modest effects of digital headache self-management interventions, largely based on small RCTs and behavioral approaches, with substantial variability in outcomes and overall certainty of evidence. However, their review did not include trials on VR–based interventions or REN (29).
Behavioral and app-based interventions—such as digital CBT, relaxation training, and biofeedback—yielded more heterogeneous outcomes. While objective improvements in headache frequency or disability were generally modest, these interventions consistently achieved high feasibility, adherence, and user satisfaction, supporting their practicality in real-world settings. Similar trends have been observed in prior meta-analyses of digital behavioral therapy for migraine (29–31), and in a recent systematic review and meta-analysis of behavioral interventions for migraine prevention, which included 50 randomized trials in adults (N = 6024) and found that CBT, relaxation training, and mindfulness-based therapies may reduce migraine or headache frequency, while education alone may improve migraine-related disability (32). Together, these findings suggest that digital behavioral tools may effectively support self-management, patient empowerment, and continuity of care.
Importantly, the current review highlights not only the diversity of technological solutions but also the growing coherence of evidence across trials using structured digital approaches, particularly those integrating remote monitoring, guided feedback, or personalized behavioral content. This convergence suggests that the field is moving from early feasibility phases toward more standardized and clinically applicable designs.
In conclusion, these findings indicate that digital and virtual interventions are emerging as valuable adjuncts rather than alternatives to conventional treatment. Their integration into multidisciplinary care pathways offers new opportunities to bridge the gap between specialist care and home-based disease management, especially in resource-limited settings.
Clinical implications
From a clinical standpoint, outcomes differed across intervention types: REN and VR-based interventions were associated with reductions in pain intensity and acute medication use, whereas digital behavioral approaches primarily demonstrated high feasibility, adherence, and patient engagement, with more modest effects on headache frequency and disability. Digital and virtual modalities hold significant promise for enhancing accessibility, personalization, and adherence in migraine care. App-based programs can support patient education and behavioral modification, while VR and REN technologies provide innovative, non-pharmacological pain management options (21–24). By complementing traditional therapy, these interventions can help address treatment gaps, improve quality of life, and facilitate continuity of care between clinic and home (2).
Future clinical frameworks should incorporate digital health tools into multimodal migraine management, supported by validated endpoints, standardized follow-up intervals, and integration within electronic health records. The increasing interoperability of wearable sensors, telemedicine, and artificial intelligence offers an opportunity to develop truly data-driven, personalized migraine care ecosystems (33).
Limitations
Several limitations should be considered when interpreting the present findings. The most important concern relates to the heterogeneity of study design and outcome reporting, which limited the feasibility of conducting a comprehensive quantitative synthesis. Although a formal meta-analysis was initially planned, the generation of forest and funnel plots was not possible because the primary outcomes were reported inconsistently across trials.
In particular, migraine frequency was expressed through multiple non-standardized metrics—such as monthly migraine days, monthly headache days, moderate/severe headache days, ratio of headache to headache-free days, or percentage of participants achieving ≥50% reduction in headache days. This variability prevented data pooling and highlights the urgent need for standardized definitions and reporting conventions in digital health research. Rather than representing an intrinsic limitation of the digital field, such heterogeneity reflects its ongoing methodological evolution. Accordingly, the results were synthesized descriptively and summarized in Table 3, to provide a transparent overview of study-level findings.
Overview of included studies Summary of the included randomized and quasi-randomized controlled trials assessing digital dBT, VR, dBF, REN, and TM in migraine management. dBT: digital behavioral therapy, VR: virtual reality, dBF: digital biofeedback, REN: remote electrical neurostimulation, TM: telemedicine, MMD: monthly migraine days, MHD: monthly headache days, VAS: visual analog scale, NRS: numeric rating scale, MIDAS: Migraine Disability Assessment, HIT-6: Headache Impact Test, MSQ: Migraine Specific Quality of Life.

Most included studies were open-label or single-blind, with only the REN trials adopting a rigorous double-blind, sham-controlled design. It should be acknowledged that achieving full blinding is inherently challenging in trials involving digital interventions and device-based therapies. Therefore, although several studies were rated as having high risk of bias according to strict Cochrane RoB 2 criteria, the overall certainty of evidence would still be considered moderate when evaluated using the GRADE framework. Control conditions were highly variable—often based on waiting-list or reduced-access comparators rather than active treatments—thus limiting internal validity. Sample sizes were generally small, with many studies designed as pilot or feasibility trials, and follow-up durations ranged from a few weeks to one year. Attrition rates were relatively high, and missing-data handling was frequently incomplete or unclearly reported. Furthermore, the limited adoption of validated outcome instruments, such as MIDAS, HIT-6, or MSQ, restricts comparability across studies.
At the review level, the inability to perform a formal assessment of publication bias due to the small number of homogeneous trials per endpoint, together with the restriction to English-language publications, represents an additional limitation. Nonetheless, these issues are typical of an expanding research field and reflect the progressive but still maturing methodological landscape of digital and virtual health interventions for migraine.
Future perspectives
Future research may build on the evidence emerging from the included trials. In particular, the consistent effects observed for REN and VR-based interventions on pain intensity and acute medication use support the need for larger, adequately powered RCTs with standardized endpoints, while the high feasibility and adherence reported for digital behavioral interventions highlight opportunities to optimize their efficacy through improved study design and longer follow-up. Looking ahead, the field of digital and virtual therapeutics for migraine is expected to evolve rapidly toward greater integration, personalization, and scientific validation. Large-scale, multicenter RCTs with harmonized endpoints and robust blinding procedures are needed to confirm clinical efficacy. Emerging technologies—such as AI-driven adaptive algorithms, wearable biosensors, and real-time digital biomarkers—offer new opportunities to monitor physiological responses and dynamically tailor interventions to individual patient profiles.
Furthermore, collaborations between clinicians, data scientists, engineers, and policymakers will be essential to establish standardized frameworks for safety, privacy, and regulatory approval, enabling the translation of digital migraine therapeutics from research prototypes to routine clinical care. Cost-effectiveness analyses and health-economic modeling should also be integrated into future trial designs to evaluate long-term sustainability and system-level impact.
Ultimately, the convergence of digital technologies with clinical neuroscience may enable a new paradigm of precision headache medicine, where behavioral, physiological, and environmental data are combined to deliver individualized, continuously optimized migraine management.
Conclusions
In conclusion, this systematic review demonstrates that digital and virtual interventions represent a promising frontier in migraine management. REN and VR-based modalities show reproducible clinical benefits, while digital behavioral tools offer strong feasibility and user engagement that may translate into long-term adherence advantages. The field is progressing rapidly toward greater standardization and clinical integration, driven by advances in remote monitoring, personalized feedback, and hybrid behavioral–neuromodulatory platforms.
Future large-scale, rigorously designed trials adopting unified endpoints and transparent reporting will be essential to consolidate the evidence base. With continued methodological refinement and interdisciplinary collaboration, digital therapeutics have the potential to become a transformative, patient-centered complement to conventional migraine care, expanding access and empowering individuals to manage their condition more effectively.
Footnotes
Article highlights
Acknowledgments
None.
Author contributions
Claudio Tana: Conceptualization, Methodology, Study Design, Data Verification, Data Interpretation, Writing – Original Draft, Supervision, Project Administration. Livia Moffa: Literature Search, Data Curation, Investigation, Methodology, Writing – Review & Editing. Bianca Raffaelli: Data Collection, Data Extraction, Formal Analysis, Validation, Writing – Review & Editing. Jonas Götz: Data Collection, Formal Analysis, Visualization, Validation, Writing – Review & Editing. Cornelius Angerhöfer: Data Verification, Risk of Bias Assessment, Data Interpretation, Writing – Review & Editing, Supervision.
Consent to participate
Not applicable
Consent for publishing
The authors agree to publish the article in Cephalalgia.
Data availability statement
All data extracted from the included studies and all other materials employed in this review are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support funding is provided through institutional resources available to Dr Bianca Raffaelli at Charité–Universitätsmedizin Berlin.
Open practices
Not applicable
