Abstract
Aim
This study aimed to identify risk factors associated with non-optimal triptan response among migraine patients, leveraging the unique combination of genetic and longitudinal data available in the FinnGen project.
Methods
We analyzed register data from 5351 consistent triptan users within the FinnGen project, focusing on patients who switched triptan medications at least once within a two-year period. We assessed 528 variables, including disease diagnoses, drug purchases and socio-economic status, aiming to evaluate clinical risk factors. Moreover, a genome-wide association study (GWAS) was conducted to explore genetic predispositions to triptan switching behavior in the same cohort of patients. Since no significant single nucleotide polymorphisms (SNPs) were found in the GWAS, a supplementary analysis based on previously reported SNPs associated with migraine susceptibility and triptan response was performed.
Results
Our cohort consisted predominantly of women (87%) with an average age of 38 years at the time of their first triptan purchase. In this population of patients who consistently use triptans, 85% of patients did not switch medications, while 9% switched once, 3% switched twice and 3% switched three or more times. Younger patients were more likely to switch medications. Clinical risk factors for switching included fibromyalgia and the use of gastro-oesophageal reflux medications for those who switched twice, and oral cavity disorders, intestinal disorders and gynecological diagnoses for those who switched three or more times. No significant genetic associations were identified.
Conclusions
Non-optimal triptan response in migraine patients is associated with several clinical characteristics, including prior medical diagnoses and the use of other medications. No genetic associations were observed in this cohort.
This is a visual representation of the abstract.
Introduction
Migraine is a common neurological disorder affecting over one billion individuals worldwide
Based on current Finnish and international guidelines, the first choice of medication in mild to moderate migraine attacks is standard non-steroidal anti-inflammatory drugs (NSAIDs) or a combination of pain and nausea medication. In case there is no treatment response, or migraine is severe, triptans are used (8,9). Triptans have been available in Europe since the 1990s, and all seven triptan medications are available in Finland (https://www.hila.fi/luettelot/korvattavat-myyntiluvalliset-laakevalmisteet). Although triptans are suitable for the vast majority of migraineurs, only 18–33% of patients achieve long-term pain and other symptom relief for 2–24 hours with triptans, and 20–34% need additional medication due to recurrence of pain (10). In addition, triptan adverse events – most commonly fatigue, nausea and dizziness (11) – may lead to avoidance of timely triptan intake (12).
According to the current guidelines from the International Headache Society (IHS), if a patient does not respond to one triptan medication – despite proper dosage, administration route and timing in at least two out of three attacks – switching to another triptan is recommended, with up to three different triptans considered before exploring alternative drug classes (9). An important concern arises from the observation that switching a triptan preparation to another does not necessarily lead to an improvement in headache-related functional capacity and may increase the costs of primary care (13–15). Furthermore, patients who have an inadequate response to two or more triptan treatments, whether they are triptan-resistant (experiencing only partial relief or insufficient improvement in headache severity) or triptan-refractory (experiencing no benefit at all from triptan use) or have contraindications to triptans, represent a group with a specific medical need for novel drug classes, including ditans or gepants (13).
Despite the significant burden migraine places on individuals and healthcare systems worldwide, as well as the availability of various triptan treatments, the underlying patient characteristics that influence treatment response remain scarcely elucidated.
Genetic factors have also been investigated in relation to triptan efficacy, although the evidence remains limited. Terrazzino et al. (16) examined a set of candidate genes and found that a single variant, rs2651899 in PRDM16, was associated with inconsistent triptan response. Similarly, Asuni et al. (17) focused on rizatriptan response and identified a DRD2 variant linked to better response in a small sample. Other studies have explored additional genetic factors with mixed results. Gentile et al. (18) looked at genes involved in how the body processes key brain chemicals, while Cargnin et al. (19) tested the predictive power of a polygenic score for migraine without aura on triptan response, finding a modest but significant effect of two alleles: TRPM8 rs6724624 and FGF6 rs1024905. Christensen et al. (20) assessed a combined genetic score across 12 single nucleotide polymorphisms (SNPs), again highlighting rs2651899 in PRDM16 as associated with efficacy. Despite these findings, no genetic predictors have been consistently replicated, and no genome-wide association study (GWAS) has yet specifically examined triptan response.
In the present study, we leverage the unique combination of genetic and longitudinal data available in the FinnGen project to identify the risk factors associated with a non-optimal triptan response. Our primary objective is to analyze triptan treatment failure, specifically through the lens of how frequently patients (with a minimum of five purchases over a two-year period) switch between different triptan medications. By examining patterns of medication switching – whether patients change triptans once, twice, three times or more – we aim to uncover which patient characteristics contribute to these treatment modifications and subsequently predict treatment response. To achieve this, we assess a comprehensive set of 528 variables encompassing a wide range of possible riskfactors. These include detailed records of disease diagnoses, drug purchases and socio-economic status, allowing us to evaluate which clinical risk factors may be driving the decision to switch triptan medications, and might predict treatment response in migraine management. In addition to clinical risk factors, we extend our analysis to explore the potential role of genetic predispositions to triptan switching behavior. We investigate whether specific genetic variants or profiles are associated with a higher likelihood of switching medications. By integrating both clinical and genetic data, our study aims to provide a comprehensive understanding of the factors predicting non-optimal triptan response. This approach has the potential to enhance migraine management and lead to more precise identification of patients who require alternative treatments such as gepants, helping to tailor care more effectively and potentially improve long-term patient outcomes.
Methods
Data resources
FinnGen (21) is a research project in genomics and personalized medicine. It comprises a large public–private partnership (all FinnGen partners are listed here: https://www.finngen.fi/en/partners; a list of FinnGen authors is provided in the
supplementary material, Table S1
Characteristics of the cohort stratified by outcome: no switch, switch 1 triptan (S1), switch 2 triptans (S2) and switch 3 or more triptans (S3).

*Missing data for 1908 subjects.
Chi-squared test.
One-way analysis of variance.
Kruskal–Wallis test.
Cohort definition and outcome used in the study
Our initial population consisted of all individuals present in FinnGen as of 31 December 2023. We considered as migraineurs all adult individuals who purchased at least one triptan medication between 1998 and 2023. Since the only other indication where triptans are used in Finland is cluster headache, we proceeded to exclude these patients to be able to capture solely migraine patients. Cluster headache patients were defined as being diagnosed with the disease (ICD-10: G44.0) and/or as using zolmitriptan (ATC: N02CC03) in the mode of nasal spray.
We included in the study all migraineurs who were alive and residing in Finland as of 1 January 2010 to ensure robustness of the analysis as a result of complete registry records. Exclusion criteria were applied to exclude existing users (those with a purchase before 1 January 2000) and late users (those with a first triptan purchase made after 31 December 2021) to avoid possible biases in the outcome definition. In addition, our cohort was composed of individuals whose first triptan purchase was made between the age of 18 and 75 years. Each individual was followed for a period of two years starting from their initial purchase of triptan medication. To study only the migraineurs who had a consistent triptan usage, individuals with fewer than five purchases within two years from their first triptan purchase were excluded (

Study cohort and outcome definition. (A) Flowchart of the study cohort definition. (B) Outcome definition based on switching status. (C) Time-window periods applied for evaluating risk factors.
To define patient's response to triptan, we categorized individuals based on their first five triptan purchases ( No switch: individuals with at least first three triptan purchases without switching Switch 1 triptan (S1): individuals who switch to another triptan after making just one purchase of their first triptan, and renewing their second purchase for at least three times Switch 2 triptans (S2): individuals who switch to a third triptan after one purchase of their second triptan, and renewing their third purchase for at least three times Switch ≥3 triptans (S3): individuals who switch to a fourth triptan after one purchase of their third triptan
All individuals who did not meet any of our triptan switching definitions were excluded from the analysis (
Patient risk factors
We gathered extensive data on prior clinical conditions from a list of curated disease endpoints available in FinnGen from the decade preceding the first triptan purchase for each patient (
Data on drug purchases and drug reimbursements during the three years prior to the first purchase of triptan medication was extracted from the Kela register (
Finally, a 30-day “washout” period was implemented in our study to ensure that any disease diagnosis or medication use was already established prior to the first triptan prescription in our cohort, thereby providing a more accurate baseline for analysis.
For all disease endpoints and medications purchases available, a prevalence check was conducted to exclude those with less than 1% prevalence within the study population, to ensure sufficient sample size for reliable estimation. To further refine the selection and avoid redundancy, a comprehensive check for correlation was performed using the Jaccard similarity index. Variables exhibiting a Jaccard index over the predefined threshold of 0.9 were excluded, ensuring that only distinct and not too correlated variables were retained in the final selection.
After all these steps, our list of risk factors was composed of 465 disease endpoints, 60 medications and three socio-economic status variables, for a total of 528 total medical risk factors assessed in our model (see supplementary material, Table S1 ).
Statistical analysis
Descriptive statistics of the study cohort
We provide descriptive statistics of the triptan user population stratified based on the type of triptan response encountered. Continuous variables are shown as mean and standard deviation (SD) or median and interquartile range [IQR] in case of non-normal data. Categorical variables are reported as absolute frequencies and proportions. The comparisons between groups of the continuous variables were performed by one-way analysis of variance (or non-parametric Kruskal–Wallis test), while chi-squared tests (or Fisher’s tests) were used for categorical variables.
High-throughput association analysis of clinical risk factors
For each type of outcome of the study (S1, S2 and S3), we applied a high-throughput association screening to identify candidate risk factors for a non-optimal triptan response. Each of the 528 predefined risk factors was tested for association with the outcomes, compared to the non-switching group, using a logistic regression model adjusted for year of birth, year of first triptan purchase and sex. In addition, to enhance the robustness of our findings and control for false positives, we applied a correction method using the false discovery rate (FDR) (22).
GWAS
We used REGENIE, version 2.2.4 (23) to run a GWAS of non-optimal triptan response (for more details about genotyping and imputation used in the analysis, see sections below on FinnGen genotyping and quality control and FinnGen imputation). Each outcome (S1, S2 and S3) was considered as a binary variable in the analysis, with non-switching users were used as controls. For all the analyses, we adjusted for sex, genotyping batch, ancestry (using the first 10 genetic principal components), year at first purchase and year of birth. The resulting summary statistics were post-processed to keep only SNPs with an INFO score ≥0.8 and MAF ≥1%.
Further analysis was conducted using the GWAS Credible Sets module of the Open Target Genetics platform (https://genetics.opentargets.org; June 2025 update version) to explore the relationships between SNPs that met statistical significance in the GWAS analysis (p < 5 × 10−8), or were close to it (p < 5 × 10−7), and a broad range of phenotypes. Our use of the platform was limited to a post-hoc evaluation of whether these SNPs had been previously reported in association with other phenotypes, thereby providing additional context to our findings.
As a supplementary analysis, we also assessed the association between each of our three outcomes (S1, S2 and S3) and a list of previously reported SNPs for triptan response and migraine susceptibility. The analysis was adjusted for sex, ancestry (using the first 10 genetic principal components), year at first purchase and year of birth. We considered eight SNPs from two distinct publications about triptan response (19,20) and 10 SNPs (most significant ones based on the reported p-value) from one meta-analysis about migraine (24).
FinnGen genotyping and quality control
FinnGen consists of prospectively recruited samples and a series of legacy cohorts with genotypes already available. Prospective samples were genotyped using the ThermoFisher Axiom custom array which tags a total of 655,973 variants. Genotype calling was performed using the Array Power Tools software. Legacy cohorts were genotyped using various Illumina arrays and genotype calling was performed using either GenCall or zCall algorithms. Additional information about FinnGen arrays and genotyping can be found at the following link: https://docs.finngen.fi/finngen-data-specifics/red-library-data-individual-level-data/genotype-data.
Samples were removed if:
Pihat was >0.9 and the samples were not monozygotic or replicates There was a discrepancy between reported sex and genetically determined sex (F ≤ 0.3 for females and F ≥ 0.8 for males) Missingness was ≥5% Heterozygosity was ±4 SDs from the population average Pihat was >0.1 with 14 or more samples Samples were ±4 SDs away from the population average according to the first two genetic principal components. Samples were tagged should there be evidence of a mendelian error or contain replicate samples with over 50,000 discrepancies.
Variants were removed if:
The variant failed the Hardy-Weinberg Equilibrium test (p < 10–6) The variant had a call rate <98%
FinnGen imputation
Pre-phasing was performed using Eagle 2.3.5 (https://alkesgroup.broadinstitute.org/Eagle/) and samples were imputed using the SiSu v3 imputation reference panel (https://docs.finngen.fi/finngen-data-specifics/red-library-data-individual-level-data/genotype-data/imputation-panel/sisu-v3-reference-panel). This reference panel is specific to the Finnish population, containing high-coverage (25− 30×) whole-genome sequencing data from 3775 Finns and 16,962,023 variants with minor allele count ≥3. After imputation, 16,387,711 variants were imputed with high quality (INFO > 0.6).
Results
Characteristics of non-optimal triptan responders
We analyzed a total of 500,348 patients. Among them, 11% (57,223) purchased triptans at least once between 1998 and 2023. After applying our exclusion criteria, such as removing patients with cluster headache and non-persistent users (individuals with less than five purchases), as well as avoiding possible time window biases (only individuals with first purchase between 2000 and 2021), our cohort consisted of 5351 consistent triptan users, representing 1% of the original FinnGen population.
The study cohort of consistent triptan users was primarily composed by women (87%) and had an average age of 38 years at the time of their first triptan medication purchase
To assess the switching patterns of different triptan medications we generated a Sankey diagram. This diagram offers a detailed and comprehensive visualization of all triptan purchases within the study cohort, illustrating the transitions between different medications over time. The analysis revealed that the majority of consistent triptan users consistently adhered to their initial medication, with relatively few transitioning between different triptans. Among those who switched therapies, no clear preference for a specific medication was observed, as users transitioned between medications without consistent patterns

Sankey diagram of medication switching. Number of patients using specific triptan medications at each purchase time point (x-axis) throughout the analysis period. Flow line represents a group of patients transitioning from one purchase to the successive, with the thickness of the lines proportional to the number of patients in each transition. Flows representing fewer than five patients were removed to preserve patient confidentiality and visual clarity.
Sumatriptan was the most frequent first triptan in our cohort (49%) and notably was mainly used by people who did not switch medication (p = 0.0001) (see supplementary material, Table S3 ). In contrast, zolmitriptan and frovatriptan were characterized by a higher percentage of patients in the switching groups (p = 0.0002 and 0.0071 respectively (see s upplementary material, Table S3 ). The least purchased drugs were eletriptan (6%) and naratriptan (2%).
Non-optimal triptan response is associated with several clinical risk factors
To identify risk factors of each type of non-optimal triptan response (S1, S2 and S3) compared to optimal treatment response (no switch), we applied a logistic regression adjusting for year of birth, year of first triptan purchase and sex.
No associated factors for switching after 1 triptan group (S1) were found after FDR correction
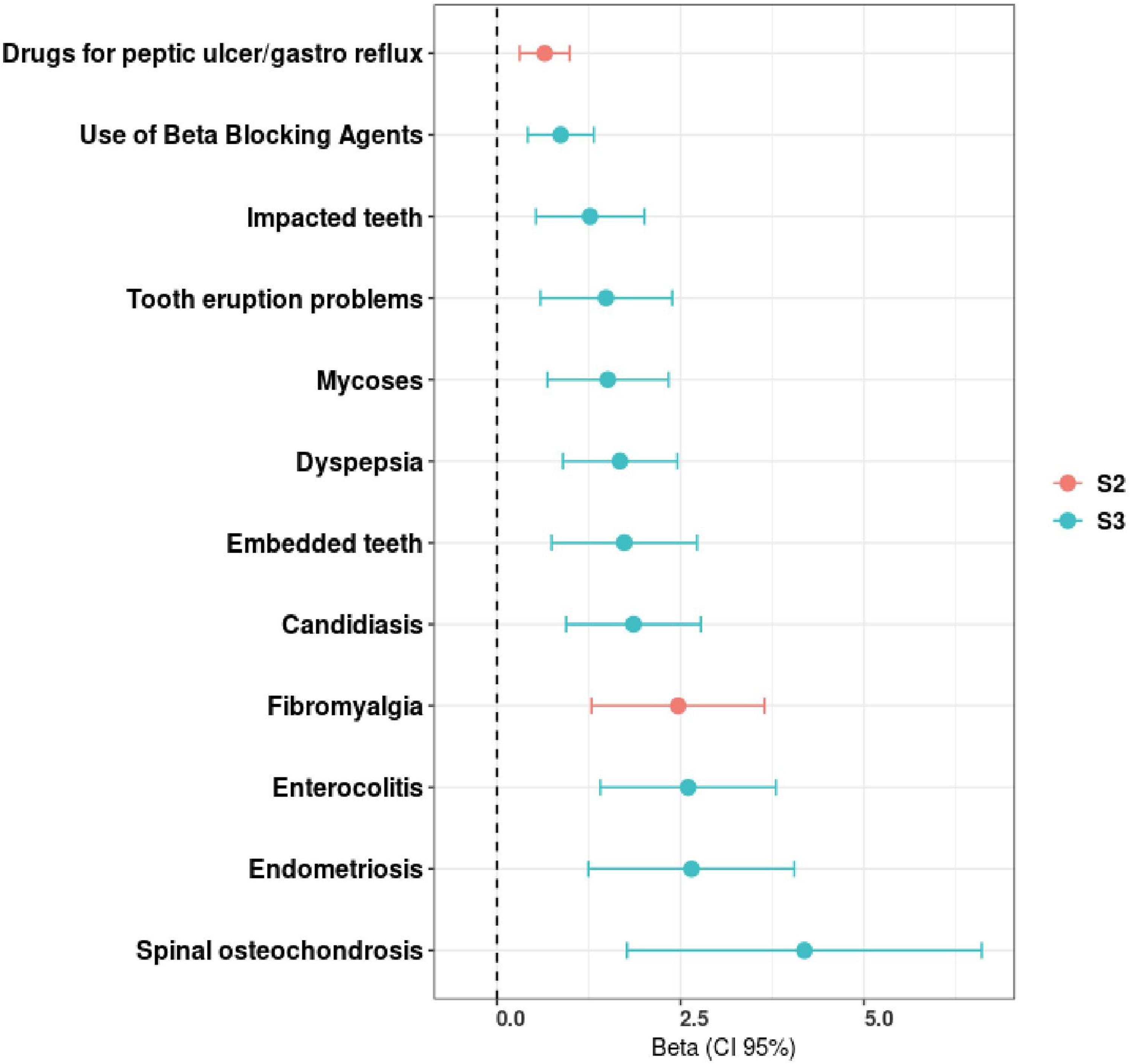
Clinical risk factors of non-optimal triptan response groups. Significant associations, following false discovery rate correction (95% confidence interval (CI)), between triptan treatment failure (sub-outcomes S1, S2 and S3) and the evaluated clinical risk factors.
We identified that individuals from the S2 group used gastro-oesophageal reflux medications more often and had higher prevalence of fibromyalgia (β = 0.65, p = 0.0321, and β = 2.47, p = 0.0075, respectively (
The S3 group presented a higher prevalence of diseases of the oral cavity, such as impacted teeth, tooth eruption problems and embedded teeth (β = 1.27, p = 0.0332; β = 1.49, p = 0.0476 and β = 1.73, p = 0.0332, respectively) (
No significant genetic factors associated with triptan response
To perform a high-throughput analysis of the genetic determinants of non-optimal triptan response we conducted a GWAS using our three outcomes (S1, S2 and S3) as phenotypes in the analysis. No genome-wide significant association was observed; however, there were indications of potential signals (

Manhattan plot of genome-wide association study (GWAS) results for each outcome. Genetic association between chromosome loci and switching triptan medication one (A), two (B) or three or more times (C). Each dot represents a single nucleotide polymorphism (SNP), with SNPs ordered on the x-axis according to their genomic position. The y-axis represents strength of their association measured as –log10 transformed p-values. Red line marks genome-wide significance threshold of p < 5 × 10−8. SNPs labels are added when close to the statistical significance threshold: p < 5 × 10−7.
For the S1 group, we identified three SNPs (rs1157881, rs142841213 and rs76076332) that approached statistical significance
In the S3 phenotype three variants were close to the statistical significance threshold
Since the lack of significant results in our GWAS may be attributed to the limited sample size, we conducted a hypothesis-driven analysis where we tested the association between each of our three outcomes (S1, S2 and S3) and a set of previously reported SNPs related to triptan response and migraine susceptibility (see Methods). Although some SNPs showed moderate associations with triptan response switching behaviors, none of the signals remained significant after correction for multiple testing (
see supplementary material, Tables S9
Discussion
The present study investigated the clinical and genetic factors influencing non-optimal triptan response in a cohort of 5351 Finnish consistent triptan users from the FinnGen research project. The study cohort was predominantly female (87%), consistent with the known prevalence of migraine among women (25), with no significant differences in triptan switching patterns between sexes. However, the users who switched more medications were generally younger, with the most frequent switching observed in the 18–29 years age group, suggesting that younger patients may be more likely to seek alternative treatments early in their migraine management.
Several factors may contribute to this pattern. Younger individuals might be more proactive in seeking optimal symptom relief, less tolerant of side effects, or more sensitive to the impact of migraine on daily functioning, education or work. They may also be more familiar with digital health resources and more inclined to advocate for changes in treatment. Additionally, younger patients may have less established treatment routines and are therefore more likely to experiment with different triptan formulations or mechanisms of action. This observation warrants further investigation because it may reflect age-related differences in treatment expectations, health-seeking behavior or migraine phenotype. Understanding these dynamics could help tailor more age-sensitive approaches to migraine care and improve treatment adherence.
Baseline characteristics such as body mass index, education level and known comorbidities of clinical interest showed no significant differences between switchers and non-switchers.
In this population of patients who consistently use triptans (a minimum of five triptan prescriptions over a two-year period), 85% remained consistent with their medication choices, with relatively few switching between medications, aligning with previously reported literature (26). When switches did occur, they appear evenly distributed across the purchase timeline and list of medications, suggesting no strong tendency toward switching at any specific time point and from/to any specific triptan. Sumatriptan was the most frequently used in our study cohort and had a higher rate within individuals who did not switch medication. In contrast, zolmitriptan and frovatriptan were characterized by a higher percentage of patients in the switching groups.
No significant factors associated with the S1 group were identified after FDR correction.
In contrast, individuals in the S2 group were more likely to have a diagnosis of fibromyalgia and demonstrated higher usage of gastroesophageal reflux medications. Fibromyalgia is a known comorbidity of migraine (27–29) and has been linked to increased sensitivity to pain as well as a higher likelihood of functional gastrointestinal disorders (30). The increased utilization of gastroesophageal reflux medications might reflect an overlap with gastrointestinal phenotypes of migraine or a heightened susceptibility to gastrointestinal side effects from triptans. A similar mechanism may explain the prevalence of dyspepsia in the S3 group. It is documented that medications such as triptans can alter gastrointestinal motility and trigger side effects such as nausea and abdominal pain (31). In addition, the S3 group showed associations with oral cavity disorders and gynecological diagnosis. Certain medications, including triptans, can contribute to conditions such as dry mouth or other forms of oral discomfort (32); however, there is no evidence to suggest that these medications are associated with tooth eruption disorders. The higher rate of gynecological diagnoses in this group might be explained by medication or hormone interactions. Endometriosis is associated with higher prevalence of comorbid conditions in migraine (33), while candidiasis can affect immune responses, complicating triptan effectiveness. Notably, triptans have demonstrated efficacy for the acute treatment of menstrual migraine but there is no clear recommendations for treatment priority (34–38). Although the genome-wide association study (GWAS) did not identify significant associations (p < 5 × 10–8), potentially because of limited statistical power, six SNPs were close to reach the threshold for statistical significance (p < 5 × 10–7). Of those, SNP rs111959732 (odds ratio = 2.953 of switching three or more times, p = 4.89 × 10–7) was also found to be positively associated with lymphocyte count. Supporting a potential genetic contribution to triptan response, a Danish population study (39) showed that a higher migraine polygenic risk score was associated with better triptan response (defined as ≥10 triptan purchases), suggesting a genetic contribution to treatment efficacy. These findings suggest that genetic predispositions may play a role in triptan response, but larger migraine cohorts with drug purchases or other measures of triptan response combined with genome-wide genetic data are needed to clarify potential associations.
Our study has several strengths and limitations. One of the key strengths is the comprehensive use of both clinical and genetic data from the FinnGen project, which allows for an in-depth examination of migraineurs. Specifically, we focused on a wide range of variables to obtain a holistic view of the factors that may contribute to non-optimal triptan response. Moreover, we created a novel definition of suboptimal triptan response utilizing data from the national drug purchase register, which provides more reliable information of drug usage compared to prescription data. Finally, finding genetic determinants for treatment response requires large-scale genomics data with detailed information of participants’ drug usage. FinnGen is one of the largest cohorts in the world with linked genome-wide genomic data and detailed real-world data with extensive follow-up and thus, applicable for this kind of analysis. However, because our GWAS showed no significant associations with triptan response, potentially due to limited statistical power, additional data are needed to evaluate whether genetic factors could contribute to triptan response. Another limitation of our study is that FinnGen participants were recruited exclusively from Finland, making it representative of a specific ancestry and environmental context. Thus, the findings may not be generalizable to other populations with different genetic backgrounds, environmental exposures or lifestyle factors. Moreover, by focusing only on patients with consistent triptan use, the study may also not be generalizable to those who discontinue after fewer than five purchases and very short follow-up periods, potentially influencing patient classification. Finally, although we do not consider the observed results to be attributable to reporting bias or missing data, given the high coverage and reliability of the Finnish national registers, we acknowledge this as a potential limitation that should be considered when interpreting the findings. Despite these limitations, the study offers valuable insights into the factors affecting triptan treatment efficacy and can help to inform personalized treatment strategies for migraine patients.
In conclusion, non-optimal response in consistent triptan users can be broadly categorized into two groups. The first group consists of individuals who switch medication once, which might reflect an individual response to initial triptan treatment: lack of efficacy, inconsistent efficacy and/or triptan related side effects. The second group includes individuals who switch triptans more than once, which may indicate the presence of concurrent comorbid pathologies that exacerbate migraine symptoms and increase susceptibility to side effects, or reflect alternate causes of migraine-like headaches. These comorbidities may include conditions such as fatigue, nausea and gastrointestinal disorders, or gynecological issues such as endometriosis, menstrual pain or menstrual related migraine, all of which might complicate migraine treatment. Understanding these factors can help tailor treatment strategies and improve outcomes for patients experiencing non-optimal triptan response.
Non-optimal triptan response was associated with several clinical risk factors such as fibromyalgia and the use of gastro-oesophageal reflux medications, oral cavity disorders, intestinal disorders and gynecological diagnoses. Our main finding that non-optimal triptan response in migraine patients is influenced by various clinical risk factors could help tailor treatment strategies for better patient outcomes. Understanding these factors may open possibility to faster identification and implementation of most suitable migraine treatments, and improved identification of patients who are more prone to undergo futile treatment trials.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024251414621 - Supplemental material for Clinical and genetic characterization of non-optimal triptan response among migraine patients
Supplemental material, sj-pdf-1-cep-10.1177_03331024251414621 for Clinical and genetic characterization of non-optimal triptan response among migraine patients by Ettore Brenna, Matteo Ferro, Marja-Liisa Sumelahti, Tatiana Cajuso, Andrea Ganna, Jari Rossi, Emmi Tikkanen and in Cephalalgia
Footnotes
Acknowledgments
We are grateful to the Finnish individuals, whose data made this study possible. The FinnGen project is funded by two grants from Business Finland (HUS 4685/31/2016 and UH 4386/31/2016) and the following industry partners: AbbVie Inc., AstraZeneca UK Ltd, Biogen MA Inc., Bristol Myers Squibb (and Celgene Corporation & Celgene International II Sàrl), Genentech Inc., Merck Sharp & Dohme LCC, Pfizer Inc., GlaxoSmithKline Intellectual Property Development Ltd, Sanofi US Services Inc., Maze Therapeutics Inc., Janssen Biotech Inc, Novartis AG and Boehringer Ingelheim International GmbH.
The following biobanks are acknowledged for delivering biobank samples to FinnGen: Auria Biobank (www.auria.fi/biopankki), THL Biobank (www.thl.fi/biobank), Helsinki Biobank (www.helsinginbiopankki.fi), Biobank Borealis of Northern Finland (https://www.ppshp.fi/Tutkimus-ja-opetus/Biopankki/Pages/Biobank-Borealis-briefly-in-English.aspx), Finnish Clinical Biobank Tampere (www.tays.fi/en-US/Research_and_development/Finnish_Clinical_Biobank_Tampere), Biobank of Eastern Finland (www.ita-suomenbiopankki.fi/en), Central Finland Biobank (www.ksshp.fi/fi-FI/Potilaalle/Biopankki), Finnish Red Cross Blood Service Biobank (www.veripalvelu.fi/verenluovutus/biopankkitoiminta), Terveystalo Biobank (www.terveystalo.com/fi/Yritystietoa/Terveystalo-Biopankki/Biopankki) and Arctic Biobank (https://www.oulu.fi/en/university/faculties-and-units/faculty-medicine/northern-finland-birth-cohorts-and-arctic-biobank). All Finnish Biobanks are members of BBMRI.fi infrastructure (www.bbmri.fi). Finnish Biobank Cooperative FINBB (![]() ) is the coordinator of BBMRI-ERIC operations in Finland.
) is the coordinator of BBMRI-ERIC operations in Finland.
Author contributions
Ettore Brenna: data curation, formal analysis, methodology, writing – original draft. Matteo Ferro: data curation, formal analysis, methodology, writing – original draft. Marja-Liisa Sumelahti: conceptualization, writing – review & editing. Tatiana Cajuso: methodology, writing – review & editing. Andrea Ganna: conceptualization, methodology, project administration, supervision, writing – review & editing. Jari Rossi: conceptualization, writing – review & editing. Emmi Tikkanen: Conceptualization, writing – review & editing.
Data availability
The code for the project is available online (https://github.com/dsgelab/project_migraine). The individual-level data in these studies is protected for data privacy, access is regulated through the biobanks. The Finnish biobank data can be accessed through the Fingenious® services (![]() ) managed by FINBB.
) managed by FINBB.
Declaration of conflicting interests
All authors have completed the ICMJE uniform disclosure form available online (![]() ). This study was conducted as a collaboration with the University of Helsinki and Pfizer. JR and EM are employees of Pfizer Inc.; AG is the founder of Real World Genetics Oy; MLS has received consultancy fees from Abbvie, Teva, Lundbeck, Novartis, Pfizer and Organon, payment honoraria for lectures and presentations from Abbvie, Teva, Lundbeck, Novartis, Pfizer and Organon, payment for expert testimony from Lundbeck, support for attending meetings from Abbvie, Teva, Lundbeck and Pfizer, participated in advisory boards for Abbvie, Teva, Lundbeck and Pfizer, is the Chair of Finnish Current Care Guidelines in Migraine, and has a role in The Finnish Medical Society Duodecim, with no other relationships or activities that could appear to have influenced the submitted work.
). This study was conducted as a collaboration with the University of Helsinki and Pfizer. JR and EM are employees of Pfizer Inc.; AG is the founder of Real World Genetics Oy; MLS has received consultancy fees from Abbvie, Teva, Lundbeck, Novartis, Pfizer and Organon, payment honoraria for lectures and presentations from Abbvie, Teva, Lundbeck, Novartis, Pfizer and Organon, payment for expert testimony from Lundbeck, support for attending meetings from Abbvie, Teva, Lundbeck and Pfizer, participated in advisory boards for Abbvie, Teva, Lundbeck and Pfizer, is the Chair of Finnish Current Care Guidelines in Migraine, and has a role in The Finnish Medical Society Duodecim, with no other relationships or activities that could appear to have influenced the submitted work.
Ethical considerations
Patients and public were not involved in the designing process of this study. The patients will not be informed individually of the study results otherwise than through possible media coverage.
Funding
The authors received no financial support for the research, authorship and/or publication of this article. The study was conducted in collaboration with the University of Helsinki and Pfizer. The study was funded by both parties.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
