Abstract
Aim
The diagnostic criteria for chronic migraine, which are based on the total number of monthly headache days, are the subject of ongoing debate. The present study aimed to investigate and compare the burden of disease and quality of life between high-frequency episodic migraine and chronic migraine, using data from the large population-based PopHEAD study.
Methods
PopHEAD is a population-based cross-sectional study in the Norwegian county of Vestfold and Telemark performed in 2023. Among 28,753 randomly selected adults (aged 18–70 years) invited to complete an electronic questionnaire, 8265 (28.7%) participants responded. The questionnaire was a modified version of the Headache-Attributed Restriction, Disability, Social Handicap and Impaired Participation questionnaire, and migraine was classified using a diagnostic algorithm that has been validated by telephone interview in this population. High-frequency episodic migraine was classified according to newly proposed criteria, and chronic migraine according to International Classification of Headache Disorders, 3rd edition (ICHD-3) criteria. Using linear regression for continuous variables and logistic regression for binary outcomes, we compared the disease burden and quality of life between participants classified with high-frequency episodic migraine and those with chronic migraine. The analysis was conducted in two steps: a primary analysis adjusted for age and sex, as well as a second analysis with an additional adjustment for monthly migraine days.
Results
Of the 8265 responders, 225 had high-frequency episodic migraine and 349 had chronic migraine. Compared to the high-frequency episodic migraine group, the chronic migraine group had more monthly migraine days (17.9 vs. 9.7, p < 0.001) and headache days (20.8 vs. 9.9, p < 0.001). The chronic migraine group also used more acute medication (p < 0.001). In analyses adjusted for age and sex, participants with chronic migraine reported greater disease burden across almost all measures, including work and social impairment, and had a lower quality of life (p < 0.01). With additional adjustment for monthly migraine days, no significant differences in disease burden were found between the two groups, except from days missed from household work (p = 0.03).
Conclusions
The higher disease burden observed in chronic migraine compared to high-frequency episodic migraine was fully explained by the higher number of monthly migraine days in the chronic migraine group. Our findings support previous suggestions to simplify the ICHD-3 criteria for chronic migraine by basing it solely on the number of monthly migraine days.
This is a visual representation of the abstract.
Keywords
Introduction
In the first edition of the International Classification of Headache Disorders (ICHD) from 1988, migraine was defined without reference to headache frequency (1). Chronic migraine (CM) was introduced in ICHD, 2nd edition (ICHD-2) from 2004 (2), where it was categorized as a complication of migraine, defined as ≥15 days per month meeting the criteria for migraine and no medication overuse headache (MOH) (3). A subsequent update to ICHD-2 broadened these criteria to allow 15 or more total headache days per month, which could include tension-type headache (TTH), provided that at least eight days met the criteria for migraine or were relieved by a triptan or ergot derivative (4). The revision also allowed the concurrent diagnosis of CM and MOH. In the present ICHD, 3rd edition (ICHD-3) from 2018 (5), CM is defined as a separate entity rather than a complication, but the criteria themselves underwent minimal changes. In contrast, the term episodic migraine (EM) has been in widespread use to refer to individuals who have migraine, but do not meet the criteria for CM. The term was first addressed by the International Headache Society in 2020 (6), which described EM as a “headache occurring on less than 15 days a month over the last three months, which on some days is migraine”.
Even before the release of ICHD-3, there was a debate about the definition of CM. Because the diagnostic criteria count total headache days rather than only migraine days, a person with 14 migraine days per month and no TTH days is diagnosed with EM, while a person with eight migraine days and seven days with mild TTH is diagnosed with CM, although the former can be assumed to have the highest disease burden. This diagnostic distinction has direct implications for many patients because healthcare systems in several countries restrict certain advanced treatments, such as calcitonin gene-related peptide (CGRP)-targeted treatment, to patients with CM (7,8).
The term high-frequency episodic migraine (HFEM) has emerged to describe patients with a high number of monthly migraine days who do not meet the diagnostic criteria for CM, although its definition is not standardized, with publications applying different lower cut-offs for monthly migraine days in the range 5–11 (9). While several studies have confirmed that HFEM carries a greater burden than low-frequency episodic migraine (LFEM), its comparison to CM is less conclusive (9–13). An important limitation that may explain this inconsistency is that most previous studies have defined HFEM based on total monthly headache days rather than migraine days (9). The importance of this distinction was shown by a Danish study that used migraine days to define HFEM, and found no difference in disease burden between the HFEM and CM groups (10). Finding a similar number of migraine days in both groups, it was suggested that the diagnostic criteria for CM should be revised to be based on a threshold of at least eight migraine days per month.
Here, we compare the burden of HFEM and CM in a large, population-based study that includes detailed information on various aspects of headache burden.
Methods
Study population
The PopHEAD study is a population-based cross-sectional digital survey conducted in the Norwegian county of Vestfold and Telemark in 2023. Details of the method and its validation have been described in detail previously (14). A random sample of 28,753 adults (aged 18–70 years) were invited to participate, of which 8265 responded (4921 women and 3344 men), corresponding to a participation rate of 28.7%. Compared to non-participants, the participants were older (mean age 47.3 vs. 42.7 years), more often female (59.5% vs. 46.3%), married (48.6 vs. 37.3) and of Scandinavian origin (96.3% vs. 89.1%).
Study instrument
The study instrument was a Norwegian modified version of the questionnaire Headache-Attributed Restriction, Disability, Social Handicap and Impaired Participation (HARDSHIP) (15) translated from English using Lifting The Burden's translation protocol for hybrid documents (16). The questionnaire consists of five parts, with questions regarding (i) demographics, (ii) headache screening, (iii) diagnosis, (iv) healthcare utilization and (v) disease burden.
Headache diagnosis
The HARDSHIP diagnostic algorithm for migraine applies modified ICHD-3 criteria as described in detail elsewhere (5,16). Briefly, participants who answered “yes” to the question “Have you had a headache the last 12 months?” were asked “During the last 30 days, on how many of these days did you have a headache?” Based on questions about the headache characteristics of their most bothersome headache, they were classified as having definite migraine, definite TTH, probable migraine, probable TTH or undetermined headache using the HARDSHIP diagnostic algorithm. As in previous HARDSHIP studies, we defined migraine as either definite or probable migraine. The frequency of the given headache was based on the question “How often do you have this type of headache? Indicate the number of days per month”. Participants with migraine were subclassified into CM (≥15 headache days and ≥8 migraine days/month), HFEM (8–14 migraine days/month and not qualifying for CM) (9,10,17) or LFEM (<8 migraine days/month). The definition of HFEM follows the recommendation from a recent review (9) and was also applied by Chalmer et al. (10) in a study comparing the burden of HFEM and CM. To test the robustness of our findings, we performed sensitivity analyses using alternative HFEM definitions (5–14 and 10–14 monthly migraine days). Only CM and HFEM were included in the analysis. MOH was diagnosed in participants with ≥15 headache days/month and medication overuse the last 30 days. Overuse was defined as ≥15 days with non-opioid analgesics and ≥10 for opioids, triptans and combination drugs. Migraine and MOH were not considered mutually exclusive. We included in the analyses both over-the-counter drugs (OTC) and combination drugs. OTC in Norway are both simple analgesics and some combination drugs (paracetamol, Paracetduo (Karo Healthcare AB, Stockholm, Sweden) (paracetamol and caffeine), Paralen (Zentiva, Prague, Czech Republic) (acetylsalicylic acid, paracetamol, caffeine), ibuprofen, naproxen and phenazone-caffeine. Combinations drugs are Paracetduo, Paralen, phenazone-caffeine and paracetamol with codeine in various strengths and forms.
To validate the questionnaire-based headache diagnoses, a random subsample of 505 participants were contacted by telephone within four weeks of participation for an interview with a headache neurologist, who was blinded to questionnaire responses (14). Sensitivity and specificity were, respectively, 77% and 85% for migraine overall, 74% and 87% for EM, 47% and 97% for CM, and 58% and 99% for MOH (14).
Disease burden
In addition to headache and migraine frequency, as well as the use of acute headache medication, other types of headache burden examined in the questionnaire were: Effects on education (“Have your headaches interfered with your education?”); career opportunities (“Do you believe your headaches have made you less successful in your career?”); income (“Have your headaches reduced your earnings?”); days absent from work (“On how many days in the last three months could you not go to work or school because of your headaches?”); household work (“On how many days in the last three months could you not do any household work because of your headaches?)”; and social commitments (“On how many days in the last three months did you miss family, social or leisure activities because of your headaches?”). The World Health Organisation Quality of Life (WHOQoL-8) sum score is designed to measure quality of life across cultures and diseases (18) and consists of a eight questions on quality of life, satisfaction with one's health, ability to perform daily activities, satisfaction with oneself, satisfaction with personal relationships, satisfaction with living conditions, energy in daily life and whether money fulfils necessary needs. Each question is scored from 1 (worst) to 5 (best), with the total score in the range 8–40. A shortened version of the questionnaire was sent to participants who required a reminder. Because this version did not include the WHOQOL-8 items (14), 36% of responses were missing for this measure.
Statistical analysis
Sociodemographic characteristics are presented as counts with percentages for categorical variables, and as the mean ± SD for continuous variables. Sex was categorized as “men” or “women”. The median household income in Norway in 2023 was NOK 806,800 (19). To facilitate interpretation, self-reported income (≤360,000; 360,000–570,000; 570,000–840,000; 840,000–1,300,000; ≥1,300,000 NOK) was re-categorized as “low” (<570,000) NOK, “medium” (570,000–840,000 NOK) and “high” (>840,000 NOK). Habitation was categorized as “urban” or “rural” based on the question “Do you live in a city/town?”. Education level was categorized as “<high school”, “high school”, “university/college <4 years” and “university/college ≥4 years”. The days last month with use of acute medications was categorized as “None”, “1–9”, “10–14” and “≥15”.
Sociodemographic factors, headache and migraine frequency and the use of acute medication were compared between HFEM and CM using unadjusted analyses, the chi-squared test for categorical variables and the t-test for continuous variables. Analyses comparing measures of headache burden between HFEM and CM were first adjusted for age and sex using logistic regression for binary variables and linear regression for continuous variables. To specifically test the hypothesis that the number of migraine days can explain the difference in disease burden between HFEM and CM, irrespective of non-migraine headache days, these analyses were then repeated with an additional adjustment for migraine days per month. Adjusted proportions and means were derived using the margins command in Stata, version 17.0 (StataCorp, College Station, TX, USA), which estimates predicted values from the fitted regression models while adjusting for covariates. We also performed sensitivity analyses (i) using two alternative definitions of HFEM (5–14 and 10–14 monthly migraine days, respectively) and (ii) stratifying CM by MOH (CM with MOH and CM without MOH). Analyses were performed using complete case data. Participants with missing values for any variable included in a given analysis were excluded from that analysis. The number of observations (n) reported in each table reflects the available data for each analysis. Pearson's correlation coefficient was used to evaluate the relationship between monthly migraine days and monthly headache days.
Stata, version 17.0, was used for all analyses. p < 0.05 was considered statistically significant.
Results
Of the 8265 participants (4921 women and 3344 men) in the PopHEAD study, 225 participants had HFEM (183 women and 42 men) and 349 participants had CM (283 women and 66 men). Of those with CM, 208 (59.6%) also fulfilled the criteria for MOH.
Demographic and socioeconomic profile
The demographic and socioeconomic characteristics of the two groups are described in Table 1. Participants with HFEM were, on average, older than those with CM (43.1 vs. 40.5 years, p = 0.02) and had a significantly higher rate of employment (56.0% vs. 45.0%, p = 0.01).
Demographic and sociodemographic characteristics.

p-values were calculated using the chi-squared test for categorical variables and the independent samples t-test for continuous variables. p < 0.05 is indicated in bold.
Migraine frequency and acute medication
As detailed in Table 2, the HFEM group reported fewer mean monthly migraine days than the CM group (9.7 vs. 17.9 days, p < 0.001). A similar difference was observed for mean monthly headache days (9.9 in HFEM vs. 20.8 days in CM, p < 0.001). For both groups, the difference between mean headache days and mean migraine days was small, measuring 0.2 days in the HFEM group and 2.9 days in the CM group, and the two measures were strongly correlated (r = 0.89).
Migraine frequency and use of acute medication.

Mean days of use of acute medication was calculated for the entire group, including non-users.
p-values were calculated using the chi-squared test for categorical variables and the independent samples t-test for continuous variables. p < 0.05 is indicated in bold.
§Migraine and non-migraine headache.
aNSAIDs = non-steroidal anti-inflammatory drugs: ibuprofen, naproxen, diclofenac and indomethacin.
bOTC = over-the-counter drugs: in Norway, these include paracetamol (500 mg), Paracetduo (500 mg paracetamol, 65 mg caffeine), Paralen (acetylsalicylic acid 250 mg, paracetamol 250 mg, caffeine 50 mg), ibuprofen (200–400 mg), naproxen (250 mg) and phenazone-caffeine (phenazone with caffeine in various strengths and forms).
cTriptans: sumatriptan, eletriptan, rizatriptan, zolmitriptan, almotriptan, frovatriptan and naratriptan.
dOpioids: tramadol, paracetamol with codeine in various strengths and forms.
eCombination drugs: Paracetduo (500 mg paracetamol, 65 mg caffeine), Paralen (acetylsalisylic acid 250 mg, paracetamol 250 mg, caffeine 50 mg), phenazone-caffeine (500 mg phenazone, 100 mg caffeine or 1000 mg phenazone and 100 mg caffeine) and paracetamol with codeine in various strengths and forms.
The use of acute medication was lower in the HFEM group compared to the CM group (mean 10.0 vs. 14.2 days, p < 0.001). The proportion using ≥15 days of acute medication in the last 30 days was 18.3% in the HFEM group and 49.6% in the CM group. The CM group reported more frequent use of several specific medication types, including paracetamol (p < 0.001), non-steroidal anti-inflammatory drugs (NSAIDs) (p < 0.001), OTC drugs (p < 0.001), opioids (p = 0.02) and combination drugs (p = 0.007).
Headache burden and quality of life
Measures of headache burden and quality of life are detailed in Table 3. After adjusting for age and sex, the CM group reported a higher burden across almost all parameters compared to the HFEM group. This included interference with education (p = 0.05), career success (p < 0.001), receipt of social benefits due to headache (p = 0.003) or any cause (p = 0.001), and days missed from work, household and social activities (all p < 0.001). The WHOQoL-8 quality of life score was also significantly lower for the CM group (p = 0.001). When the analyses were additionally adjusted for monthly migraine days, no difference was observed between the two diagnostic groups except for days missed from household work (p = 0.03). Sensitivity analyses confirmed that the pattern of higher burden in the CM group compared to the HFEM group, which was explained by monthly migraine days, was consistent across alternative HFEM definitions (5–14, 8–14 and 10–14 monthly migraine days) (see supplementary material, Table S1) and when the CM group was stratified by the presence of MOH (see supplementary material, Table S2). For most burden measures, participants with CM and concurrent MOH tended to report higher burden than those with CM without MOH.
Measures of headache burden and quality of life, adjusted for age, sex and migraine days.
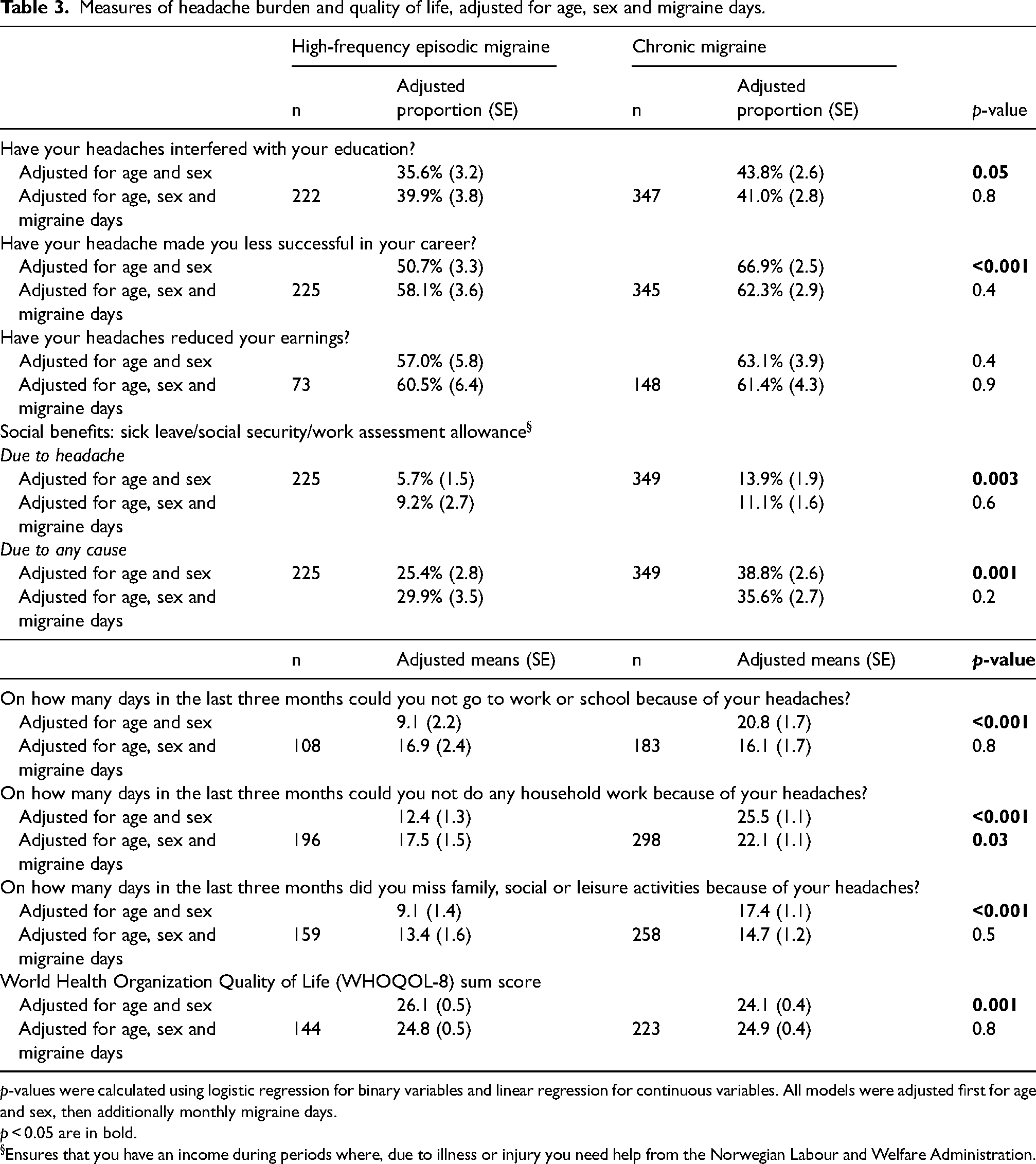
p-values were calculated using logistic regression for binary variables and linear regression for continuous variables. All models were adjusted first for age and sex, then additionally monthly migraine days.
p < 0.05 are in bold.
§Ensures that you have an income during periods where, due to illness or injury you need help from the Norwegian Labour and Welfare Administration.
Discussion
Most previous studies have identified HFEM as an intermediate state between LFEM and CM, in terms of disease burden indicators such as disability and quality of life (9). A smaller number of studies have found comparable levels of burden between HFEM and CM (10,11,13,20). Supporting the prevailing evidence, we found a higher disease burden associated with CM than with HFEM across almost all measures of disease burden, after adjustment for age and sex. Participants with CM reported more days when headache interfered with work, school, household work or social activities and were more likely to report that headache had negatively affected their education and career and had slightly lower overall quality of life scores. About 60% of participants in both groups reported that headaches had reduced their income. The proportion receiving social benefits was high in both groups (25.4% in HFEM, 38.8% in CM), but only a minority attributed these benefits to headaches (5.7% and 13.9%), suggesting a high degree of comorbidity. Headache and migraine frequency, and the use of acute treatments, were also significantly higher among those with CM.
A major limitation of most previous studies comparing the burden of HFEM and CM is that HFEM was defined based on monthly headache days rather than monthly migraine days. Of the 26 studies included in a recent review by Cammarota et al. (9), only two studies used migraine days to define HFEM and directly compared it with CM using current diagnostic criteria. The first of these studies, as performed by Chalmer et al. (10), found similar burden in HFEM and CM across a range of measures. Interestingly, it also found a similar number of migraine days in both groups, suggesting that the distinction between a CM and HFEM diagnosis in this cohort was attributable to a difference in non-migraine headache days. The second study, as performed by Barbanti et al. (21) likewise found no difference in headache burden between HFEM and CM, as measured by the Headache Impact Test (HIT-6), but a higher level of psychiatric comorbidities among those with CM. This study did not report separate numbers for migraine and headache days.
In the present study, the CM group had a higher number of both migraine and headache days than the HFEM group, in contrast to the clinic-based findings of Chalmer et al. (10) However, in line with their conclusions, we found that differences in disease burden were explained by the number of migraine days, rather than by the diagnostic classification itself. After adjusting for monthly migraine days, no significant differences remained between the groups across measures including impact on education, career success, income, work or social functioning, receipt of social benefits, or overall quality of life. The only exception was a higher number of days of household work lost to headache in the CM group. These findings were supported by sensitivity analyses, which showed the same pattern across alternative definitions of HFEM and after stratifying the CM group by the presence of MOH. In both analyses, the higher burden observed in the CM group was explained by monthly migraine days, indicating that this relationship was consistent across classification definitions and clinical subgroups.
Our findings support earlier proposals to revise the diagnostic criteria for CM to be based solely on the frequency of migraine days. Work on ICHD, 4th edition (ICHD-4) has begun (22) and there is an ongoing debate as to whether HFEM should be defined as a separate entity or be integrated into CM. The current classification not only requires at least eight monthly migraine days for a CM diagnosis but also includes a threshold for the total number of headache days. We propose removing the reference to non-migraine headache days, and defining CM solely on the basis of at least eight monthly migraine days, an approach also suggested by Chalmer et al. 10 Based on our data and the two previous studies (10,21), this revision would align the diagnostic criteria for CM more closely with the observed disease burden. A direct consequence of this change would be an increase in the number of individuals diagnosed with CM. While Chalmer et al. (10) estimated that this would double the number of patients with CM, our population-based study found a smaller increase of 64%.
In the present study, there was only a modest difference between mean monthly headache days (9.9 days in HFEM; 20.8 days in CM) and mean monthly migraine days (9.7 days in HFEM; 17.9 days in CM). Few studies have reported both measures within the same cohort, making it difficult to compare limiting direct comparisons of these numbers across different populations and methodological approaches. The discrepancy between headache days and migraine days likely depends on how a migraine day is defined, a topic of ongoing debate (23). The most stringent approach is to prospectively record, day-by-day, detailed headache characteristics and associated symptoms, enabling precise classification of both migraine and TTH days. Two studies using this approach found a greater discrepancy between headache days and migraine days than we observed. Van der Arendt et al. (23), using a detailed electronic diary, found that patients with HFEM had 10.5 headache days and 6.0 migraine days per month, while those with CM had 19.8 and 13.7 days, respectively. Similarly, Raffaelli et al. (24) reported 8.2 headache days and 5.6 migraine days for episodic migraine, and 21.1 and 15.8 days for CM. In routine clinical practice, this stringent approach is rarely used because it requires a high level of patient compliance and is often impractical. Instead, clinicians often use a more pragmatic approach, in which the patient describes each headache type, the clinician assigns one or more diagnoses, and the patient then reports the frequency of each headache type, typically with the aid of a headache calendar. This approach closely resembles the method used in the HARDSHIP questionnaire, where participants are asked whether they experience one or multiple headache types, followed by detailed questions about their most bothersome headache and the frequency of these, as well as the total frequency of all headaches (15). A key difference from routine clinical practice and also a limitation of the present study is that we relied on patient recall rather than using a headache calendar. Previous studies have shown that recall-based estimates tend to underestimate headache frequency compared with diary recordings, and that the discrepancy increases with higher headache frequency (25,26). Consequently, the true number of headache and migraine days in our sample may be higher than reported, and the difference between the HFEM and CM groups potentially larger than observed. Nevertheless, our findings indicate that recall-based migraine frequency remains closely associated with multiple measures of disease burden.
A clinically important secondary finding was the high rate of acute medication use in this population-based cohort. The proportion of participants taking acute medication on at least 10 days per month, comprising a risk factor for progression to higher headache frequency (27), was 46.0% in those with HFEM and 68.1% in those with CM. As many as 59.6% of individuals with CM also had MOH. These findings are consistent with previous reports (28) and emphasize the need for clinicians to screen for and address medication overuse in patients with high-frequency headaches, as successful treatment of MOH can lead to a significant reduction in headache frequency (29).
The strengths of the present study include the population-based design, large sample size, use of validated questionnaire-based diagnoses and detailed assessment of disease burden using the HARDSHIP instrument, which was developed specifically for this purpose. Several limitations should also be noted. First, migraine and headache frequencies were based on retrospective self-report, which is likely to be less accurate than data collected prospectively using a headache calendar, as discussed above. This limitation is closely related to the population-based design and emphasizes the need for studies with complementary designs. We find it reassuring that two clinic-based studies that did use headache calendars reached similar conclusions to this study. Second, the headache diagnoses were based on questionnaires rather than clinical interviews. However, validation against clinical interviews showed high specificity (87% for EM; 97% for CM; and 99% for MOH), indicating a low rate of false positive diagnoses. Thirdly, although the modest participation rate of 28.7% is comparable to previous population-based HARDSHIP studies in Europe (11.2 31.1%) (14,30), there is a risk of selection bias favouring participants with more severe headache, which may lead to inflated burden estimates and limit the generalizability of our findings. An overrepresentation of highly burdened individuals in the HFEM group could narrow the apparent difference in disease burden between HFEM and CM. However, this is unlikely to fully account for the results, as the findings were consistent across sensitivity analyses using alternative HFEM definitions (5–14 and 10–14 monthly migraine days). Lastly, the extent to which patient-reported outcome measures can accurately capture headache burden is debated. Headache disorders affect multiple domains of life and no single instrument can capture all aspects (31). Patient-reported outcomes rely on subjective recall and interpretation and may be influenced by factors such as current pain state, education level, or motivation. A strength of the HARDSHIP questionnaire is that it was specifically developed to assess headache burden in population-based studies, covers a broad range of relevant domains, and has been used in several countries worldwide (14,15,32,33).
Conclusions
The observed differences in disease burden between HFEM and CM were fully explained by the number of self-reported migraine days, independent of diagnostic classification. These findings support previous suggestions to revise the ICHD criteria by defining CM solely on the basis of monthly migraine days, rather than on total headache days.
Clinical implications
Chronic migraine had a higher disease burden compared to high-frequency episodic migraine. The difference was fully explained by the higher number of monthly migraine days in the chronic migraine group.
The findings support revising the ICHD criteria to define CM based on monthly migraine days only.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251406103 - Supplemental material for The burden of high-frequency episodic migraine and chronic migraine in the population-based PopHEAD study
Supplemental material, sj-docx-1-cep-10.1177_03331024251406103 for The burden of high-frequency episodic migraine and chronic migraine in the population-based PopHEAD study by Helene Engstrand, Maria Bengtson Argren, John-Anker Zwart, Erling Tronvik and Bendik Slagsvold Winsvold in Cephalalgia
Supplemental Material
sj-docx-2-cep-10.1177_03331024251406103 - Supplemental material for The burden of high-frequency episodic migraine and chronic migraine in the population-based PopHEAD study
Supplemental material, sj-docx-2-cep-10.1177_03331024251406103 for The burden of high-frequency episodic migraine and chronic migraine in the population-based PopHEAD study by Helene Engstrand, Maria Bengtson Argren, John-Anker Zwart, Erling Tronvik and Bendik Slagsvold Winsvold in Cephalalgia
Footnotes
Author contributions
Helene Engstrand: conceptualization, methodology, formal analysis, data curation, writing – original draft. Maria B. Argren: conceptualization, methodology, data curation. John-Anker Zwart: conceptualization, methodology, funding acquisition. Erling Tronvik: methodology, funding acquisition. Bendik S. Winsvold: conceptualization, methodology, formal analysis, supervision, project administration. All authors have critically reviewed and approved the final version of the manuscript submitted for publication.
Data availability
The datasets generated and analysed during the current study are not publicly available due to national privacy regulations but are available from the corresponding author on reasonable request, and with the necessary approvals.
Declaration of conflicting interests
HE has received personal fees for lectures from Pfizer and Novartis. MA has received personal fees for lectures from Lundbeck and Teva. ET has received personal fees for lectures and advisory boards from Novartis, Eli Lilly, Abbvie, TEVA, Roche, Lundbeck, Pfizer, Biogen and Organon. ET is a consultant for and holds stocks and intellectual property in Man & Science and Nordic Brain Tech and holds shares in Keimon Medical. ET has received non-personal research grants from European Commission, Norwegian Research Council, KlinBeForsk and Nordic Innovation, and has conducted commissioned (non-personal) research for Lundbeck. ET is an editorial board member of Cephalalgia and board member of European Headache Federation.
Ethical considerations
The study was approved by the Regional Committee for Medical and Health Research Ethics (REK #355800). The STROBE guidelines for observational studies were followed in this study. Participation was based on informed consent.
Funding
This work was funded by the Norwegian Research Council (#328615). Norges Forskningsråd, (grant number 328615).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
