Abstract
Background
This systematic review and meta-analysis synthesized migraine-related disability outcomes according to headache frequency subclassifications, including low-frequency episodic migraine (LFEM), high-frequency episodic migraine (HFEM) and chronic migraine (CM).
Methods
We searched the PubMed and Cochrane Library (CENTRAL) up to 16 October 2024 for peer-reviewed non-interventional studies reporting migraine-related disability outcomes in CM and subclassifications of episodic migraine (e.g. LFEM and HFEM). Eligible studies with an HFEM subgroup were grouped by headache frequency measure (monthly migraine days [MMD] or monthly headache days [MHD]), HFEM subgroup, disability parameter and study setting. Random-effects meta-analyses were conducted on groups with three or more studies, with the results presented in forest plots. Risk of bias was assessed using Joanna Briggs Institute tools.
Results
Of the 32 included studies, 27 were grouped, yielding five meta-analysis groups containing three or more studies. All five groups had an HFEM subgroup of 8–14 MHD. Accordingly, we classified LFEM as 0–7 MHD and CM as ≥15 MHD. Ten studies contributed data to the five meta-analysis groups. Estimated pooled values are reported by headache frequency subgroup for each meta-analysis group: LFEM (95% confidence interval [CI]), HFEM (95% CI) and CM (95% CI). For meta-analysis group 1, four population-based studies reported Migraine Disability Assessment (MIDAS) Grade IV proportions, with pooled values of 11.1% (7.1–17.1), 43.9% (31.8–56.9) and 57.5% (42.7–71.1), respectively. For meta-analysis group 2, five population-based studies documented Work Productivity and Activity Impairment (WPAI) outcomes. Pooled mean overall work productivity impairment (OWPI) scores were 36.9% (30.8–43.1), 44.7% (38.4–51.0) and 52.0% (47.7–56.3), respectively. Pooled mean activity impairment (AI) scores were 36.4% (33.7–39.1), 46.4% (42.8–50.0) and 53.5% (52.1–54.8), respectively. For meta-analysis group 3, three clinic-based studies presented MIDAS Grade IV proportions. Pooled values were 15.0% (4.4–40.5), 42.4% (17.1–72.4) and 65.7% (30.3–89.4), respectively. For meta-analysis group 4, three clinic-based studies reported WPAI outcomes. Pooled mean OWPI scores were 28.8% (21.6–35.9), 40.2% (36.2–44.2) and 49.6% (46.3–52.9), respectively. Pooled mean AI scores were 29.4% (21.9–36.9), 43.5% (39.0–48.1) and 51.6% (48.8–54.4), respectively. For meta-analysis group 5, three clinic-based studies detailed MIDAS scores. Pooled mean scores were 10.7 (3.9–17.5), 23.9 (9.4–38.4) and 49.6 (15.8–83.3), respectively.
Conclusions
This study showed a pattern of increasing migraine-related disability with rising headache frequency. Regardless of study setting, our meta-analyses also suggested a severe level of disability in many individuals (42%–44%) with HFEM, highlighting an unmet need for more effective migraine management. Disability burden and headache frequency should both be considered when determining treatment needs and therapy access, particularly for patients with HFEM.
Study Registration: INPLASY2024120039.
Keywords
Introduction
Migraine is a leading cause of disability worldwide, responsible for 88.3% of the burden of headache disorders and 4.7% of the total global years lived with disability in 2021 (1). The International Classification of Headache Disorders, 3rd edition (ICHD-3; 2018) defines chronic migraine (CM) as 15 or more monthly headache days (MHD) for more than three months, with the characteristics of a migraine headache on at least eight days per month (2). A recent update to the guidelines formalized the definition of episodic migraine (EM) as fewer than 15 MHD during the preceding three months, with some headaches being migraine (ICHD-4 alpha; 2020) (3). Approximately 2.5% of people with EM transition to CM each year (4), with higher headache frequency being a key risk factor for chronification (5,6). Some individuals also fluctuate spontaneously between EM and CM (7,8).
Historical characterizations of migraine-associated disability have been shaped largely by the 15 MHD threshold between CM (≥15 MHD) and EM (<15 MHD), with evidence consistently showing a greater severity of disability in CM compared to EM (9–12). However, EM is a heterogeneous condition with a wide range of headache frequencies (1–14 MHD) and associated disease burdens. The traditional approach to migraine classification overlooks the high burden of disease experienced by many individuals with a higher frequency of headaches that do not meet the ≥15 MHD criterion for a CM diagnosis. Accordingly, there is growing interest in evaluating the disability burden in EM subclassifications. Researchers typically differentiate between low-frequency episodic migraine (LFEM) and high-frequency episodic migraine (HFEM), with the threshold between these categories commonly set at 8, 9 or 10 MHD.
Although several studies have recently published cross-sectional analyses examining migraine-related disability in CM and subclassifications of EM, to the best of our knowledge, a comprehensive review synthesizing these findings through meta-analysis is lacking. To address this evidence gap, we conducted a systematic review and meta-analysis of migraine-related disability according to headache frequency subclassifications, with a focus on characterizing disability in the subset of migraineurs with HFEM.
Methods
Eligibility
Articles were eligible for inclusion if they reported migraine-related disability outcomes from a non-interventional study that included adults with CM and subclassifications of EM (e.g. LFEM and HFEM). Studies were not required to adhere to ICHD criteria. Disability outcomes were required to be either migraine-specific, be validated for use in migraine populations, or reflect migraine impact or impairment. Publications were excluded if they were not peer-reviewed; had no full-text manuscript available (e.g. conference proceedings); presented no original data (e.g. data duplication, protocol and technical descriptions, commentaries and review articles); or relevant data could not be extracted for the population(s) of interest.
Search strategy
We systematically searched the PubMed and Cochrane Library (CENTRAL) electronic databases from inception up to 16 October 2024, using the built-in advanced search tools within each platform. Other sources included Google Scholar and reference lists of retrieved publications. The detailed search strategy is presented in the supplementary material (Tables S1 and S2). We reported our review according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) (13). The study was registered with INPLASY (ID: INPLASY2024120039; DOI: 10.37766/inplasy2024.12.0039).
Selection process
A PhD-level reviewer (DE) screened all titles and abstracts to identify articles eligible for further review. Subsequently, full-text articles were obtained and assessed for compliance with the eligibility criteria. A second reviewer (RC; MSc) independently assessed a random sample of screened records (14). Disagreements on eligibility were resolved by discussion until consensus was achieved, with a third reviewer available to resolve disagreements if necessary (MSM).
Data extraction and outcomes
Two reviewers (DE and RC) extracted the following data from eligible publications: study design and setting variables, sample size variables, MHD or monthly migraine days (MMD) subgroup definitions, and migraine-related disability data.
Risk of bias assessment
Two reviewers (DE and RC) independently assessed the risk of bias for each included study using the Joanna Briggs Institute (JBI) critical appraisal tool for analytical cross-sectional studies (see supplementary material, Table S3). Disagreements were resolved through discussion until consensus was achieved, with a third reviewer available to resolve disagreements if necessary (MSM). The overall level of bias in each study was rated according to the number of “yes” answers: high risk (≤4), moderate risk (5–6) or low risk (7–8).
Statistical analysis
Headache frequency thresholds for LFEM, HFEM and CM, along with disability parameters, were determined post hoc based on the available studies after grouping according to headache frequency measure, HFEM subgroup, disability parameter and study setting. A meta-analysis was conducted for groups that contained at least three studies to ensure validity in the presence of heterogeneity (15). If studies reported the number of headache or migraine days per three months, we converted these to MHD and MMD, respectively. For each study that contributed data to the meta-analyses, we extracted the mean and standard deviation (SD) values for continuous variables or proportions for categorical variables. Missing SDs were approximated using the standard error (SE), confidence interval (CI) and sample size or median and interquartile range (IQR), or imputed from other studies that reported the SD by group (16). Standard techniques, as described in Luo et al. (17), were applied to convert median, Q1 and Q3 into mean and SDs using the metamean() function of the R-package meta (https://cran.r-project.org/package=meta). Each meta-analysis included three headache frequency ranges, defined according to the HFEM category. For example, for an HFEM subgroup of 8–14 MHD, LFEM would be categorized as 0–7 MHD and CM as ≥15 MHD. Subranges (e.g. 0–3 and 4–7 MHD) were combined to form a single category as necessary.
For continuous disability variables, we performed a random-effects meta-analysis of single means to calculate the overall mean and account for possible between-study variance. Meanwhile, for categorical disability variables, we performed a random-effects meta-analysis for single proportions. All data pooling was performed using inverse variance weighting. We used the I² test to assess heterogeneity between studies, adhering to the typical thresholds of low (25%), moderate (50%) and high (75%) for interpretation (18,19). Comparisons of mean values and proportions between (i) study settings (population-based and clinic-based) and (ii) headache frequency subgroups (LFEM, HFEM and CM) were conducted using linear and logistic mixed models, with the study setting or frequency subgroup as a categorical predictor and publication as a random effect to account for between-study heterogeneity. p-values were corrected for multiple testing (n = 3) using Bonferroni correction.
A sensitivity analysis was performed by evaluating and comparing the pooled estimates from a fixed-effect and random-effects meta-analysis. We also performed an additional sensitivity analysis that examined the effect of removing studies at high risk of bias (if applicable). All statistical analyses were performed using R, version 4.2.1 (R Foundation). We used the R-package “meta” for the meta-analyses. The resulting estimates and 95% CIs of the meta-analyses are presented in forest plots organized by study setting and disability measure.
Meta-analyses outcome measures
Two disability instruments were included in the meta-analyses after study selection and grouping: the Migraine Disability Assessment (MIDAS) and the Work Productivity and Activity Impairment Questionnaire (WPAI). The MIDAS questionnaire assesses the number of days in the last three months that migraine headaches limited or prevented activity, with disability classified according to total score: Grade I (0–5 points, little or no disability), Grade II (6–10 points, mild disability), Grade III (11–20 points, moderate disability) and Grade IV (21+ points, severe disability) (20). The WPAI questionnaire evaluates the impact of a health condition on an individual's work and daily activities over the past week, providing scores for absenteeism, presenteeism, overall work productivity impairment (OWPI; a combination of absenteeism and presenteeism) and activity impairment (AI). The WPAI questionnaire has been validated in both EM and CM (except for the absenteeism domain) (21).
Results
Study selection
Our systematic search of the PubMed and Cochrane Library (CENTRAL) databases retrieved 769 unique citations (Figure 1). Of these, we excluded 685 based on their title and abstract content. After a full-text evaluation of the remaining 84 citations (all English language), 27 articles met our eligibility criteria. Five additional articles met our eligibility criteria after searching the Google Scholar database and the reference lists of relevant manuscripts. In total, 32 study reports were included in our review (22–53).
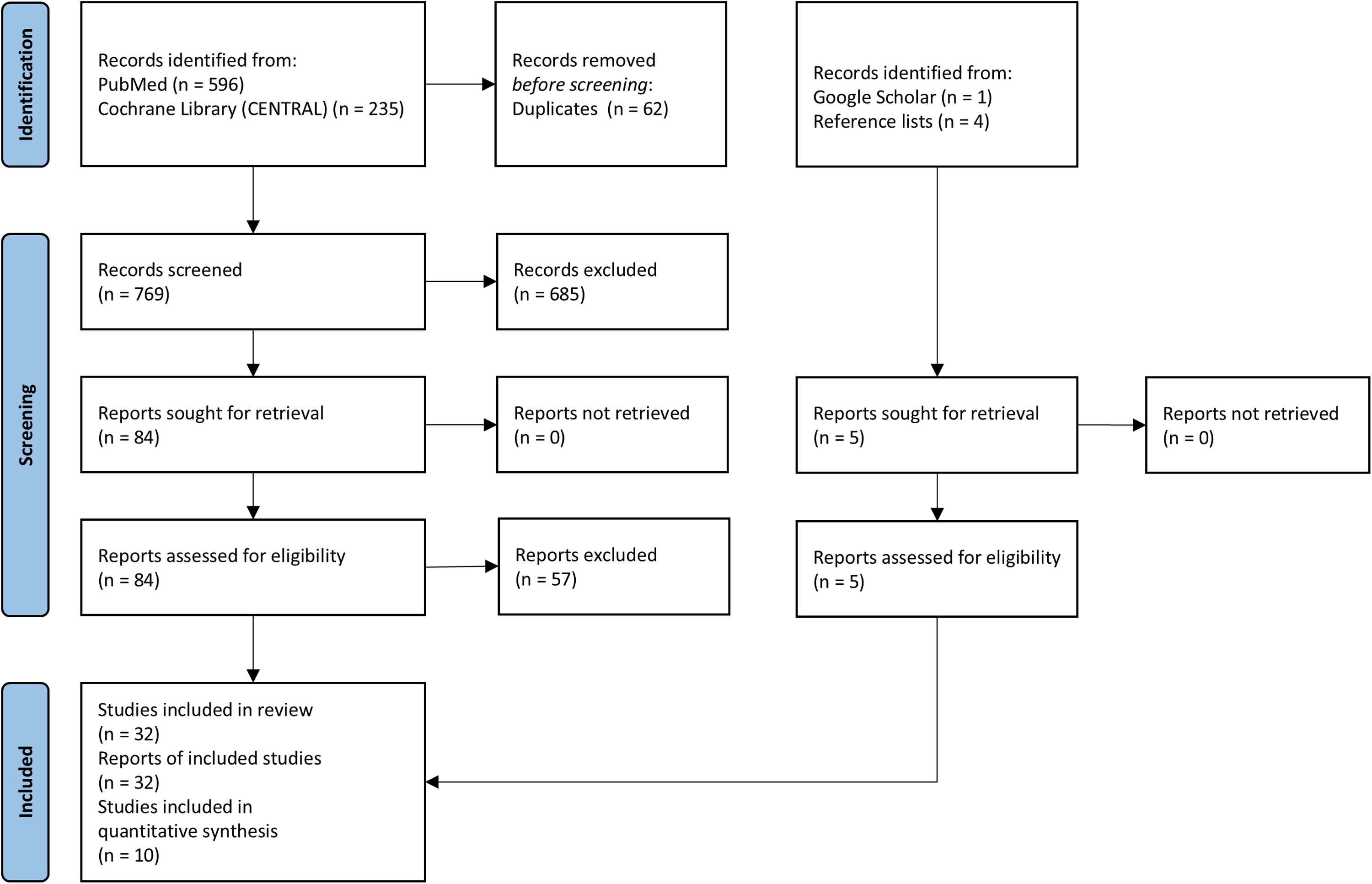
Preferred reporting items for systematic reviews and meta-analyses (PRISMA) reporting guideline flowchart (13). Registration number: INPLASY2024120039. DOI: 10.37766/inplasy2024.12.0039.
Among the 32 studies, five were excluded from the meta-analysis grouping process due to undefined headache frequency ranges (40), unclear HFEM classification (34,44,47) or unclear frequency measure (MHD or MMD) (46). The remaining 27 studies were grouped according to headache frequency measure (MHD or MMD), HFEM subgroup, disability parameter and study setting (see supplementary material, Table S4) (22–33,35–39,41–43,45,48–53). Five groups met the threshold for meta-analysis (i.e. more than three studies), comprising data from 10 studies, with some studies contributing to more than one meta-analysis (22–31). Groups with fewer than three studies (comprising data from 17 studies) were excluded from meta-analysis (for study details, see supplementary material, Table S5) (32,33,35–39,41–43,45,48–53).
Study characteristics and meta-analysis groups
Table 1 provides an overview of the main characteristics and findings of the 10 studies that contributed data to the meta-analyses. All studies performed a cross-sectional analysis and were published between 2017 and 2024. Geographical settings (exclusively or multinationally) included France, Germany, Italy, Spain, the UK (EU5), Japan and the USA. The analyzed sample sizes ranged from 218 to 21,143 participants, with most studies (nine of 10) having analysis populations of 500 or more (22–27,29–31). All studies measured headache frequency using MHD and had an HFEM subgroup of 8–14 MHD.
Characteristics and main findings of the 10 studies that contributed data to the meta-analyses.

Abbreviations: AI = Activity Impairment; ARMR = American Registry for Migraine Research; CM = Chronic Migraine; DSP = Disease Specific Programmes; EF = Emotional Function; HFCM = High-Frequency Chronic Migraine; HFEM = High-Frequency Episodic Migraine; ICHD = International Classification of Headache Disorders; LFCM = Low-Frequency Chronic Migraine; LFEM = Low-Frequency Episodic Migraine; MFEM = Medium/Moderate Frequency Episodic Migraine; MHD = Monthly Headache Days; MIBS = Migraine Interictal Burden Scale; MIDAS = Migraine Disability Assessment; MSQ = Migraine Specific Questionnaire; NHWS = National Health and Wellness Survey; OWPI = Overall Work Productivity Impairment; OVERCOME = Observational Survey of the Epidemiology, Treatment and Care of Migraine; RFP = Role Function-Preventive; RFR = Role Function-Restrictive; WPAI = Work Productivity and Activity Impairment. DE = Germany; ES = Spain; FR = France; UK = United Kingdom; IT = Italy; JP = Japan; USA = United States of America; EU5 = FR, DE, IT, ES, UK. *Headache frequency subgroup names (e.g. LFEM, MFEM, HFEM, CM) were applied to studies that reported only numerical ranges – typical thresholds were used.
Seven of the 10 studies were population-based (22–28), with data drawn from the American Migraine Prevalence and Prevention Study (AMPP: 2005 (22)), the National Health and Wellness Survey (NHWS: EU5, 2016 (28); EU5, 2017 (23); Spain, December 2019 to April 2020 (24)) and the ObserVational survey of the Epidemiology, tReatment and Care of MigrainE (OVERCOME: USA, September to November 2018 (25); Japan, July to September 2020 (26); Spain, October 2020 to February 2021 (27)). The remaining three studies were clinic-based (29–31), with data drawn from the Adelphi Migraine Disease Specific Programme (USA, January to March 2014 (29); Japan, January to March 2014 (31)) and the American Registry for Migraine Research (ARMR: February 2016 to March 2020 (30)). Although some surveys were conducted in the same country or region, no overlap in the timeframes was evident between the 10 studies.
Four of the 10 population-based studies reported the proportion of participants classified with severe disability (MIDAS Grade IV), forming the first meta-analysis group (22,25–27). The pooled sample included 49,071 participants in the LFEM subgroup, 5451 in the HFEM subgroup and 4924 in the CM subgroup. Five of the 10 population-based studies contributed WPAI data to the second meta-analysis group, with separate analyses of OWPI (LFEM: n = 20,191; HFEM: n = 2163; CM: n = 1538) and AI (LFEM: n = 25,951; HFEM: n = 2868; CM: n = 2186) (23,24,26–28).
All three clinic-based studies reported the proportion classified with severe disability (MIDAS Grade IV), forming the third meta-analysis group (LFEM: n = 1158; HFEM: 367; CM: n = 431) (29–31). These studies also provided WPAI data to form the fourth meta-analysis group, with separate analyses of mean OWPI scores (LFEM: n = 1016; HFEM: n = 269; CM: n = 258) and mean AI scores (LFEM: n = 1227; HFEM: n = 313; CM: n = 282). Additionally, these three studies all reported MIDAS scores to form the fifth meta-analysis group (LFEM: n = 1268; HFEM: n = 380; CM: n = 445).
Risk of bias in studies
Our risk of bias assessment judged three of the seven population-based studies included in the meta-analyses as having a high risk of bias (see supplementary material, Table S6) (25–27). None of these studies described strategies to address potential confounding. Another three of the seven population-based studies were rated as having a moderate risk of bias (23,24,28). Although these studies reported strategies to control for confounding, they relied on a self-reported physician diagnosis of migraine without specifying whether standard ICHD diagnostic criteria were consistently applied to all participants. We assessed the remaining population-based study as having a low risk of bias (22). Among the three clinic-based studies, two were rated as having a high risk of bias (29,31). These studies did not describe any methods to address potential confounding. The remaining clinic-based study was deemed to have a moderate risk of bias (30). Although this study did not describe methods to control for confounding, a headache specialist consistently applied ICHD diagnostic criteria to all participant questionnaire data (30).
We also evaluated the risk of bias within each of the five meta-analysis groups. In the first population-based meta-analysis analyzing the proportions classified with severe disability (MIDAS Grade IV), three of four studies were assessed as having a high risk of bias (25–27), while the remaining study was judged to have a low risk of bias (22). In the second population-based meta-analyses of mean WPAI scores, two of five studies were judged to be at high risk of bias (26,27) and three of five at moderate risk of bias (23,24,28). For each of the three clinic-based meta-analyses (the proportions classified with severe disability [MIDAS Grade IV], mean WPAI scores and mean MIDAS scores), two of the three studies were rated as having a high risk of bias (29,31), with the remaining study judged to have a moderate risk of bias (30).
Migraine-related disability in population-based studies
In the population-based meta-analysis of the pooled proportions with severe disability (MIDAS Grade IV), the estimated proportion was 11.1% (95% CI = 7.1–17.1) for LFEM, 43.9% (95% CI = 31.8–56.9) for HFEM and 57.5% (95% CI = 42.7–71.1) for CM, with high heterogeneity for all subgroups (I2 = 99.6%, 98.8% and 99.1%, respectively) (Figure 2). Pairwise comparisons of estimates showed a significant difference between LFEM and HFEM (p < 0.001) and between LFEM and CM (p < 0.001), but not between HFEM and CM (p = 0.48). Extracted data for this meta-analysis group are presented in the supplementary material (Table S7).

Forest plots of the estimated overall proportions of the population sizes with severe disability (MIDAS Grade IV) from population-based studies included in the meta-analysis. The 95% Wilson score CI for single proportions is reported for each study. The estimated between-study heterogeneity I-squared statistic (I2) is also reported. Abbreviations: CI = Confidence Interval; CM = Chronic Migraine; HFEM = High-Frequency Episodic Migraine; LFEM = Low-Frequency Episodic Migraine; MHD = Monthly Headache Days; MIDAS = Migraine Disability Assessment.
The population-based meta-analysis of pooled WPAI scores estimated a mean OWPI score of 36.9% (95% CI = 30.8–43.1) for LFEM, 44.7% (95% CI = 38.4–51.0) for HFEM and 52.0% (95% CI = 47.7–56.3) for CM, with high heterogeneity for all subgroups (I2 = 99.7%, 96.5% and 85.2%, respectively) (Figure 3). We observed no significant difference between estimates for LFEM and HFEM (p = 0.17) or HFEM and CM (p = 0.26); however, LFEM and CM were significantly different (p < 0.001). For the WPAI AI domain, the estimated mean score was 36.4% (95% CI = 33.7–39.1) for LFEM, 46.4% (95% CI = 42.8–50.0) for HFEM and 53.5% (95% CI = 52.1–54.8) for CM (Figure 4). Heterogeneity was high for LFEM (I2 = 98.9%) and HFEM (I2 = 91.1%) but low for CM (I2 = 19.5%). All pairwise comparisons of estimates were statistically significant (LFEM vs. HFEM: p < 0.001; LFEM vs CM: p < 0.001; HFEM vs CM: p = 0.003). Extracted data for this meta-analysis are available in the supplementary material (Table S8).
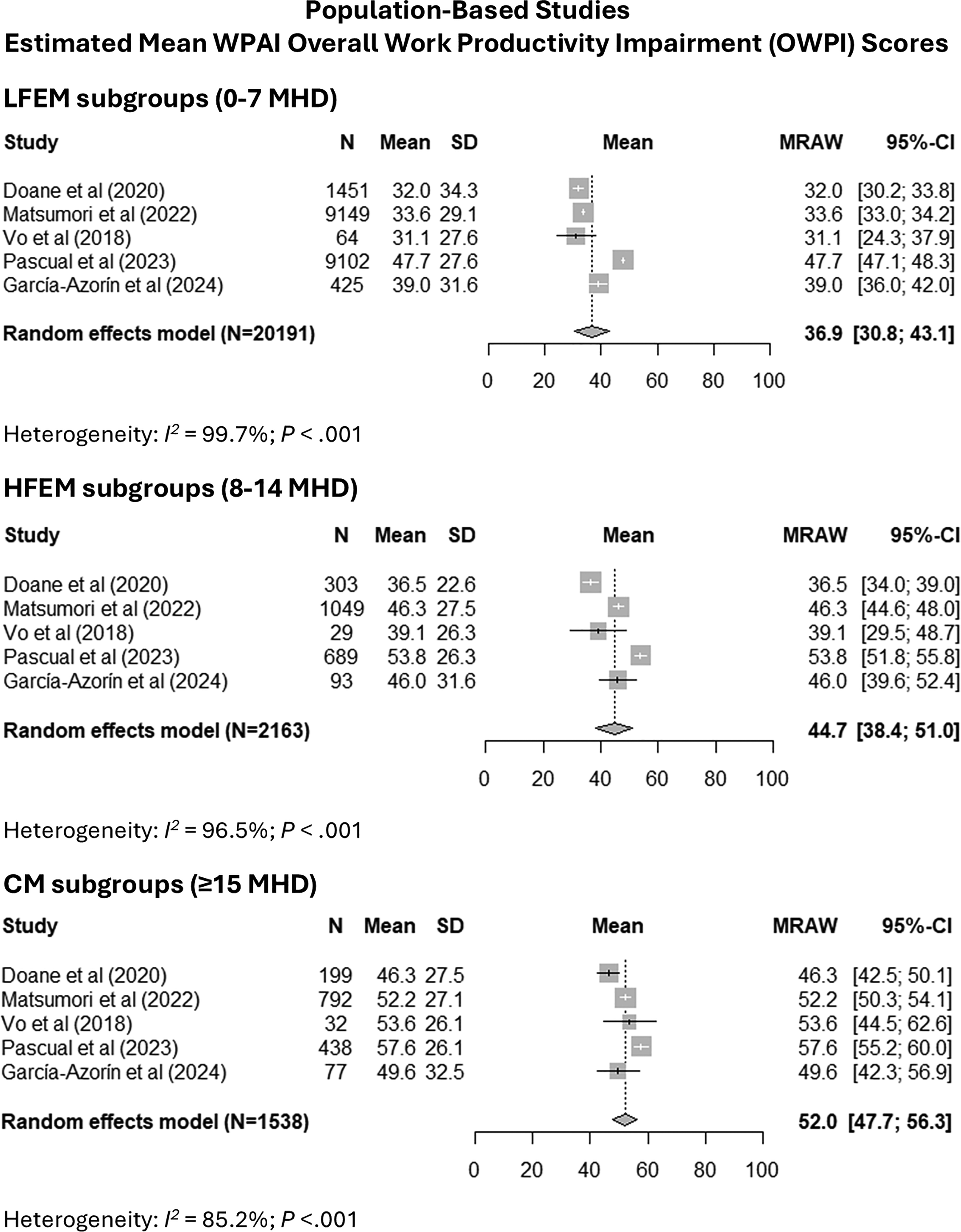
Forest plots of the estimated overall mean scores for the WPAI OWPI domain for population-based studies included in the meta-analysis. The 95% CI for the mean is reported for each study. The estimated between-study heterogeneity I-squared statistic (I2) is also reported. Abbreviations: CI = Confidence Interval; CM = Chronic Migraine; HFEM = High-Frequency Episodic Migraine; LFEM = Low-Frequency Episodic Migraine; MHD = Monthly Headache Days; N = population size; OWPI = Overall Work Productivity Impairment; SD = Standard Deviation; WPAI = Work Productivity and Activity Impairment.

Forest plots of the estimated overall mean scores for the WPAI AI domain for population-based studies included in the meta-analysis. The 95% CI for the mean is reported for each study. The estimated between-study heterogeneity I-squared statistic (I2) is also reported. Abbreviations: AI = Activity Impairment; CI = Confidence Interval; CM = Chronic Migraine; HFEM = High-Frequency Episodic Migraine; LFEM = Low-Frequency Episodic Migraine; MHD = Monthly Headache Days; N = population size; SD = Standard Deviation; WPAI = Work Productivity and Activity Impairment.
Migraine-related disability in clinic-based studies
In the clinic-based meta-analysis of the pooled proportions with severe disability (MIDAS Grade IV), the estimated proportion was 15.0% (95% CI = 4.4–40.5) for LFEM, 42.4% (95% CI = 17.1–72.4) for HFEM and 65.7% (95% CI = 30.3–89.4) for CM, with high heterogeneity for all subgroups (I2 = 98.6%, 97.1% and 95.0%, respectively) (Figure 5). Estimates for LFEM differed significantly from CM (p = 0.047), but not for LFEM and HFEM (p = 0.43) or HFEM and CM (p = 0.10). Extracted data for this meta-analysis are detailed in the supplementary material (Table S9).

Forest plots of the estimated overall proportions of the population sizes with severe disability (MIDAS Grade IV) from clinic-based studies included in the meta-analysis. The 95% Wilson score CI for single proportions is reported for each study. The estimated between-study heterogeneity I-squared statistic (I2) is also reported. Abbreviations: CI = Confidence Interval; CM = Chronic Migraine; HFEM = High-Frequency Episodic Migraine; LFEM = Low-Frequency Episodic Migraine; MHD = Monthly Headache Days; MIDAS = Migraine Disability Assessment.
The clinic-based meta-analysis of pooled WPAI scores estimated a mean OWPI score of 28.8% (95% CI = 21.6–35.9) for LFEM, 40.2% (95% CI = 36.2–44.2) for HFEM and 49.6% (95% CI = 46.3–52.9) for CM (Figure 6). Heterogeneity was high for LFEM (I2 = 94.8%), moderate for HFEM (I2 = 39.2%) and not observed for CM (I2 = 0%). Estimates for LFEM differed significantly from both HFEM (p = 0.01) and CM (p < 0.001), but not for HFEM and CM (p = 0.22). For the WPAI AI domain, the estimated mean score was 29.4% (95% CI = 21.9–36.9) for LFEM, 43.5% (95% CI = 39.0–48.1) for HFEM and 51.6% (95% CI = 48.8–54.4) for CM (Figure 7). Heterogeneity was high for LFEM (I2 = 97.0%), moderate for HFEM (I2 = 60.1%) and not observed for CM (I2 = 0%). Estimates for LFEM differed significantly from both HFEM (p = 0.002) and CM (p < 0.001), but not for HFEM and CM (p = 0.14). Extracted data for this meta-analysis are presented in the supplementary material (Table S10).
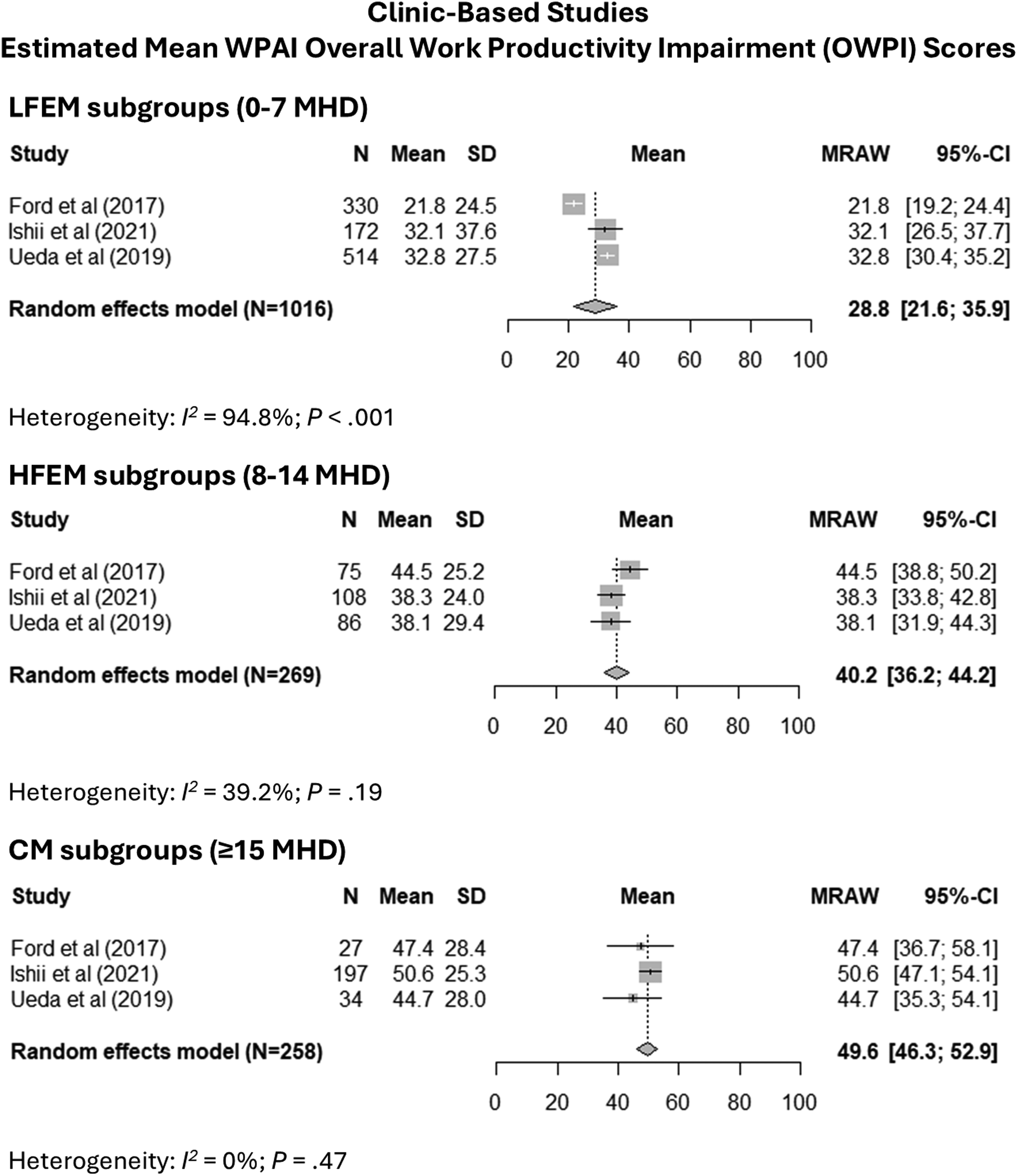
Forest plots of the estimated overall mean scores for the WPAI OWPI domain for clinic-based studies included in the meta-analysis. The 95% CI for the mean is reported for each study. The estimated between-study heterogeneity I-squared statistic (I2) is also reported. Abbreviations: CI = Confidence Interval; CM = Chronic Migraine; HFEM = High-Frequency Episodic Migraine; LFEM = Low-Frequency Episodic Migraine; MHD = Monthly Headache Days; N = population size; OWPI = Overall Work Productivity Impairment; SD = Standard Deviation; WPAI = Work Productivity and Activity Impairment.

Forest plots of the estimated overall mean scores for the WPAI AI domain for clinic-based studies included in the meta-analysis. The 95% CI for the mean is reported for each study. The estimated between-study heterogeneity I-squared statistic (I2) is also reported. Abbreviations: AI = Activity Impairment; CI = Confidence Interval; CM = Chronic Migraine; HFEM = High-Frequency Episodic Migraine; LFEM = Low-Frequency Episodic Migraine; MHD = Monthly Headache Days; N = population size; SD = Standard Deviation; WPAI = Work Productivity and Activity Impairment.
In the clinic-based meta-analysis of pooled MIDAS scores, the estimated mean score was 10.7 (95% CI = 3.9–17.5) for LFEM, 23.9 (95% CI = 9.4–38.4) for HFEM and 49.6 (95% CI = 15.8–83.3) for CM, with high heterogeneity in all subgroups (I2 = 97.8%, 97.4% and 98.6%, respectively) (Figure 8). Estimates for LFEM differed significantly from both HFEM (p < 0.001) and CM (p < 0.001), but not for HFEM and CM (p = 0.10). Extracted data for this meta-analysis are shown in the supplementary material (Table S11). Figure 9 summarizes the meta-analyses for all disability outcomes and study settings.

Forest plots of the estimated overall mean MIDAS scores for clinic-based studies included in the meta-analysis. The 95% CI for the mean is reported for each study. The estimated between-study heterogeneity I-squared statistic (I2) is also reported. Abbreviations: CI = Confidence Interval; CM = Chronic Migraine; HFEM = High-Frequency Episodic Migraine; LFEM = Low-Frequency Episodic Migraine; MHD = Monthly Headache Days; MIDAS = Migraine Disability Assessment; N = population size; SD = Standard Deviation.
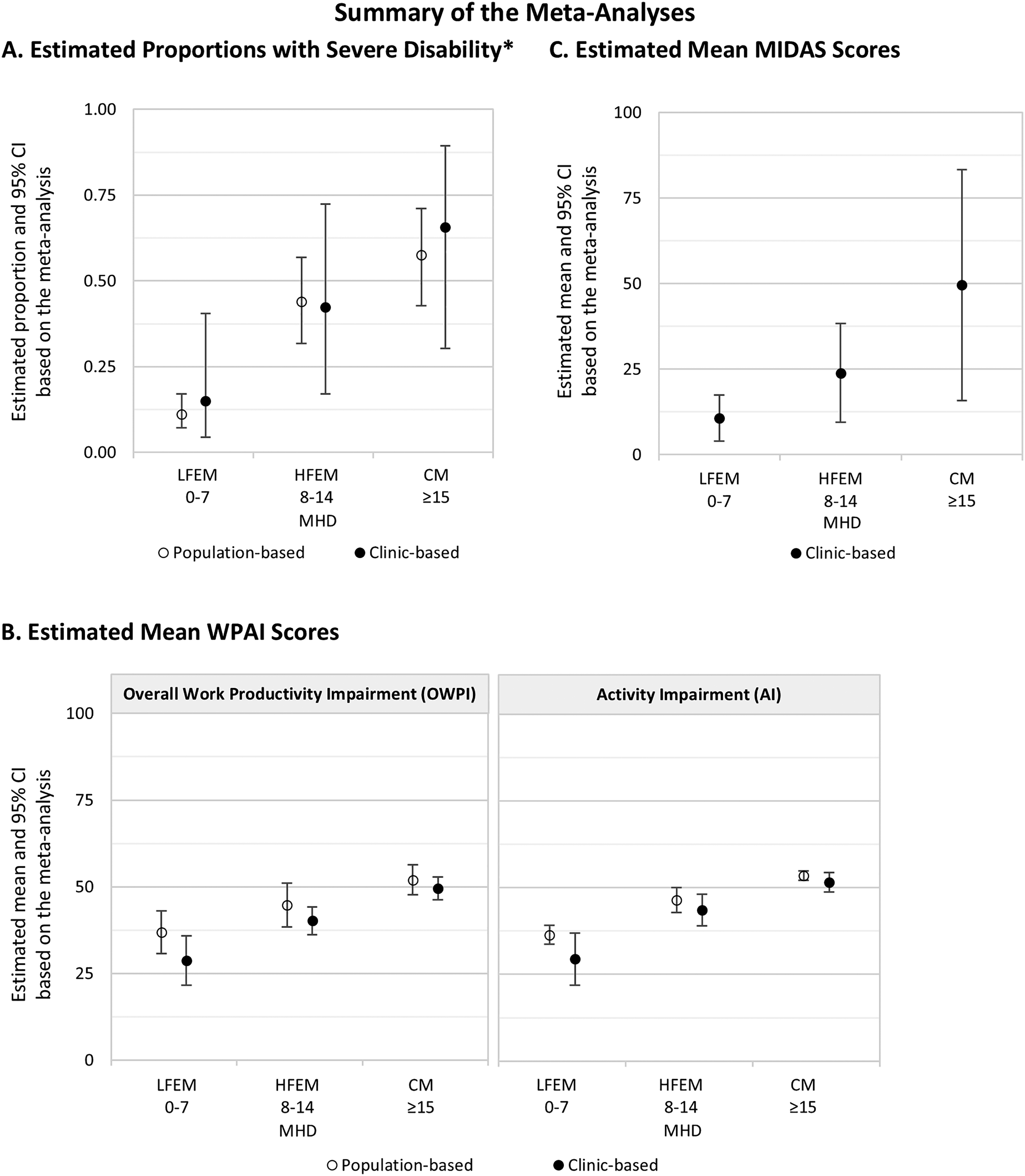
Summary of the meta-analyses showing estimated overall mean and 95% CI or estimated overall proportion and 95% CI for (A) Estimated proportions with severe disability (MIDAS Grade IV), (B) estimated mean WPAI scores, and (C) estimated mean MIDAS scores. Abbreviations: AI = Activity Impairment; CI = Confidence Interval; CM = Chronic Migraine; HFEM = High-Frequency Episodic Migraine; LFEM = Low-Frequency Episodic Migraine; MHD = Monthly Headache Days; MIDAS = Migraine Disability Assessment; OWPI = Overall Work Productivity Impairment; WPAI = Work Productivity and Activity Impairment. *MIDAS Grade IV.
Migraine-related disability in population-based vs clinic-based studies
Meta-regression found no statistically significant differences between population-based and clinic-based estimates for any subgroup (LFEM, HFEM and CM) for the proportion classified with MIDAS Grade IV (p = 1.0 for all comparisons), mean WPAI OWPI score (p = 0.31, p = 1.0 and p = 0.98, respectively), or mean WPAI AI score (p = 0.10, p = 1.0 and p = 0.82, respectively). For MIDAS Grade IV and mean MIDAS score, wide CIs were observed, which could be expected given the low number of studies and high study heterogeneity.
Sensitivity analyses
A fixed-effect meta-analysis was performed as a sensitivity analysis for each metric and study setting (see supplementary material, Figures S1–S5) (14,54). The point estimates of the overall means and proportions were similar in all but one analysis, supporting the robustness of our findings. As expected, in the presence of between-study heterogeneity, the confidence intervals of the fixed-effect model were narrower, showing that the random-effects model is the most conservative approach in this context. In the sensitivity analysis of the clinic-based proportion classified with MIDAS Grade IV in the CM subgroup, we observed a difference between the fixed-effect and random-effects models, likely stemming from the much larger sample size in one of the clinic-based studies. Given the high heterogeneity in this analysis, the random effects model remains the preferred approach.
It was possible to perform an additional sensitivity analysis on the population-based group of studies that documented mean WPAI scores because three studies were rated as having a moderate risk of bias (23,24,28) and two studies were judged to have a high risk of bias (26,27). Removing the studies with a high risk of bias from the meta-analysis yielded generally lower between-study heterogeneity and similar or higher estimated mean WPAI scores compared to the complete group (see supplementary material, Figure S6), suggesting that the observed mean scores from the complete group were not an overestimation caused by the inclusion of studies with a high risk of bias.
Discussion
To our knowledge, this is the first systematic review to synthesize migraine-related disability outcomes across headache frequency subclassifications using meta-analytic techniques, with a particular focus on HFEM. Our meta-analyses suggest a pattern of worsening disability as headache frequency increased, based on subgroups of LFEM (0–7 MHD), HFEM (8–14 MHD) and CM (≥15 MHD). This trend was consistent across study settings (population-based and clinic-based) and all analyzed disability measures (the proportions classified as MIDAS Grade IV, mean WPAI scores and mean MIDAS scores).
When we compared pooled estimates between headache frequency subgroups in population- and clinic-based studies, LFEM had a significantly lower disability burden than HFEM in most comparisons (5 of 7), and a significantly lower disability burden than CM in all comparisons. In contrast, the difference between HFEM and CM was statistically significant in only one comparison (1 of 7). However, the small number of studies in both settings and the smaller sample sizes in the clinic-based studies may have limited our ability to detect true differences between the HFEM and CM subgroups. Similarly, we observed no statistically significant differences in comparisons between population- and clinic-based samples. However, in this case, confounding factors such as participation bias may also have contributed to this result; for example, individuals with a higher symptom burden from the general population may be more inclined to complete disability questionnaires, resulting in an overall higher level of disability more akin to that seen in a clinic-based setting.
Consistent across study settings, our results suggest that many individuals with HFEM (42–44%) may experience severe disability and substantial impairment at work and in daily activities. To address this unmet treatment need, recommendations and decisions regarding access to migraine therapies for patients with HFEM should consider both headache frequency and the severity of migraine-related disability.
Severe disability in HFEM substantially impacts healthcare resource utilization (HCRU), as demonstrated in a US study of 7662 patients with EM (55). In a specialist care setting, patients with severe disability (MIDAS score >20) had over 42% higher mean healthcare visit costs and over 50% higher mean pharmacy costs compared to those with moderate disability (MIDAS score 11–20). Additionally, several of the 32 studies included in our review found no statistically significant difference between HFEM and CM in at least one disability measure, suggesting a similar level of disability in the two groups (Table 1; see also supplementary material, Table S4) (22,30,35,36,38,52). Some researchers have proposed merging the HFEM and CM categories (36), while others have reservations about widening the definition of CM (40). Adding to this complexity, two analyses proposed a threshold of 10 MHD to identify the most severely disabled migraine patients (42,52). In our review, the definitions of HFEM varied across the 32 studies, including 8–14, 9–14 and 10–14 MHD, which may have contributed to the small inconsistencies in outcomes.
While the literature lacks a clear consensus on whether the HFEM and CM categories should be combined based on migraine-related disability burden, a recent narrative review from Cammarota et al. (56) identified distinct clinical and pathophysiological features of HFEM compared to LFEM, along with evidence of more severe disability and a higher risk of disease chronification. Accordingly, the authors proposed formal recognition of HFEM as a separate migraine subtype (defined as 8–14 MHD for more than three months, with migrainous features on more than four days per month), suggesting that this would raise awareness of the high disease burden in HFEM, improve access to preventive treatments, and potentially reduce the incidence of chronification. Our meta-analytic findings support this position, suggesting a severe disability burden in HFEM defined as 8–14 MHD and a significant difference in disability burden between LFEM and HFEM. In the meantime, pending further evidence or updated ICHD guidelines to settle the debate over migraine subclassification based on headache frequency thresholds, a pragmatic approach to treatment access that considers both disability burden and headache frequency would be beneficial.
Strengths and limitations of the review
The main strengths of this study are its systematic review of the literature, application of meta-analytic techniques and sensitivity analyses, and adherence to the PRISMA reporting guidelines (13). However, our study also has some limitations. High heterogeneity (85.2–99.7%) was observed in 16 of the 21 pooled subgroup analyses, likely arising from differences in study populations and methodologies. For example, most studies did not report the consistent application of ICHD diagnostic criteria to all participants. Considering the high level of heterogeneity across the meta-analyses, our pooled estimates should be interpreted with caution. We did not conduct subgroup analyses or meta-regression to identify possible sources of heterogeneity, other than risk of bias (all studies versus low to moderate bias), because of the small number of studies in each of our meta-analyses (54).
Our use of cross-sectional data also restricts causal inference between migraine and disability. Cross-sectional studies are also particularly susceptible to non-response and recall biases for patient-reported outcomes. Furthermore, they exclude the possibility of examining longitudinal changes in migraine burden over time that may arise from other factors, including disease progression, fluctuations in disease severity, and changes in migraine treatment. However, our use of non-interventional studies may better reflect the characteristics of the general migraine population compared to more selected cohorts.
Among the 32 studies included in the review (prior to the grouping process), we observed variations in the headache frequency measure (MHD vs. MMD). While this distinction may not be critical, we grouped studies according to the reported headache frequency measure for consistency. This approach reduced the size of some meta-analysis groups and excluded MMD disability parameter groups from meta-analysis (all contained fewer than three studies). Similarly, there was variation in the HFEM subgroup (typically 8–14, 9–14 or 10–14 MHD) and study settings. We also considered these factors in the study grouping for consistency, which further reduced the size of some meta-analysis groups. Additionally, creating migraine subgroups using specific headache frequency thresholds will always involve some inherent subjectivity, with the possibility of finding similarities and differences between groups regardless of the threshold used.
Among the 10 studies included in our meta-analyses, only seven countries were represented, which limits the generalizability of our findings to other countries and regions. Of the five meta-analysis groups, four were dominated by studies rated as having a high risk of bias, which could lead to an overestimation of the disease burden. We were unable to conduct sensitivity analyses that excluded high-risk-of-bias studies for these four groups due to the small number of studies.
The data format in some articles may have led to minor inaccuracies in the source data for our meta-analyses. For example, some studies required data pooling to create a standard set of categories (e.g. pooling 0–3 MHD and 4–7 MHD categories to form a single LFEM group; see supplementary material, Tables S7–S11). Some studies also omitted essential data points (e.g. SDs and numerator and denominator for proportions), necessitating approximation or imputation using standard methods (see supplementary material, Tables S7–S11). Other studies provided data graphically, which required extraction using tools such as WebPlotDigitizer (see supplementary material, Tables S7–S11) (57). In one study, the analysis excluded the group with 0–3 MHD, resulting in an LFEM group of 4–7 MHD (28).
Another potential limitation of our study relates to the measurement properties of the MIDAS and WPAI instruments. The MIDAS 3-month recall period may introduce recall bias (58). Additionally, patients may be confused by subjective questions, such as estimating the number of days in which productivity was reduced by half or more (59,60). In contrast, the WPAI instrument uses a seven-day recall. This shorter window may improve recall accuracy, but may not represent the true migraine burden in LFEM, which can vary considerably from week to week. Furthermore, since the WPAI work productivity domain excludes the unemployed, it misses individuals who have recently left work due to frequent migraine-related absenteeism (21). This may lead to an underrepresentation of the most severely disabled populations.
Finally, our study also analyzed migraine frequency according to categories of MHD, in line with the included articles. Analyzing the linear relationship between the number of headache days per month and disability measures would provide a more nuanced examination of disability across the headache frequency spectrum.
Implications for practice, policy, and future research
Current treatment guidelines from the International Headache Society (IHS) and the American Headache Society recommend initiating preventive therapy considering both headache frequency and the degree of migraine-related disability or impact (61,62). However, some countries limit access to therapies such as onabotulinumtoxin A based on a CM diagnosis (63), typically defined with a threshold of ≥15 MHD. A more pragmatic approach to treatment access that also considers disability burden in addition to headache frequency would help physicians tailor treatment strategies by disease severity (64). Earlier interventions that effectively manage and reduce migraine frequency may alleviate disease burden and lower the risk of chronification (65). This has important implications for healthcare systems, given the association between increased headache frequency and higher HCRU and direct/indirect costs (35,41,49).
While our results suggest that disability burden increases with rising headache frequency, our analysis was constrained by the categorical classifications used in the included literature. It is more likely that migraine-related disability increases with each additional headache day per month, rather than at specific frequency thresholds. Future large-scale, longitudinal, prospective cohort studies, ideally conducted in population-based settings, will help clarify the relationship between headache frequency and disability in migraine. We also suggest analyzing the linear relationship between the number of headache days per month and disability measures instead of headache frequency categories. The consistent use of a single definition of HFEM and a validated disability measure would improve the comparability of studies for future data synthesis.
A more pragmatic approach to future clinical trials of preventive therapies is also needed. Specifically, trials should combine highly disabled HFEM and CM cohorts into a single population, with prespecified subgroup subanalysis. Such an approach would mirror the recent IHS guidelines for migraine preventive trials among children and adolescents, which recommend removing the 15 MHD threshold and including all headache frequencies, with a preplanned subanalysis of migraine frequency during the initial screening phase (66).
Finally, there is an urgent need to raise awareness among healthcare professionals about the potential for a high disability burden in patients with ≥8 MHD. This heightened awareness could lead to earlier intensification of migraine preventive treatment in patients with HFEM, potentially improving their quality of life, preventing chronification and mitigating the socioeconomic impact of lost work days among those in employment.
Conclusions
This study showed a pattern of increasing disability in people with migraine as headache frequency increased. Our meta-analyses also indicate that disability may be severe in many individuals (42–44%) with HFEM, suggesting an unmet need for more effective migraine management in this population. Accordingly, disability burden and headache frequency should ideally both be considered when recommending migraine treatment plans and deciding about coverage for migraine therapies. Such a shift in clinical practice and policy may support timely interventions tailored to disease severity that can improve patients’ quality of life and reduce the risk of chronification.
Clinical implications
This systematic review and meta-analysis showed that migraine-related disability increases as headaches become more frequent. Our results also suggest that high-frequency episodic migraine (HFEM; 8–14 MHD) is a severely disabling migraine subtype. Treatment decisions and therapy access should consider both headache frequency and disability burden, especially for those with HFEM.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251385965 - Supplemental material for Migraine-related disability according to headache frequency subclassifications: A systematic review and meta-analysis
Supplemental material, sj-docx-1-cep-10.1177_03331024251385965 for Migraine-related disability according to headache frequency subclassifications: A systematic review and meta-analysis by Manjit Singh Matharu, Stephen Silberstein, Hsiangkuo Yuan, Deborah Edgar, Roos Colman, Todd J. Schwedt, Michel Lanteri-Minet and Mark Obermann in Cephalalgia
Footnotes
Acknowledgements
In a protocol amendment, we updated our analysis approach to account for the potential impact of study setting (i.e. sample type) on study outcomes, compare estimates between study settings, and compare estimates between headache frequency subgroups. We also revised our JBI checklist.
Author contributions
Conceptualization: MSM, DE and RC; Methodology: MSM, DE and RC; Meta-analyses: RC; Writing – original draft preparation: DE; Writing – review and editing: MSM, SS, HY, TS, ML, MO and RC. All authors have read and agreed to the final version of the manuscript submitted for publication.
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MSM is chair of the medical advisory board of the CSF Leak Association; has served on advisory boards for AbbVie, Eli Lilly, Lundbeck, Pfizer, Salvia and TEVA; has received payment for educational presentations from AbbVie, Eli Lilly, Lundbeck, Organon, Pfizer and TEVA; has received grants from Abbott, Medtronic and Ehlers Danlos Society; and has a patent on system and method for diagnosing and treating headaches (WO2018051103A1, issued). Within the past 24 months, HY has received funding from AHS Early-Stage Investigator Research Award; institutional support for serving as an investigator from Teva, AbbVie, Ipsen, Pfizer and Parema; consultant/ad-board fees from Salvia, AbbVie, Pfizer and Cerenovus; and royalties from Cambridge University Press and MedLink. DE received consulting fees from Salvia BioElectronics BV in her capacity as an independent medical writer. RC received consulting fees from Salvia BioElectronics BV in her capacity as an independent biostatistician. TS, within the prior 24 months, has received consulting fees from AbbVie, Amgen, Eli Lilly, Linpharma, Lundbeck, Salvia BioElectronics, Scilex and Theranica, and royalties from UpToDate. He holds/held stock options in Allevalux, Aural Analytics and Nocira. He has received research funding from the American Heart Association, Flinn Foundation, Henry Jackson Foundation, National Headache Foundation, National Institutes of Health, Patient-Centered Outcomes Research Institute, Pfizer, Spark Neuro and United States Department of Defense. MLM reports personal fees for advisory boards, speaker panels, or investigation studies from Allergan, Amgen, Astellas, ATI, BMS, Boehringer, Boston Scientific, CoLucid, Convergence, Eli Lilly, GlaxoSmithKline, Grunenthal, Eli Lilly, IPSEN Lundbeck, Medtronic, MSD, Novartis, Perfood, Pfizer, Reckitt Benckiser, Saint-Jude, Salvia BioElectronics, Sanofi-Aventis, Teva, UCB, UPSA and Zambon. MO received scientific support, travel support, and/or consulting fees from Biogen Idec, Novartis, Sanofi/Genzyme, Pfizer, Teva, Ely Lilly, Noema Pharma and Salvia BioElectronics. He received research grants from Allergan, Electrocore, Heel and the German Ministry for Education and Research (BMBF).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Salvia BioElectronics BV funded this study. The funder facilitated an external clinical advisory committee, an independent biostatistician, and an independent medical writer to develop and execute this study. The funder did not direct the data analysis, results interpretation, drafting of the manuscript, final review of the manuscript, or decision to submit the manuscript for publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
