Abstract
Background
Many guidelines list migraine with aura (MwA) as a contraindication to estrogen-containing combined hormonal contraceptives (CHCs) due to vascular risks. However, current evidence is based on small sample studies with potential influence by confounding factors. Additionally, few studies have examined the vascular risk associated with modern CHCs with lower dose estrogen, particularly in relation to aura status. The present study aims to investigate the vascular risk of modern CHCs in women with migraine with and without aura.
Methods
We used a de-identified electronic medical record database with 120 million patients across multiple health systems in the United States of America. We included female patients aged 18–45 years who received a migraine diagnosis code, had at least three office visits within three years, and were prescribed at least one migraine-specific medication within 6 months following the first outpatient visit. All data after 2010 were included. Patients with prior cardiovascular events were excluded. Our composite endpoint consisted of acute ischemic stroke, acute myocardial infarction, deep vein thrombosis/pulmonary embolism and intravenous thrombolytic administration. We stratified our analysis according to CHC exposure and aura status and compared the incidence of the endpoint with high-dimensional propensity score-matching between CHC users and non-users in (i) the overall cohort, (ii) MwA and (iii) migraine without aura (MwoA); between MwA and MwoA in (iv) patients prescribed CHCs; and in (v) those without CHC prescriptions.
Results
We included 5535 patients who received CHC prescriptions and 21,520 who did not. 114 (2.06%) of CHC users and 547 (2.54%) of CHC non-users had at least one vascular event. With propensity score-matched comparison, the composite endpoint did not significantly differ between CHC and non-CHC in the overall migraine group, those with MwA and the MwoA group. In those prescribed CHC, MwA and MwoA did not differ in all the outcomes. For CHC non-users, MwA was associated with a higher incidence of acute ischemic stroke (hazard ratio = 2.45; 95% confidence interval = 1.58–3.78; p < 0.001; n = 6201 in each group) and the composite endpoint (hazard ratio = 1.34; 95% confidence interval = 1.08–1.67; p = 0.008).
Conclusions
Our real-world study showed that exposure to modern CHC was not associated with a significant increase in vascular risk in women aged 18–45 years with migraine, MwA or MwoA who have no prior cardiovascular events. However, in those who never received CHC, MwA was associated with higher vascular risks compared to MwoA. While limitations exist using large scale electronic medical record databases for analysis, our results suggest that carefully designed prospective studies should be conducted to reassess the vascular risk associated with CHC use in women with migraine, especially MwA.
This is a visual representation of the abstract.
Introduction
Migraine is a highly prevalent neurological disorder in women, particularly those of reproductive age (1–3) for whom contraceptives measures are often prescribed for family planning and treatment of gynecological conditions. Migraine, especially migraine with aura (MwA) is an established independent risk factor for vascular events, such as acute ischemic stroke (AIS) (4,5), acute myocardial infarction (AMI) (6–8), pulmonary embolism (PE), and deep vein thrombosis (DVT) (9,10). Estrogen-containing combined hormonal contraceptives (CHCs) are also independent risk factors for venous, and to a lesser extent, arterial thromboembolic events (5,11,12). Based on limited existing literature, concerns have been raised about further increasing vascular event risk (13–17) with CHC use in persons with migraine, especially MwA. Many guidelines recommend against the use of CHCs in patients with MwA (13,14,16,18–21). For example, the World Health Organization (13) lists MwA as a contraindication for CHC in women at any age, given the increased risk of stroke in patients with MwA compared to those with migraine without aura (MwoA), and the increased risk of ischemic stroke in oral CHC users with migraine compared to CHC non-users with migraine. Similarly, the American College of Obstetricians and Gynecologists states that CHCs are not recommended for women with MwA, as they list MwA, increasing age and CHC use as independent risk factors for stroke, even though the absolute risk of ischemic stroke is considered low in women of reproductive age (14).
Importantly, due to the low prevalence of vascular events in young womens, previous evidence has been based on small cohorts often without specifying migraine subtypes and lacking sub-group comparisons within MwA and MwoA (16,22). Many studies compared those with migraine and CHC prescription to healthy controls without CHC use, lacking information on the effect of individual factors. Additionally, most studies did not conduct stratified analysis with propensity-matched cohorts, and the results could have been influenced by various confounding factors, such as demographic factors and comorbidities that increase vascular risks (23–25).
Moreover, the putative mechanism for the increased vascular risks associated with CHC use in women with MwA is hypercoagulability, which is linked to estrogen levels in a dose-dependent manner (5,23,26). The amount of estrogen in modern CHCs (typically <50 μg) is substantially lower compared to when first introduced in the 1960s (approximately 150 μg) (23,27–30). Therefore, it is possible that vascular risks in patients with MwA may not significantly change with modern CHCs.
A large proportion of women of reproductive age use contraceptives. In the United States (U.S.), during 2017–2019, 65.3% of women aged 15–49 years used some method of contraception, and 14.0% used oral contraceptive pills (31). Additionally, CHCs are often used to treat conditions such as dysmenorrhea and endometriosis. Menstrual migraine is also sometimes treated with CHCs, although the evidence level remains low (32). The limitations in previous studies and changes in CHC formulations over the last few decades warrant further investigation of vascular risk in women with migraine, especially MwA, who use CHCs. In the present study, we leveraged a nationwide electronic medical record database to assess the vascular risk associated with modern CHCs in a female migraine cohort.
Methods
Study design and ethics
The study utilized a nationwide de-identified database and the analysis of group-level results without the storage of individual patient-level information. The Mayo Clinic Institutional Review Board (IRB) determined that formal ethics review was not required, as confirmed by the Mayo Clinic IRB Human Subjects Research Wizard tool.
Data source
We used the Eos Database as part of Atropos Health's Evidence Network (33), which is an Electronic Health Record Integrated Database that includes deidentified data from over 120 million patients in the U.S. The data come from point-of-care entry of patients with all payer types seen by all levels of providers, including primary care clinicians, neurologists and headache specialists, and are sourced from more than 30 health systems that collectively include >2000 outpatient/ambulatory health centers and >500 hospitals, including both academic centers and private practices. Data were restricted to the years 2010–2023.
Inclusion and exclusion criteria
Eligible patients were women aged 18–45 years with an International Classification of Diseases (ICD)-9 or ICD-10 diagnosis code for migraine. A list of ICD codes used to determine MwA and MwoA is provided in the supplementary material (Table S1). As patients with MwA can have attacks with or without aura, patients who had received a diagnostic code of both MwA and MwoA at any time were classified as MwA. The date of the first outpatient visit was used as the index date and date of inclusion. To ensure patients included were actively being followed or treated for migraine, only patients with at least three office visits within the first three years, and who had at least one prescription for a migraine-specific medication in the first six months following the index date were included. Migraine-specific medications included sumatriptan, rizatriptan, naratriptan, frovatriptan, eletriptan, zolmitriptan, almotriptan, ergotamine, dihydroergotamine, ubrogepant, rimegepant, atogepant, zavegepant, lasmiditan, erenumab, galcanezumab, fremanezumab and eptinezumab. Finally, eligible patients were required to have at least one prescribed medication (i.e. a prescription for either a CHC or another non-CHC medication) within one year of inclusion. Patients with a history of heart failure, myocardial infarction, peripheral vascular disease, cerebrovascular disease and paraplegia before the index date were excluded (see supplementary material, File S1). Patients were further required to have at least 90 days of history prior to the index date.
Exposures
CHCs were defined as any combination contraceptive, including oral, vaginal and transdermal routes of administration. Prescriptions were identified from Anatomical Therapeutic Chemicals codes (34) and RxNorm codes (35), which included 24 oral, three topical, 11 transdermal and eight vaginal CHC treatments (see supplementary material, File S2). Eligible women with a CHC prescription within one year of the index date were classified in the CHC exposure group. The control group consisted of eligible women with no CHC prescription but had at least one non-CHC prescription within one year of the index date. Requiring patients to have received a prescription in the one-year window, CHC or non-CHC, was to ensure the prescription was relevant to the index visit and patients were actively receiving healthcare.
Outcomes
We defined the following individual outcomes based on ICD-9 or ICD-10 codes assigned to medical encounters after the index date: AIS, AMI and DVT/PE. To ensure including patients with the above acute conditions, we defined intravenous thrombolytic administration as another outcome to be included in the composite endpoint, based on prescription records. Finally, we defined a composite endpoint as the combination of all the above individual outcomes (see supplementary material, File S1). We chose not to include transient ischemic attack, as, in clinical practice, many patients who present with transient neurological episodes from non-vascular causes are often mislabeled as transient ischemic attack. Intracerebral hemorrhage and subarachnoid hemorrhage were not included in the composite outcome given their lower prevalence compared to other outcomes, and the association between hemorrhage and MwA is less certain (36).
Covariates
We used patient demographics, procedure, and prescription codes prior to the index date to define covariates of interest. Covariates included age, sex, race/ethnicity, comorbid illnesses, concurrent prescriptions and healthcare utilization. A patient's comorbidities were quantified by the Charlson Comorbidity Index (37,38), which is a scoring system commonly used in electronic health record-based analysis to quantify the burden of a patient's comorbid conditions, including malignancy, solid tumor, diabetes mellitus with or without complications, chronic pulmonary disease, dementia, mild/severe liver disease, renal disease, peptic ulcer disease, connective tissue disease, congestive heart failure, peripheral vascular disease, leukemia/lymphoma and acquired immunodeficiency syndrome. Although some of these comorbidities overlap with those listed in the exclusion criteria, we first excluded patients based on the predefined exclusion criteria and subsequently extracted the remaining comorbidities for propensity score matching.
Statistical analysis
To best assess the effect of CHC exposure and aura status on vascular outcomes, we conducted the following five analyses and compared the rates of the composite endpoint and individual endpoints between the two groups in each of the following comparisons.
CHC vs. non-CHC in the overall migraine group. CHC vs. non-CHC in MwA. CHC vs. non-CHC in MwoA. MwA vs. MwoA in CHC. MwA vs. MwoA in non-CHC.
Here, CHC represents patients who were prescribed CHCs; non-CHC represents patients who were never prescribed CHCs; MwA represents patients with migraine with aura; and MwoA represents patients with migraine without aura (Figure 1).

Number of patients included in each sub-group. CHC = combined hormonal contraceptives; MwA = migraine with aura; MwoA = migraine without aura.
For each analysis, we compared the baseline covariates between the two groups. For each individual outcome, and for the composite outcome, we used Cox proportional hazards models to compare time-to-event between the treatment and control group, or between those with MwA vs. MwoA. Models were adjusted for covariates using high-dimensional propensity score matching (39). We fit exposure propensity score models using lasso regression (40) and then matched the study and control groups women using a 1:1 ratio based on the propensity score. All cohort selection and statistical analysis were performed using R, version 4.2 (R Foundation, Vienna, Austria) and the detailed methodology of high-dimensional propensity score matching was previously published (41–43).
Results
Population
From a total of >160 million patients in the database, we used data of 27,055 female patients with migraine according to the inclusion/exclusion criteria. We identified 5535 patients who were prescribed CHCs (CHC) and 21,520 patients who were not prescribed CHCs (non-CHC). The average age was 28.1 ± 7.9 years in CHC and 33 ± 8 in non-CHC. In CHC, 1514 (27.35%) patients had MwA and, in non-CHC, 6763 (31.43%) had MwA (Figure 1 and Table 1). The self-identified race/ethnicity distribution for CHC was: White 2880 (52%), other or unknown 2270 (41%), Black 278 (5%), Asian 107 (1.9%) and Hispanic 329 (5.9%). The distribution for non-CHC was: White 10,643 (49.5%), other or unknown 8864 (41.2%), Black 1552 (7.2%), Asian 461 (2.1%) and Hispanic 1307 (6.1%). Mean follow-up days were 1745.7 ± 1244.1 in CHC and 1448.2 ± 1147.8 in non-CHC. The detailed demographics and comorbidity scores included in the Charlson Comorbidity Index of both groups (CHC vs. non-CHC) are summarized in Table 1.

(I) to (V) Survival probability in the composite outcome. The number of events in each propensity score-matched group is indicated within parentheses. Purple and green lines represent survival probabilities with 95% confidence intervals shown with shaded areas. CHC = patients who were prescribed CHCs; non-CHC = patients who were not prescribed CHCs; HR = hazard ratio; MwA = patients with MwA; MwoA = patients with MwoA.
Demographic data and comorbidities of the overall cohort.

Age and follow-up days are reported as the mean
CHC = combined hormonal contraceptives; SD = standard deviation.
Results comparing CHC vs. non-CHC in the (i) overall migraine cohort, (ii) MwA subgroup and (iii) MwoA subgroup
In the overall female migraine cohort, the propensity score-matched comparison included 4720 patients in each group and showed no significant difference in the composite outcome with 104 events in CHC and 89 events in non-CHC and a hazard ratio (HR) of 1.2 (95% confidence interval (CI) = 0.89–1.61; p = 0.23). Comparison of each individual outcome also did not show any significant difference between CHC and non-CHC (AIS, HR = 1.05; 95% CI = 0.59–1.86; p = 0.86; AMI, HR = 1.43; 95% CI = 0.57–3.57; p = 0.44; DVT/PE, HR = 1.2; 95% CI = 0.8–1.78; p = 0.38). (Figures 1 and 2 and Table 2).
Comparison of survival probability (i) between CHC and non-CHC in the overall female migraine cohort; (ii) between CHC and non-CHC in MwA; (iii) between CHC and non-CHC in MwoA; (iv) MwA and MwoA in CHC; and (v) MwA and MwoA in non-CHC.
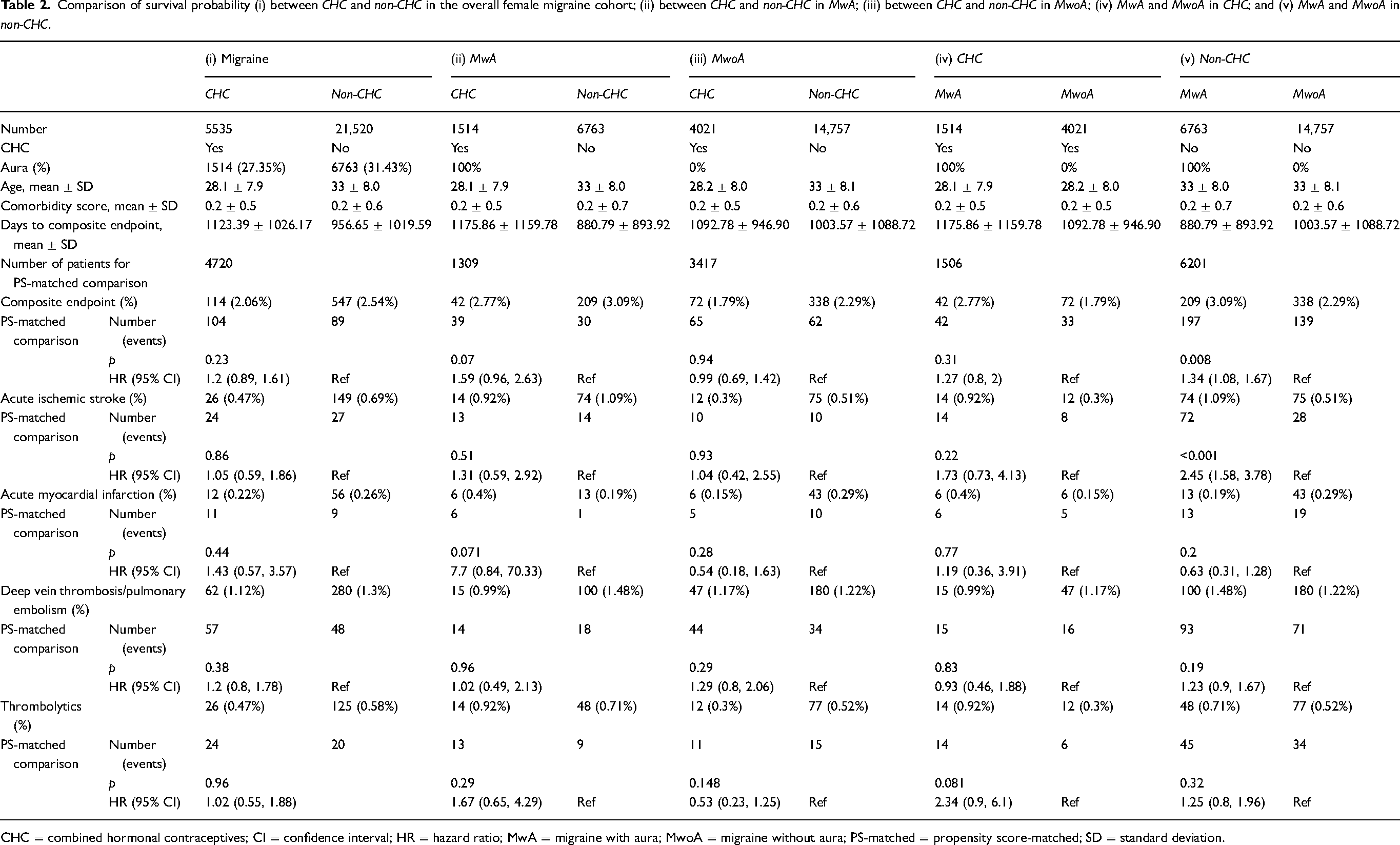
CHC = combined hormonal contraceptives; CI = confidence interval; HR = hazard ratio; MwA = migraine with aura; MwoA = migraine without aura; PS-matched = propensity score-matched; SD = standard deviation.
Among the 8277 patients with MwA, 1514 patients were prescribed CHCs, and 6763 were never prescribed CHCs. The propensity score-matched comparison of 1309 patients from each group included 39 events in CHC and 30 events in non-CHC among those with MwA and revealed no significant difference in the composite outcome (HR = 1.59; 95% CI = 0.96–2.63; p = 0.07). We then examined the 1514 patients with MwA who were prescribed CHCs. Among them, 368/1514 (24.3%) had CHC prescriptions after the date of MwA diagnosis and 8/368 (2.17%) of them experienced at least one composite outcome. For 1146 (75.7%) patients, CHC prescriptions were written before MwA was diagnosed and 34/1146 (2.96%) had at least one composite outcome.
As for the 18,778 patients with MwoA, 4021 patients had CHC and 14,757 did not. The composite outcome occurred in 65 in CHC and 62 in non-CHC in the propensity score-matched group of 3417 patients in each category, and no significant difference was observed (HR 0.99; 95% CI, 0.69–1.42; p = 0.94) (Table 2).
Results comparing MwA vs. MwoA in (iv) those who were prescribed CHC and (v) non-CHC patients
For the 5535 who had been prescribed CHC, 1514 had MwA and 4021 had MwoA. The composite outcome occurred in 42 in MwA and 33 in MwoA in propensity score-matched groups of 1506 patients and did not show any significant difference between MwA and MwoA (HR = 1.27; 95% CI = 0.8–2; p = 0.31).
For the 21,520 who had never been prescribed CHC, 6763 had MwA and 14,757 had MwoA. The propensity score-matched comparison included 6201 patients in each group and demonstrated a significantly higher incidence of AIS (72 events vs. 28 events; HR = 2.45; 95% CI = 1.58–3.78; p < 0.001) and the composite endpoint (197 events vs. 139 events; HR = 1.34; 95% CI = 1.08–1.67; p = 0.008) in those with MwA compared to MwoA. In Table 2, we list the detailed numbers of patients included in each of the five analyses listed above, the number of patients used for each propensity score-matched comparison, and the number of each vascular outcome analyzed in each comparison.
The survival probability curve of the comparisons are presented in Figure 2. The characteristics of propensity-matched groups are summarized in the supplementary material (File S3). The standardized mean differences were <0.1, except for the following factors: (i) mean follow-up days; (ii) mean pre-index days and mean follow-up days; (iii) mean follow-up days; and (iv) race/ethnicity – Black. The number of patients at risk is provided in the supplementary material (File S4).
Discussion
Our real-world analyses on a nationwide de-identified electronic medical record database utilizing high dimensional propensity score matching comparisons demonstrated that there was no statistically significant difference in the risk of developing the composite vascular endpoint, or any of the individual outcomes, including AIS, AMI and DVT/PE in women with migraine, aged 18–45 years, who have been prescribed estrogen-containing CHCs compared to those who have never received a CHC prescription in the overall migraine group, and separately in the MwA and MwoA subgroups. Furthermore, we found that the risks of the composite outcome and AIS were significantly increased in MwA compared to MwoA for those never exposed to CHCs, but not for patients who had CHC prescriptions.
Despite some guidelines suggesting that MwA is a contraindication to CHC, we found that 368 patients had CHC prescribed after the MwA diagnosis date, and 8/368 (2.17%) developed the composite endpoint, which was similar to the rate of composite endpoint of the overall cohort (114/5535 (2.06%) in CHC and 547/21,520 (2.54%) in non-CHC). This numbers highlights the importance of our findings in real-world practice, as many female patients received a CHC prescription even after MwA was diagnosed.
Previous studies evaluating the risk of CHC in migraine overall suggested an increased risk of ischemic stroke or cardiovascular outcomes, especially in patients with MwA and CHC use. However, most studies compared the risk of patients with migraine using CHCs to healthy controls not on CHCs, while very few studies have directly compared the risk of using CHCs to not using CHCs in patients with migraine, MwA and MwoA (16,22). A population-based, case-control study including 25,887 patients with ischemic stroke was conducted from 2006 through 2012 and showed that those with MwA using CHC were associated with a six-fold increased risk of ischemic stroke compared to those without migraine without CHC use, emphasizing the joint effect of CHCs and MwA (17), Our results are consistent with the data reported, such that using CHC did not significantly increase ischemic stroke risks in those with MwoA. However, for those with MwA, our results suggested no significant difference in the risk of vascular outcomes between CHC users and non-users, and there was an increased risk of ischemic stroke in MwA compared to MwoA in those who were never prescribed CHC, suggesting that MwA is likely the main contributor of the vascular risk. These differences could be due to differences in covariates, outcome definitions, the types of CHCs and inclusion criteria, which were limited to patients with private insurance in the prior study. Another study in 1998 investigated the risk of stroke associated with the use of low-dose CHCs with <50 μg estrogen by conducting a pooled analysis of two US population-based case–control studies and concluded that low-dose CHCs could increase risks of ischemic stroke in those with a history of migraine (44), but not in those without migraine. However, the number of patients with migraine in the case group was low (n = 51 for ischemic stroke; n = 50 for hemorrhagic stroke), migraine subtypes were not specified and only participants who could be directly interviewed were included in the study, introducing the possibility of non-participation bias.
In 2018, the European Headache Federation and the European Society of Contraception and Reproductive Health published a consensus statement about the association between hormonal contraceptives and risks of ischemic stroke (16,22). They demonstrated that the absolute risk of ischemic stroke in women aged 20–44 years without migraine without CHCs is 2.5/100,000 per year, while, in MwA without CHCs, it is 5.9/100,000 per year, an, in MwA with CHCs, it is further increased to 14.5/100,000 per year. The statement recommends “a clinical evaluation for the presence of migraine, for the definition of migraine subtype (i.e., with or without aura) and migraine frequency together with the ascertainment of conventional vascular risk factors before prescription of combined hormonal contraceptives” and considers that CHCs with >35 μg ethinyl estradiol are “high-risk products”, CHCs containing ≤35 μg and combined contraceptive patch as “medium risk products” and progestogen-only contraceptives as “no risk products”. After the EHF statement, several recent studies were published on this topic. A systematic review by Sheikh et al. (24) summarized relevant literature until January 2016 regarding the risks of stroke associated with migraine and CHCs, focusing on whether the risk of stroke is associated with estrogen dose and whether there is synergism between migraine and CHCs. It was concluded that previous evidence suggests an additive increase in risks of stroke with using CHCs in those with MwA, although insufficient sample sizes might result in lower level of evidence of prior studies. Additionally, a case-control study by Batur et al. (26) on 127 patients with confirmed ischemic stroke while on CHCs, including 14 patients with MwA and 20 with MwoA, revealed that stroke risks were higher with CHCs with ≥30 μg ethinyl estradiol compared to those with <30 μg dose, highlighting dose-dependency of vascular risks with ethinyl estradiol. Notably, they revealed that compared to those without migraine, stroke risk increased only in those with MwoA, and not in those with MwA.
Notably, the risk of the composite outcome or AIS was not significantly increased in our propensity-matched comparisons for those exposed to CHC, in the overall migraine group, MwA and MwoA. This finding would corroborate the findings of a systematic review published by Ornello et al. (25), such that the increased risks of ischemic stroke in young female patients with migraine using CHCs is driven mainly by MwA, rather than by low-dose CHCs. Similar to the study findings presented by Batur et al. (26), it is also possible that for patients with MwA, only those with a lower vascular risks profile were prescribed CHCs. In addition, based on the previous hypothesis and reports that the vascular risks associated with CHC in migraine are dose-dependent (23), it is possible that the differences observed in our study compared to prior studies are resulting from a much lower dose of ethinyl estradiol included in CHC products over the past 15 years, reducing from 150 μg in the 1960s to mostly lower than 50 μg currently (23,27–30). Our results suggest the use of modern CHC might not significantly increase vascular risks in patients with migraine, regardless of migraine subtypes, although a limitation of our study is that we do not have the dosage information in the database to conduct additional evaluations of the effect of the dose of estrogen on vascular risk.
Our study depicted the increased vascular risks in patients with MwA compared to MwoA in patients who did not use CHCs, but not in patients who used CHCs. The increase in vascular risks in patients with MwA compared to MwoA has been repeatedly shown in previous studies (4,6,8,45–48). However, it is intriguing that this finding was not observed in those who received CHCs. Given the large electronic medical record database, we employed strict inclusion criteria to ensure patients included were being actively treated and followed, and only women aged 18–45 years were included, comprising a population typically with a lower risk of vascular events. Since the absolute number of vascular events included in CHC group (MwA, 42 (2.77%); MwoA, 72 (1.79%)) is smaller compared to those observed in non-CHC group (MwA, 209 (3.09%); MwoA, 338 (2.29%)), the insignificant result in MwA population could be attributed to a lack of statistical power issue and does not necessarily exclude increased vascular risks of CHCs in those with MwA. Additionally, it might be possible that mildly increased estrogen levels with CHC use exhibit protective effects for cardiovascular systems by reducing oxidative stress (49,50) or having anti-inflammatory effects (51). It has been suggested that continuing ultra-low dose formulations of ethinyl estradiol could potentially decrease the frequency of aura (23). As an association between aura or migraine frequency with the risk of stroke or cardiovascular diseases has been suggested (52–54), a reduction in aura frequency from CHC use could have led to decreased stroke risks. Additionally, given the guideline suggestions (21), it is possible that clinicians only prescribed CHC to patients with MwA who had a low baseline vascular risk profile, resulting in potential selection bias.
The strengths of our study include the large number of patients across different electronic medical record systems from multiple healthcare systems in the U.S. By employing high-dimensional propensity score matching, we were able to adjust for multiple potential confounding factors, including demographics and comorbidities. We also included multiple vascular events, including both arterial and venous thromboembolism, to capture most possible adverse outcomes associated with estrogen use. Furthermore, leveraging our carefully designed group-based comparison methodologies on this large cohort of patients, we were able to conduct five separate analyses, stratified by CHC exposure and aura status, to investigate the effect of CHC exposure and aura status on vascular outcomes in each subgroup. Our results add to the literature because most previosuly published studies reporting the vascular risk of estrogen in migraine did not specifically compare between migraine subtypes or stratify patients by estrogen exposure.
Our study has several limitations. Given the concerns of vascular outcomes associated with MwA, it is likely that patients who were prescribed CHC had an overall low vascular risk profile or had better access to care. Although we have maximized ways to mitigate this selection bias by ensuring all patients had at least three office visits and received migraine-specific medications, and taking into account of their age, race/ethnicity, number of encounters and comorbidities in our high dimensional propensity matching, there might be intrinsic limitations of such retrospective electronic medical record research that are difficult to capture, such as socioeconomic status and access to care. Although our study utilized a large database, each subgroup comparison for the composite vascular outcome included fewer than 200 events, and individual outcomes such as AMI involved smaller event counts. This likely reflects our strict inclusion criteria, restricting the cohort to women aged 18–45 years who were actively receiving treatment for migraine, as well as the inherently low incidence of vascular events in this age group. Nevertheless, the limited number of events may have reduced the statistical power of our analyses. Previous studies suggested the combination of smoking and CHC significantly increased the risk of stroke in patients with MwA. However, structured information on smoking is not available in the current electronic medical record database and therefore not included. Future studies, especially those involving smaller cohorts with detailed chart review, or prospective data collection including smoking as a factor, could address this limitation more directly. Additionally, other factors that could influence the vascular risk were not specifically stratified in our analysis, such as hypertension, hyperlipidemia, inherited or acquired hypercoagulable states, as those were not specifically included in the Charlson Comorbidity Index. However, our analysis excluded patients with a history of heart failure, myocardial infarction, peripheral vascular disease, cerebrovascular disease and paraplegia before the index date. As previous studies suggest that the risk of vascular events is dose-dependent and more common during the first year of use (55), more studies are needed to investigate the vascular risk among CHC products of different estrogen dose ranges and duration of estrogen exposure, which were not available in the current database. Lastly, we used ICD codes to ascertain aura status and vascular events, which could result in inaccuracies in determining aura status or the diagnosis of vascular events. Although previous studies suggest that ICD codes could diagnose incident stroke with high specificity (56), the accuracy of migraine subtype diagnosis based on ICD codes might be limited, especially when patients are seen by non-headache specialists. Given the nature of the de-identified electronic medical record database, clinical note information was not available nor accessible to conduct chart review.
In conclusion, our large-scale observational study suggests that modern estrogen-containing CHCs did not significantly increase the risk of vascular events, including AIS, AMI and DVT/PE in young women with migraine who have no prior cardiovascular events, regardless of aura status. Our results suggest that the need to list MwA as a contraindication of modern CHC use should be carefully reassessed, and future prospective research evaluating the risk of adverse vascular events with low dose estrogen use in women with migraine could be conducted.
Article highlights
We conducted a large-scale observational study using a deidentified electronic medical record database that includes patients from multiple healthcare systems in the U.S. and compared the risk of adverse vascular outcomes in women with migraine, aged 18–45 years, stratified by CHC exposure and aura status.
Our results suggest that modern estrogen-containing CHCs did not significantly increase the risk of vascular events, including acute ischemic stroke, acute myocardial infarction and deep vein thrombosis/pulmonary embolism in those with migraine, regardless of aura status.
The vascular risk associated with CHC use in women with migraine should be carefully reassessed, especially in those with MwA.
Supplemental Material
sj-xlsx-1-cep-10.1177_03331024251404924 - Supplemental material for Estrogen exposure from modern contraceptives and vascular risk in women with migraine: A nationwide electronic medical record database study
Supplemental material, sj-xlsx-1-cep-10.1177_03331024251404924 for Estrogen exposure from modern contraceptives and vascular risk in women with migraine: A nationwide electronic medical record database study by Keiko Ihara, C. William Pike, Gavin Hui, Saurabh Gombar, Michael L. Jackson, Alison Callahan, Gretchen E. Tietjen and Chia-Chun Chiang in Cephalalgia
Supplemental Material
sj-docx-2-cep-10.1177_03331024251404924 - Supplemental material for Estrogen exposure from modern contraceptives and vascular risk in women with migraine: A nationwide electronic medical record database study
Supplemental material, sj-docx-2-cep-10.1177_03331024251404924 for Estrogen exposure from modern contraceptives and vascular risk in women with migraine: A nationwide electronic medical record database study by Keiko Ihara, C. William Pike, Gavin Hui, Saurabh Gombar, Michael L. Jackson, Alison Callahan, Gretchen E. Tietjen and Chia-Chun Chiang in Cephalalgia
Supplemental Material
sj-xlsx-3-cep-10.1177_03331024251404924 - Supplemental material for Estrogen exposure from modern contraceptives and vascular risk in women with migraine: A nationwide electronic medical record database study
Supplemental material, sj-xlsx-3-cep-10.1177_03331024251404924 for Estrogen exposure from modern contraceptives and vascular risk in women with migraine: A nationwide electronic medical record database study by Keiko Ihara, C. William Pike, Gavin Hui, Saurabh Gombar, Michael L. Jackson, Alison Callahan, Gretchen E. Tietjen and Chia-Chun Chiang in Cephalalgia
Supplemental Material
sj-xlsx-4-cep-10.1177_03331024251404924 - Supplemental material for Estrogen exposure from modern contraceptives and vascular risk in women with migraine: A nationwide electronic medical record database study
Supplemental material, sj-xlsx-4-cep-10.1177_03331024251404924 for Estrogen exposure from modern contraceptives and vascular risk in women with migraine: A nationwide electronic medical record database study by Keiko Ihara, C. William Pike, Gavin Hui, Saurabh Gombar, Michael L. Jackson, Alison Callahan, Gretchen E. Tietjen and Chia-Chun Chiang in Cephalalgia
Footnotes
Author contributions
Conceptualization: CC. Data curation: WP, GH, SG, MJ and AC. Formal analysis: WP, GH, SG, MJ and AC. Methodology: WP, GH, SG, MJ, AC and CC. Writing-original draft: KI and CC. Writing-review and editing: KI, CCC, GT, WP, GH, SG, MJ and AC.
Data availability
Due to contractual limitations, the data analyzed in the current study are not immediately available from Atropos, but may be available from the upstream data provider. Restrictions apply to the availability of these data, which were used under license for this study. Data are available from the authors with the permission of Atropos Health.
Declaration of conflicting interests
KI received American Heart Association Postdoctoral Fellowship Grant 2025, with funds paid to her institution; WP, GH, SG, AC and MJ, are employees of Atropos Health; GT serves as the Medical Advisory Board of Impel Pharmaceuticals and H. Lundbeck A/S. Spouse owns common stock in Johnson & Johnson; CC has served as a consultant for Pfizer, AbbVie, Amneal, Satsuma and eNeura. She has received research support from the American Heart Association, with funds paid to her institution.
Ethical considerations
As this study utilized de-identified databases and only involved the receipt of analyzed results without the storage of patient-level information, the Mayo Clinic Institutional Review Board (IRB) determined that formal ethics review was not required, as confirmed by the Mayo Clinic IRB Human Subjects Research Wizard tool.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
